Elementary Chemistry Exam 3
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
amides
carbonyl group (C=O) attached to a nitrogen atom (N attached to H or alkyl)
primary amide
nitrogen attached to one carbon
secondary amide
nitrogen attached to 2 carbons
tertiary amide
nitrogen attached to 3 carbons
lactones
cyclic esters
lactams
cyclic amides
nomenclature for carboxylic acids
carboxylic acids have highest priority over all other functional groups
use suffix -oic acid
carboxylic acids don’t need a number; always at C1
condensed form: COOH
ex: 4,5-dimethylhexanoic acid
ex: benzoic acid
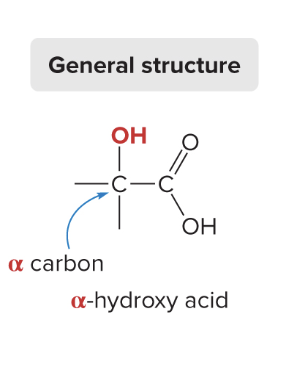
greek letters used to refer to carbon positions relative to the carboxylic acid carbon (a - carbon attached to the carboxylic acid carbon; b - carbon attached to the a carbon)
ex: a-hydroxy acid
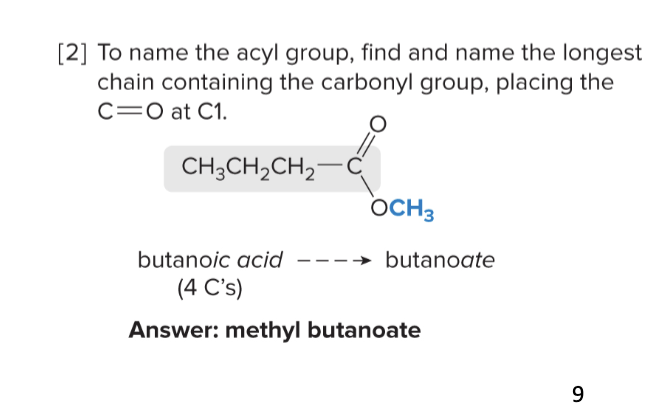
nomenclature for esters
use suffix -oate
1) name alkyl group as substituent (group directly attached to the oxygen)
2) name acyl chain as the parent chain name
ex: methyl butanoate
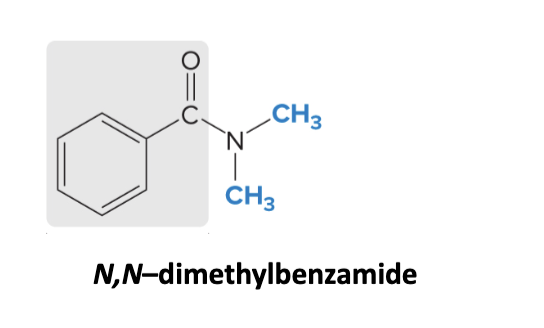
