MCAT GEN CHEM
1/41
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
42 Terms
Nucleons refer to
Protons and neutrons, forming the nucleus of the atom
The mass of 1 electron is approximately
0 AMU (atomic mass units), or 0 e
The number of ____ determines the element a specific atom is, and is refered to as the ____ _____.
The number of protons determines the element a specific atom is, and is refered to as the atomic number.
Atoms of the same atomic number but with differing numbers of neutrons are _______
Atoms of the same atomic number but with differing numbers of neutrons are isotopes
What is the atomic weight of an element?
The atomic weight is the average mass of all naturally occurring isotopes of an element
What keeps the protons held so closely together in a stable way in the nucleus, even though like charges naturally repel one another?
The strong nuclear force is a fundamental force that prevents this. In particular, it is an attractive force that holds protons and neutrons together—despite any alike positive charge.
The weaker the nuclear force, the more radioactive decay occurs.
Per the Aufbau Principle, what is the correct order in which the orbitals are filled?
Write it out with arrows!
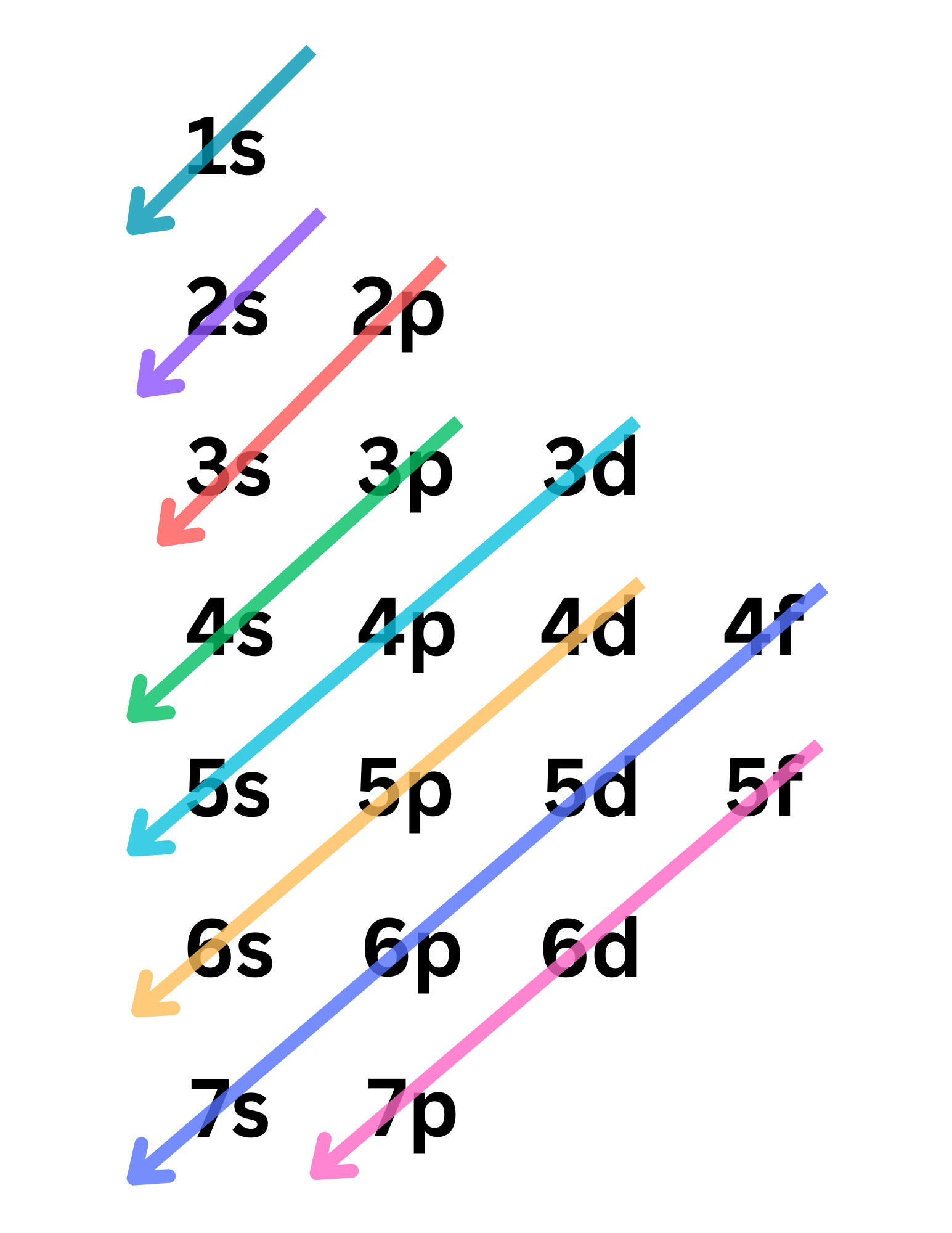
Electron configuration can be organized into s, p, d, and f, which are _______. How many orbitals does each one have, which translates to how much electron capacity for each?
