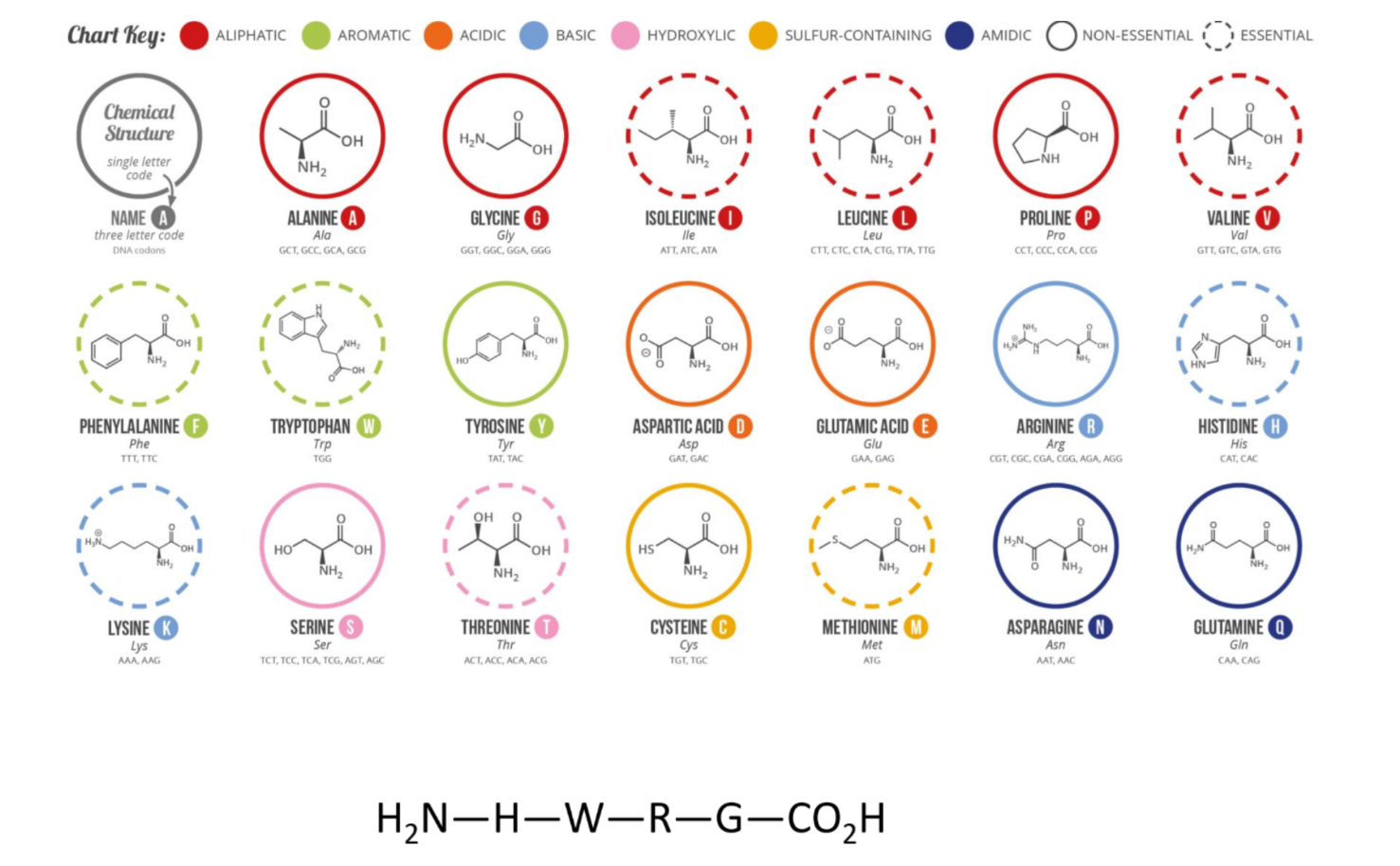
4 biomolecules - amino acids and proteins
1/31
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
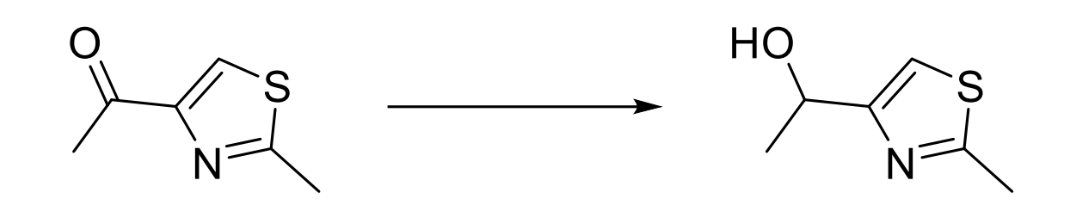
reagents
NaBH4
give 3 aspects of a cell’s life cycle that proteins play a role in
signalling, proliferation and repair
what are peptides
chains of multiple amino acids