nomenclature for amides
use suffix -amide
1) name alkyl groups as substituents (the group directly attached to the nitrogen)
add N or N,N at the beginning to indicate alkyl groups on the nitrogen (treat them like numbers)
2) name the acyl chain as the parent chain name
ex: N,N-dimethylbenzamide
physical properties of carboxylic acids
have very high boiling points
double hydrogen bonding b/w 2 carboxylic acids
physical properties of esters
esters can’t hydrogen bond in pairs like carboxylic acids
physical properties of amides
primary/secondary amides can hydrogen bond
tertiary amides can’t hydrogen bond; have lower boiling points
acidity of carboxylic acids
carboxylic acids are acids
acid increases the amount of H3O+ when added to water
carboxylic acids serve as a proton (H+) donor
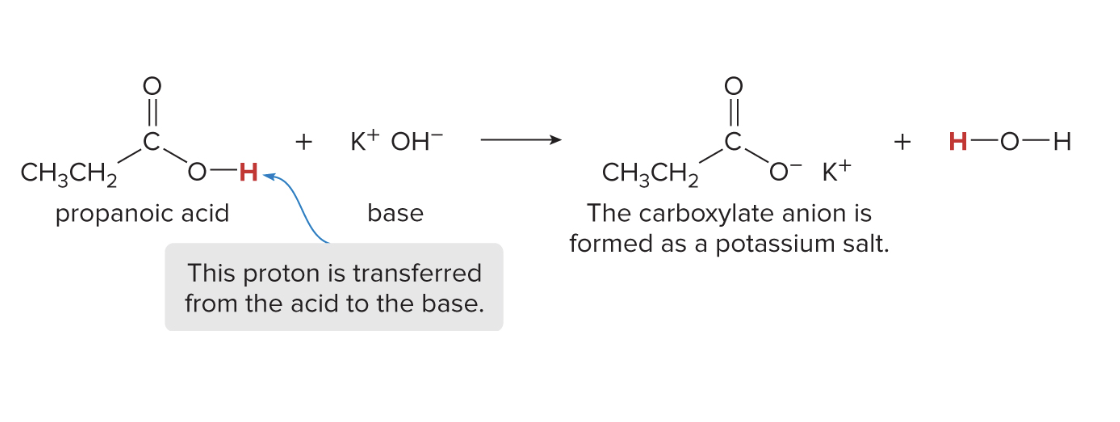
acid-base reactions w/ carboxylic acids
when carboxylic acids are deprotonated (lost their H+ proton) they form carboxylate salts
whatever the counter ion for the base was (Na+ or K+), that becomes associated w/ the carboxylate (COO-)
water also forms as a product