s, p, d, and f are subshells. Each orbital contains a max of 2 electrons with opposite spins (Pauli Exclusion Principle). So…
s → 1 orbital → 2 electrons
p → 3 orbitals → 6 electrons
d → 5 orbitals → 10 electrons
f → 7 orbitals → 14 electrons
What does the Pauli Exclusion Principle state about an electron’s quantum numbers within the same atom?
The Pauli Exclusion Principle states that NO electron in an atom can have the same four quantum numbers.
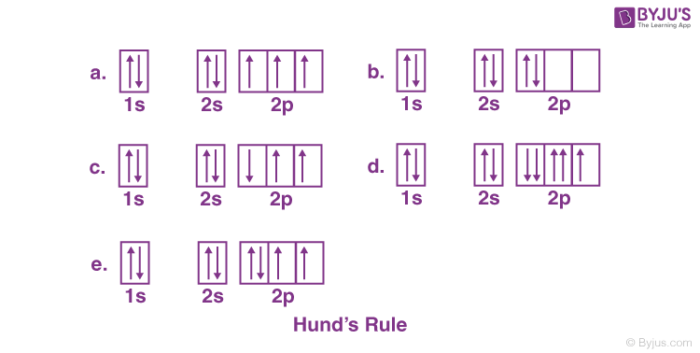
Based on Hund’s Rule, which option shows the correct electron configuration?
Choice A
Per Hund’s Rule, electrons within a subshell occupy orbitals singly with parallel spins before pairing up.
Explain how the concepts of shells, sub-shells, and orbitals can be explained relative to each other.
Shells AKA principle energy levels are denoted by n. On the periodic table, elements in each PERIOD (horizontal row) is found within the same shell/energy level (1-7).
Each of these shells have sub-shells (s/p/d/f AKA the Azimuthal quantum number) each labeled with the n value of the corresponding shell (e.g. 3s, 3p, 3d in the 3rd shell).
Each of these sub-shells have a corresponding number of orbitals that can exist within it, including 1 orbital for s, 3 orbitals for p, 5 orbitals for d, and 7 orbitals for f. Each orbital then includes max 2 electrons with opposite spins.
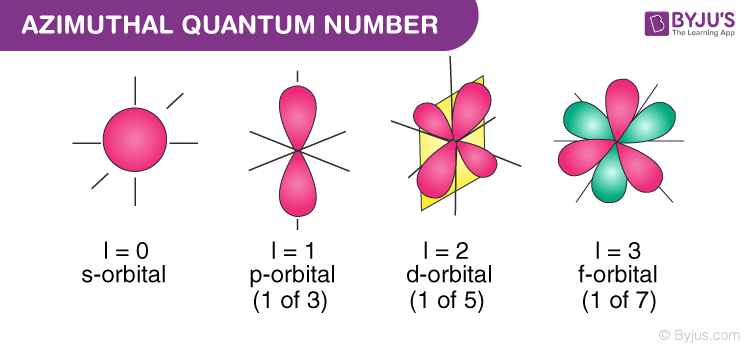
What are the azimuthal quantum numbers, and what are they describing?
Azimuthal quantum numbers, denoted by l, determines the sub-shell.
0 → s
1 → p
2 → d
3 → f
Describes the shape of the orbital and the orbital angular momentum.
