Unit 6: Chemistry (Part 2)
1/65
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
66 Terms
Bonding
electrons are exchanged or shared to fill outer energy levels
What are the types of bonds (Hint 4 types)
1. ionic bonds
2. covalent bonds
3. hydrogen bonds
4. Van Der Waals forces
Which bonds are associated with bonding between elements (Hint 2 types)
1. ionic bonds
2. covalent bonds
Which bonds are associated with bonding between molecules (Hint 2 types)
1. hydrogen bonds
2. Van Der Waals forces
What is the difference between ionic + covalent bonding
ionic --> electrons transferred
covalent --> electrons shared
Ionic Bonding
bonding where elements transfer their electrons
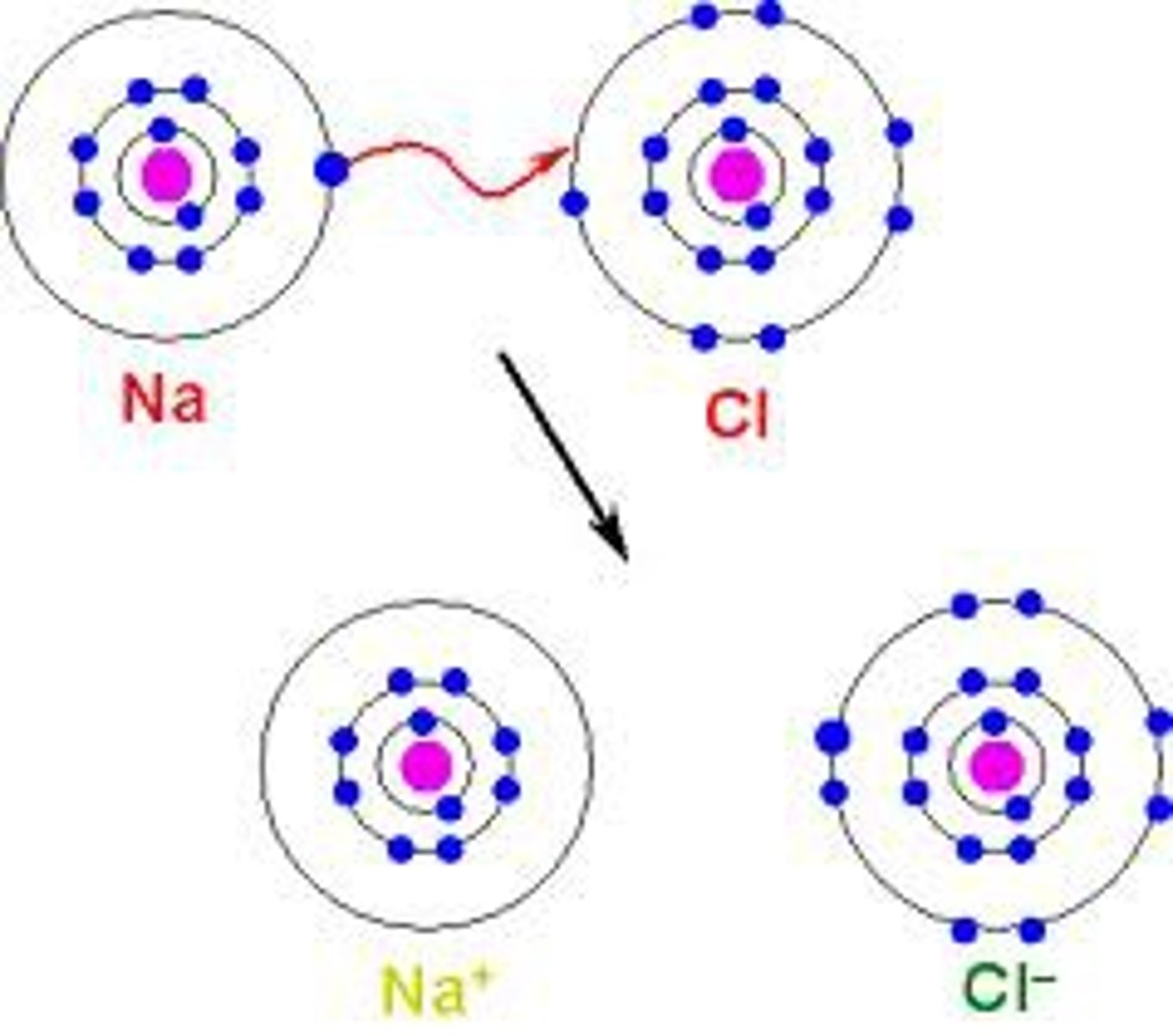
Ions
form when atoms gain or lose electrons (charged electrons)
What are the 2 types of ions
1. cation --> positive
2. anion --> negative
Cation
loses electrons = positive ion
Anion
gains electron = negative ion
Which types of elements usually bond through ionic bonding (Hint 2 pairs)