when do peptides become proteins
a molecular weight of > 10000
10,000 Da = 10 kDa
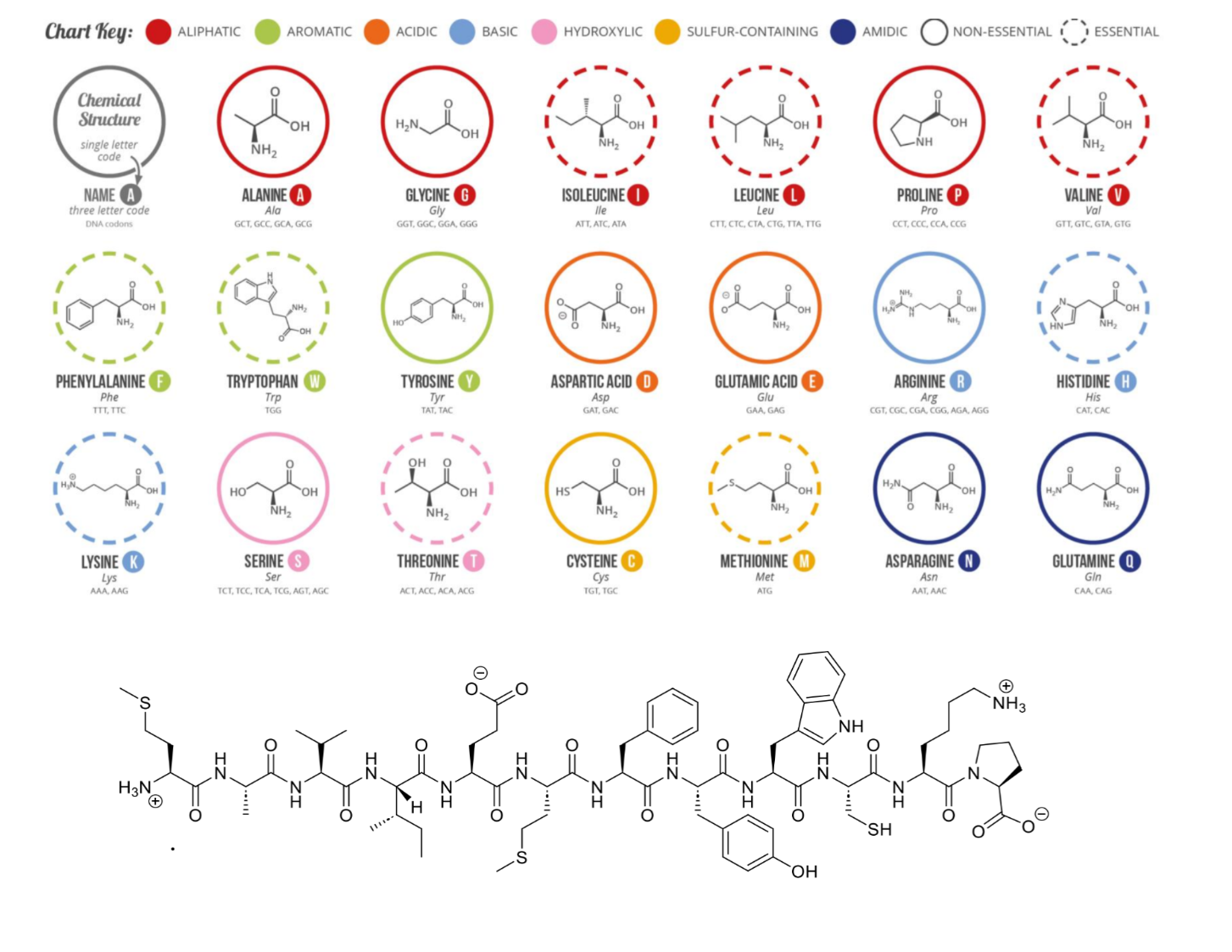
describe this peptide with 3-letter and 1-letter abbreviations
include the ends of the peptide
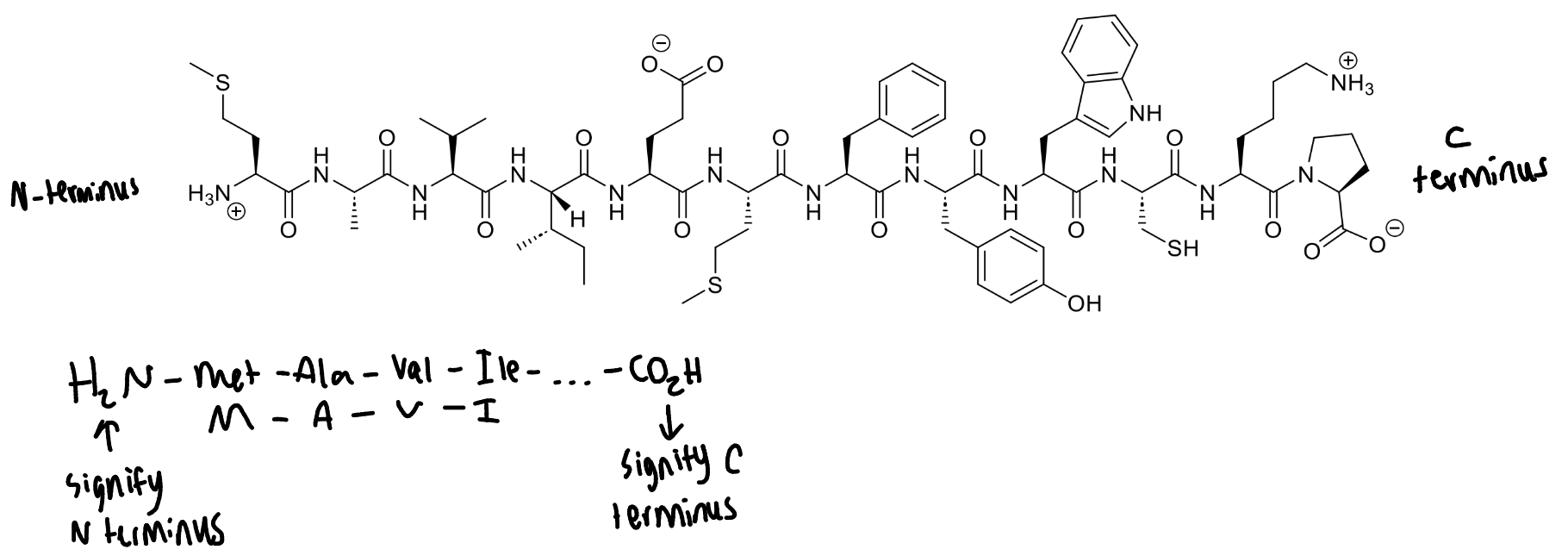
based on the charges on the peptide, what is the pH of the solution?