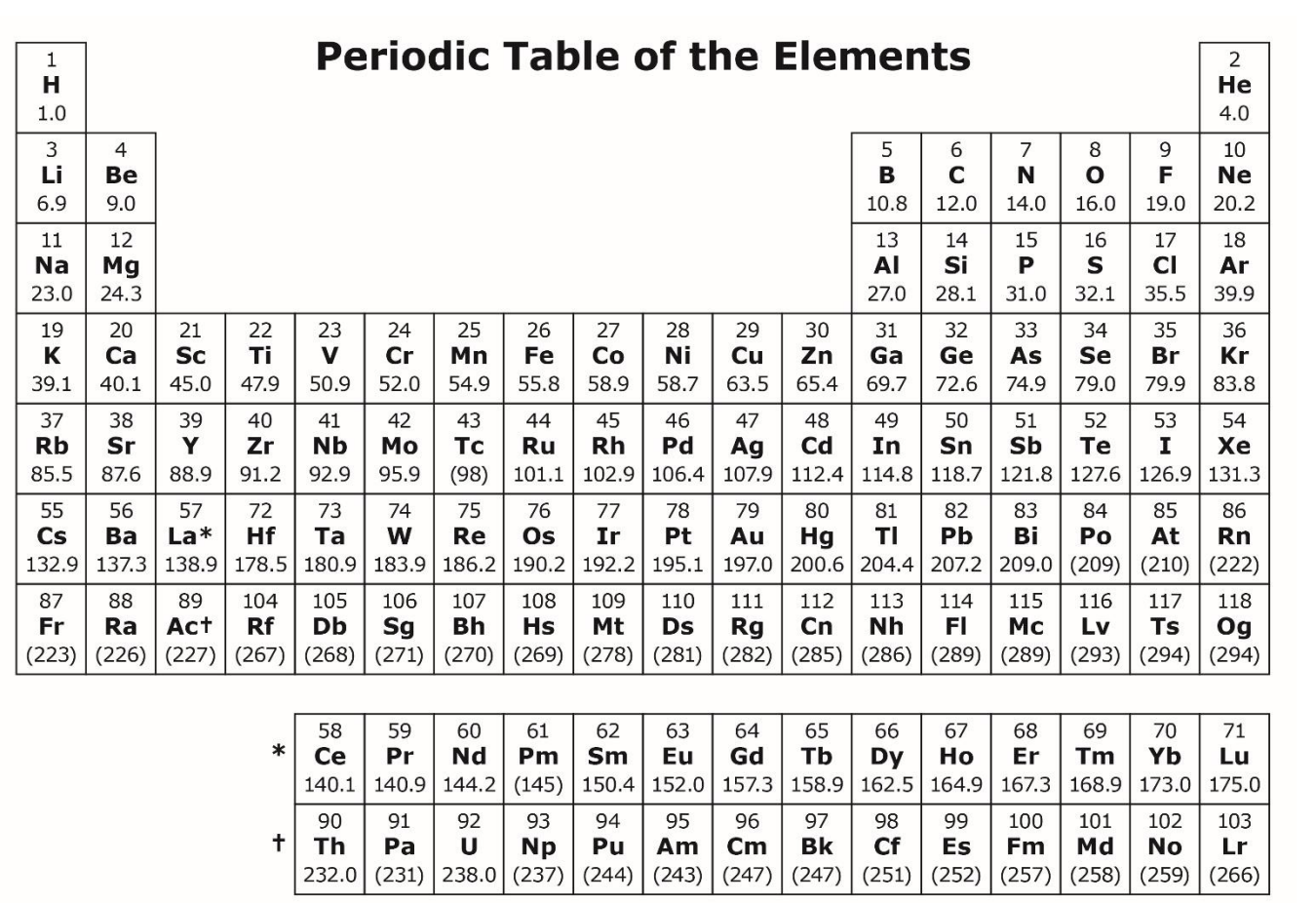
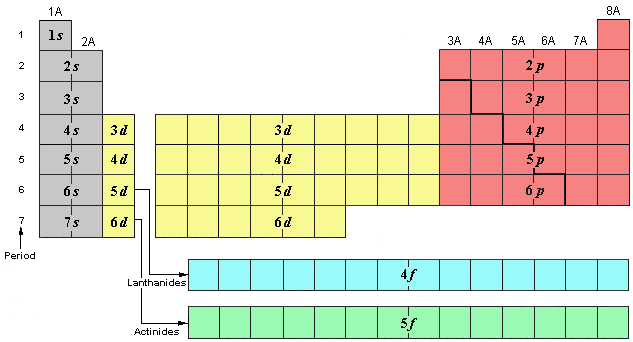
ID which areas are the: s-block, p-block, d-block, and f-block.