1. metals + nonmetals
2. metaloids + nonmetals
Covalent Bonding
bonding where elements share electrons
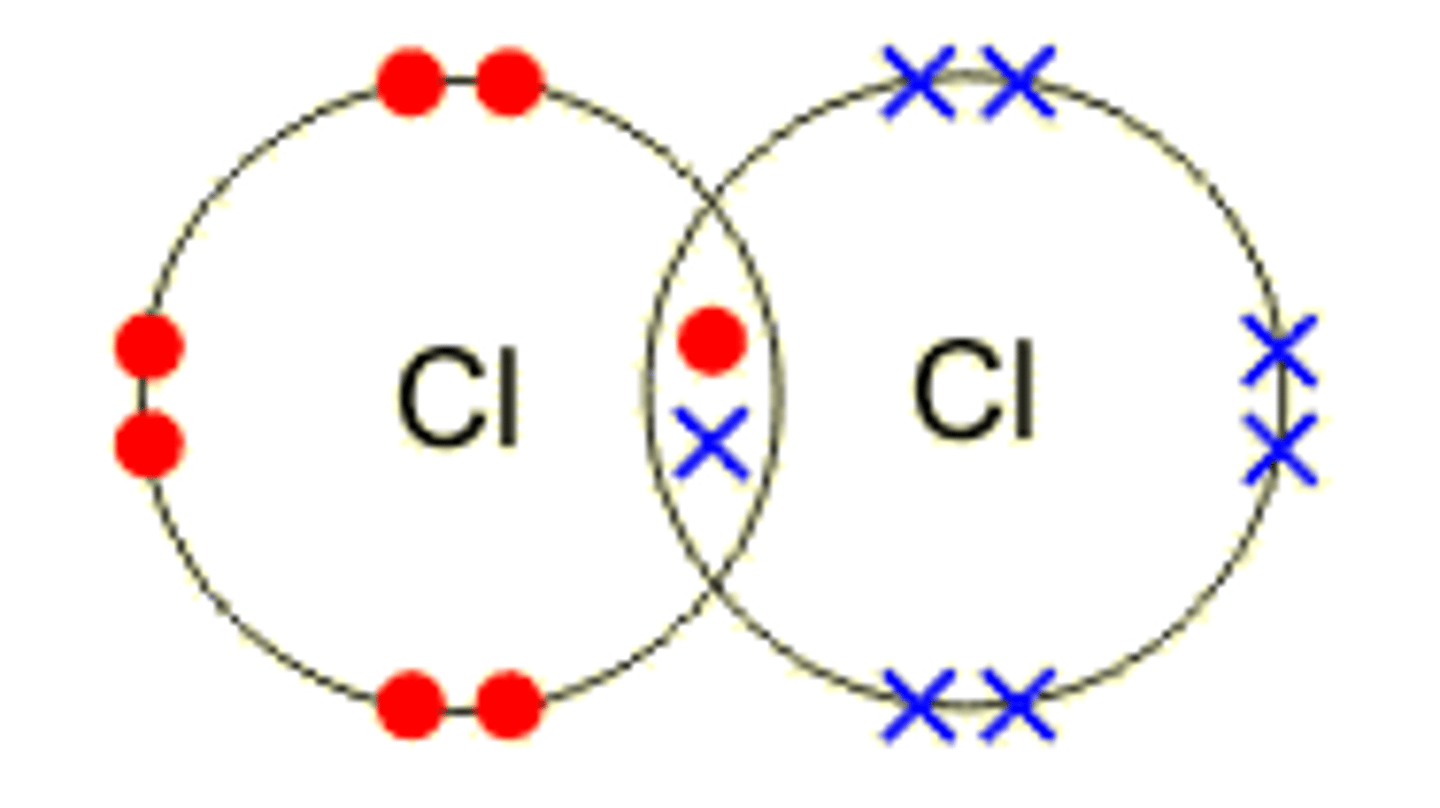
Which types of elements usually bond through covalent bonding (Hint 1 pair)
1. nonmetals + nonmetals
What are the 2 types of covalent bonds
1. polar covalent
2. nonpolar covalent
Polar Covalent Bond
unequal sharing of electrons
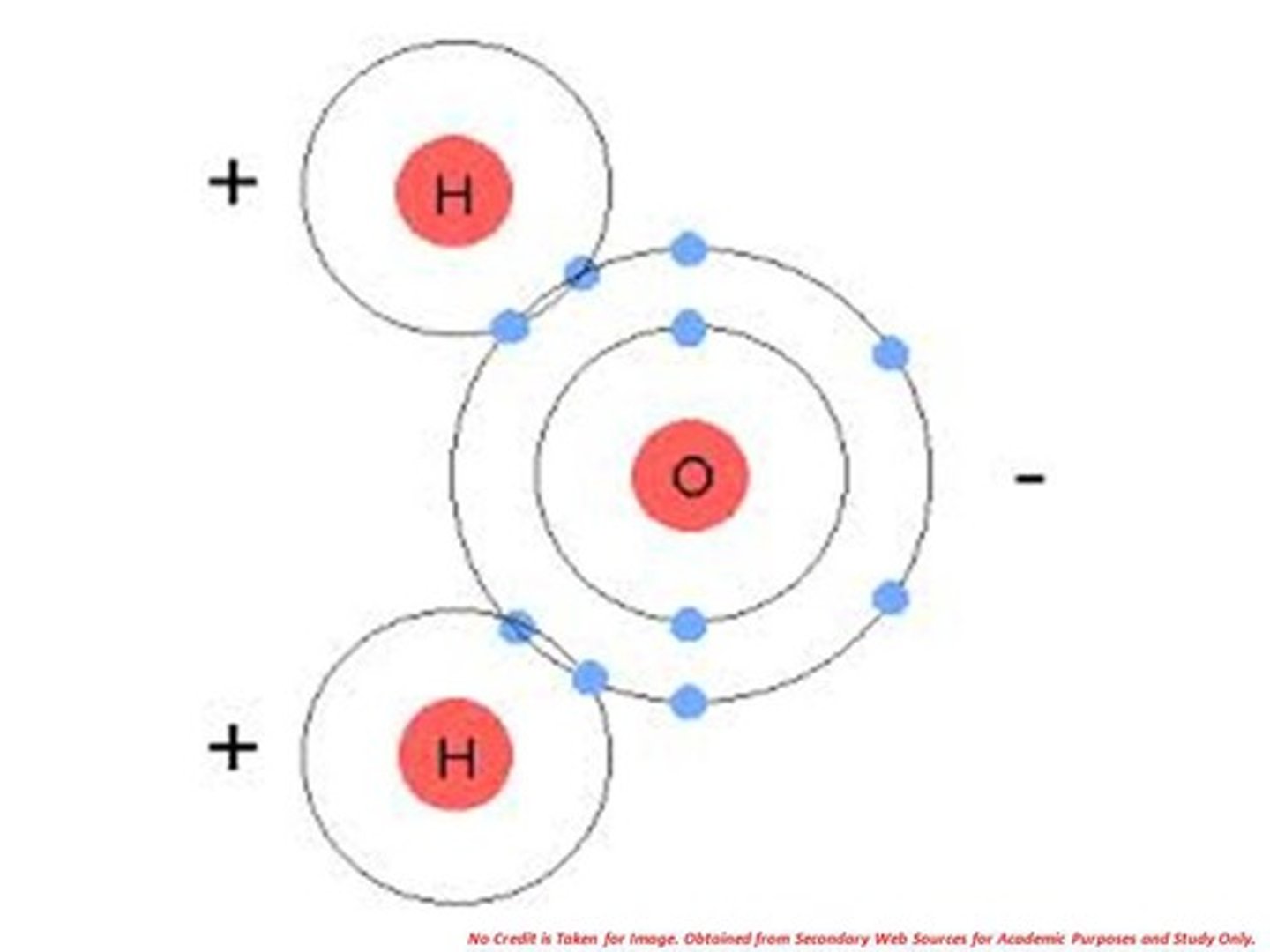
True or False: Water is an example of polar covalent
True --> bc has a slightly negative charge on oxygen + slightly positive charge on hydrogens
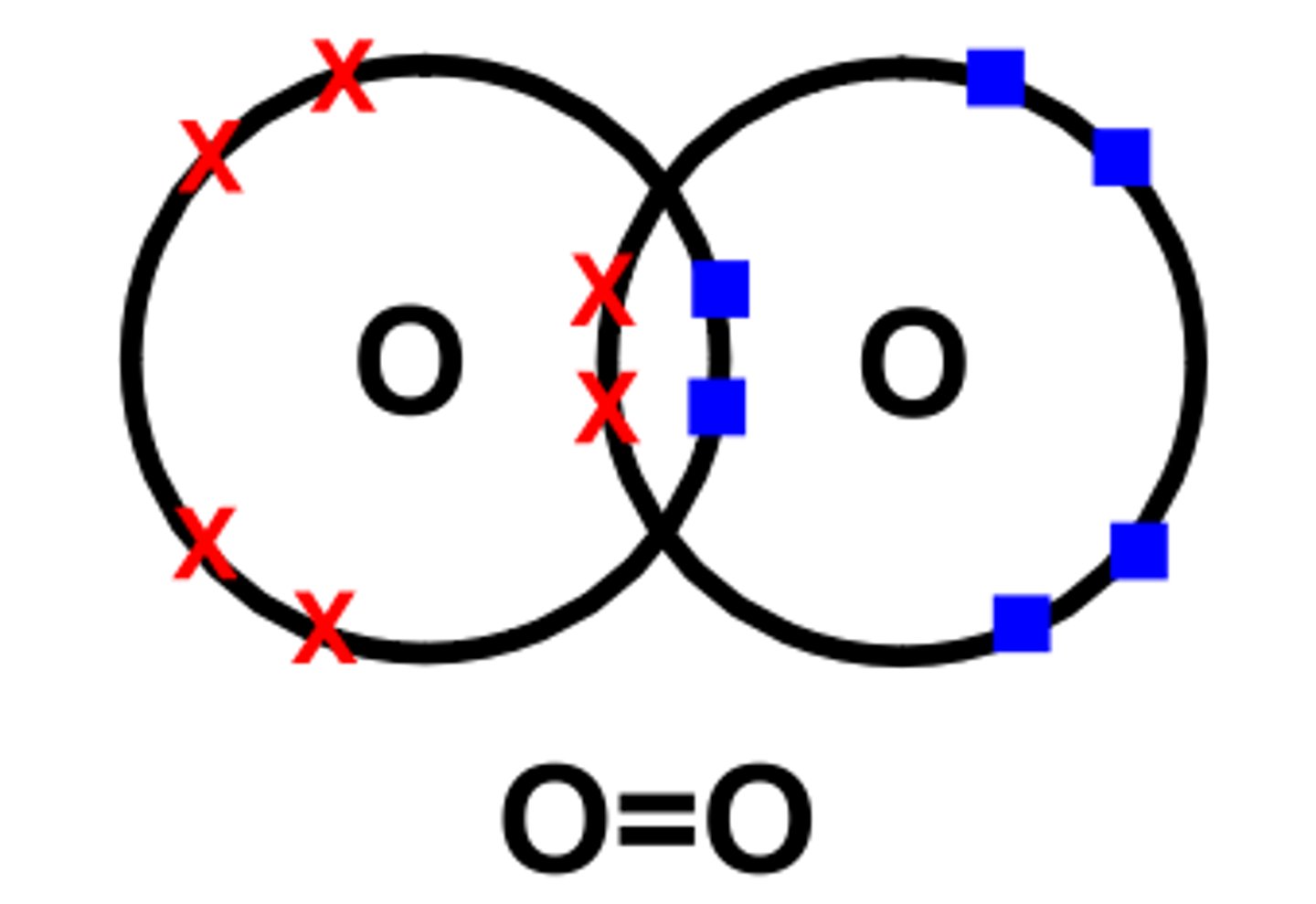
Nonpolar Covalent Bond
equal sharing of electrons
True or False: Oxygen is an example of nonpolar charge
True --> oxygen is shared evenly
Van Der Waals forces
intermolecular attractions between 1 molecule + another molecule
Hydrogen Bonding
bonding between 2 water molecules
Explain the structure and bonding of a water molecule (Hint 2 things)