Glu will be deprotonated - pKa = 4.5
Amines are protonated - pKa 9-10
If Glu fully deprotonated, pH must be at least one log unit higher = > 5.5
so pH 6-8
which direction is it common to write peptide sequences in
N-terminus to C-terminus
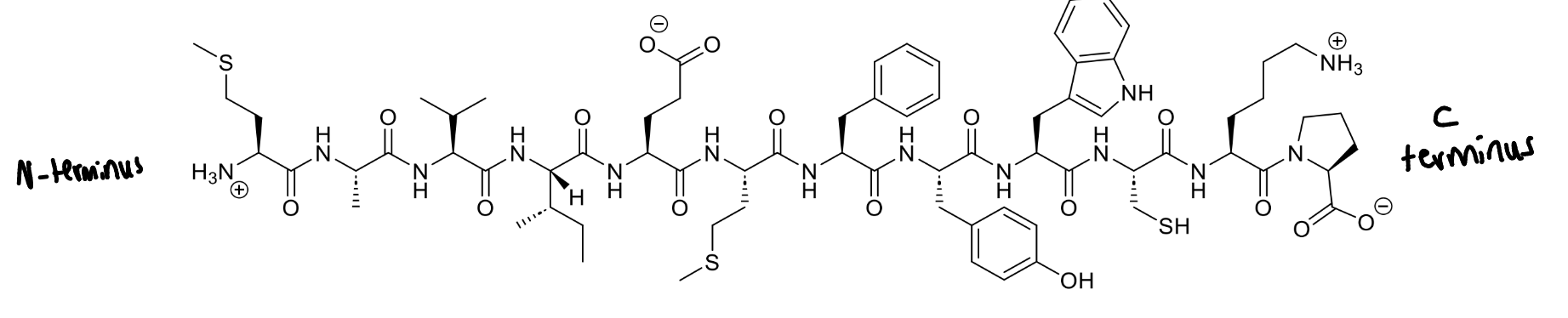
steps to draw the full structure of a peptide
draw backbone first with correct number of amines
include correct stereochemistry for the branches, with wedges pointing up and dashed pointing down
add side chains
add charges
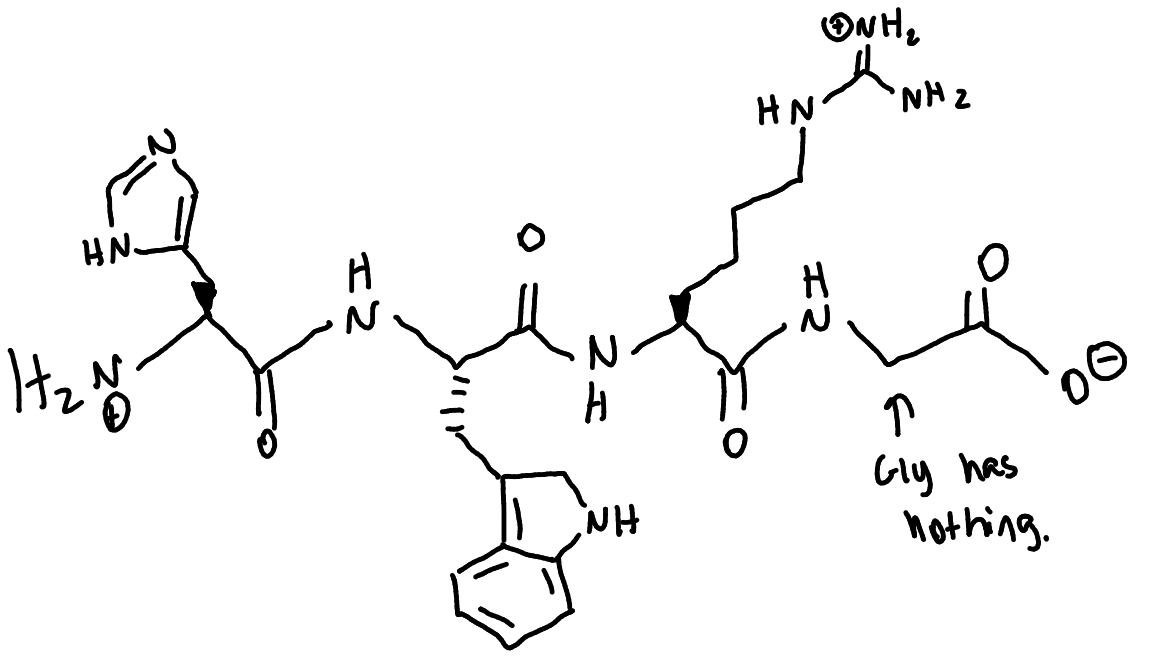
draw the full structure of this
what is the primary structure of a protein
the sequence of amino acids
what is the secondary structure of a protein
