3.1. Fundamentals of Chemistry
1/77
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
78 Terms
1. Which statement about electronegativity is most accurate?
A. A salt typically forms from two elements with a small difference in electronegativity.
B. Electronegativity and electron affinity are identical quantities.
C. Fluorine has the lowest electronegativity.
D. In a covalent bond, the tendency of an atom to attract the shared electron pair is described by its electronegativity.
E. Hydrogen has the highest electronegativity.
D. In a covalent bond, the tendency of an atom to attract the shared electron pair is described by its electronegativity.
2. Which statement about the periodic table of the elements (PSE) is correct?
A. Na and F are in the same period.
B. There are 10 main groups.
C. C and S are in the same main group.
D. In the transition elements, the electrons added within a period occupy an orbital of the outermost shell.
E. The transition elements are metals.
E. The transition elements are metals.
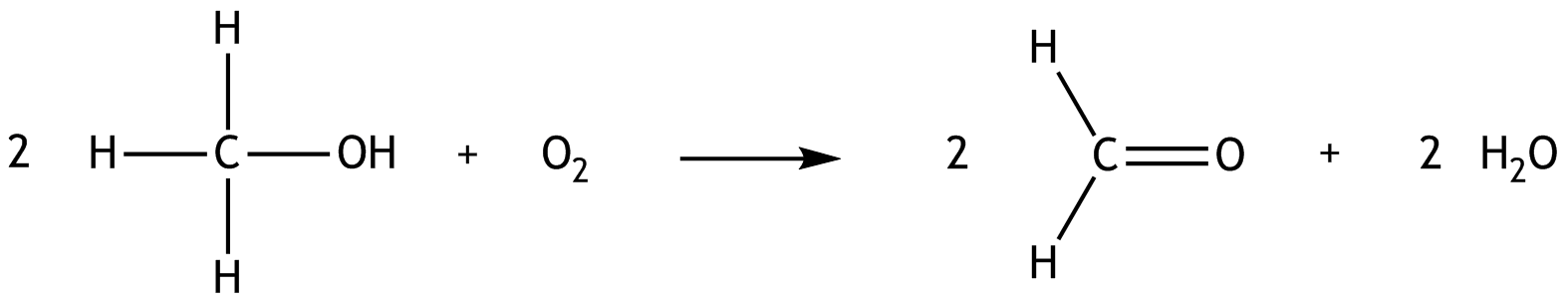
3. The oxidation of methanol can be described by the following reaction equation:
The standard enthalpies of formation ΔH° of the compounds are as follows:
Methanol: –239 kJ·mol⁻¹
Oxygen: 0 kJ·mol⁻¹
Formaldehyde: –109 kJ·mol⁻¹
Water: –242 kJ·mol⁻¹
From the resulting ΔH° of the given reaction, it follows directly that the reaction
A. is endergonic
B. is endogenous
C. is endothermic
D. is exothermic
E. is accompanied by a decrease in entropy
D. is exothermic
4. The pKₐ value of acetic acid is about 4.8, and the pK_b value of ammonia is about 4.8.
10 mL of an aqueous solution of acetic acid with a concentration of 1 mol/L are mixed with 20 mL of an aqueous solution of ammonia with a concentration of 0.5 mol/L.
Approximately what pH value does the resulting solution have?
A. 2.4
B. 4.8
C. 7.0
D. 9.2
E. 11.6
C. 7.0
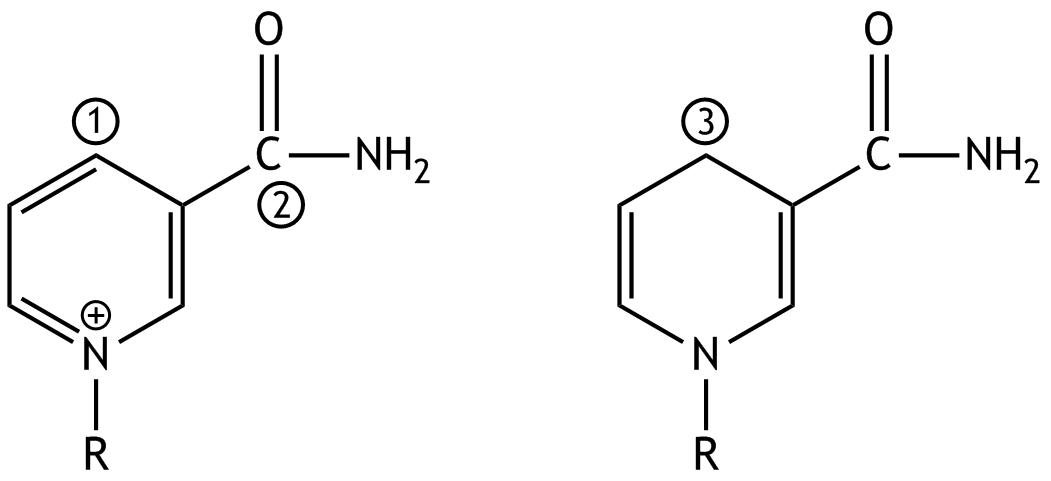
5. Which statement about the oxidation numbers (ON) of the carbon atoms ① to ③ is correct?
A. ① has the oxidation number +1.
B. ② has the oxidation number –3.
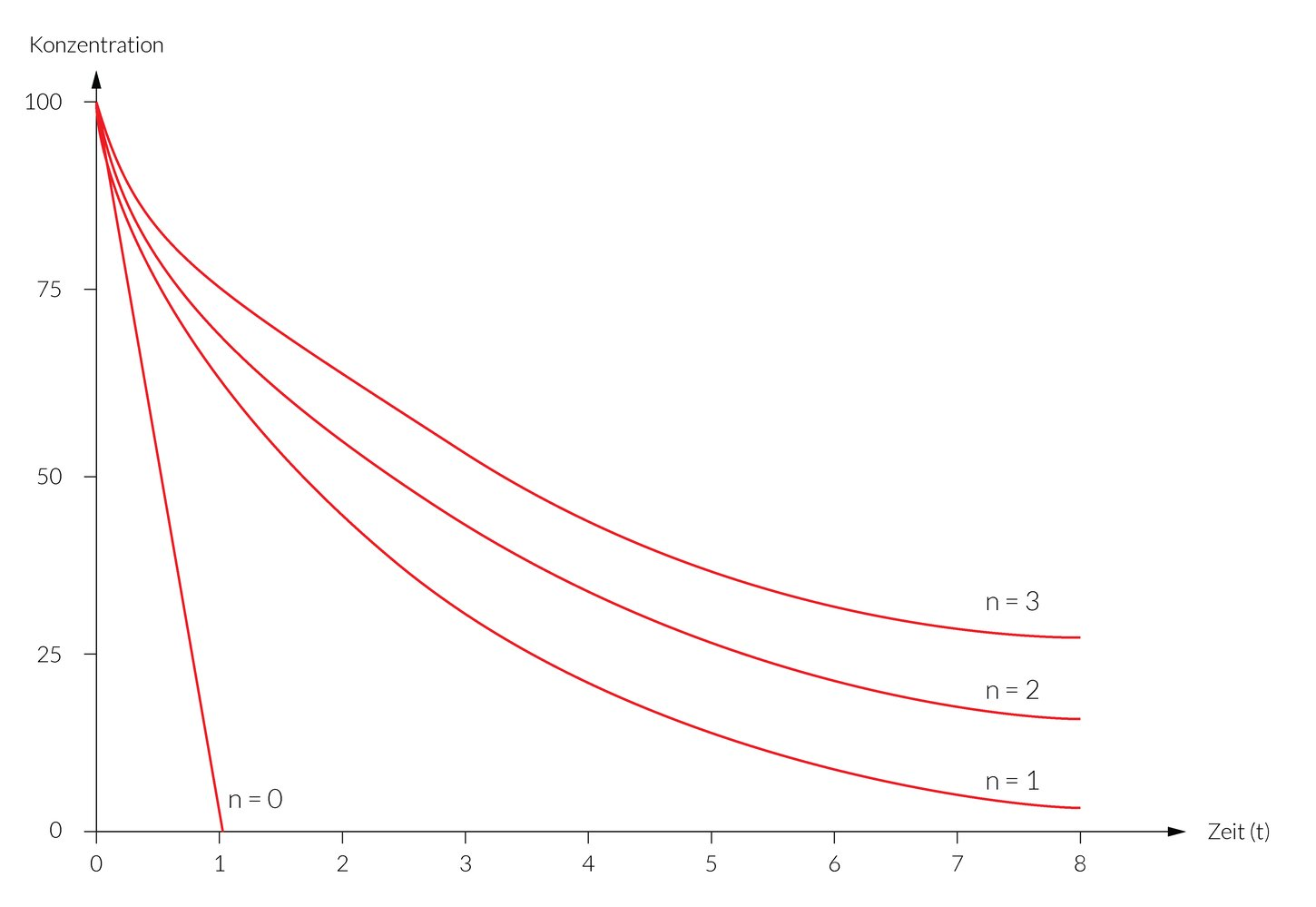
C. ③ has the oxidation number 0.
D. The oxidation number of ① is greater than that of ③.
E. The oxidation number of ③ is greater than that of ②.
D. The oxidation number of ① is greater than that of ③.
6. Which statement about alkanes is correct?
A. The liquids hexane and octane are good solvents for salts.
B. Hexadecane consists of 16 carbon atoms and 34 hydrogen atoms.
C. Hexane is a very polar liquid.
D. n-Butane has a higher boiling point than n-butanol (butan-1-ol).
E. n-Butane has a lower boiling point than isobutane (2-methylpropane).
B. Hexadecane consists of 16 carbon atoms and 34 hydrogen atoms.
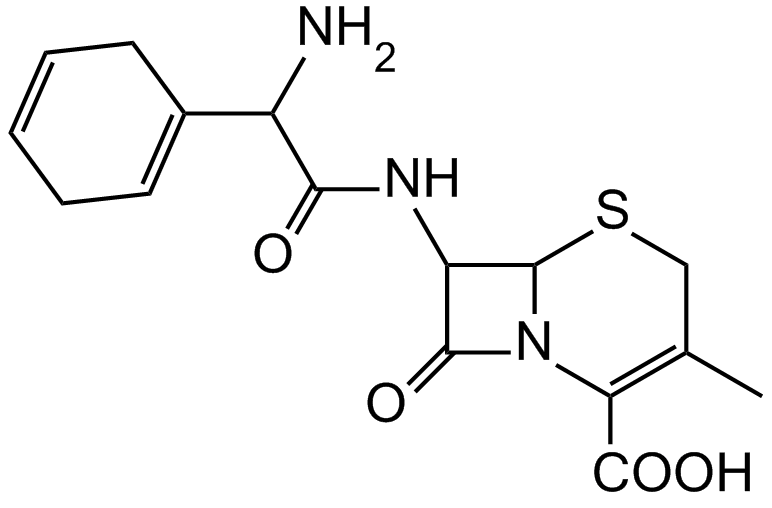
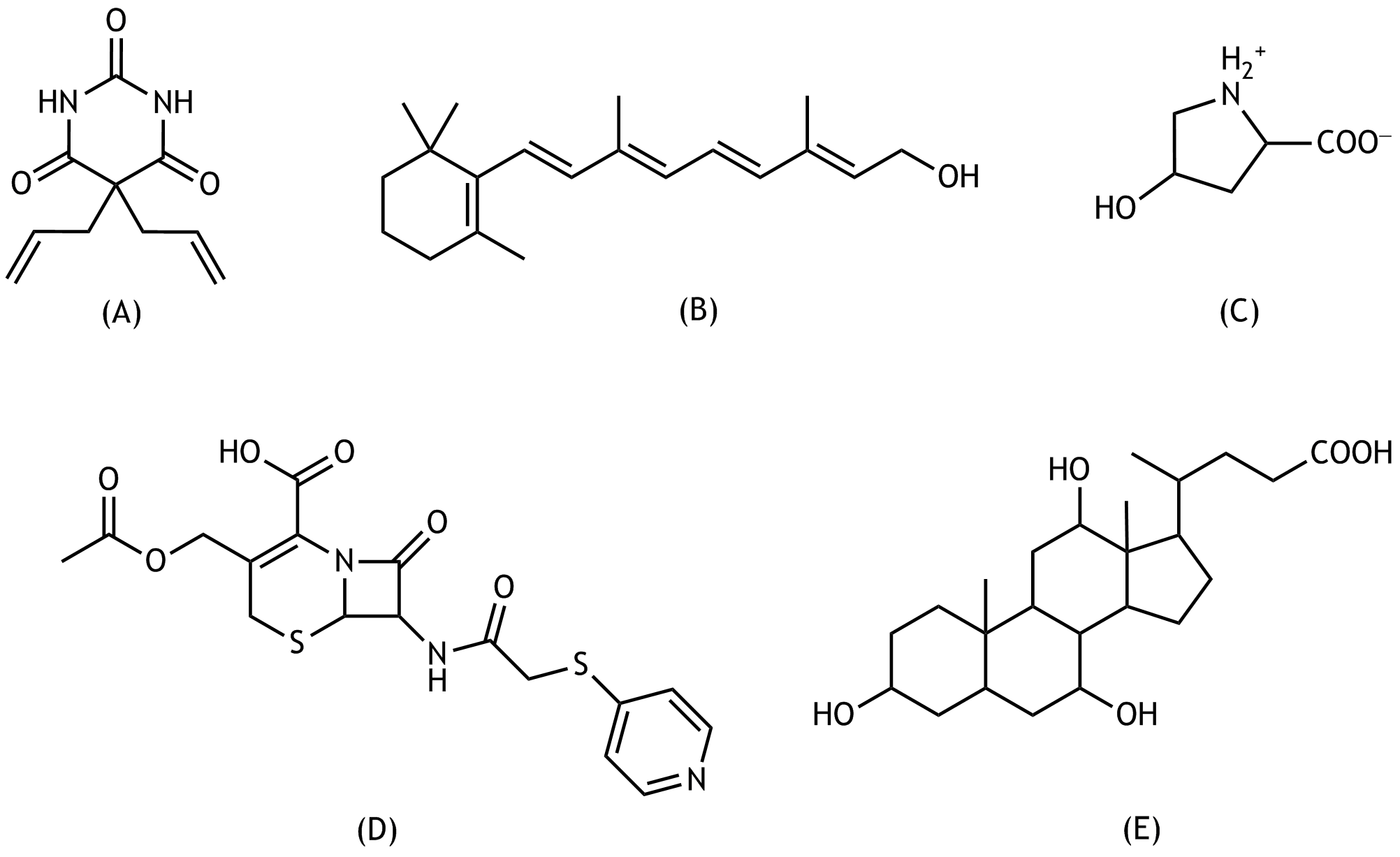
7. Which statement about the depicted antibiotic cephalosporin is correct?
A. The compound has a lactam ring (cyclic amide).
B. The compound has a thiol group (mercapto group).
C. The compound has two isoelectric points.
D. The compound contains 2-amino-2-phenylacetic acid as a component.
E. The compound has three amide bonds.
A. The compound has a lactam ring (cyclic amide).