1. between 1 oxygen + 2 hydrogens
2. bonding = polar covalent
Electronegativity
measure of tendency of atom to attract shared pair of electrons in chemical bond
Which way does electronegativity travel
lower-left --> upper-right
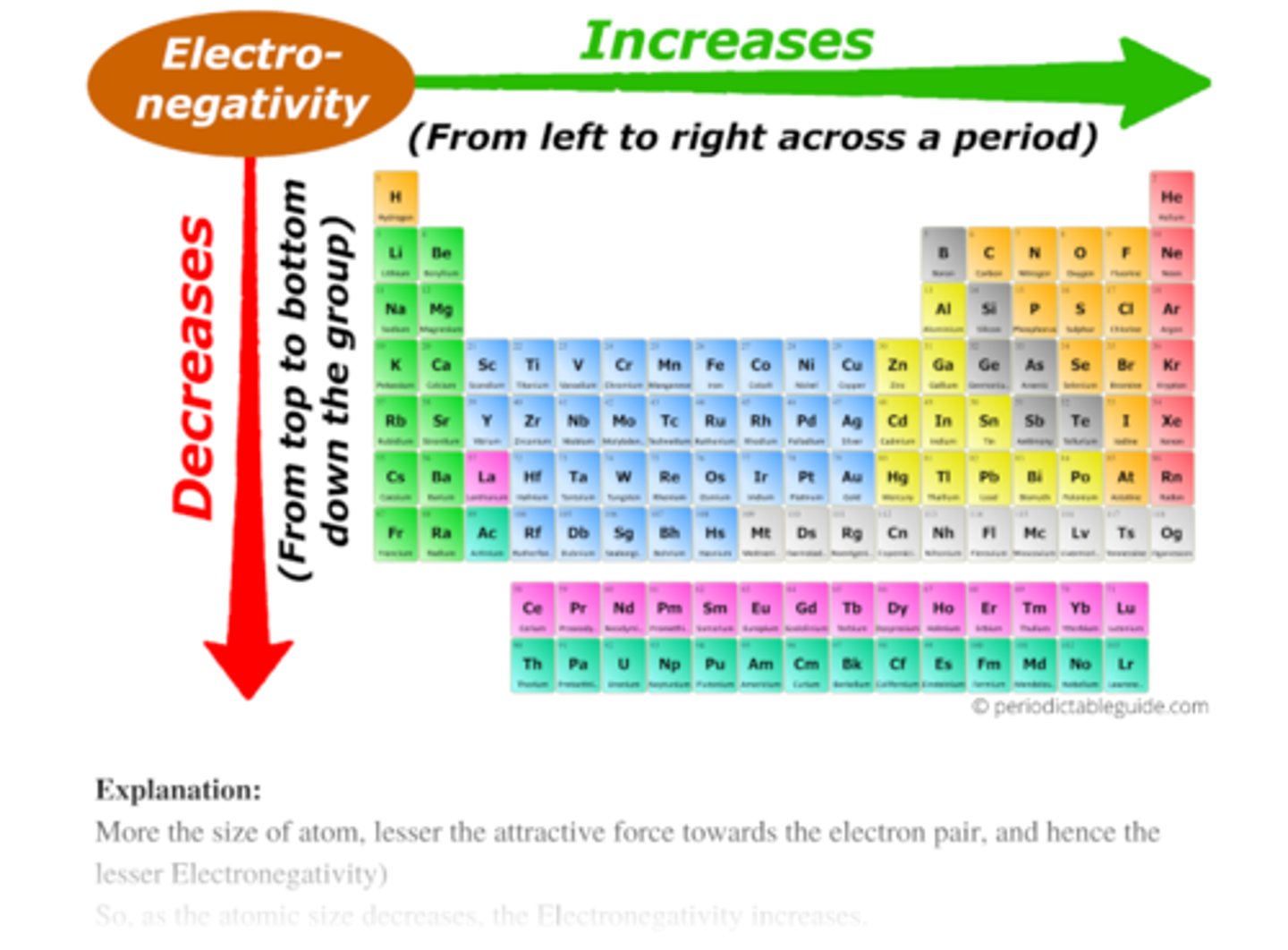
Explain why water is polar covalent (Hint 2 main things)
1. hydrogens = more postivie bc have less electronegativity (don't hold on to electrons as long --> more positive)
2. oxygen = more negative bc has more electronegativity (holds on to electrons longer --> more negative)
What type of bonding does water have with other molecules
hydrogen bond
Properties of water (Hint 6 things)
1. universal solvent
2. specific heat capacity
3. adhesion
4. cohesion
5. surface tension
6. capillary action
Universal Solvent
Water's ability to dissolve more substances than any other liquid
Why is water a universal solvent
1. water = polar + has charge
2. able to dissolve like bases (dissolve other polar/charged substances)