the 3D shape made by a chain of amino acids
what is the key criteria for the secondary structure
what does this lead to
every carbonyl group and amide proton in the peptide chain should have a hydrogen-bonding partner
leads to 2 main types of secondary structure - α-helix and β-sheet
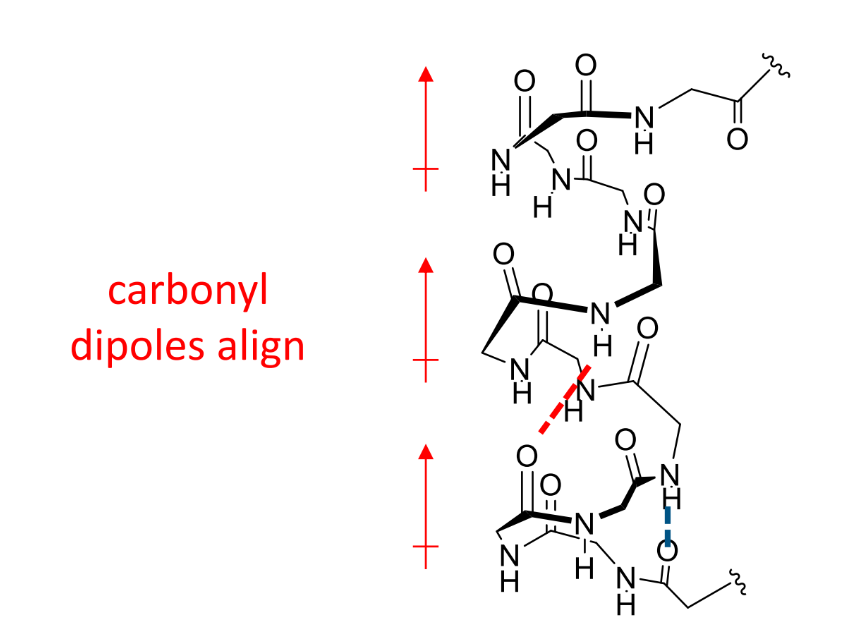
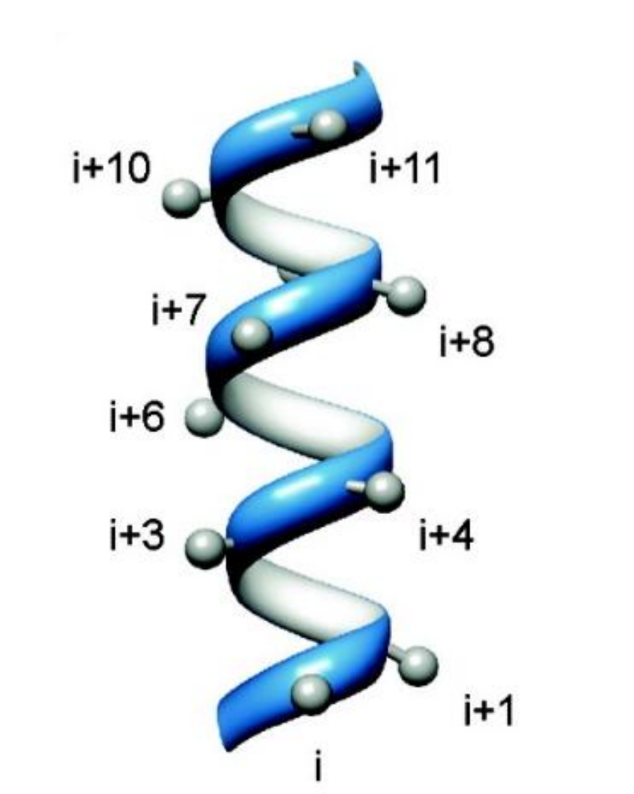
what is an α-helix
a coil of amino acids with hydrogen bonds between every third or fourth residue
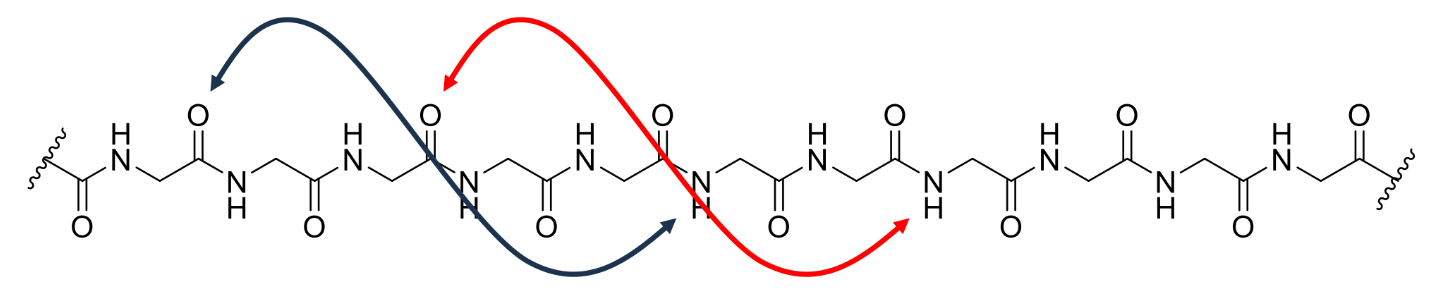
where do the side chains of an α-helix point
point outwards into space
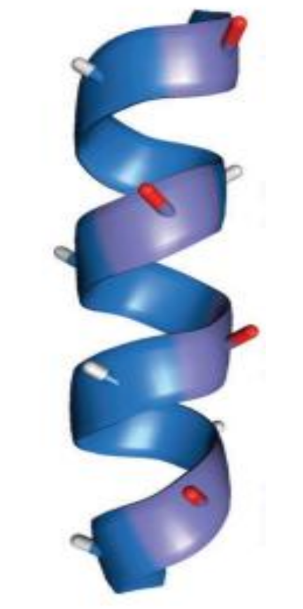
carbonyl dipoles in an α-helix?