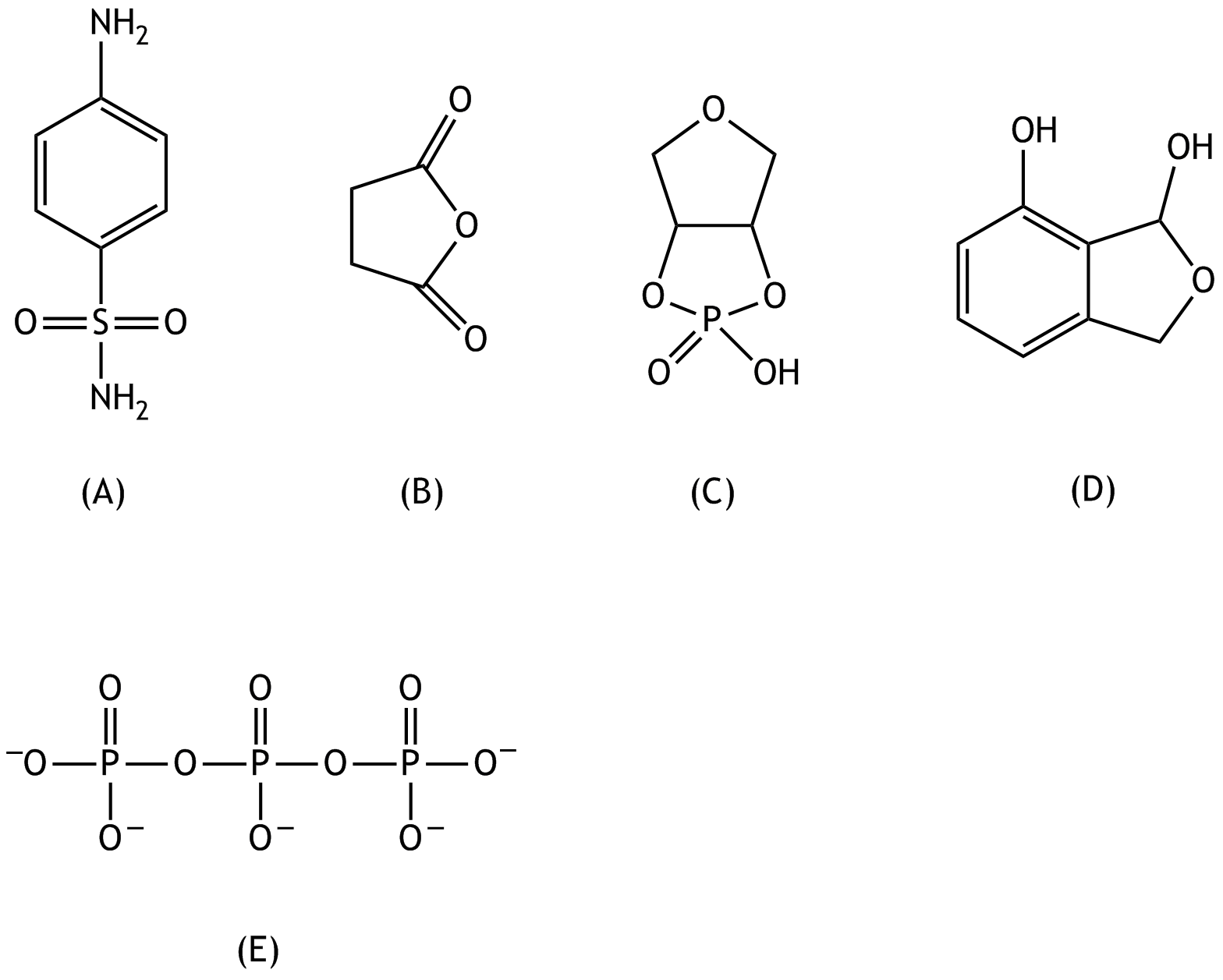
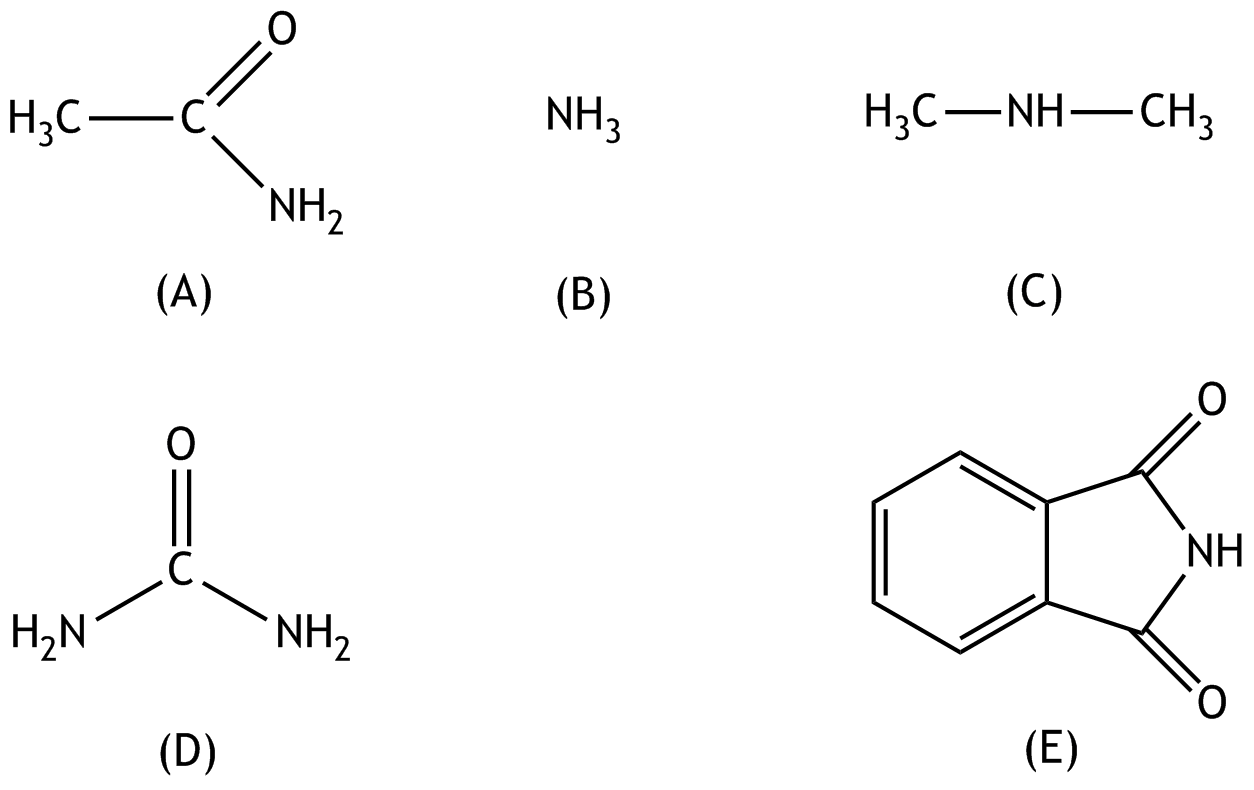
8. For which of the following compounds (A) to (E) does it best apply that it belongs to the group of esters?
C
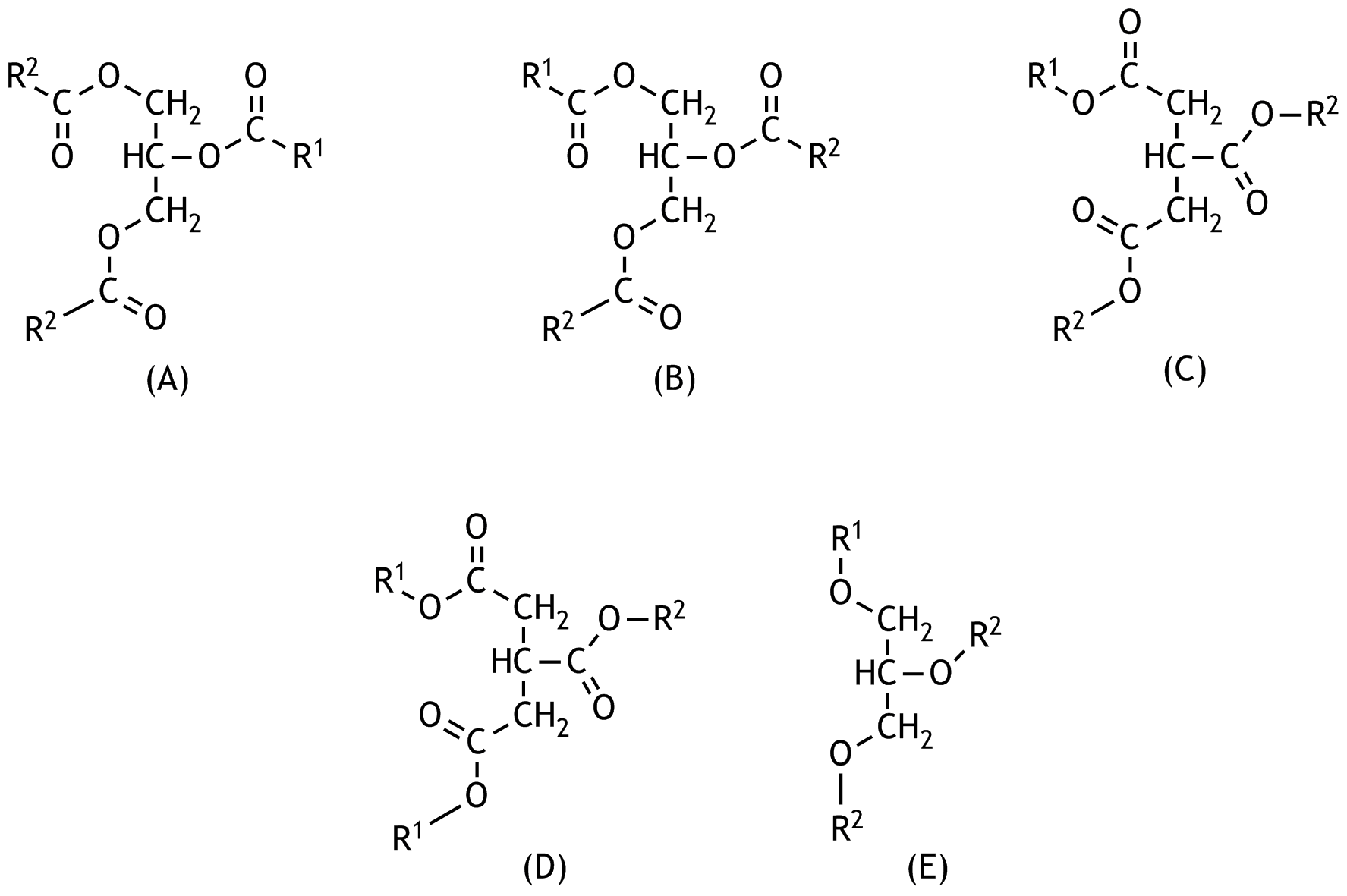
9. Which of the following structural formulas represents a triacylglycerol in which one of the depicted carbon atoms is a chiral center, assuming R₁ and R₂ represent two different alkyl residues of the fatty acids?
B
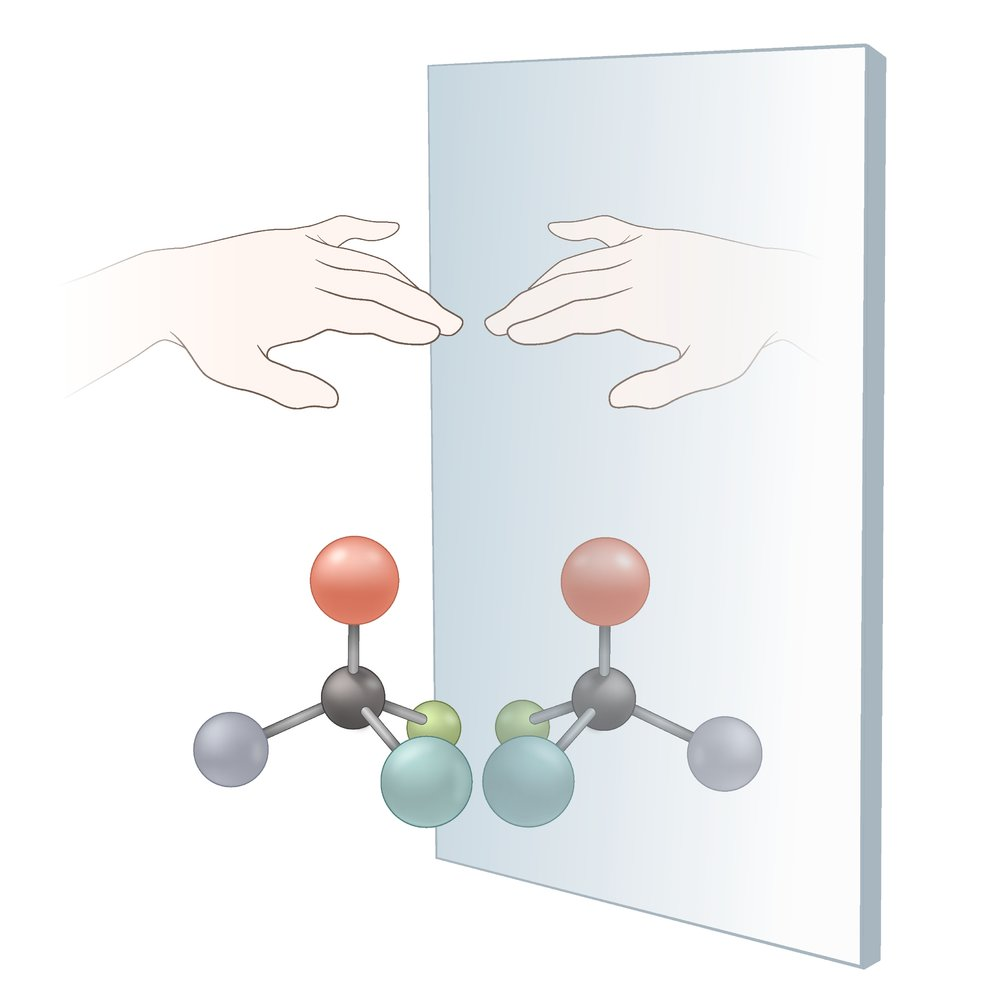
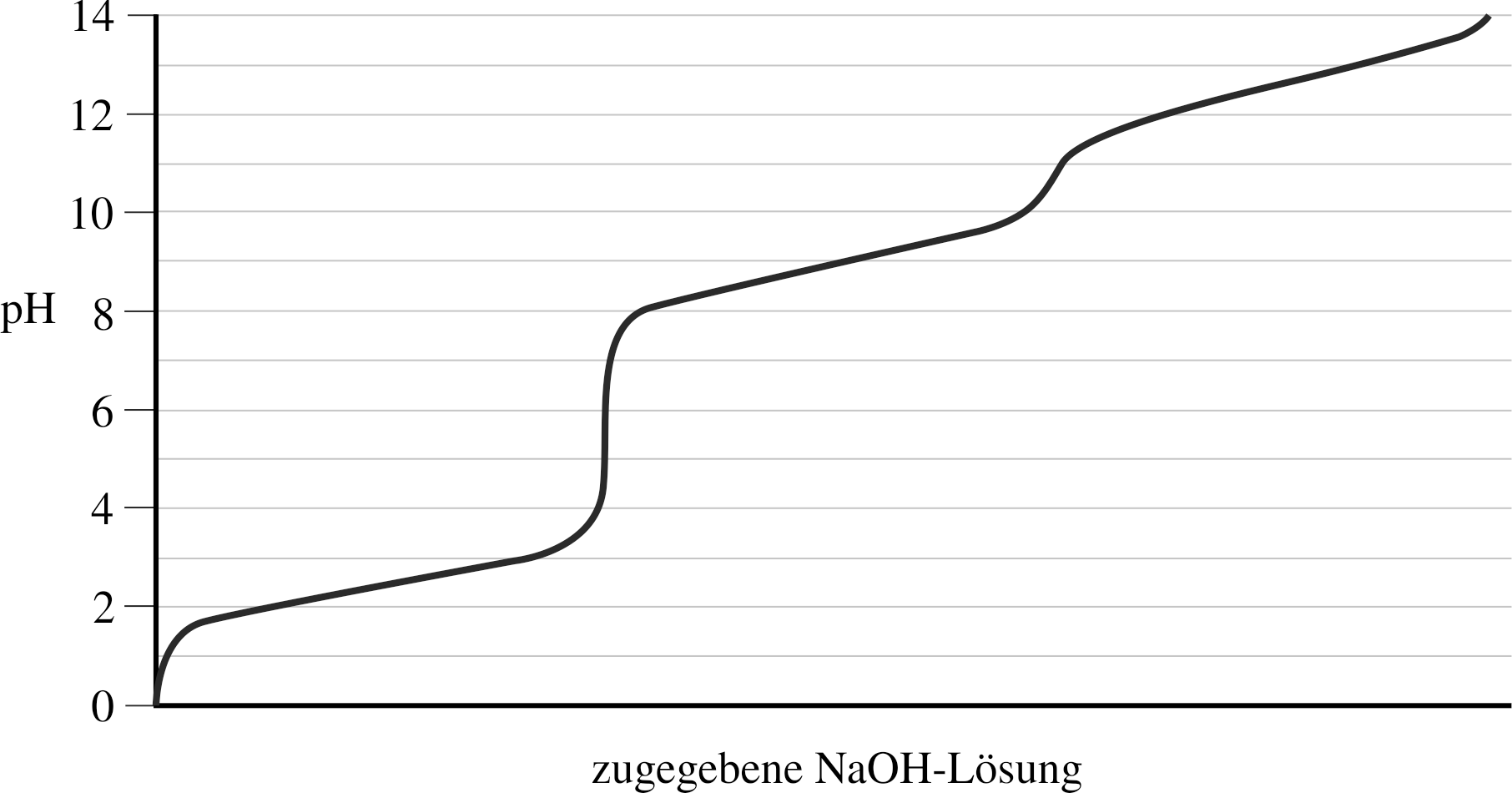
10. For which of the following amino acids is the depicted titration curve most characteristic?
A. Arginine
B. Glutamic acid
C. Glycine
D. Histidine
E. Isoleucine
A. Arginine
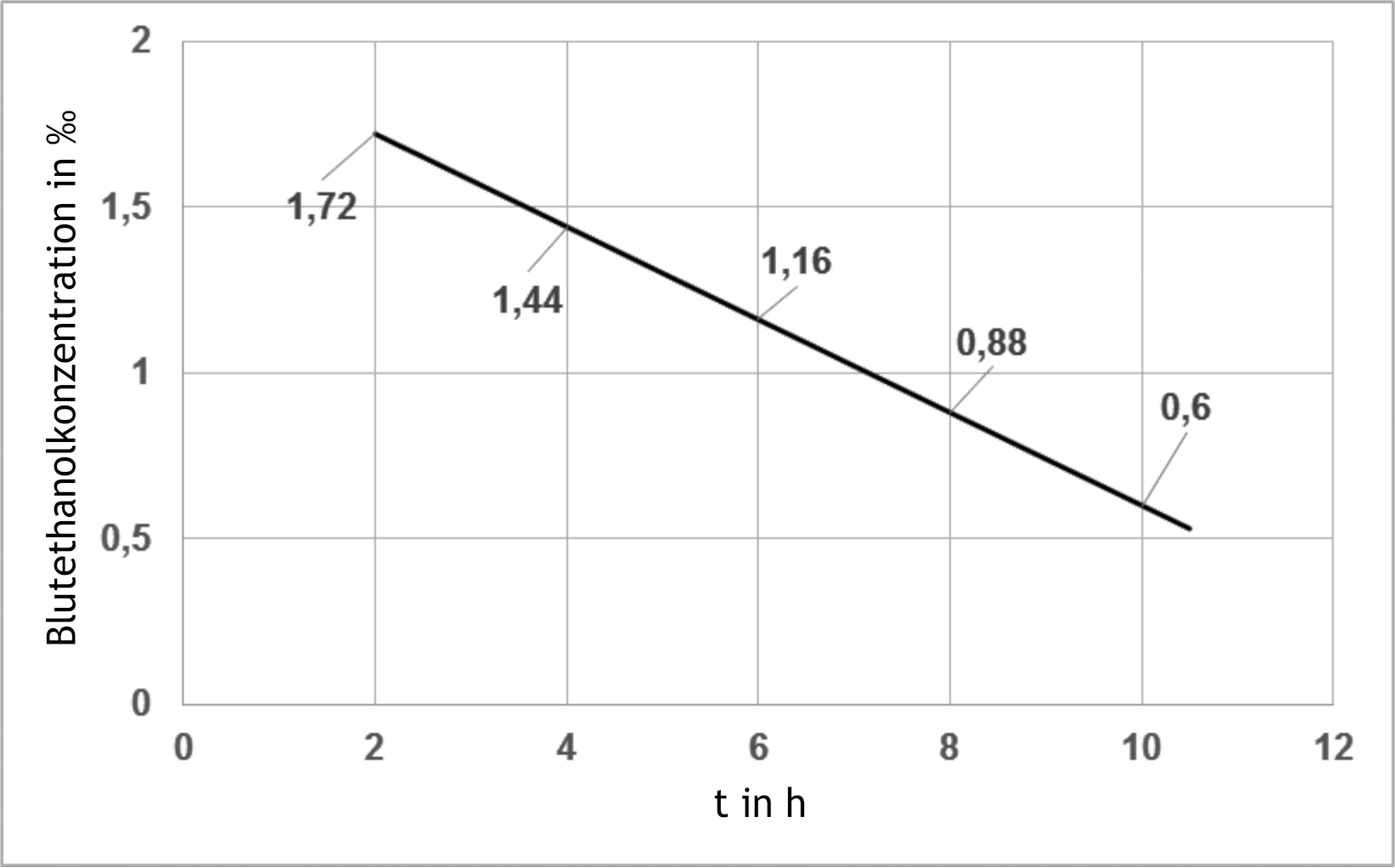
11. The ethanol degradation of an intoxicated patient is quantitatively described by the following diagram (blood ethanol concentration in ‰ vs. time). The blood ethanol concentration was measured starting 2 hours after ethanol ingestion. Which statement can be derived for the measurement period shown in the diagram?
A. At a higher initial ethanol concentration, the reaction rate would be higher than that shown in the graph.
B. The half-life is constantly 6 hours.
C. The reaction rate is directly proportional to the existing ethanol concentration.
D. The rate constant corresponds to a first-order reaction.
E. The reaction rate is constant.
E. The reaction rate is constant.
12. Which of the following elements has the highest mass fraction in the human body?
A. Carbon
B. Sodium
C. Phosphorus
D. Oxygen
E. Hydrogen
D. Oxygen
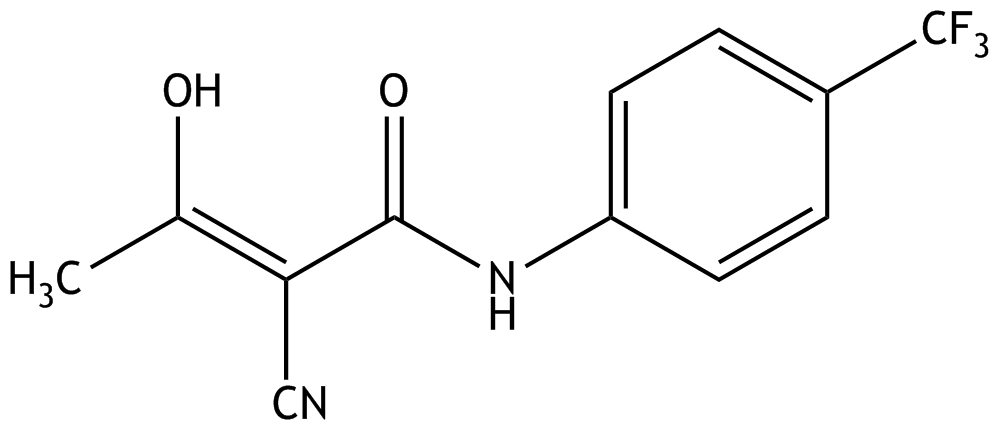
13. Teriflunomide, an inhibitor of dihydroorotate dehydrogenase, is an active substance approved for the treatment of multiple sclerosis.
Which of the chemical elements contained in this compound has the highest electronegativity?