Explain "like" dissolves "like"
polar dissolves polar
nonpolar dissolves nonpolar
(Like in math when you combine lile terms)
Is water able to dissolve sugar, explain (Hint 1 thing)
1. both substances are polar solvents
Is water able to dissolve salt (NaCl), explain (Hint 4 things)
both substances are polar solvents
1. sodium = positive + chloine = negative
2. oxygen = negative + hydrogens = positive
3. opposites attract + breaks bond between sodium + chlorine
Is water able to dissovle oil, explain (Hint 2 things)
1. water = polar + oil = nonpolar
2. not like bases --> can't combine
Specific Heat Capacity
amount of heat energy needed to raise temp of one gram of a substance by one degree Celsius
What does it mean if something has a low specific heat capacity
substance needs less energy to change its temperature (heats up faster)
What does it mean if something has a high specific heat capacity
substance needs more energy to change its temp (heats up slower)
Why does water have a high specific heat capacity
it needs more heat to break apart strong hydrogen bonds
Adhesion
attraction between molecules of different substances (water attracted to something else)
Give 2 examples of adhesion with water
1. water + paper towels
2. water + glass
Cohesion
Attraction between molecules of the same substance (water attacted to water)
Give an example of cohesion with water
1. water + water
Surface Tension
property of water that allows its surface to resist external forces
(stretchy "skin" on surface of liquid)

How does cohesion relate to surface tension
1. water molecules at surface don't have any molecules above them
2. experience greater downward pull + make surface tension
How is a paper clip able to float in water
surface tension --> cohesion makes bonds between water stronger + helps support paper clip's weight
What happens to paper clip when surface tension is disrupted
paper clip sinks --> surface tension = disrupted + strong cohesive bonds break/weakens + no longer support objects