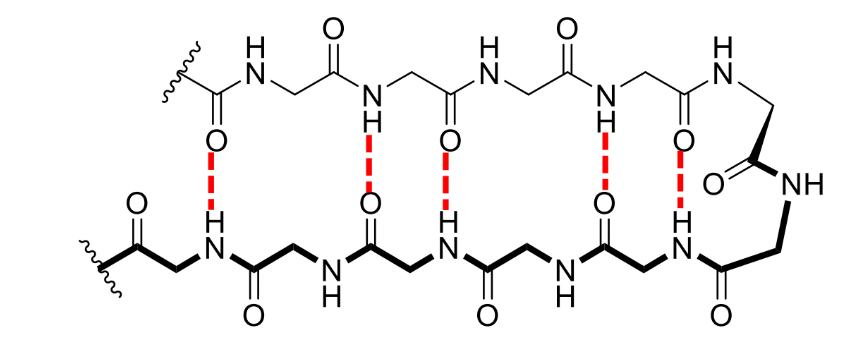
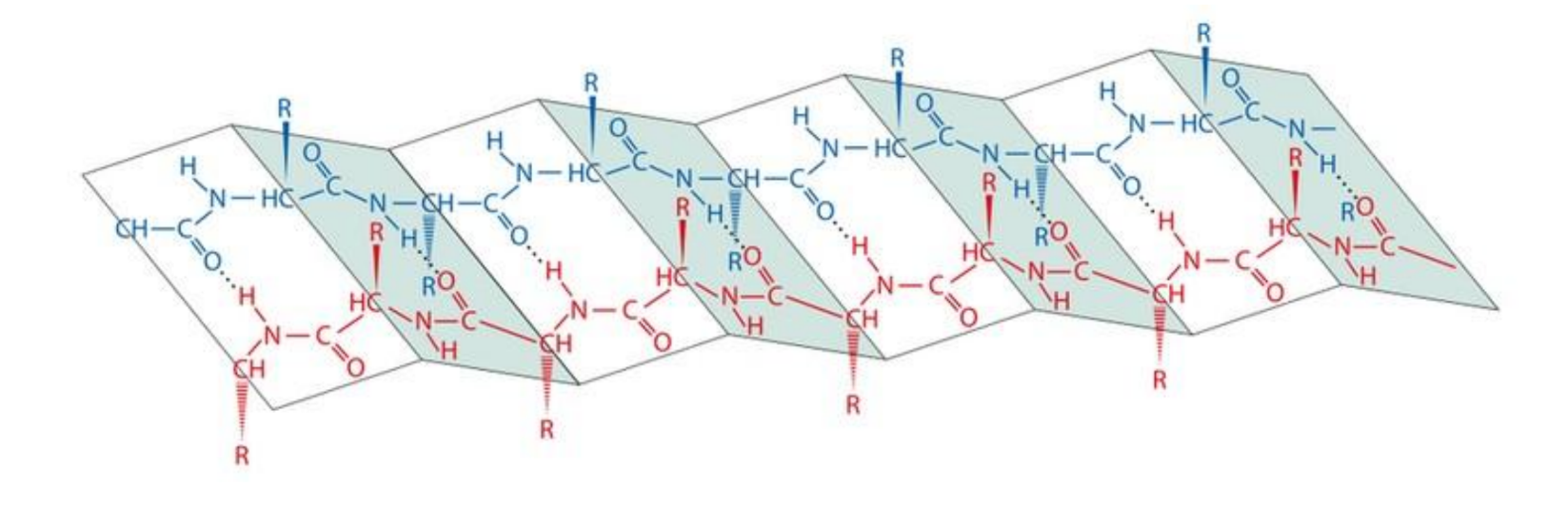
what is a β-sheet
a sequential, linear sheet of amino acid chains forming a generally flat surface
where does hydrogen bonding occur in a β-sheet
in between the individual β-strands
2 alternative names for the turning point of a β-sheet
β-turn or β-hairpin
what does the turning region of a β-sheet usually include#
example?