A. Fluorine
B. Carbon
C. Oxygen
D. Nitrogen
E. Hydrogen
A. Fluorine
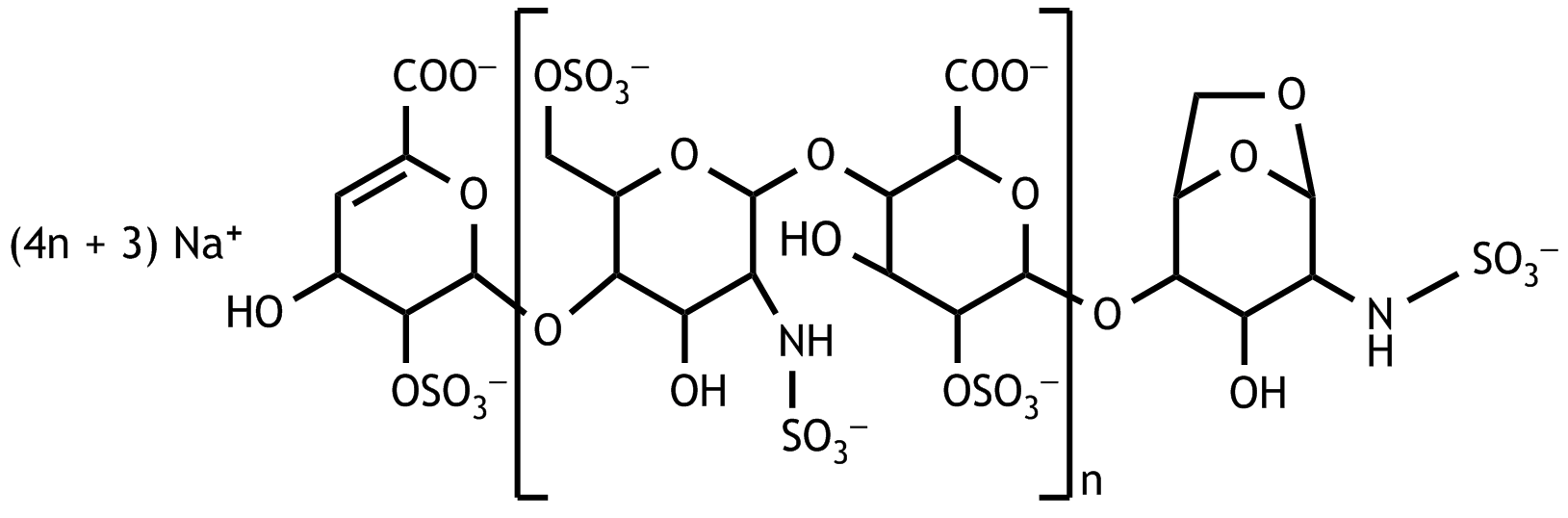
14. Low-molecular-weight heparins are used for thrombosis prophylaxis. For simplification, it is assumed that the following oligomers are present uniformly as the sodium salt: furthermore, let n in the above formula be equal to 2 (n = 2). An amount of this sodium salt of the oligomer is dissolved in (pure) water so that the solution is isoosmolar to blood plasma.
Approximately what is the molar concentration of the sodium salt of the oligomer in this solution?
A. 25 mmol/L
B. 50 mmol/L
C. 100 mmol/L
D. 200 mmol/L
E. 600 mmol/L
A. 25 mmol/L
15. To estimate the amount of HCl produced per day by the gastric glands in grams, the following assumptions are made:
The gastric glands produce 3 L of gastric juice per day.
The gastric juice is simplified as hydrochloric acid with a pH value of 1.
The molar mass of HCl is approximately 37 g/mol.
Approximately how many grams of HCl are produced by the gastric glands per day?
A. 0.37 g
B. 0.81 g
C. 1.1 g
D. 3.7 g
E. 11 g
E. 11 g
16. Which aqueous solution of the following salts has the lowest pH value at a concentration of 100 mmol/L?
A. Ammonium chloride
B. Potassium chloride
C. Sodium acetate
D. Sodium propionate
E. Sodium trichloroacetate
A. Ammonium chloride
17. For which of the following compounds is protonation of the nitrogen atom in aqueous solution most likely?
C
18. Kidney stones are often calcium oxalate stones.
Which is the correct molecular formula for calcium oxalate?
A. CaCO₃
B. CaC₂O₄
C. Ca(C₂H₅O)₂
D. Ca₂C₂O₄
E. CaO
B. CaC₂O₄
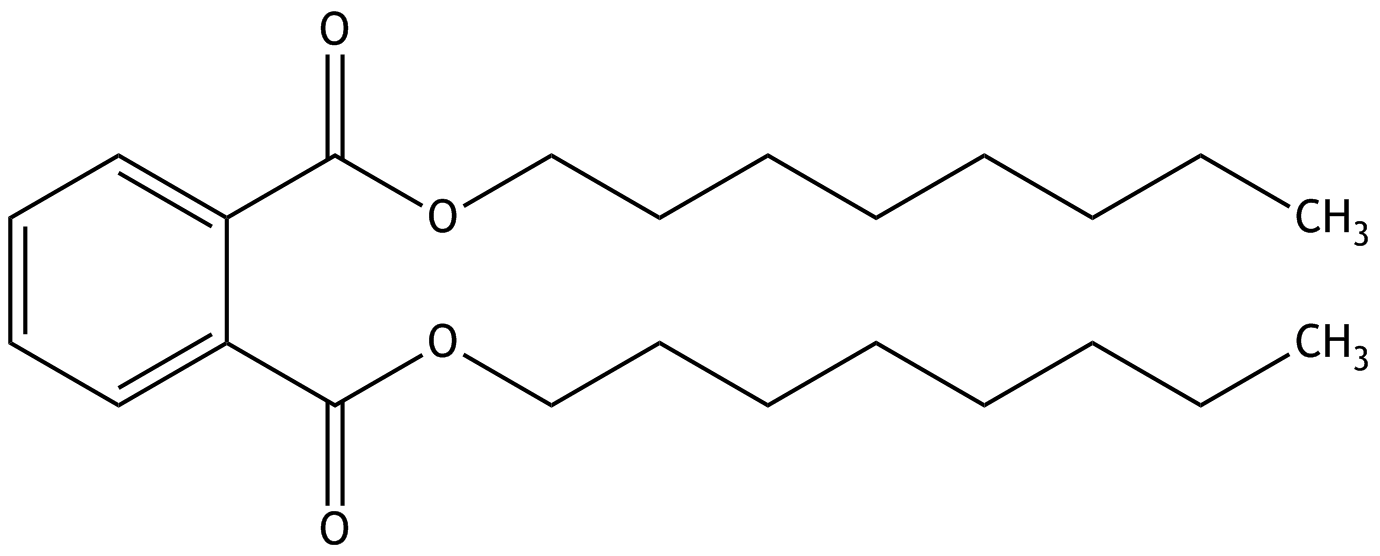
19. The di-n-octyl phthalate (DnOP) shown here is a plasticizer from the group of phthalates and a component of various plastics.
After oral intake of DnOP, various degradation products of the compound can be detected in the urine. What happens in the case of a presumed complete hydrolysis of the compound?
A. One of the reaction products formed is a dicarboxylic acid.
B. Two anhydride bonds are cleaved.
C. Two ether bonds are cleaved.
D. Two fatty acids are released.
E. A quinone is cleaved.
A. One of the reaction products formed is a dicarboxylic acid.
20. Benzene is labeled according to the Hazardous Substances Ordinance as “H350: May cause cancer.” Although the mechanism of action is very complex, it is assumed that an initial oxidation by cytochrome P450–dependent mixed-function monooxygenases occurs, forming benzene oxide, which is responsible for the carcinogenic effect.
Toluene (methylbenzene) is currently not classified as carcinogenic (although this does not mean that toluene poses no health risks).
Which mechanism best explains the lower carcinogenic potential of toluene?
A. Toluene is significantly more hydrophilic than benzene and can therefore be excreted directly via the kidneys.
B. Toluene is cleaved into phenol and methanol.
C. Toluene is reduced in a NAD(P)H-dependent manner to methylcyclohexane.
D. Toluene is oxidatively cleaved into dihydroxyacetone and D-threose.
E. Toluene is oxidized to benzoate.
E. Toluene is oxidized to benzoate.
21. Which of the following substances is a medically relevant material (e.g., used in hip socket implants)?
A. Ammonium hydrogen carbonate
B. Manganese dioxide
C. Cadmium oxide
D. Polyethylene
E. Tetrachlorosilane
D. Polyethylene
22. Which interaction characteristically underlies the β-pleated sheet structure in a protein?
A. Hydrophobic interactions between uncharged sections of the polypeptide chain
B. Ionic interactions between atoms of the protein backbone
C. Ionic interactions between basic and acidic amino acid residues
D. Stabilizing intramolecular disulfide bridges
E. Hydrogen bonds between adjacent sections of the polypeptide chain
E. Hydrogen bonds between adjacent sections of the polypeptide chain
23. How many valence electrons does the nitrogen atom have?
A. 2
B. 3
C. 4
D. 5
E. 6
D. 5
24. Which statement about the spatial arrangement of molecules or complex ions is correct?
A. CO₂ is a linear molecule.
B. The carbon atoms in benzene each form the center of a tetrahedron.
C. Formaldehyde has a tetrahedral structure.
D. H₂O is a linear molecule.
E. Ozone is a linear molecule.
A. CO₂ is a linear molecule.
25. In the event of impending acute barium poisoning after oral ingestion of barium chloride, the oral administration of a sodium sulfate (Na₂SO₄, Glauber’s salt) solution may be indicated.
The antidotal effect of Na₂SO₄ is most likely based on the fact that
A. Na₂SO₄ oxidizes barium ions to metallic barium.
B. Na₂SO₄ neutralizes barium chloride in an acid-base reaction.
C. Na₂SO₄ increases the solubility of barium chloride.
D. Na₂SO₄ forms a chelate complex with barium chloride that is excreted.
E. poorly soluble BaSO₄ is formed, which is excreted in the stool.
E. poorly soluble BaSO₄ is formed, which is excreted in the stool.
26. Which of these structural formulas contains an aromatic system?
D
27. Which of the following molecules contains an ester bond?
A. Adenosine
B. Glucose-6-phosphate
C. Glutathione
D. 5-Methylcytosine
E. Pyrophosphate (diphosphate)
B. Glucose-6-phosphate
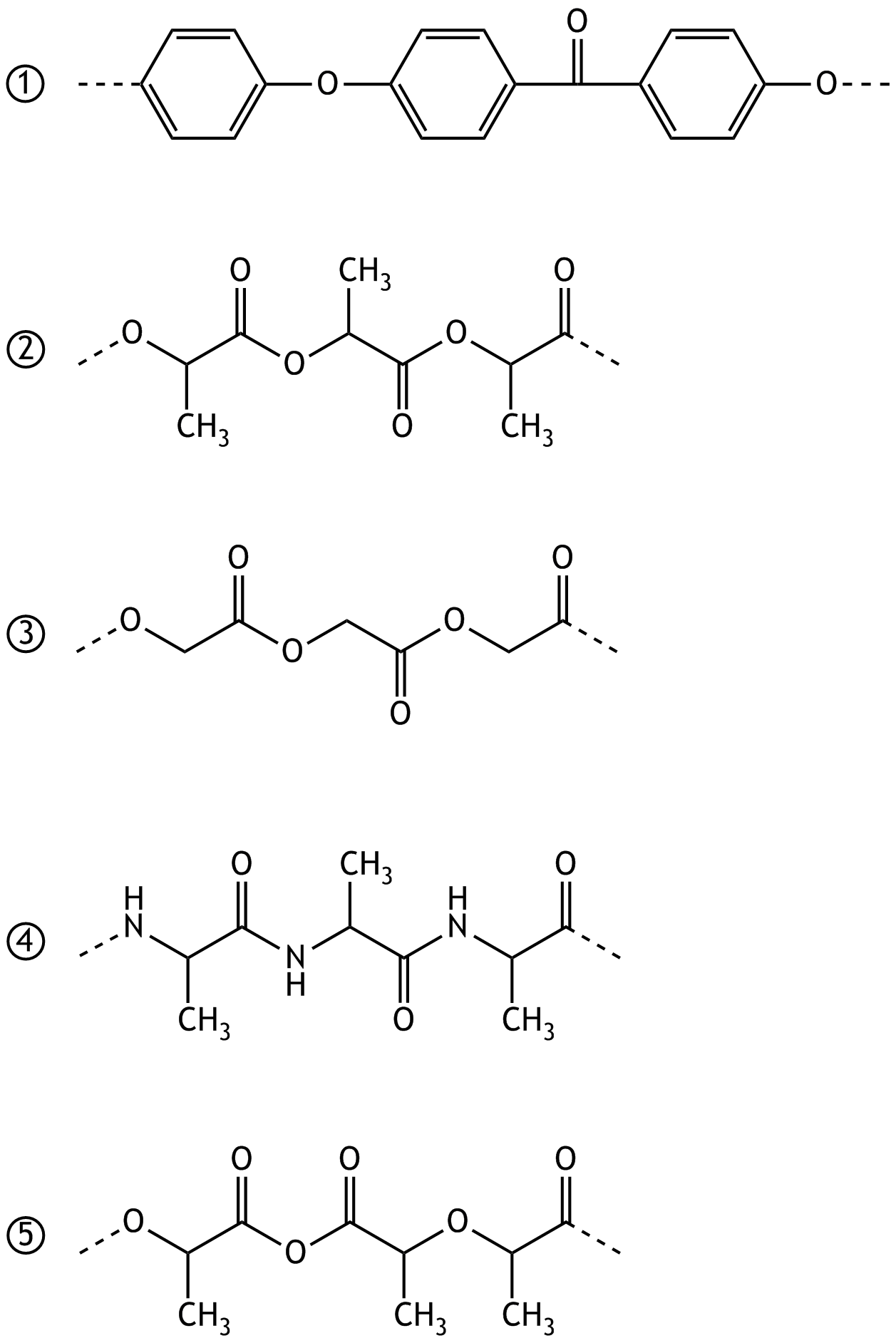
28. In tissue engineering, porous structures made of polylactide (“polylactic acid,” a polyester derived from lactic acid molecules) are used because polylactide can be degraded by the human body.
Which of the following formulas ① to ⑤ represents a portion of this polylactide?
A. ①
B. ②
C. ③
D. ④
E. ⑤
B. ②
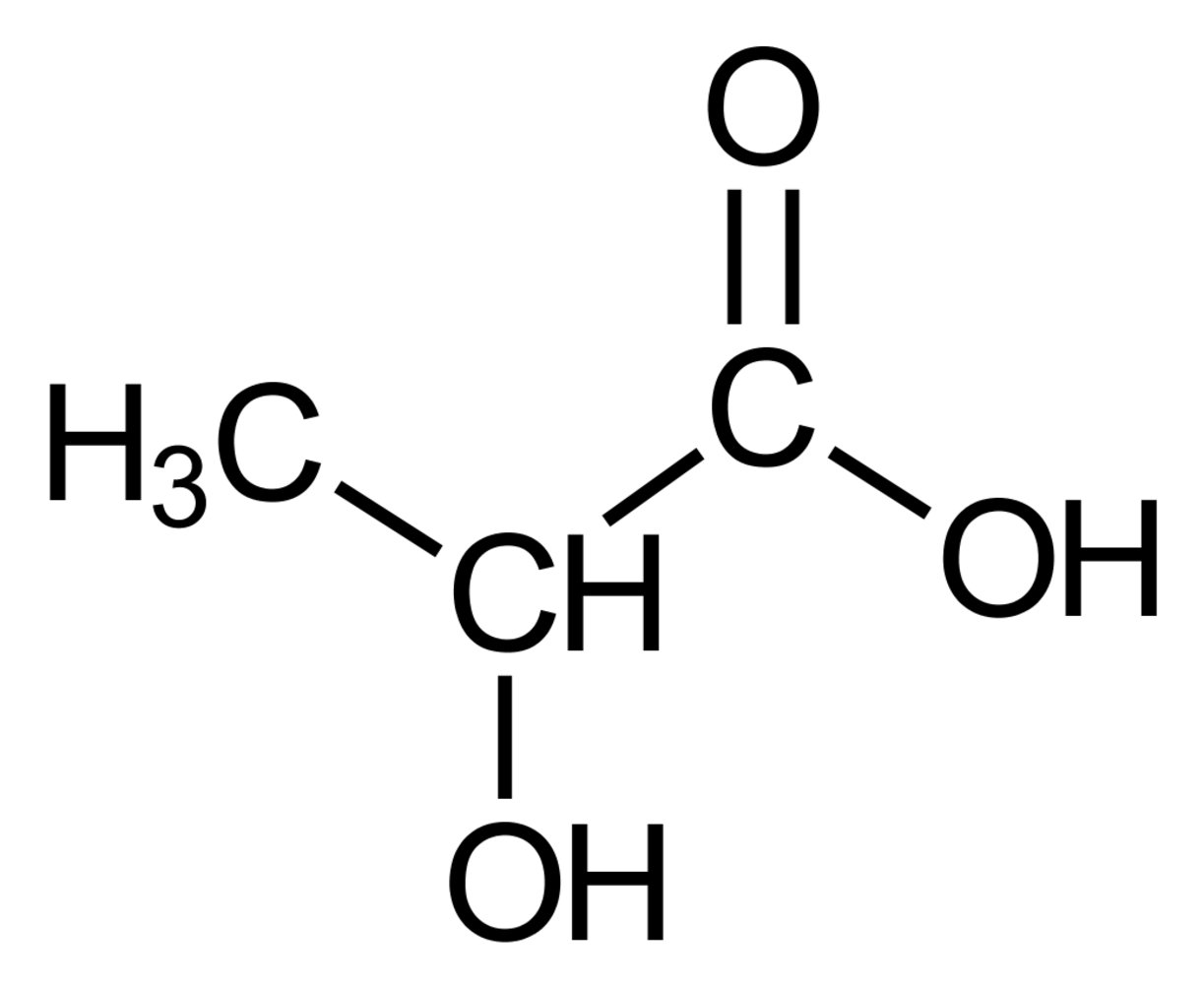
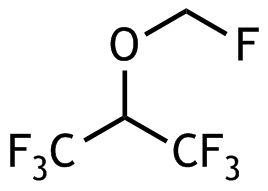
29. The inhalation anesthetic sevoflurane with the following chemical structure
A. has a chiral center
B. is an alkane
C. is an alcohol
D. is an ether
E. is a hemiacetal
D. is an ether
30. Which redox cofactor can, in the form mentioned, act as an oxidizing agent in enzymatic reactions?
A. FADH₂
B. Fe³⁺
C. FMNH₂
D. NADH
E. Ubiquinol (ubihydroquinone)
B. Fe³⁺
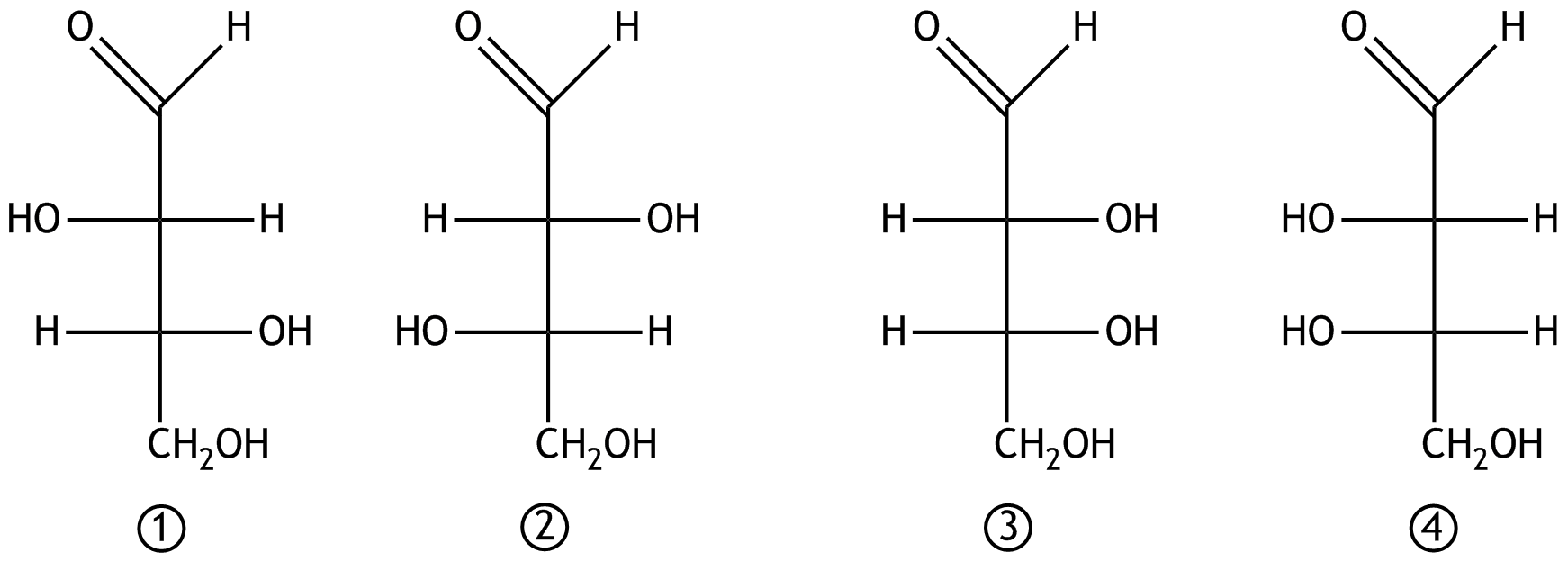
31. The following structural formulas (Fischer projections) of four monosaccharides are shown.
Which statement about the isomerism of the depicted monosaccharides is correct?