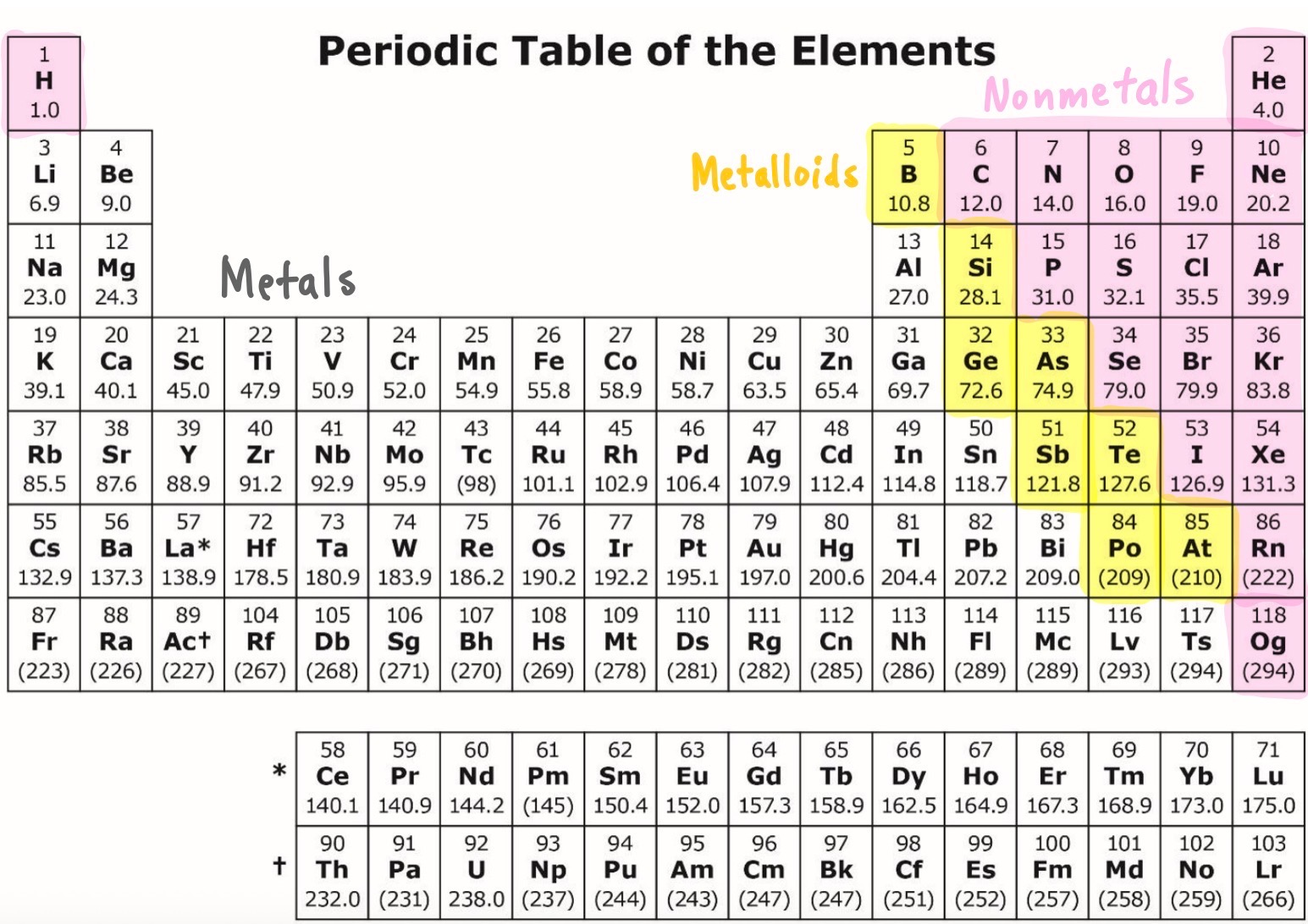
ID which areas are metals, metalloids, and nonmetals?