amino acids with unique conformational properties, e.g Gly or Pro
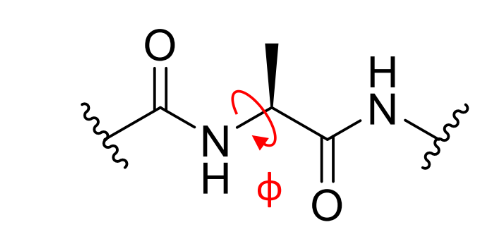
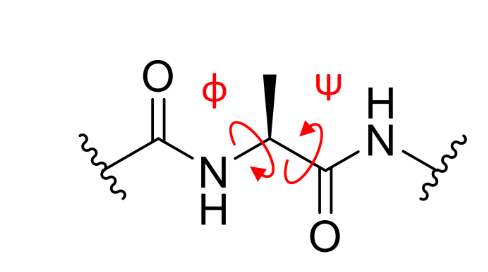
what is ϕ on a peptide linkage?
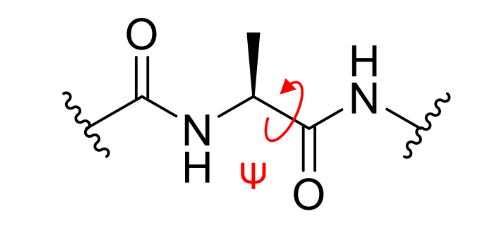
what is ψ on a peptide linkage?
what is ω on a peptide linkage?
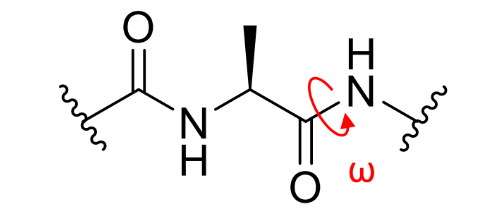
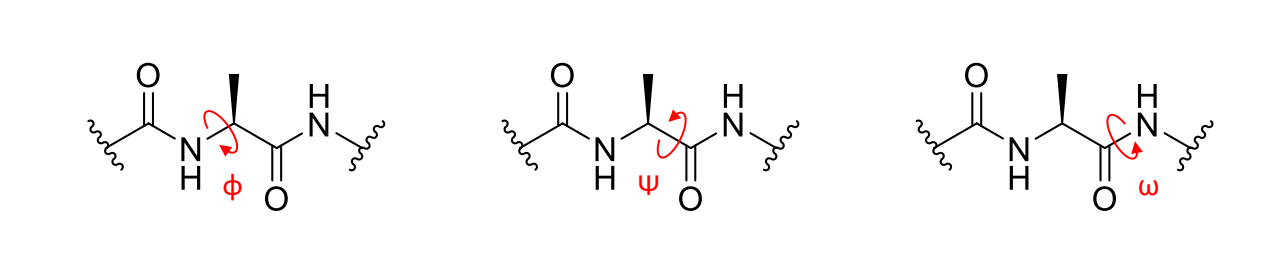
what are/can be the values of ϕ,ψ,ω?