A. Compounds ① and ② are diastereomers.
B. Compounds ① and ③ are constitutional isomers.
C. Compounds ① and ④ are epimers.
D. Compounds ② and ③ are enantiomers.
E. Compounds ② and ④ are conformers.
C. Compounds ① and ④ are epimers.
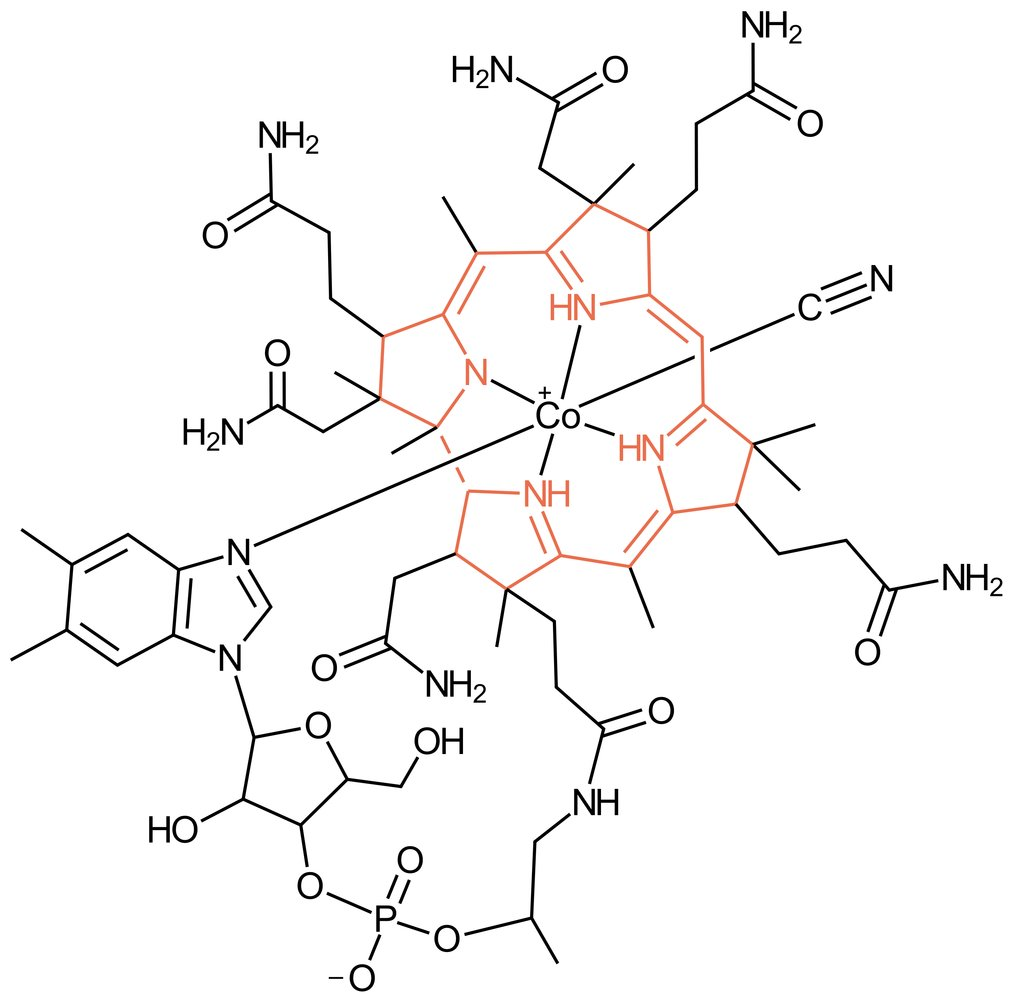
32. In a patient with vitamin B₁₂ deficiency, a resorption test is performed using an orally administered vitamin B₁₂ preparation in which a chemical element characteristically contained in vitamin B₁₂ is present as a radioactive isotope.
Which of the following elements is it?
A. Cobalt
B. Iron
C. Magnesium
D. Manganese
E. Zinc
A. Cobalt
33. In a human, one nanoliter (nL) of blood contains about 300 platelets. The average lifespan of a platelet in the blood is about 9 days. The total blood volume is about 6 liters.
Approximately how many newly formed platelets are released into the blood each day under steady-state conditions?
A. 3 × 10⁴
B. 2 × 10⁵
C. 1.6 × 10⁷
D. 3 × 10¹⁰
E. 2 × 10¹¹
E. 2 × 10¹¹
34. The partial pressure of an inhalation anesthetic in a patient’s inspired gas mixture is 1 kPa (7.5 mmHg). The total pressure of the inspired gas mixture is approximately equal to the average atmospheric pressure at sea level. The mixture can be approximated as an ideal gas mixture.
Approximately what is the volume fraction of the inhalation anesthetic in the total volume of the inspired gas mixture, in percent?
A. 1 %
B. 2 %
C. 4 %
D. 8 %
E. 16 %
A. 1 %
35. A patient is to be injected with a solution of an isolated monoclonal antibody. The concentration of this solution is 1 mmol/L. The molecular mass of the antibody is 145 kDa.
How much antibody is contained in 1 mL of the solution?
A. 1.45 μg
B. 14.5 μg
C. 145 μg
D. 1.45 mg
E. 145 mg
E. 145 mg
36. What is meant by ionization energy?
A. The absolute measure of the tendency to donate electrons in a redox reaction
B. The relative measure of a bonding partner’s tendency to attract the shared electron pair in a covalent bond
C. The numerical value of the charge of anions
D. The energy released or required when an atom takes up a proton
E. The energy that must be supplied to remove the highest-energy electron from the electron shell
E. The energy that must be supplied to remove the highest-energy electron from the electron shell
37. How many valence electrons does the nitrogen atom have?
A. 2
B. 5
C. 6
D. 7
E. 14
B. 5
38. What is the maximum number of atoms that a carbon atom can be covalently bonded to in stable compounds (under physiological conditions)?
A. 2
B. 4
C. 6
D. 8
E. 12
B. 4
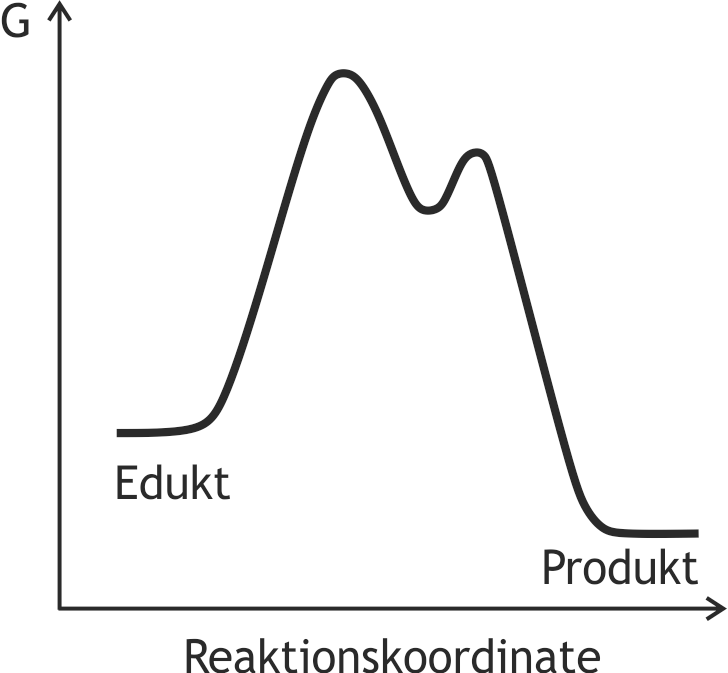
39. The following figure represents the energy diagram of a hypothetical chemical reaction.
Which statement about the course of this reaction can be clearly derived from this energy diagram?
(G = Gibbs free energy)
A. It is an endothermic reaction.
B. It is an irreversible reaction.
C. It is a catalyzed reaction.
D. It is a reaction with an intermediate step.
E. It is a second-order reaction.
D. It is a reaction with an intermediate step.
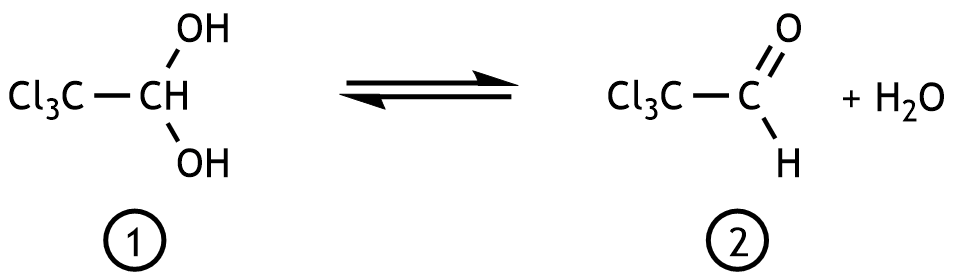
40. Substance ① is used for the short-term treatment of sleep disorders. Under physiological conditions, the following equilibrium is established at the beginning of its metabolism: In this reaction, substance ① is
A. dehydrated
B. dehydrogenated
C. hydrolyzed
D. isomerized
E. reduced
A. dehydrated
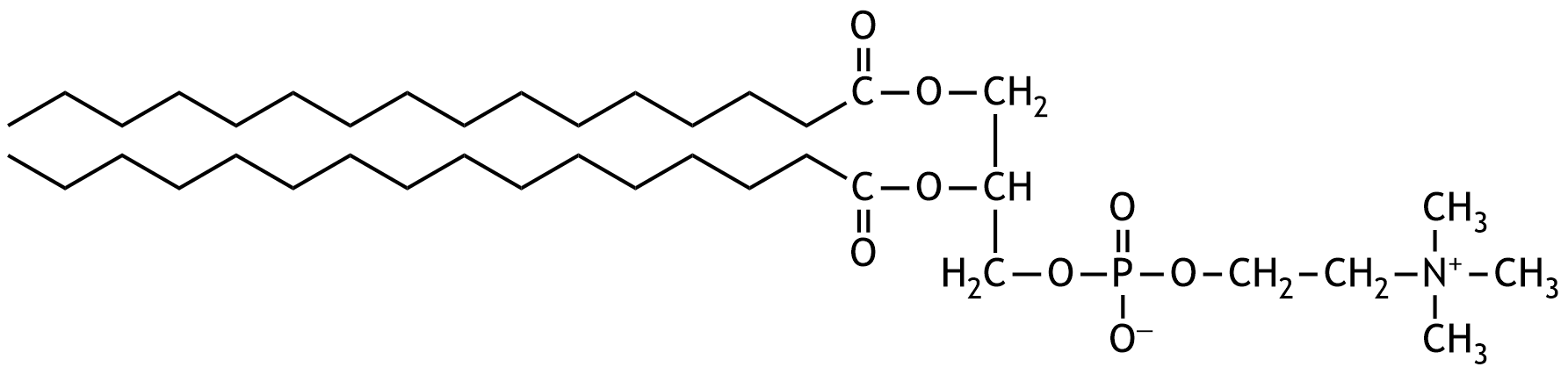
41. Dipalmitoylphosphatidylcholine (DPPC) is an important component of surfactant.
Which of the following types of chemical compounds or functional groups is present in DPPC?