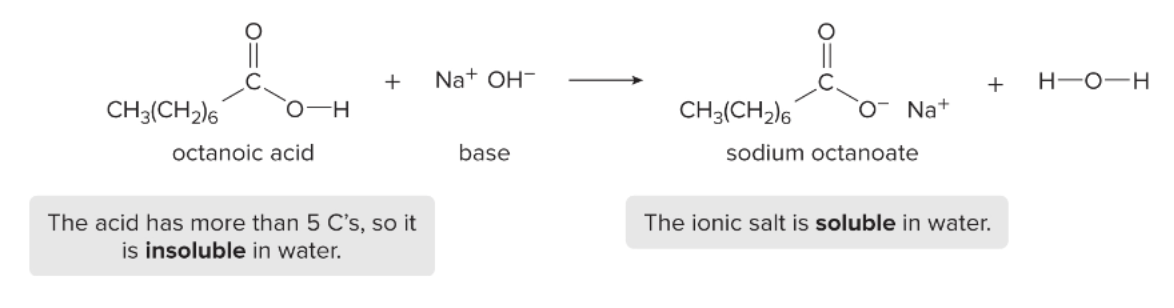
naming carboxylate salts
name of metal cation + parent + -ate (suffix)
ex: sodium octanoate
properties of carboxylic salts
much more soluble in water than the original carboxylic acid b/c they are charged
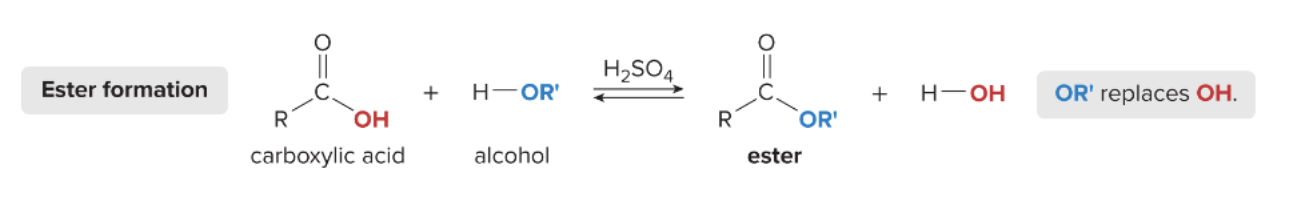
Fisher Esterification
esters formed by reacting a carboxylic acid w/ an acid catalyst + alcohol
1) carboxylic acid reacts w/ alcohol (H—OR’)
2) OR’ replaces OH of the carboxylic acid
3) ester and H2O are formed
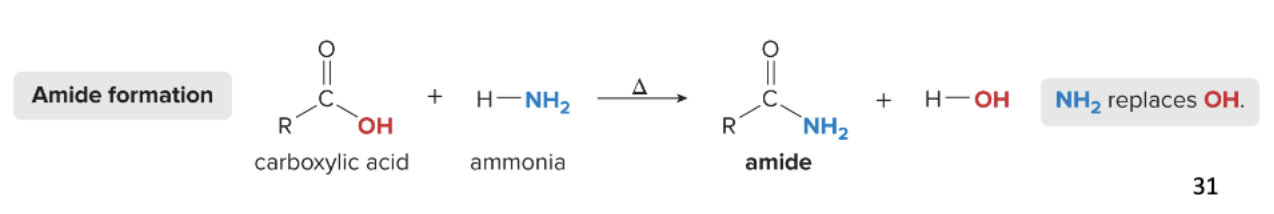
amide formation reaction
amides are formed from carboxylic acids through substitution reactions
requires heat (Δ)
1) carboxylic acid reacts w/ ammonia (H—NH2)
2) NH2 replaces the OH of the carboxylic acid
remove only one H from the nitrogen
3) amide and H2O are formed
hydrolysis
esters/amides can be converted back to carboxylic acids
uses excess water to cleave C-OR or C-NR2 bonds
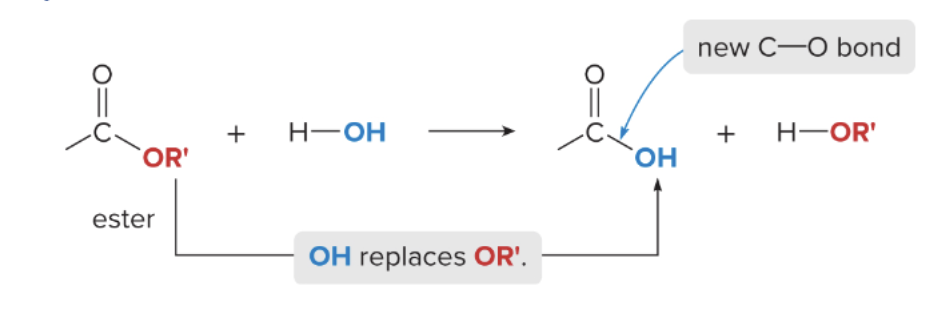
ester hydrolysis
converts ester back to carboxylic acid
water is used to cut the C-OR bond
1) ester reacts with H2O
2) OH of the water replaces the OR’ of the ester