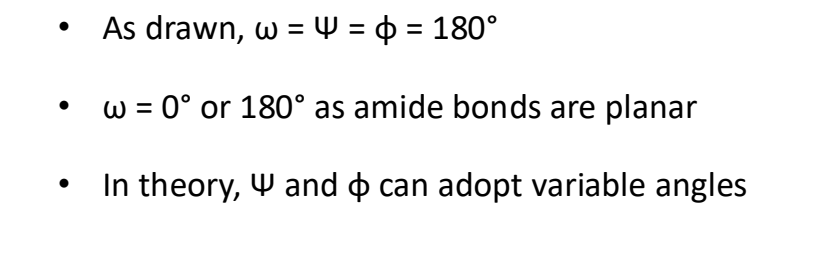
what does plotting these angles give
a Ramachandran plot
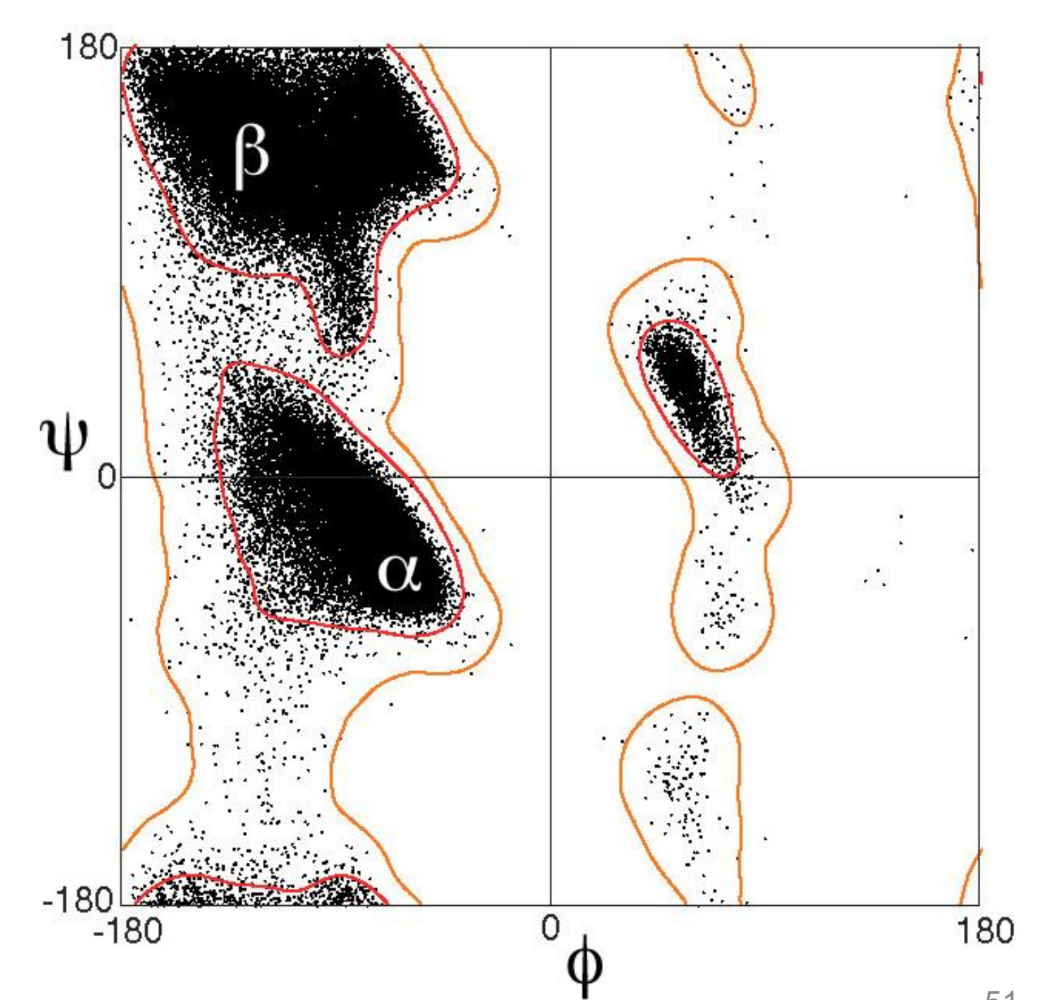
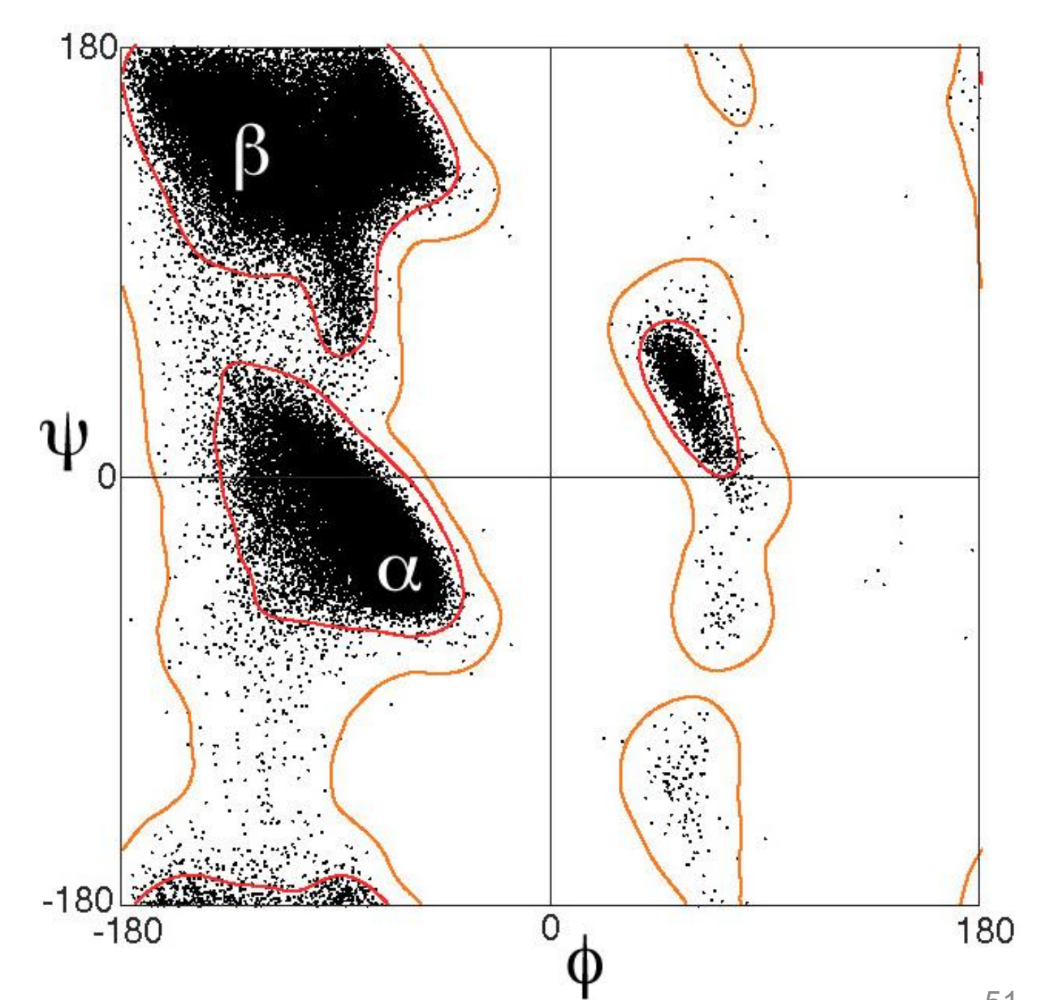
what is demonstrated by a Ramachandran plot
bonds in peptides and proteins do not rotate freely and adopt very specific conformations
what does the Ramachandran plot lead to for β-sheets?