A. Asymmetric ether
B. Carboxylic acid ester
C. Phosphoric acid anhydride
D. Secondary alcohol
E. Tertiary amine
B. Carboxylic acid ester
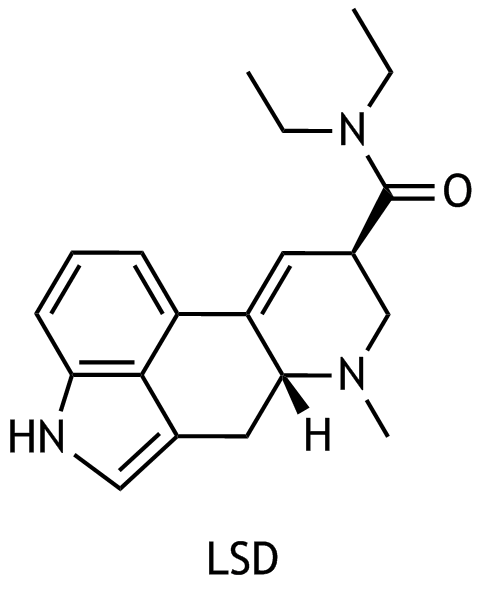
42. LSD, a derivative of tetracyclic lysergic acid, is a psychoactive compound.
LSD is
A. Lysergic acid diethylamide
B. Lysergic acid diethylamine
C. Lysergic acid diethylammonium
D. Lysergic acid diethylketimine
E. Lysergic acid dimethylammonium
A. Lysergic acid diethylamide
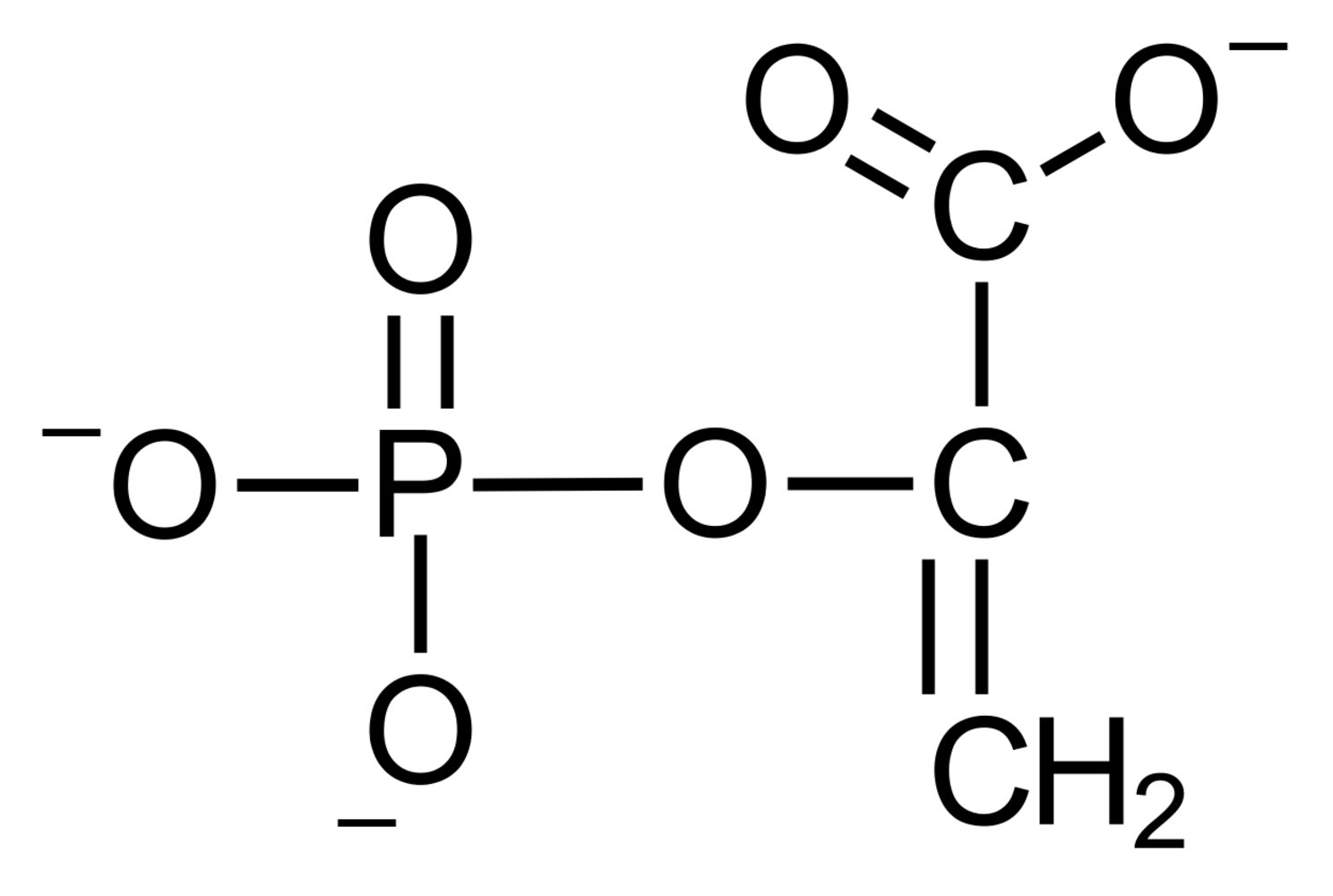
43. Which of the following compounds is an ester?
A. Glucuronic acid
B. Inosine
C. Isomaltose
D. Phosphoenolpyruvate
E. Sucrose
D. Phosphoenolpyruvate
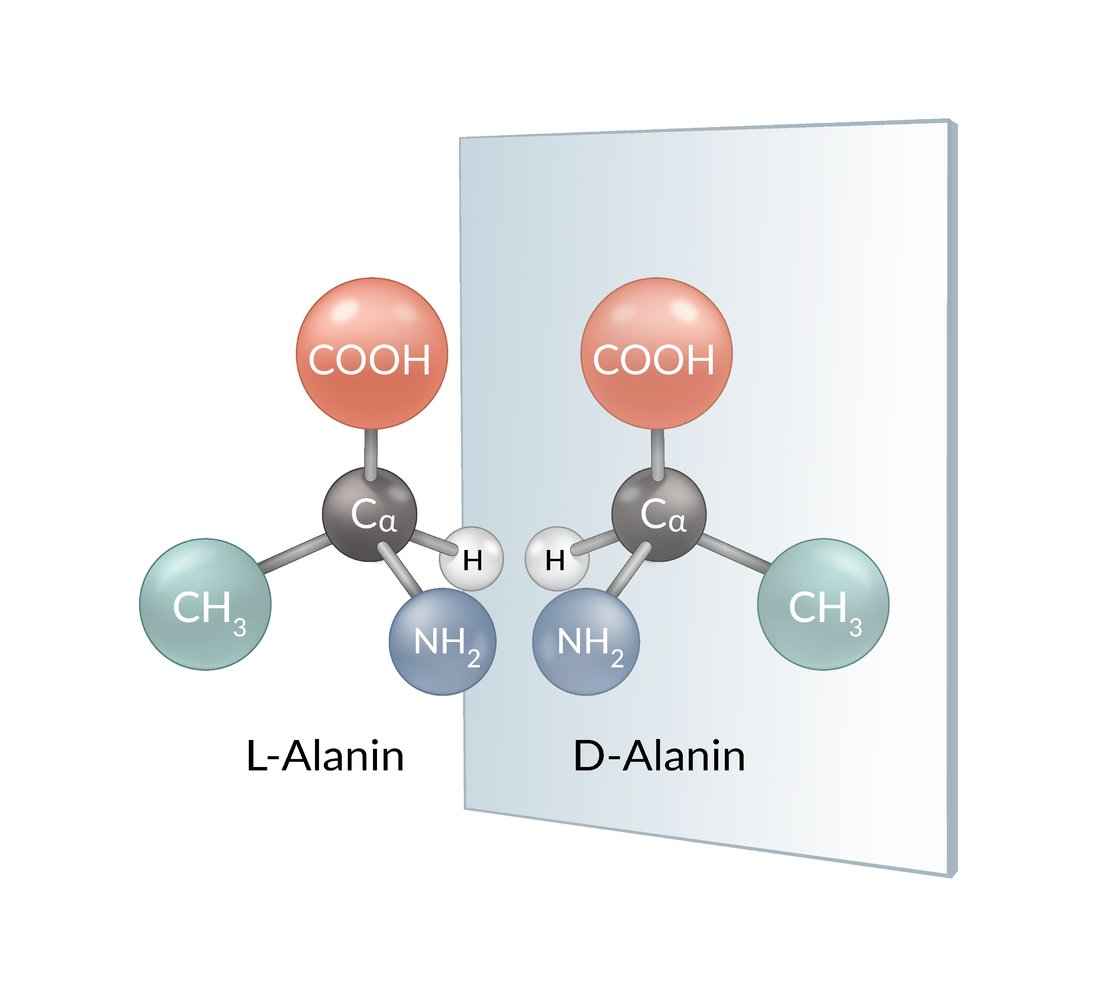
44. Which two compounds are enantiomers?
A. Adenosine-5’-monophosphate and adenosine-3’-monophosphate
B. 11-cis-retinal and all-trans-retinal
C. β-D-galactopyranose and β-D-mannopyranose
D. β-D-glucopyranose and α-D-glucopyranose
E. L-alanine and D-alanine
E. L-alanine and D-alanine
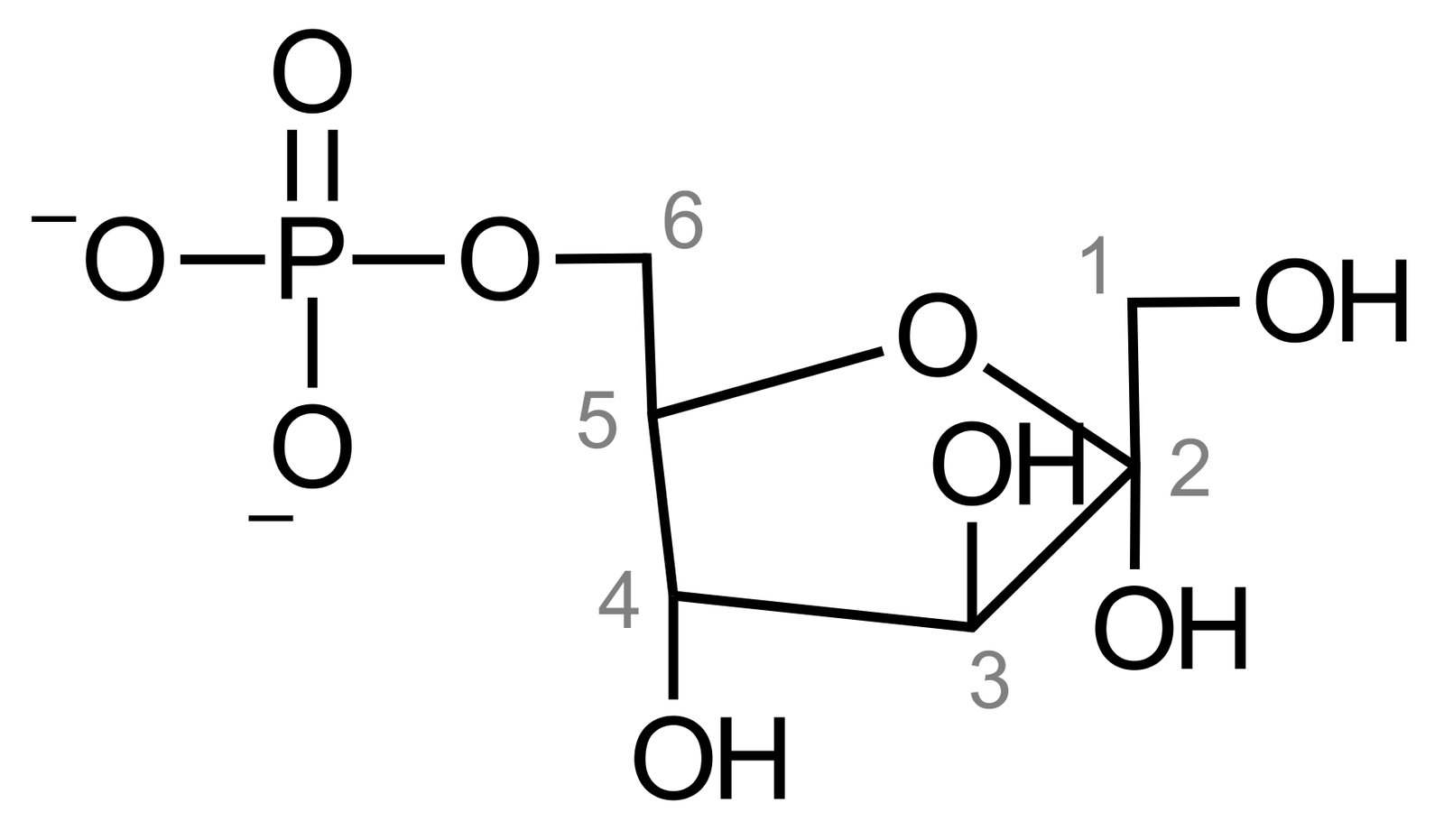
45. Which of the following substances is isomeric with galactose-1-phosphate?
A. 6-Phosphogluconolactone
B. Fructose-6-phosphate
C. Glucuronic acid-1-phosphate
D. Ribose-5-phosphate
E. Sorbitol-1-phosphate
B. Fructose-6-phosphate
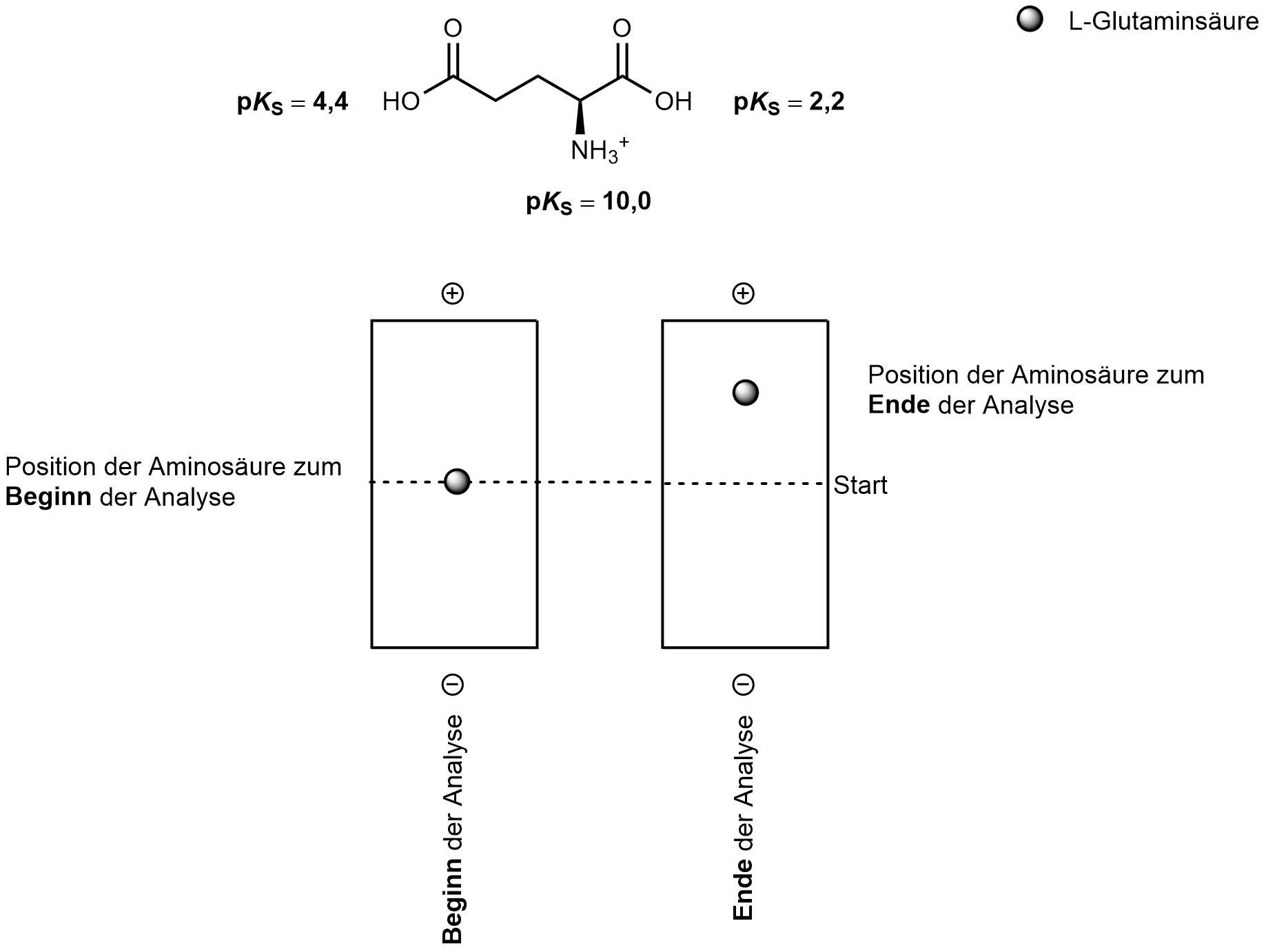
46. The following figure shows an idealized electrophoretic analysis of L-glutamic acid:
At which of the following pH values was the analysis most likely performed?
A. 0
B. 1
C. 2
D. 3
E. 7
E. 7
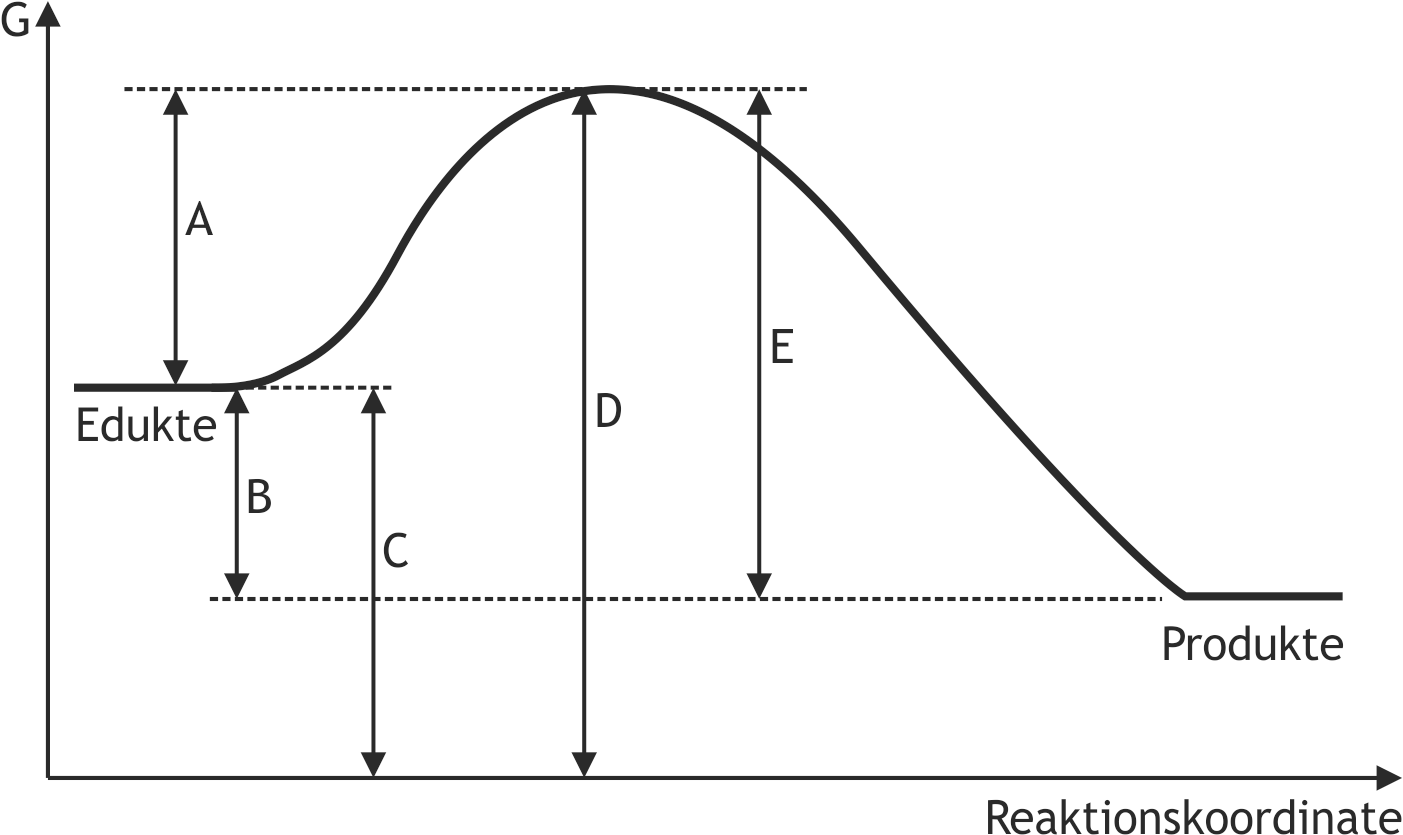
47. The drawing shows the energy diagram of a hypothetical chemical reaction.
Which of the arrows A to E in this diagram represents the free reaction enthalpy (Gibbs free energy)?
(G = Free enthalpy, Gibbs energy)
A
B
C
D
E
B
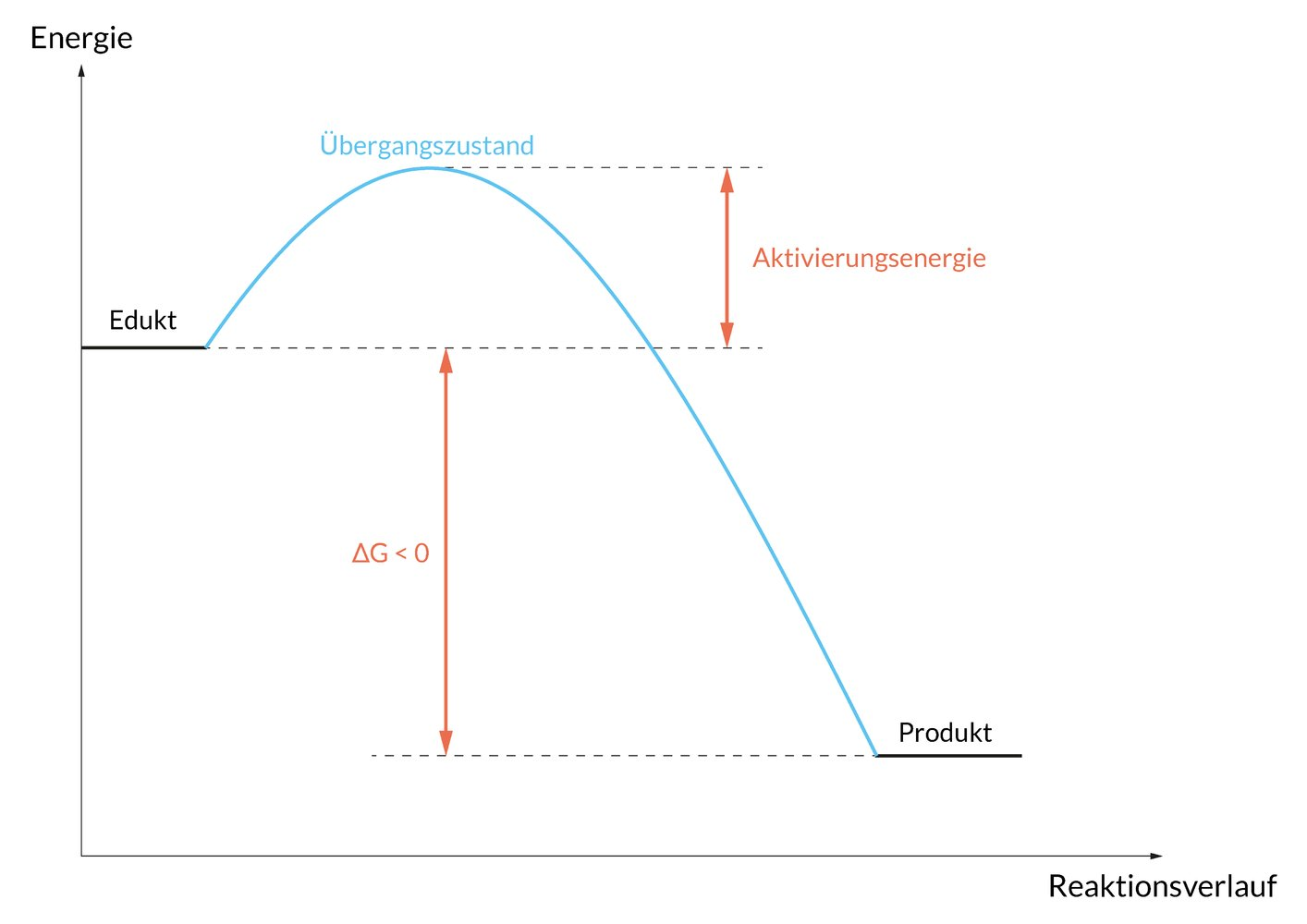
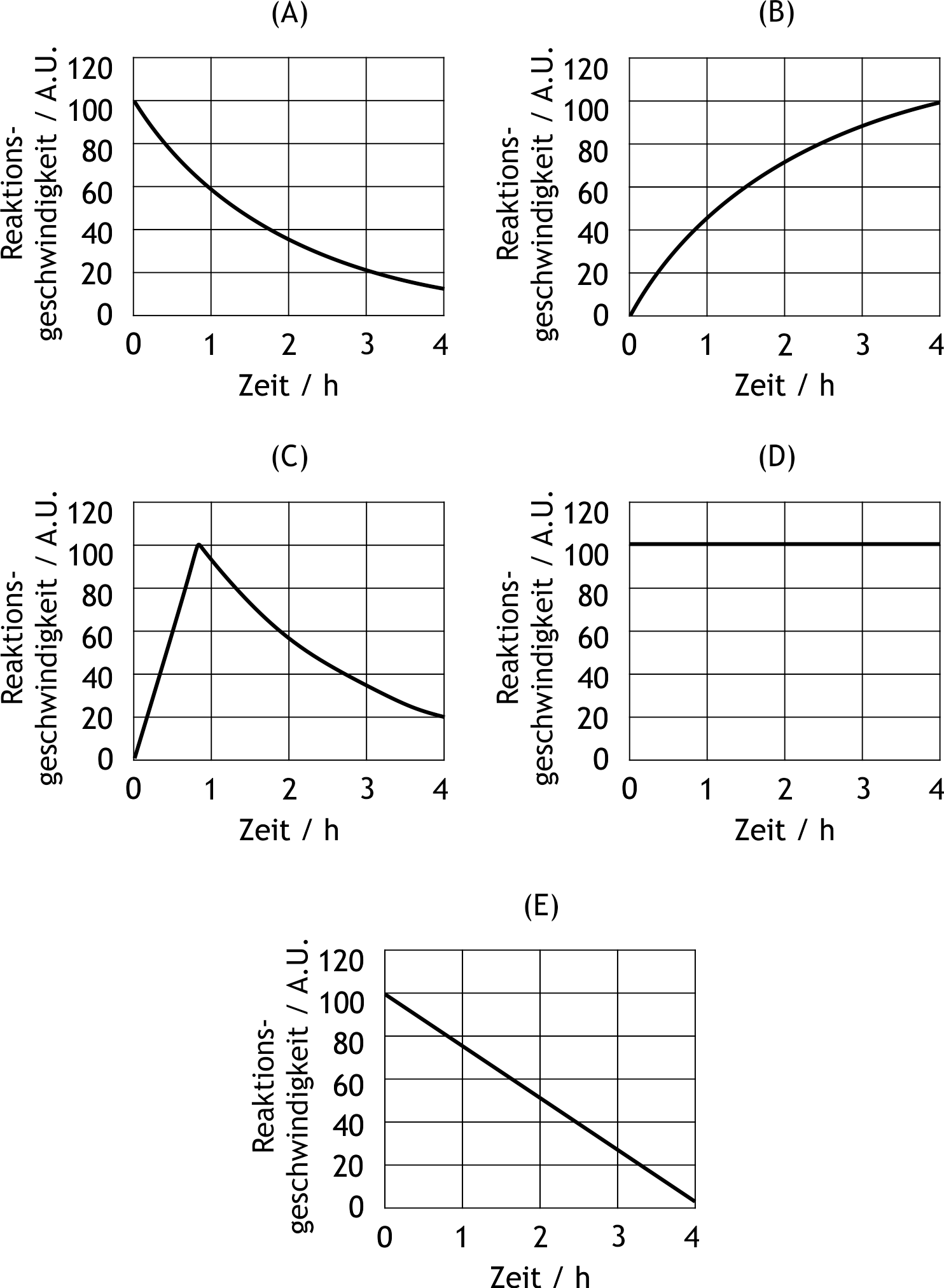
48. The breakdown of alcohol in the body follows a pseudo–zero-order reaction because alcohol dehydrogenase remains saturated throughout the observation period.