3) carboxylic acid and alcohol is formed
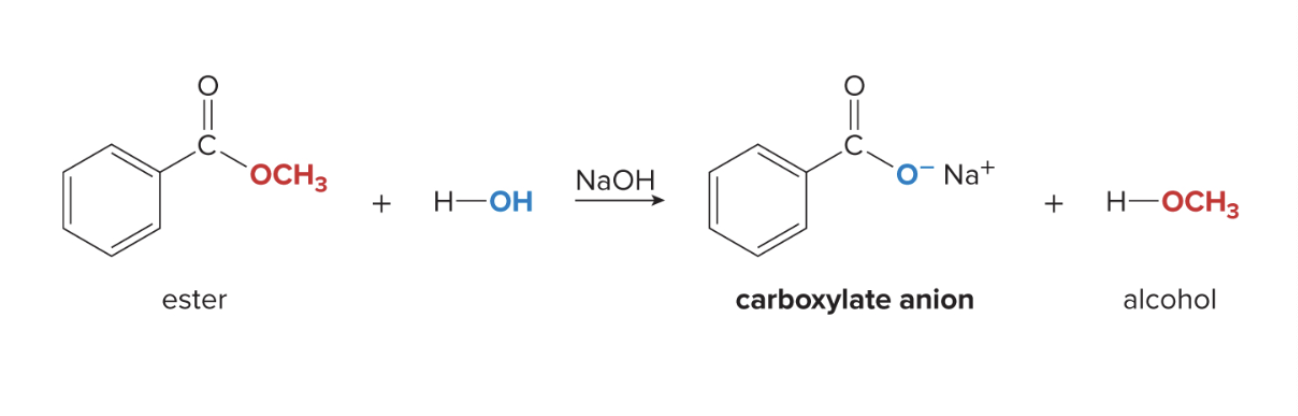
ester hydrolysis under basic conditions
ester hydrolysis can occur w/ a basic catalyst (NaOH)
a carboxylate salt is formed instead of a carboxylic acid
OR’ group of the ester become O- and Na+
carboxylate anion + alcohol is formed
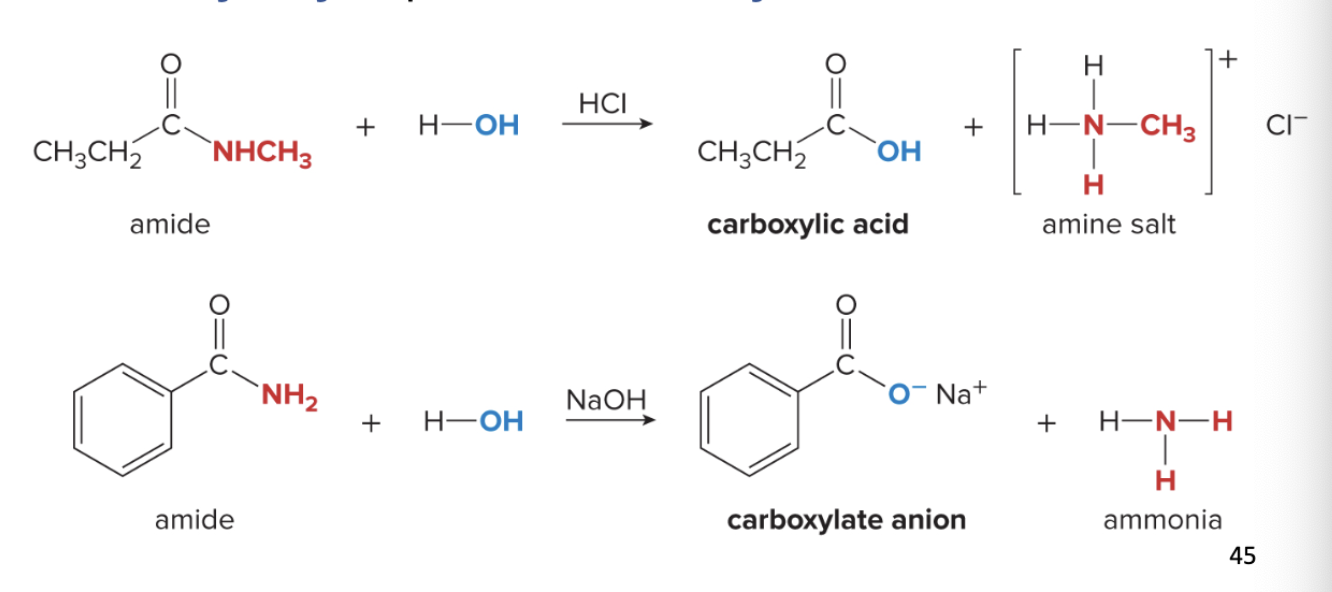
amide hydrolysis
amide hydrolysis can be done under acidic or basic conditions
1) acid hydrolysis produces carboxylic acid + amine salt
amide reacts w/ H2O; OH replaces N group
nitrogen group bonds w/ H to form amine salt
2) base hydrolysis produces carboxylate salt + amine
amide reacts w/ H2O
NH2 becomes O- and Na+
NH2 bonds w/ H to form ammonia
interesting esters and amides
esters are commonly pleasant/sweet smelling; many fruits contain esters to attract animals
melatonin contains amides
esters/amides present in many pain relievers
amines
nitrogen attached to carbon
R—NH2, R2—NH, or R3—N
primary amine
nitrogen is attached to one carbon
secondary amine
nitrogen attached to 2 carbons
tertiary amine
nitrogen attached to 3 carbons