What is important to remember about the organization of the periodic table’s periods (vertical rows) with respect to energy levels in the d-block?
Instead of 3d being on the 3rd period/row like 3s or 3p, it is actually down on the 4th row, because 3d has higher energy than 4s. This trend continues down the d-block, where 4d is on the 5th row, 5d is on the 6th row, and 6d is on the 7th row.
What does it mean when an atom is diamagnetic, and what is its defining property with how it interacts with magnets?
If an atom’s electrons are all paired (e.g., in pairs of one spin up and one spin down electron), it is considered to be diamagnetic.
Diamagnetic materials do not interact strongly with magnets.
What does it mean when an atom is paramagnetic, VS more specifically ferromagnetic, and what are the defining properties with how they interact with magnets?
Paramagnetic atoms have unpaired electrons, with more spin up than down.
Most paramagnetic materials are usually strongly affected by nearby magnetic fields, but do NOT possess a permanent magnetic charge.
However, ferromagnetic materials are paramagnetic with an additional property: the electron spins of unpaired electrons can spontaneously align in the same direction. As a result, the ferromagnetic material creates its own magnetic field.
What does the magnetic quantum number represent and how can it be found?
The magnetic quantum number describes the possible spatial orientations of an s, p, d, or f orbital within a single shell.
Any number between -ℓ and +ℓ, with ℓ = azimuthal quantum number indicating the sub-shell type (s, p, d, or f).
ex:
If electron is in the d orbitals, then ℓ = 2. So, mℓ = -2, -1, 0, +1, +2 → 5 different orientations.
Halogens are always found in a _____ state and are ____(barely vs highly) reactive.
Halogens are always found in a diatomic state (2 atoms per molecule) and are highly reactive as they only need one more electron to fulfill the perfect octet rule.
D-block elements AKA _____ ____
D-block elements AKA transition metals
What is electronegativity?
On the periodic table, what is the trend for electronegativity?
Electronegativity refers to the attractive force the nucleus of an atom exerts to attract an electron.
Elements with nearly full valence shells have a higher electronegativity compared to elements with only one or two electrons in the valence shell, as gaining just a few/one more valence electrons would most easily get these near complete shells to the perfect octet, vs. it just being easier to lose just the one or two in near empy shells.
Elements with smaller atomic radius have a higher electronegativity, because smaller size leads to less electron shielding and higher effective nuclear charge. The valence shell and the desired electron(s) are going to be closer to the nucleus that is the origin of this force.
^Therefore, the electronegativity increases upwards and across to the right of the periodic table. Fluorine (F), a halogen, is highly electronegative, whereas barium (Ba), an alkaline earth metal, is not very electronegative.
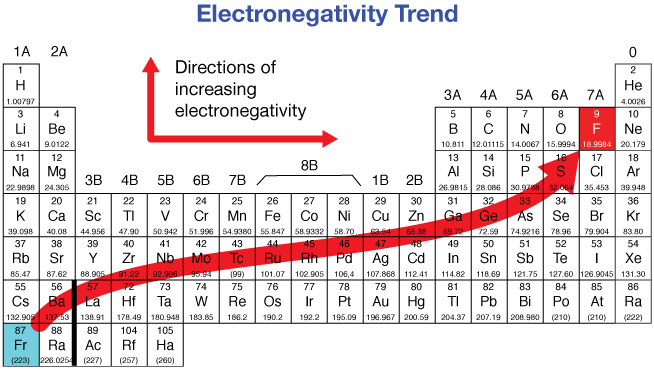
What is ionization energy?
What is its trend on the periodic table?
Ionization energy refers to the amount of energy needed to remove an electron from the valence shell of an atom.
From a stability standpoint, it is extraordinarily favorable to gain one last electron and complete the valence shell with a nearly full shell in a halogen. On the other hand, an alkali metal like potassium has one lone electron in the valence shell. From a stability standpoint, it is extraordinarily favorable to lose this electron—thus reverting the valence shell to the full electron shell of one lower period.
The smaller the atom, the closer and stronger the attractive energy from the nucleus. Therefore, more energy is needed to remove an electron from the valence shell of a smaller atom.
