Hydrogels and Natural Polymers
1/13
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
Hydrogels
Polymeric networks that absorb large amounts of water while remaining insouluble in aqueous hydrogel
Crosslink density (elastic Modulus)
low E = more flexibility
Higher E = Stiffer material
Hydrogel Molecular Weight
High crosslink density → Low Mc → Larger G or E → stiffer Hydrogel.
Basically Mw for Mc
Predicting diffusion through a network

Mesh size >1
Fast diffusion
Mesh size ~1
Slow diffusion
Mesh size < 1
Immobilization → Deformation/Degradation/swelling
Chemistry influences hydrophilicity
Changing the chemistry of the hydrogel can change the swelling ratio
Hydrophilic functional groups: •
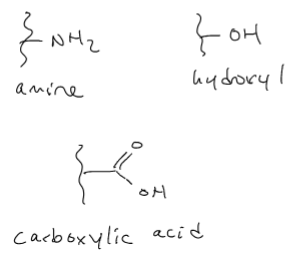
Chemistry influences hydrophilicity Alternative
Monomer sol. weight
Monomer functionality
[Monomer]
Monomer : crosslinker ratio
Swelling ratio
Q = Vswollen/ Vdry
V = volume
pH-responsive hydrogels
• Crosslinked polymer networks containing ionizable functional groups
Temperature-responsive hydrogels
Crosslinked polymer networks that undergo a phase transition at a specific temperature (i.e., lower critical solution temperature (LCST))
Natural polymers
Natural polymers (e.g., cotton, silk, natural rubber) harvested, processed and used by humans since at least several millennia BCE