Which of the following graphs correctly describes the time course of a (pseudo-)zero-order degradation reaction?
(A.U. = arbitrary units = appropriate unit of measurement)
A
B
C
D
E
D
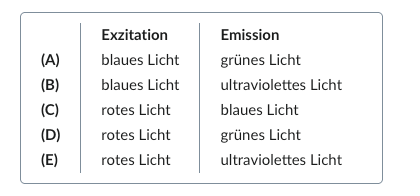
49. To diagnose epithelial defects of the cornea, a fluorescent dye is applied to the patient’s eye and stimulated with light.
Which combination of exciting (excitation) and dye-emitted (emission) light is most likely possible?
A. (A)
B. (B)
C. (C)
D. (D)
E. (E)
A. (A)
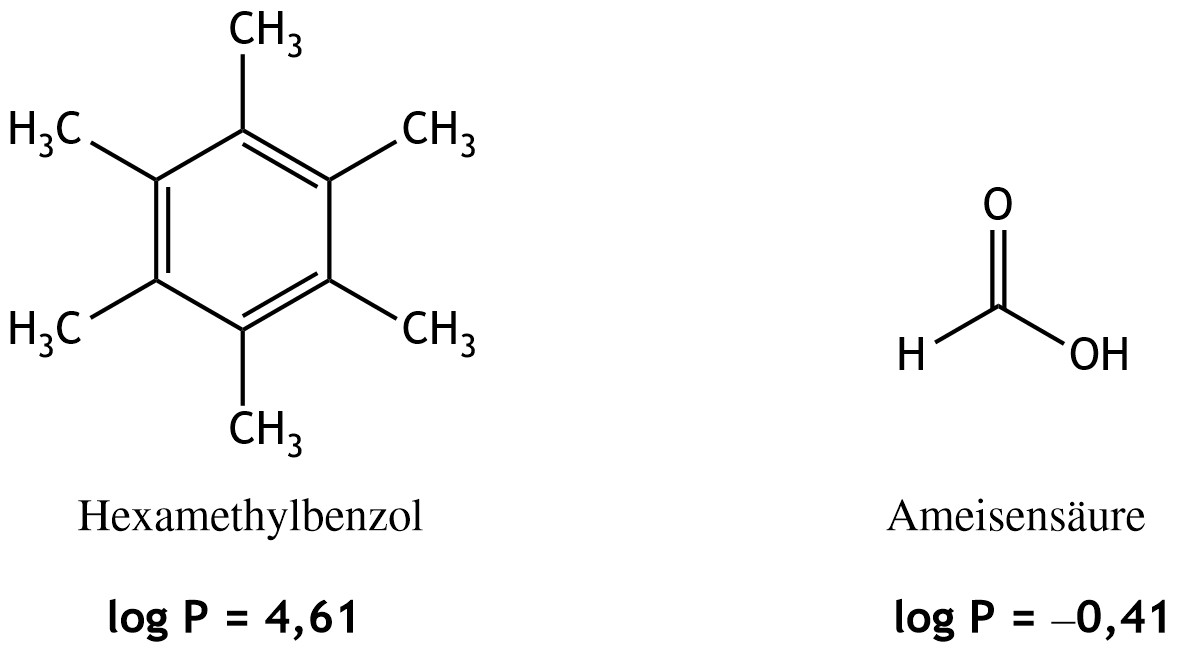
50. The physiologically relevant lipophilicity (hydrophobicity) of a substance is characterized by the decimal logarithm of the n-octanol–water partition coefficient (log P), which is defined as: log P = log Kₒw = log (cₒ / c𝓌)
In this formula, cₒ represents the concentration of the substance in the octanol-rich phase, and c𝓌 represents the concentration of the substance in the water-rich phase.
For the compounds hexamethylbenzene and formic acid, the following log P values were determined at 25 °C:
Which statement regarding their water or fat solubility can be made based on these values?
A. Hexamethylbenzene is hydrophilic and formic acid is hydrophobic.
B. Hexamethylbenzene is hydrophilic and formic acid is lipophilic.
C. Hexamethylbenzene is lipophilic and formic acid is hydrophilic.
D. Hexamethylbenzene is lipophilic and formic acid is hydrophobic.
E. Hexamethylbenzene is lipophobic and formic acid is lipophilic.
C. Hexamethylbenzene is lipophilic and formic acid is hydrophilic.
51. For the absorption and distribution of drugs in the body, distribution equilibria are important and can be quantified by distribution laws.
In an extraction experiment, an aqueous solution of a substance with a concentration of 160 mmol/L is mixed with the same volume of a pure organic solvent. After equilibrium is established, the concentration of the substance in the aqueous phase is 120 mmol/L and in the organic phase 40 mmol/L. The aqueous phase is then separated and extracted again with the same volume of the pure organic solvent.
After the second equilibrium is established, the concentration of the substance in the aqueous phase is:
A. 60 mmol/L
B. 72 mmol/L
C. 80 mmol/L
D. 90 mmol/L
E. 100 mmol/L
D. 90 mmol/L
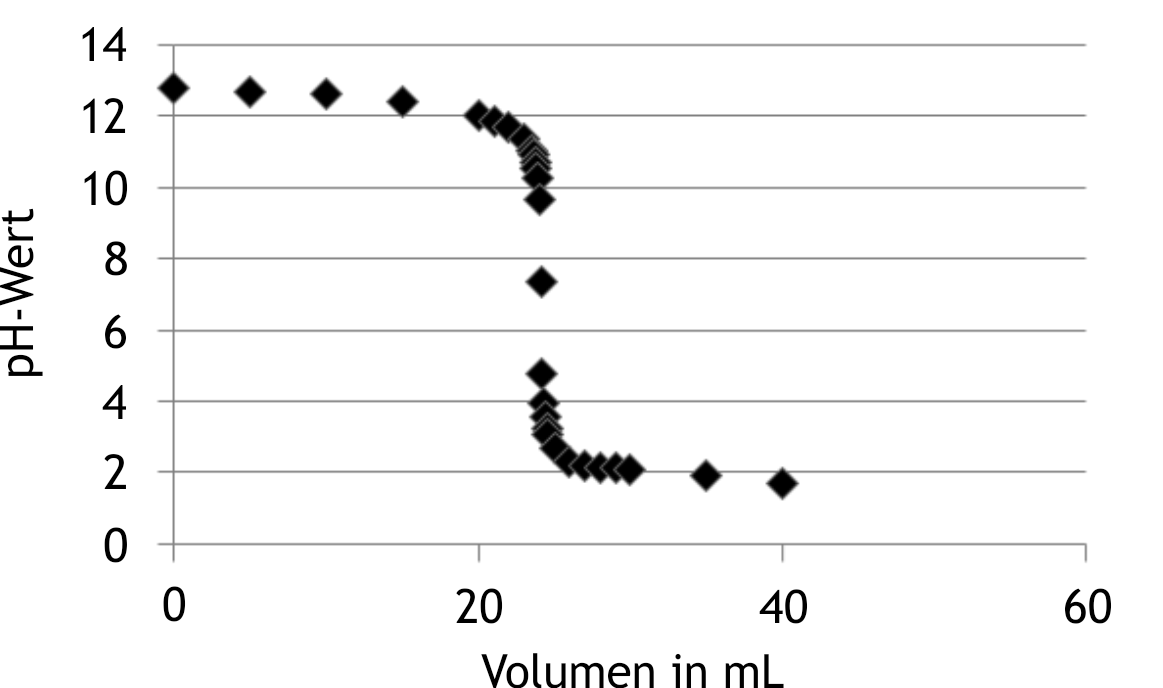
52. In the following diagram, the values of a titration curve are shown: Which process does this curve represent?
A. Addition of a weak acid to a weak base
B. Addition of a weak acid to a strong base
C. Addition of a strong base to a weak acid
D. Addition of a strong base to a strong acid
E. Addition of a strong acid to a strong base
E. Addition of a strong acid to a strong base
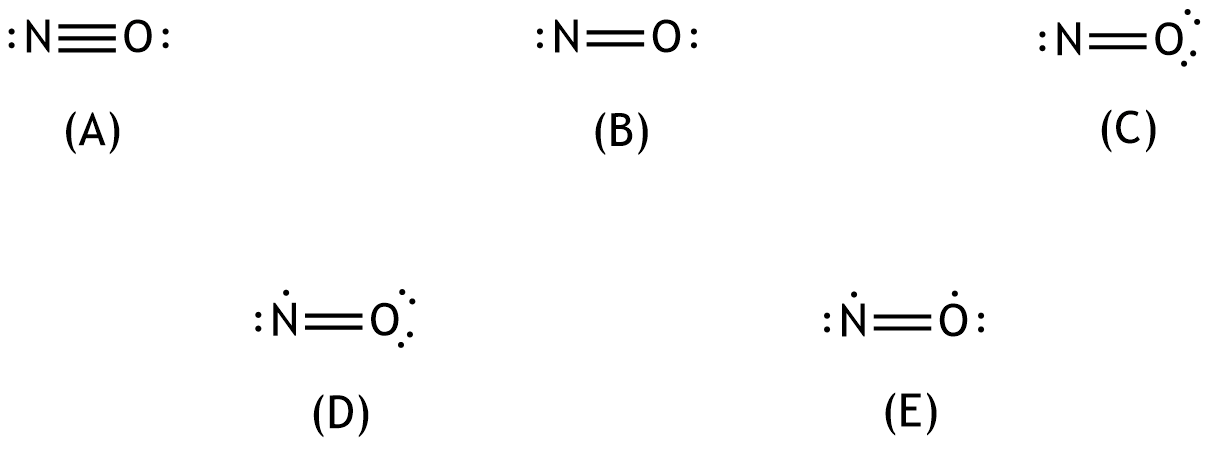
53. Nitric oxide is an important signaling molecule in the body that, among other effects, causes vasodilation.
Which of the following structures represents a (Lewis) formula for the bonding structure of nitric oxide with the correct number of electrons?
A. (A)
B. (B)
C. (C)
D. (D)
E. (E)
D. (D)
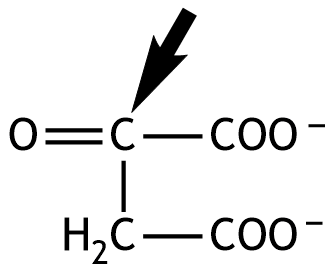
54. Oxaloacetate is formed in the citric acid cycle by oxidation.
What is the oxidation number of the carbon atom marked with an arrow?
A. –2
B. –1
C. 0
D. +1
E. +2
E. +2
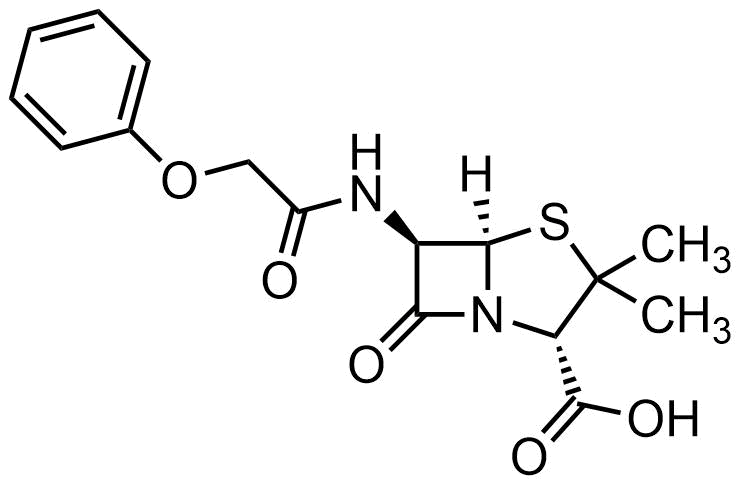
55. Penicillin V:
To which of the following classes of compounds can Penicillin V be assigned?
A. Aldehyde
B. Amide
C. Ester
D. Ketone
E. Lactone
B. Amide
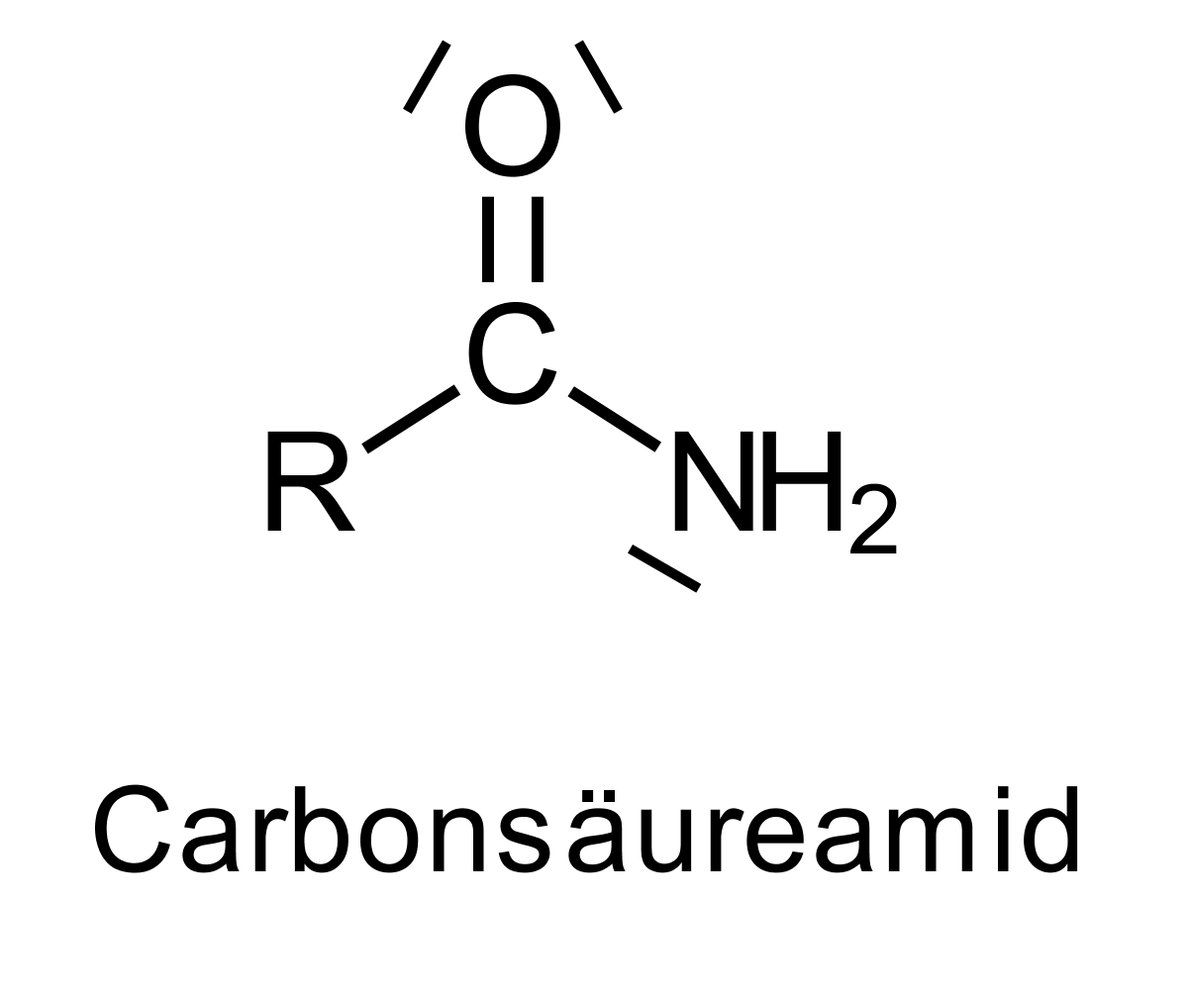
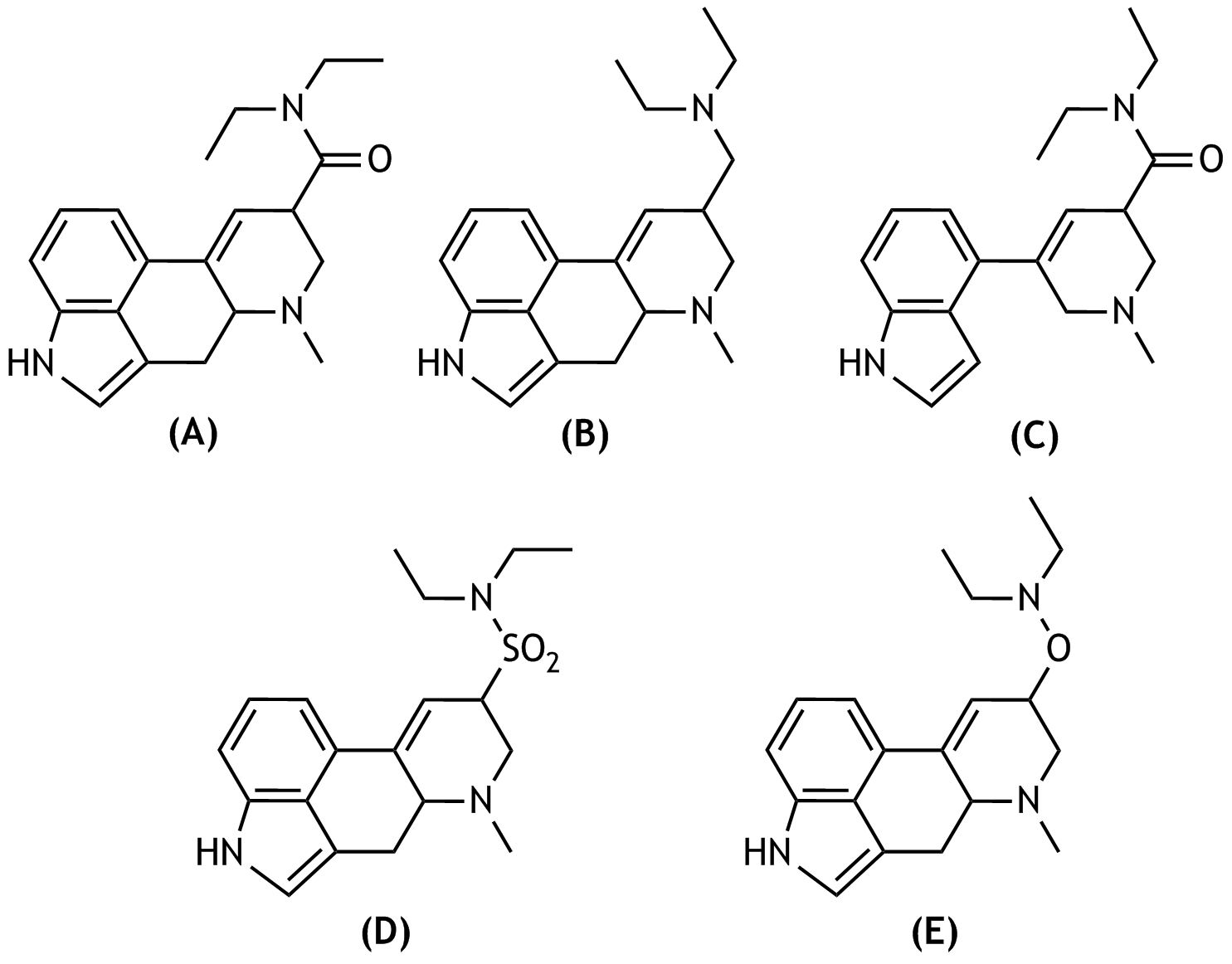
56. Lysergic acid diethylamide — better known by the abbreviation LSD — is a psychoactive derivative of lysergic acid. The latter is a tetracyclic compound that contains a β-amino carboxylic acid unit.