Thus, ionization energy increases upwards and across to the right of the periodic table.
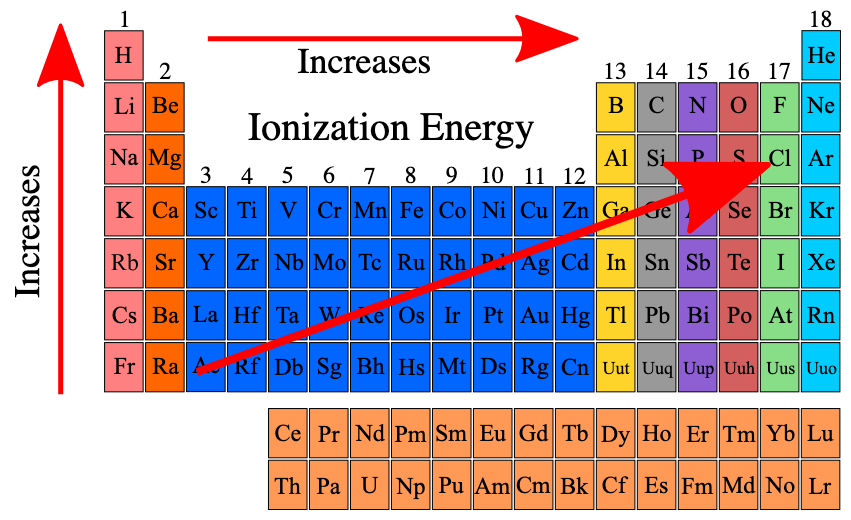
What is electron affinity?
What is its trend on the periodic table?
Electron affinity refers to the amount of energy released by an electron when it attracts an electron.
This is because attractive forces between the nucleus and valence electrons increase as distance to the valence shell decreases, and the addition of protons to a nucleus while holding this distance constant will increase the electrostatic attraction.
The closer to full the valence shell is, the more stabilizing it would be to attract the last remaining electrons needed to complete the shell. More stabilizing = more energy released.
Thus, electron affinity increases upwards and across to the right of the periodic table.
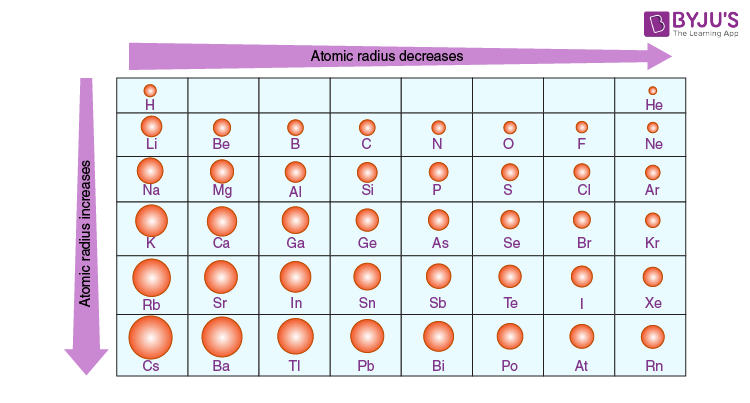
What is the trend of atomic radii on the periodic table?
Decrease from left to right across a period → if you add more electrons to the same shell, you are also accounting for more protons and more effective nuclear charge by the nucleus, pulling the electrons closer and making the atom more compact/small.
Increase down a group → more shells to the electron adds more size
What is effective nuclear charge and what are the two factors you must consider to gauge how strong it might be?
Effective nuclear charge (Zeff) is a measure of the net positive charge that electrons in an atom experience. Generally, this charge is applied to valence electrons when discussing the electrostatic attraction to the atomic nucleus. Two primary factors determine Zeff:
The number of protons in the nucleus of the atom
More protons = more Zeff for valence e-, since more positive charge can be emitted from the nuclus.
The presence of shielding electrons
More shielding electrons = less Zeff for valence e-, since there would be more distance/e- in between nucleus and valence shell.
Negative vs Positive ions → which can be expected to be bigger vs smaller than the original atom?
Negative ions are larger than the original atom as the attractive forces exerted by the nucleus are dispersed over a greater number of electrons.
Positive ions are smaller than the original atom because there are more positive nuclear forces to attract electrons toward the center of the atom
What is an acronym that can be used to remember the four quantum numbers for an electron, and what do they represent?