What happens to surface tension when soap is added to water
soap = distrupts cohesive bonds --> bonds become weak + break
Capillary Action (Hint 3 things)
1. water sticks to narrow tube
2. sticks to itself
3. moves upward
How does capillary action relate to adhesion and cohesion
Ahesion --> water sticks to sides of glass
Cohesion --> water sticks to itself
(then can slowly move upwards)
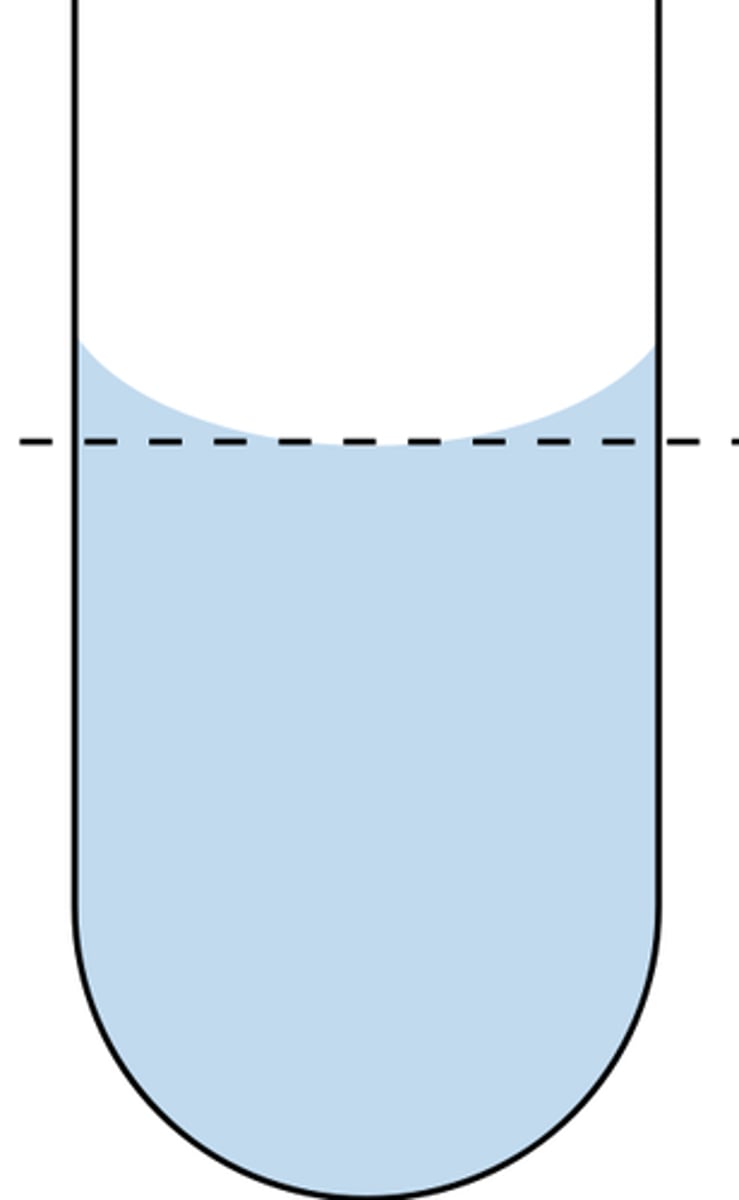
Meniscus
curved surface of water you see in container
How does structure of water influence its functions
1. universal solvent = allows to dissolve like vs like substances
2. hydrogen bond = strong bonds --> high specific heat capacity + surface tension + capillary action
pH
degree of acidity in solution
True or False: pH is a ratio between hydrogens (H+) + hydoroxides (OH-)
True --> ratio determines whether substance is acidic, nuetral, or basic
Describe the pH scale
ranges from 0 - 14
0 --> acidic
7 --> neutral
14 --> basic
What does it mean to be acidic
OH- less than H+ (range between 0-6)
What does it mean to be nuetral
OH - equal to H+ (range = 7)
What does it mean to be basic
OH - greater than H+ (ranger between 8-14)
Describe when pH is acidic
1. H+ > OH-
2. pH < 7
Describe when pH is neutral
1. H+ = OH- ions
2. pH = 7
Describe when pH is basic
1. H+ < OH-
2. pH > 7
Give 2 examples of acidic
1. lemon juice
2. vinegar
Give 2 examples of neutral
1. water
2. distilled water
Give 2 examples of basic
1. baking soda
2. soap
What are pH indicators (Hint 2 types)
1. pH meter
2. litmus paper
pH Meter
uses probe + measures voltage to determine pH
Litmus Paper
changes color depending on pH
How to maintain pH using buffers
cancel out any other reactivity to make it around regular pH level
What is an example of maintaining pH levels using buffers
sugar on apple --> doesn't make it brown
1. stops enzymes (blocks H+) from working + combining w/ oxygen (keeps it from browning)