Which of the following structural formulas correctly represents LSD, disregarding stereochemistry?
A. (A)
B. (B)
C. (C)
D. (D)
E. (E)
A. (A)
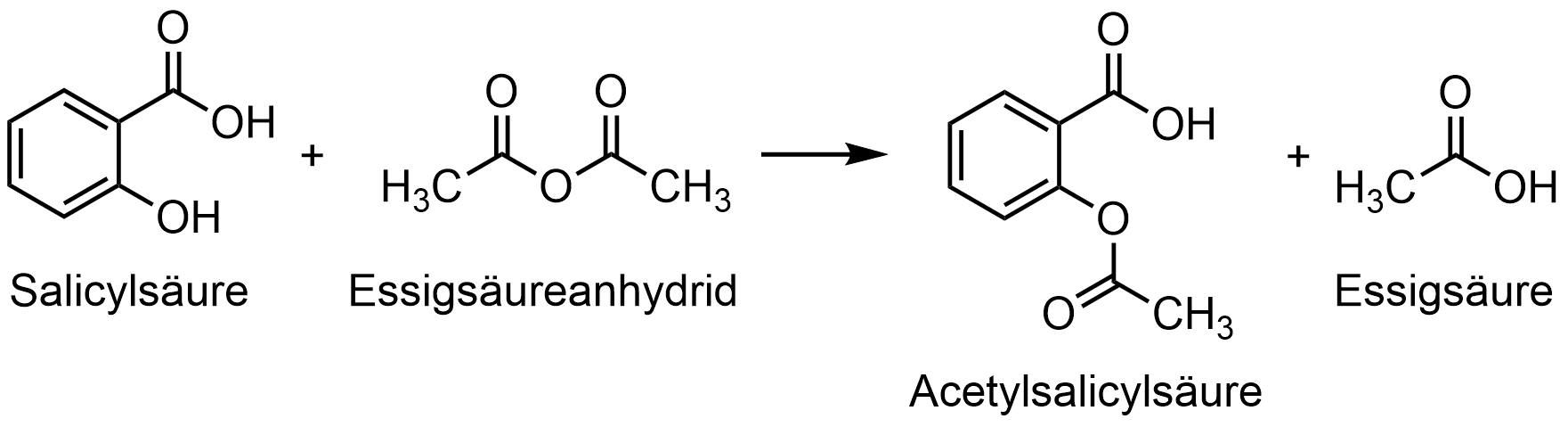
57. The chemical reaction shown below between salicylic acid (2-hydroxybenzoic acid) and acetic anhydride represents the synthesis of acetylsalicylic acid.
What type of reaction is this?