PAMS is the acronym
Principle energy level: the electron’s shell, denoted by n.
Azimuthal quantum #: the electron’s subshell, denoted by ℓ, in either the s, p, d, or f orbitals. Think of it as the SHAPE.
Magnetic quantum #: the number of possible orbitals in the electron’s subshell, denoted as mℓ. Think of it as the ORIENTATION.
Spin quantum #: the direction of the electron’s spin, denoted by ms. Either +1/2 or -1/2. Think of it as the MOVEMENT.
What phenomenon is described by the photoelectric effect?
Photoelectric effect: when an electron absorbs enough energy to match the energy of the next orbital and is able to move to a higher energy level.
When the electron releases energy to return to its ground state, photons can be emitted.
What are the equations for the energy of a photon/electron that can be applied to the photoelectric effect?
Include the relationship between the frequency, wavelength, and Rydberg equation.
E = hf = hc/λ = RH ((1/ni2) - (1/nf2))
Where:
E = energy of photon
h = Planck’s constant 6.626 × 10-34 J*s
f = frequency
c = speed light 3 × 108 m/s
λ = wavelength
RH = 1.097 × 107 m-1
ni = initial orbital level
nf = final orbital level
What does the Heisenberg uncertainty principle state?
The Heisenberg uncertainty principle states that both the position and velocity of a photon cannot be known at the same time.
This happens because the process of observing the particle itself disturbs its motion.
If we use light of very short wavelength, we can determine the position of the electron more accurately.
However, short wavelength light has high energy photons, which disturb the electron strongly and make its momentum uncertain.
On the other hand, if we use light of a longer wavelength, the disturbance is smaller, but the exact position of the electron cannot be determined accurately.
What kind of isotopes in terms of the # of neutrons relative to the # of protons are known to be more stable?
As a general rule of thumb, isotopes with an amount of neutrons approximately equal to the atomic number are more stable than their lighter variants.
Alpha decay involves the release of what kind of particle?
If 3015 P goes through alpha decay, what can you write out for the full reaction (including each mass/atomic number)?
Alpha decay involves the release of an alpha particle, AKA a helium nucleus with mass number 4 and atomic number 2.
3015 P → 42 He + 2613 Al
Phosphorus → Helium nucleus + Aluminum
Beta decay involves the release of what kind of particles?
If 3015 P goes through beta decay through both types of possible beta particles, what can you write out for the full reactions (including each mass/atomic number)?
What changes occur between neutrons and protons in the nucleus that is going through beta decay?
Beta decay involves the release of an beta particles, AKA an electron (0-1 e) OR positron (0+1 e).
Electron beta decay: 3015 P → 0-1 e + 3016 S
When an electron is ejected from the nucleus, a neutron (0) becomes a proton (+1)
Positron beta decay: 3015 P → 0+1 e + 3014 Si
When a positron is ejected from the nucleus, a proton (+1) becomes a neutron (0)
Gamma decay involves the release of what kind of particle?
If 3015 P goes through gamma decay, what can you write out for the full reaction (including each mass/atomic number)?
Gamma decay involves the release of a gamma particle, which is massless but with great amounts of energy, AKA 00 y
3015 P → 00 e + 3015 P
What is considered to be the most destructive type of radioactive decay?
Gamma decay
Even though it appears to lead to the least change as it involves a release of a massless particle, a gamma particle has higher amounts of energy than an alpha or beta particle.
Gamma rays are high-frequency, high-energy electromagnetic waves that pass through, damage, and ionize cells deep within the body.
The ____ bond forms between a nonmetal and metal.