what is the consequence for the properties of the molecule?
the amino acid side chains are positioned on opposite faces of the sheet, ie every other amino acid side chain is pointing up
this gives two faces of the molecule with different properties
what is the sequence of amino acids on the same face for an α-helix?
what does this allow?
i, i+4, i+7
means you can code for a chain with very specific properties on one side
what is the tertiary structure of a protein and how is it formed
the full 3D shape of the protein, formed from the properties of the secondary structure combining
how do proteins tend to fold
such that the hydrophobic residues are all buried into the core of the structure and hydrophilic residues point out into the solvent
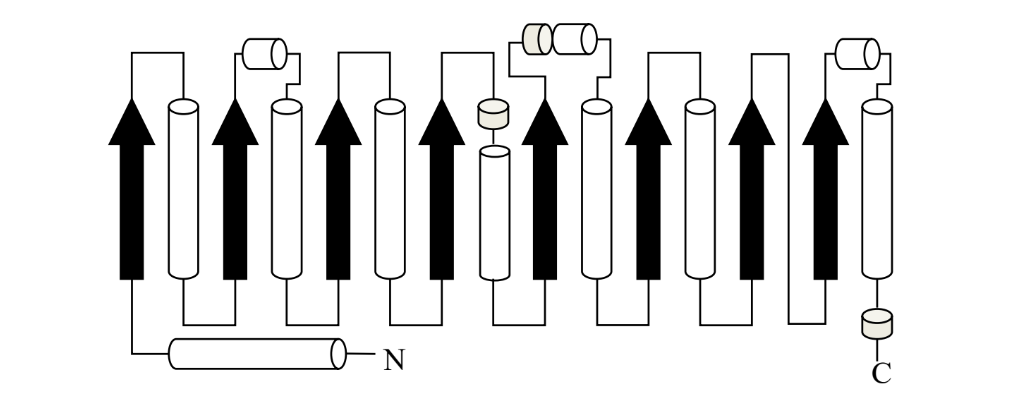
what is this protein fold called?
TIM barrel
what is the quaternary structure of a protein?
many proteins assemble into higher order structures
each colour = individual protein - all pack together
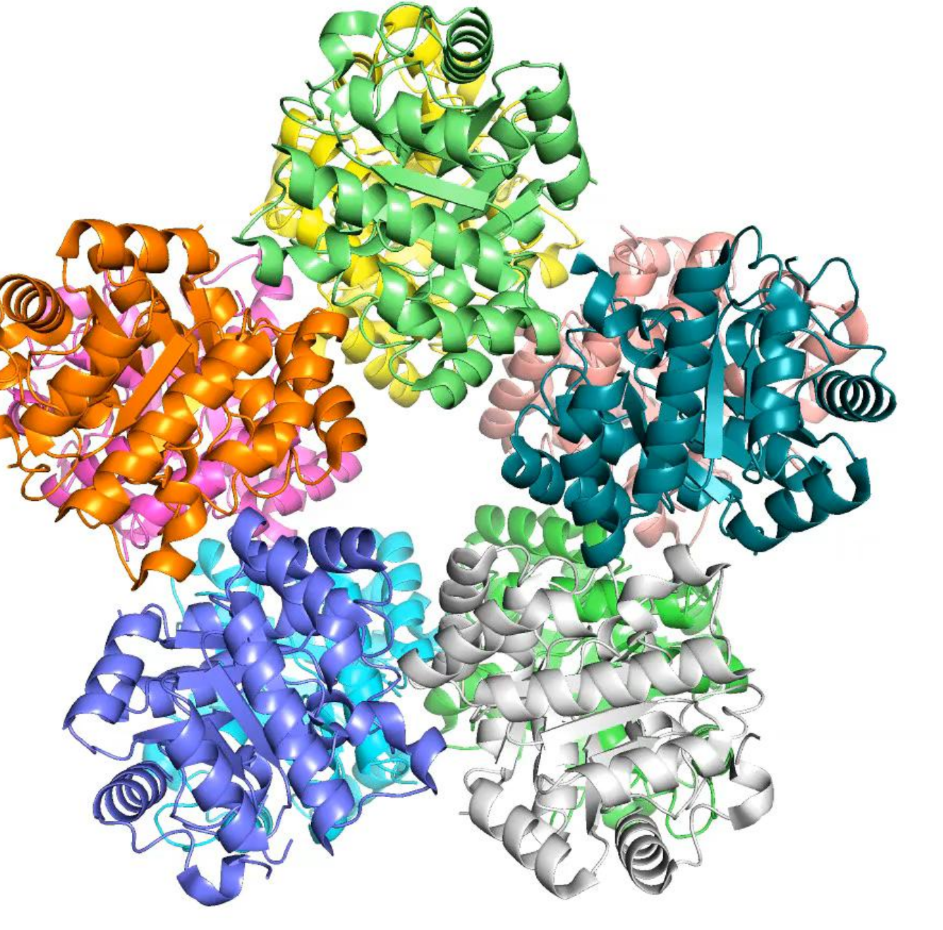
give 4 examples of higher-order quaternary structures that can be formed
dimers
trimers
tetramers
pentamers