Ionic bond
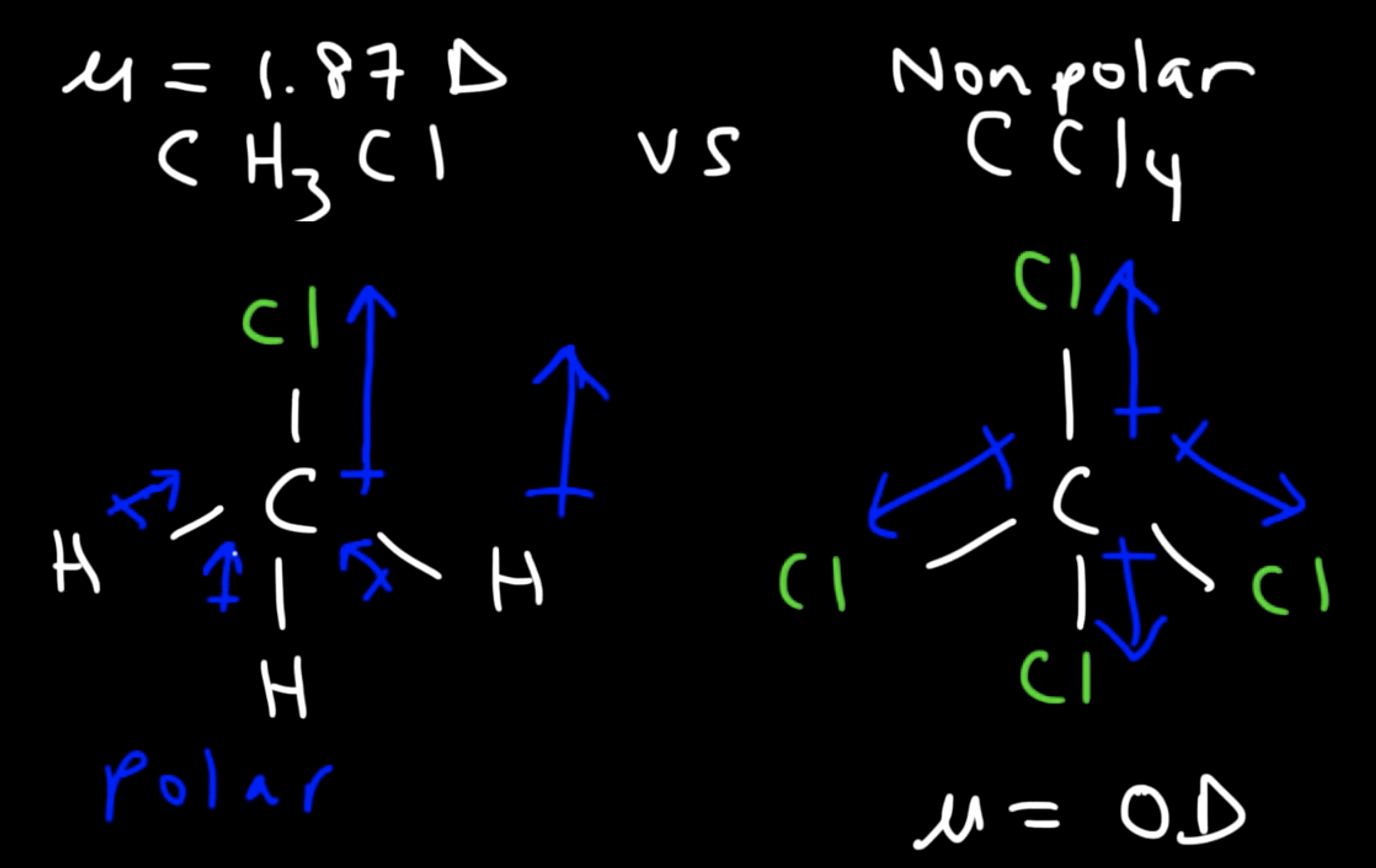
What is described by a dipole moment? What is the equation to calculate a dipole moment?
A dipole moment is a vector quantity that describes the polarity of a covalent bond.
p = qd
p = dipole moment
q = net charge
d = distance between the partial charges
When considering the polarity of an entire covalent molecule, how can you correctly use information about the dipole moment found in the molecule?
Find the NET charge and add up the dipole moments (magnitude AND direction). Spatially account for the dipole moment between each covalent bond in the molecule.
If there is a significantly greater electronegativity difference between two bonded atoms in the molecule compared to another, but this difference and big dipole moment is canceled out by another existing dipole moment in the same molecule going the opposite direction, then the net polarity is zero.
Ex shown in the image. The dipole moment between C and Cl is greater than between C and H, and there are more of these C-Cl bonds in the carbon tetrachloride, but they are all canceled out with each other, making the molecule overall nonpolar. Meanwhile the