A. Ether formation
B. Ethylation
C. Hydrolysis
D. Methylation
E. Esterification
E. Esterification
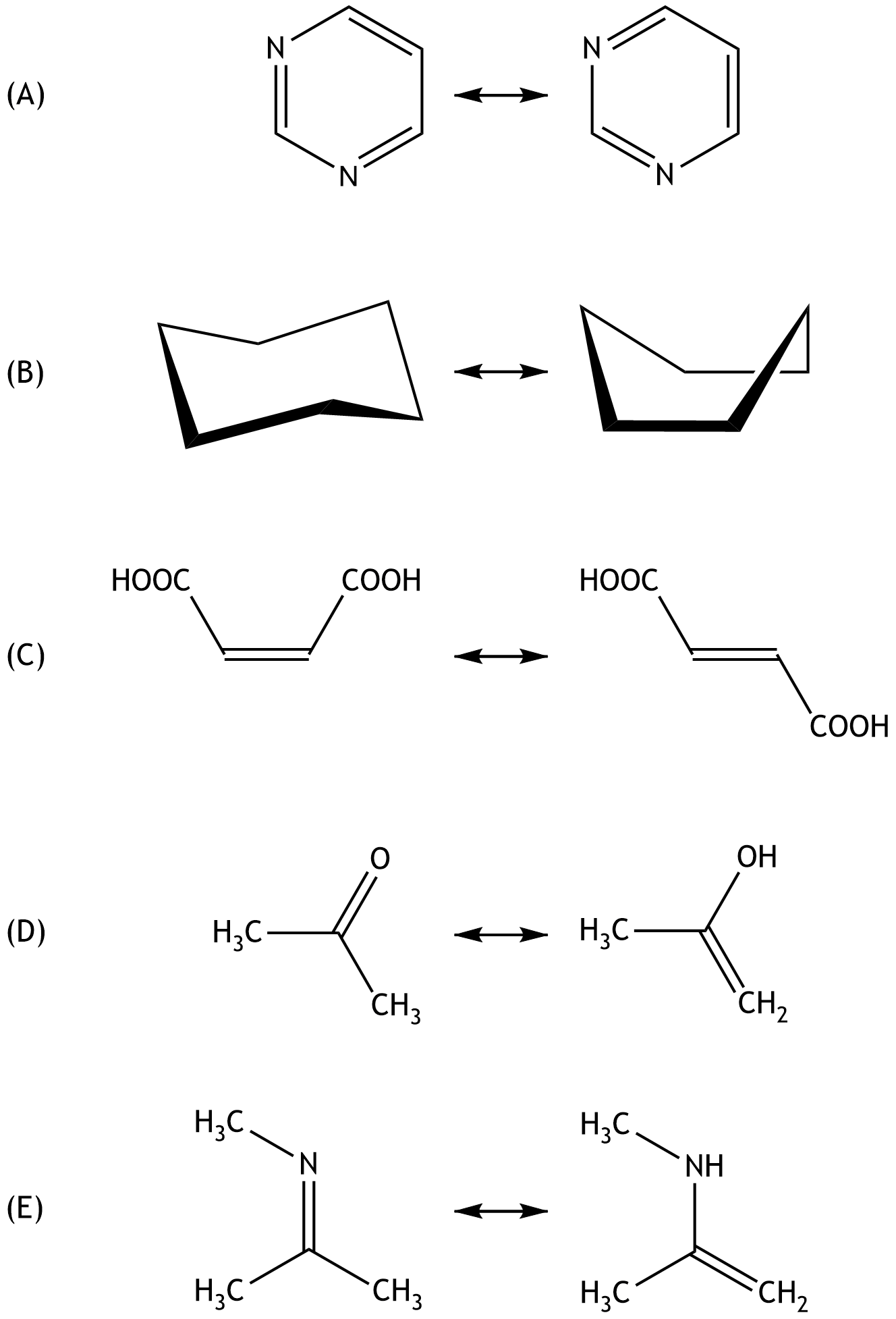
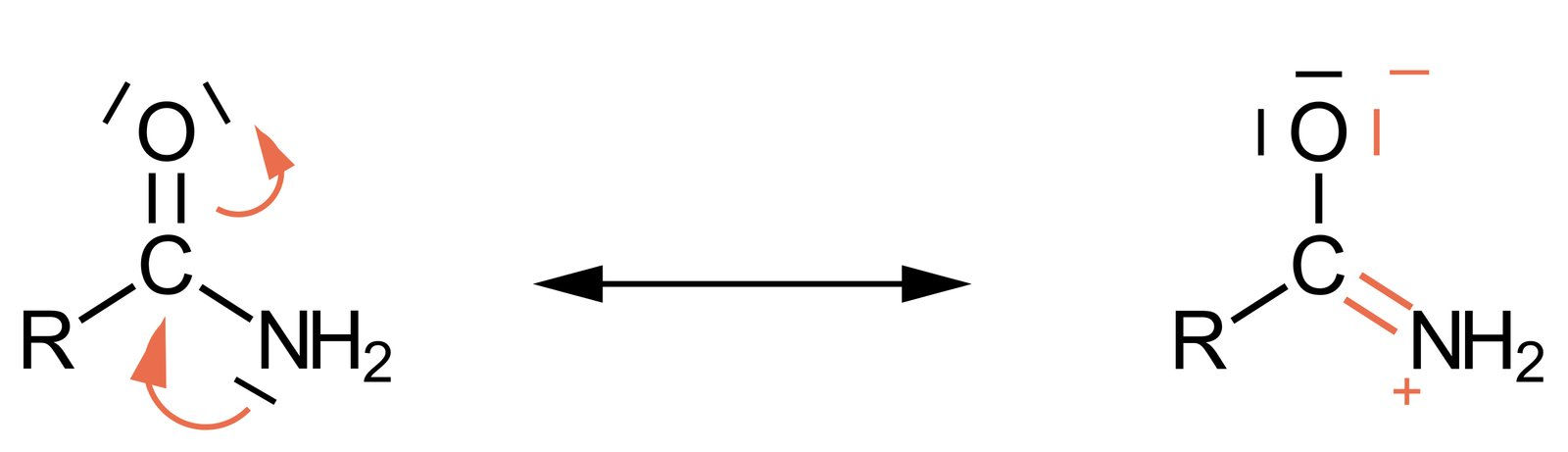
58. Which of the answer choices (A) to (E) represents mesomerism (resonance)?
A. (A)
B. (B)
C. (C)
D. (D)
E. (E)
A. (A)
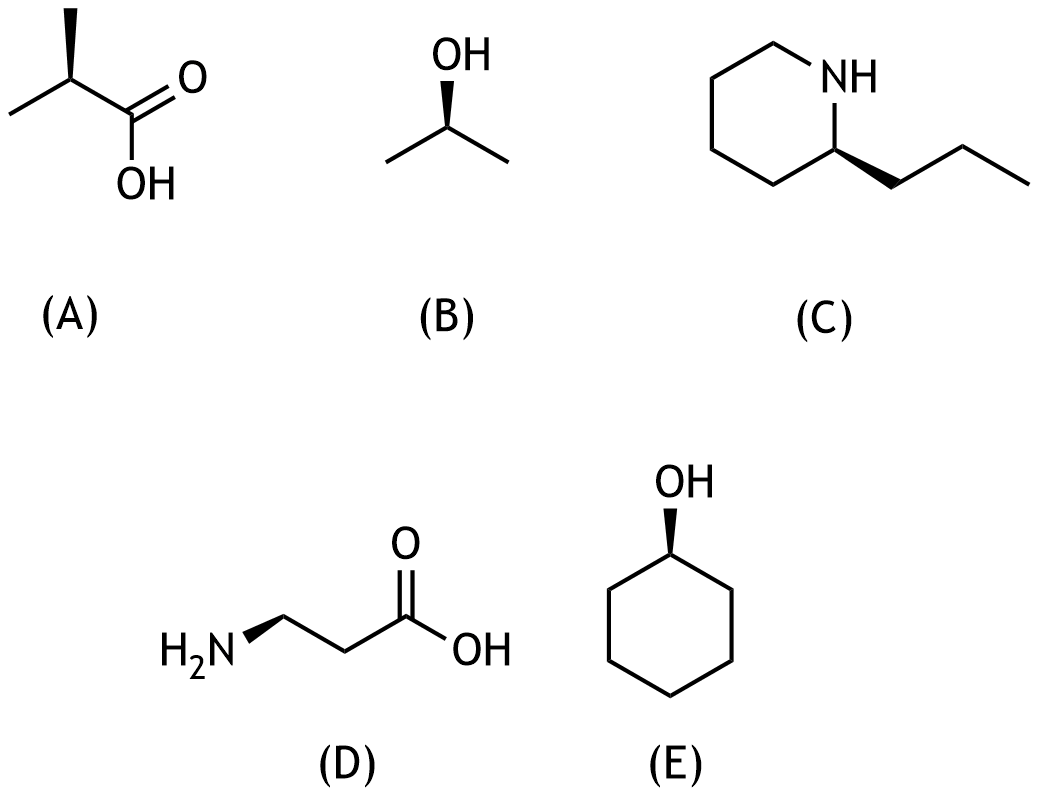
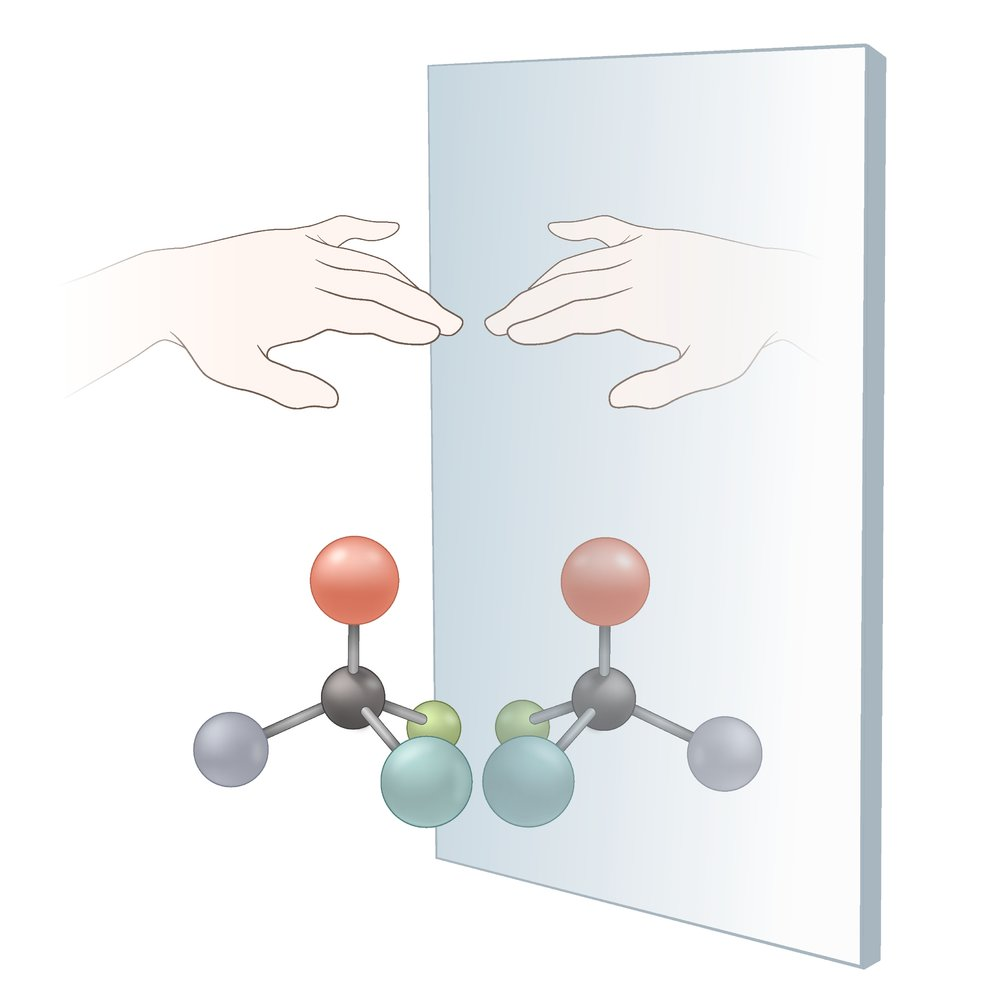
59. Which of the following chemical compounds is optically active?
A. (A)
B. (B)
C. (C)
D. (D)
E. (E)
C. (C)
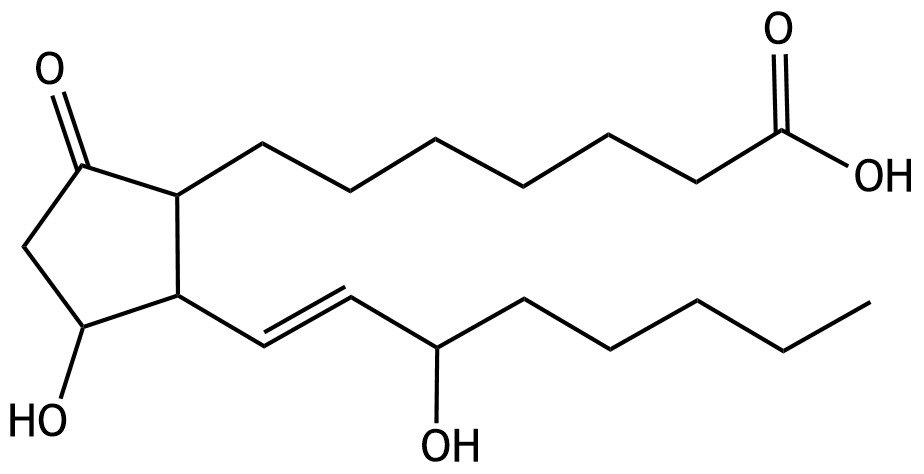
60. Prostaglandin E₁ (PGE₁) has the following chemical structure: How many chiral centers does PGE₁ contain?
A. 3
B. 4
C. 5
D. 6
E. 7
B. 4
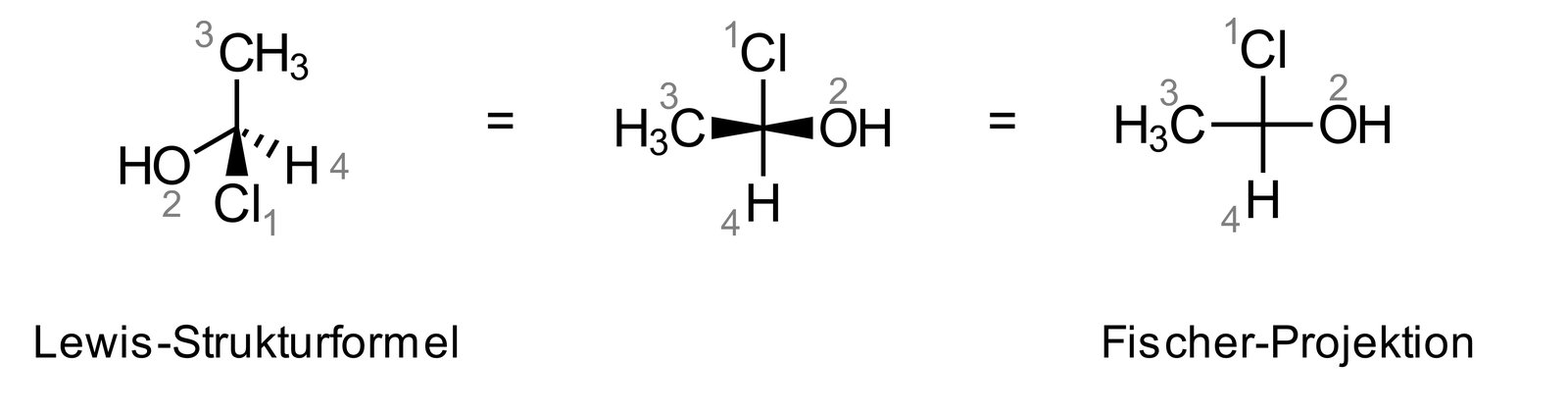
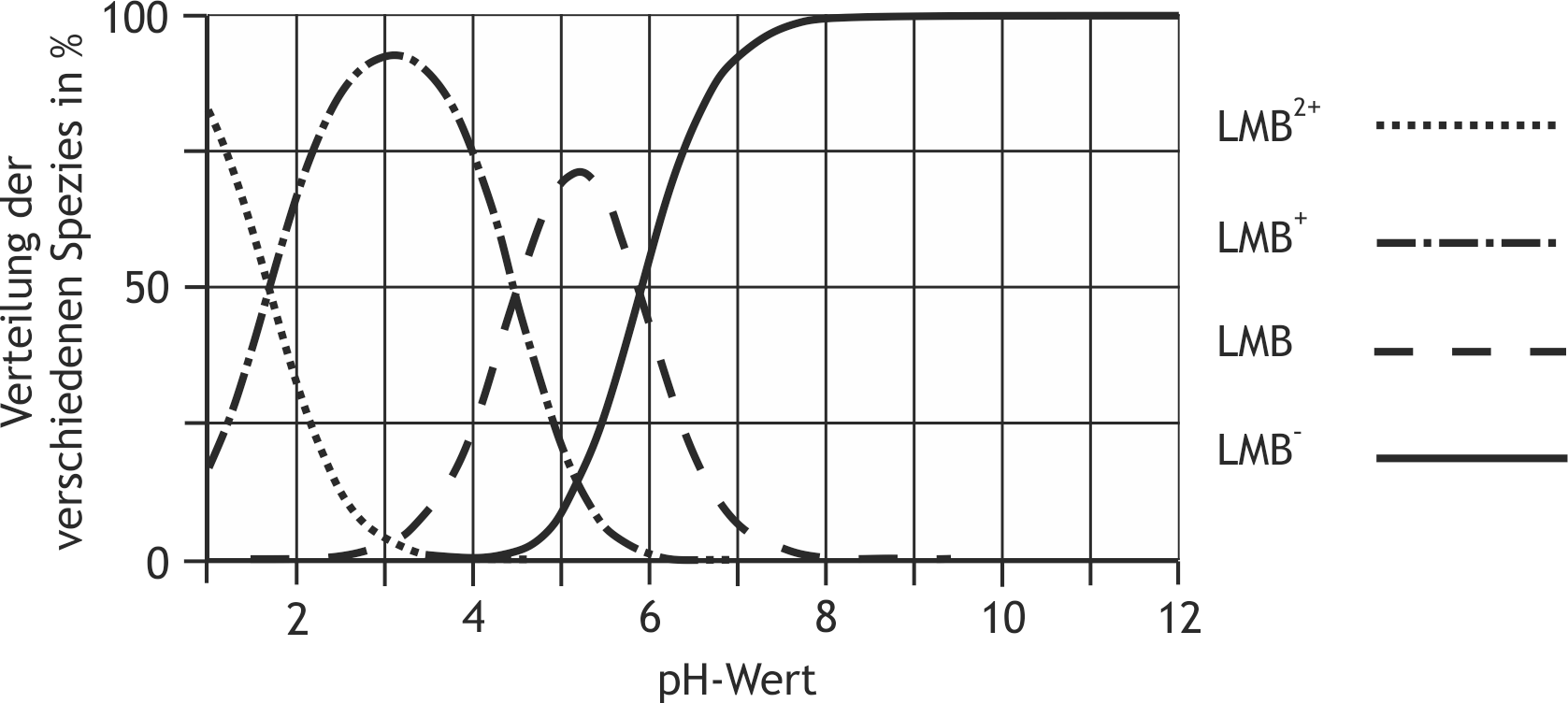
61. Leucomethylene blue (LMB) converts methemoglobin to hemoglobin and therefore acts as an antidote in nitrite poisoning. For the absorption and elimination of drugs, the charge state is often important, and its pH dependence can be predicted using the isoelectric point.
The following figure shows the pH-dependent distribution of the various LMB species that differ in their degree of protonation: What is the isoelectric point of leucomethylene blue?
A. 1.7
B. 3.3
C. 4.5
D. 5.2
E. 6.1
D. 5.2
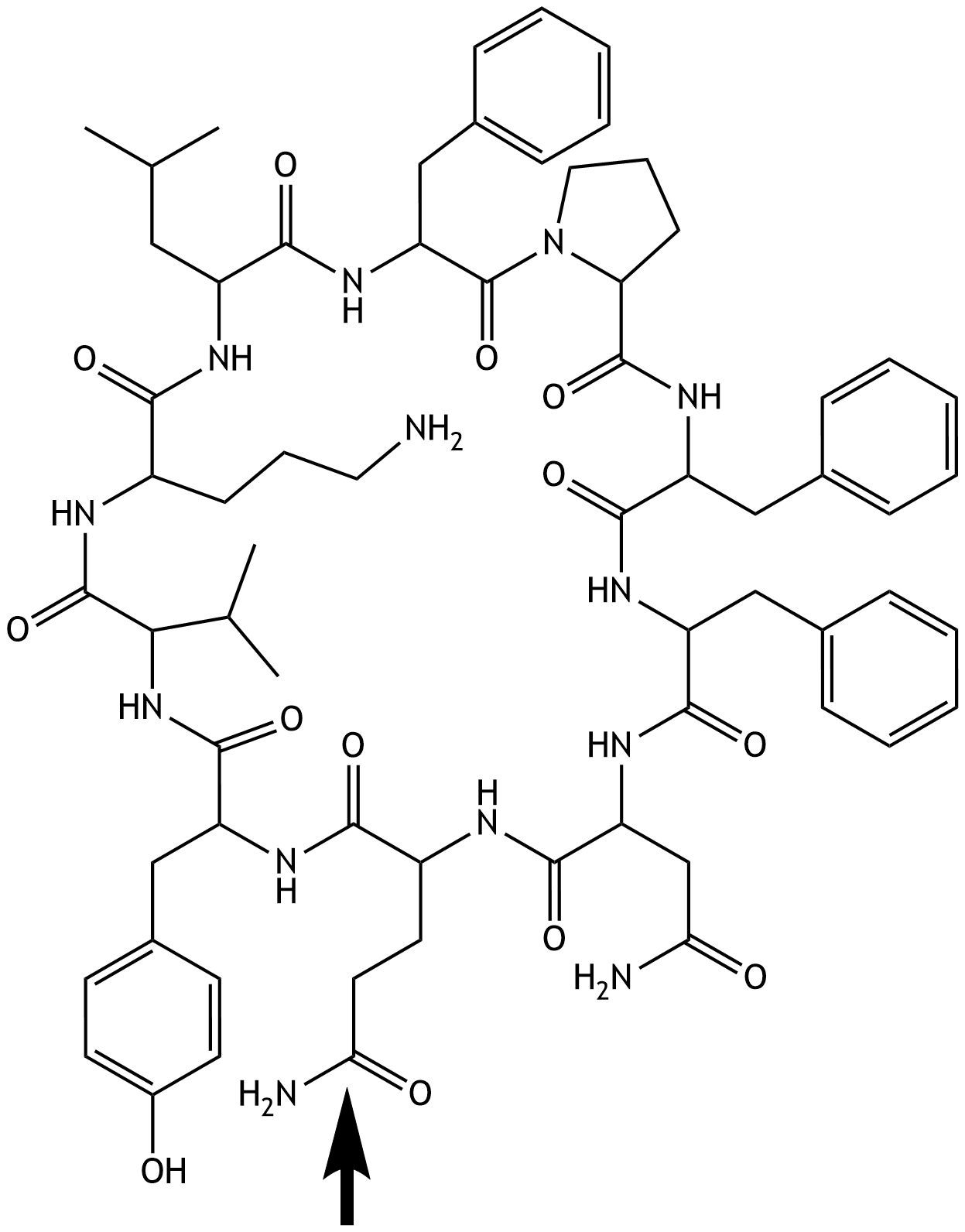
62. The structural formula of a cyclic polypeptide, the antibacterial agent tyrocidine A (found, for example, in lozenges used to treat inflammation of the pharyngeal mucosa), is shown. The residue of which amino acid is marked with an arrow in the depicted formula of tyrocidine A?
A. Arginine
B. Asparagine
C. Aspartate
D. Glutamate
E. Glutamine
E. Glutamine
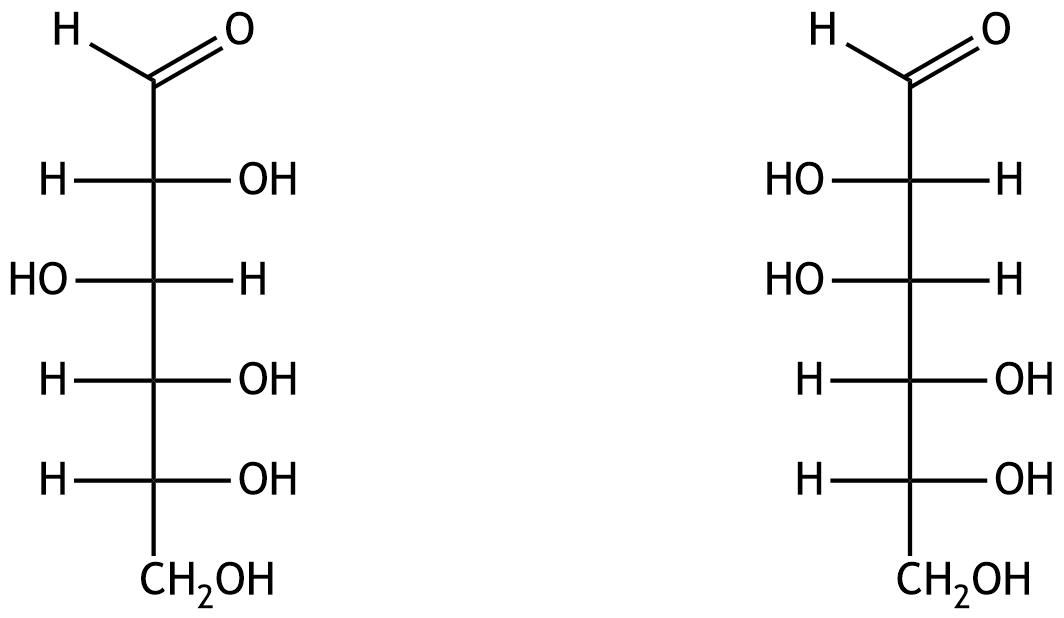
63. In what isomeric relationship do the two aldohexoses shown in the Fischer projection stand to each other?
A. Anomers
B. Enantiomers
C. Epimers
D. Conformers
E. Constitutional isomers
C. Epimers
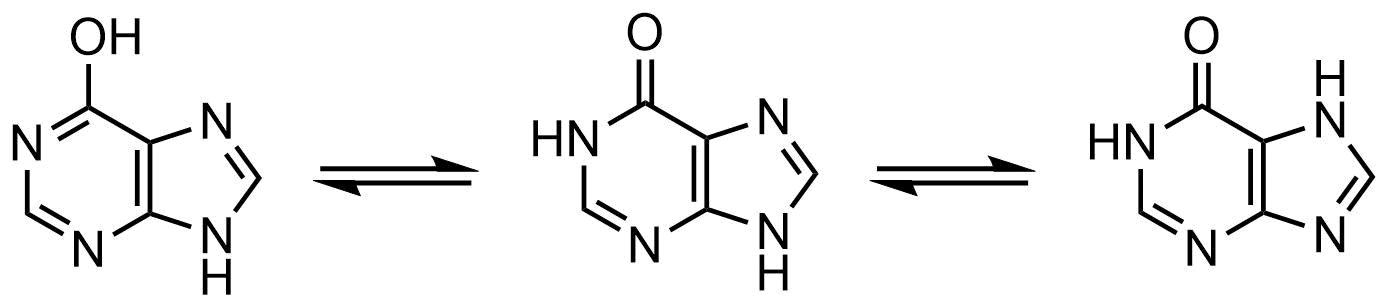
64. The naturally occurring purine derivative hypoxanthine exists in several isomeric forms that are in equilibrium with one another.
What is the relationship between these isomers?
A. Anomers
B. Enantiomers
C. Epimers
D. Conformers
E. Constitutional isomers
E. Constitutional isomers
65. Which of the following statements about the properties of the elements in the periodic table is most accurate?
A. Metallic character increases from bottom to top.
B. Atomic radius increases from left to right.
C. Ionization energy increases from bottom to top.
D. Ionic radius increases from left to right.
E. Electronegativity decreases from bottom to top.
C. Ionization energy increases from bottom to top.
66. Which common property do the elements radon (Rn), neon (Ne), argon (Ar), and xenon (Xe) most likely share?
A. They are very reactive.
B. They exist as gases at room temperature and standard pressure.
C. They have 5 valence electrons.
D. They form ions with an oxidation number of –2.
E. They belong to group 7 of the periodic table.
B. They exist as gases at room temperature and standard pressure.
67. According to the valence shell electron pair repulsion (VSEPR) model, molecules adopt a specific spatial structure due to repulsion between electron pairs.
Which molecular geometry does NH₃ most likely have according to this model?
A. Trigonal pyramidal
B. Linear
C. Bent
D. Trigonal planar
E. Tetrahedral
A. Trigonal pyramidal
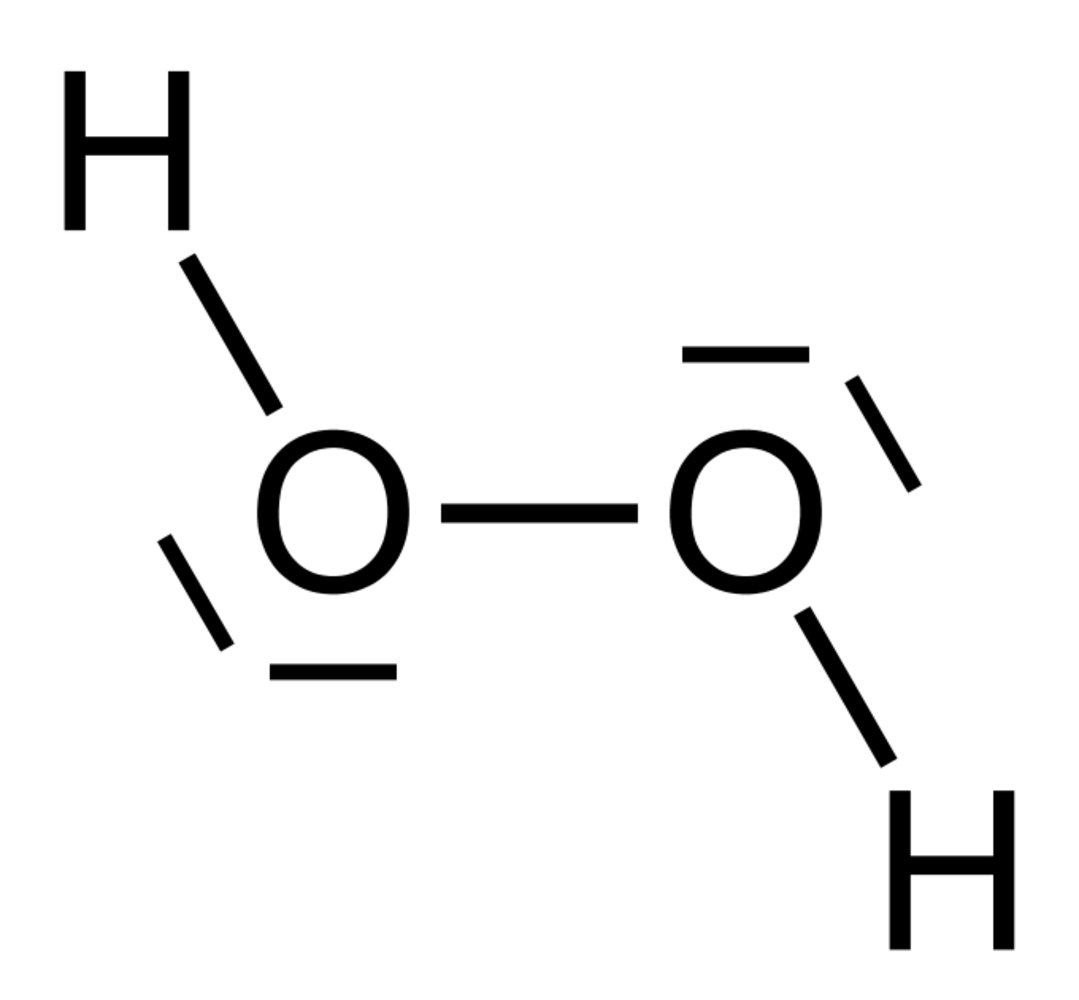
68. During the breakdown of fatty acids within peroxisomes, hydrogen peroxide is produced, which is then converted by catalase into its constituent elements, hydrogen and oxygen.
What are the most likely valences of the atoms in the hydrogen peroxide (H₂O₂) molecule?
A. H: 2; O: 1
B. H: 2; O: 2
C. H: 1; O: 3
D. H: 3; O: 1
E. H: 1; O: 2
E. H: 1; O: 2
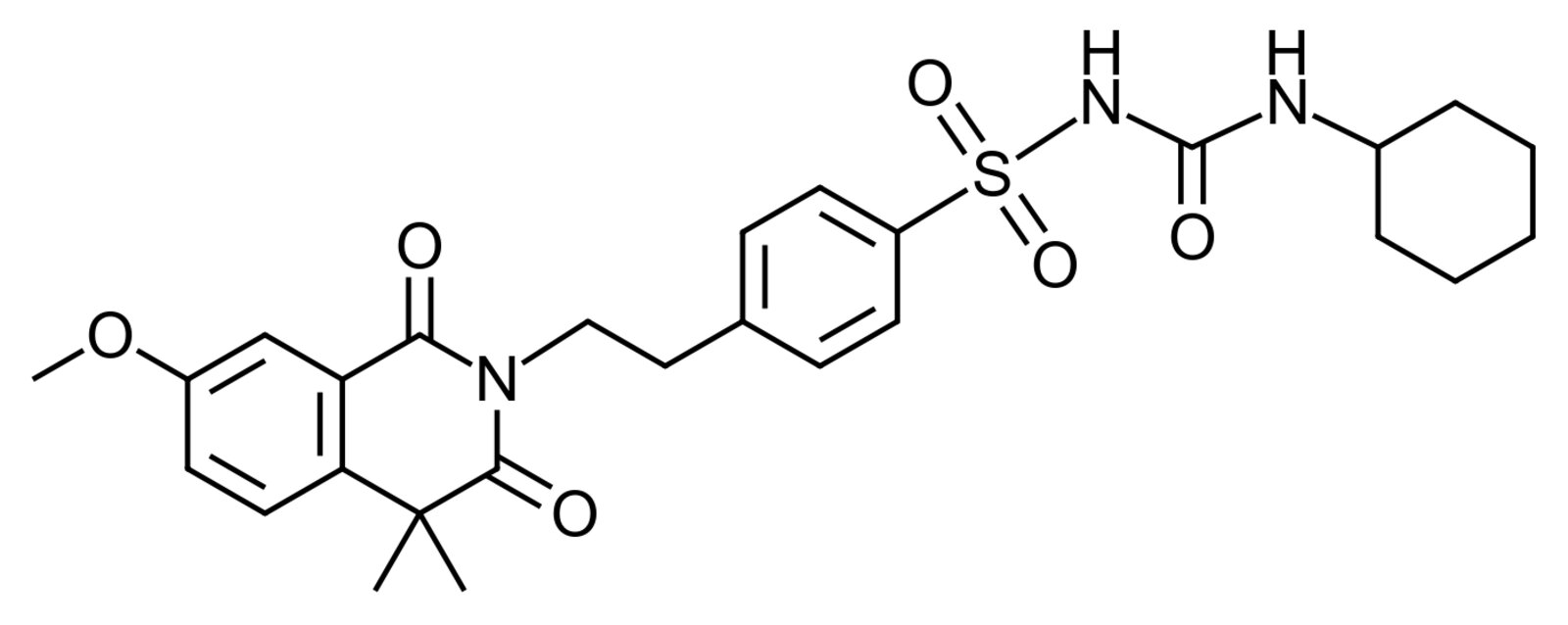
69. The structural formula of the oral antidiabetic drug gliquidone is shown. Which functional group is essential for the pharmacological activity of the molecule?
A. Carboxylic acid ester
B. Carboxylic acid amide
C. Carboxylic acid anhydride
D. Sulfonamide
E. Phosphoric acid anhydride
D. Sulfonamide
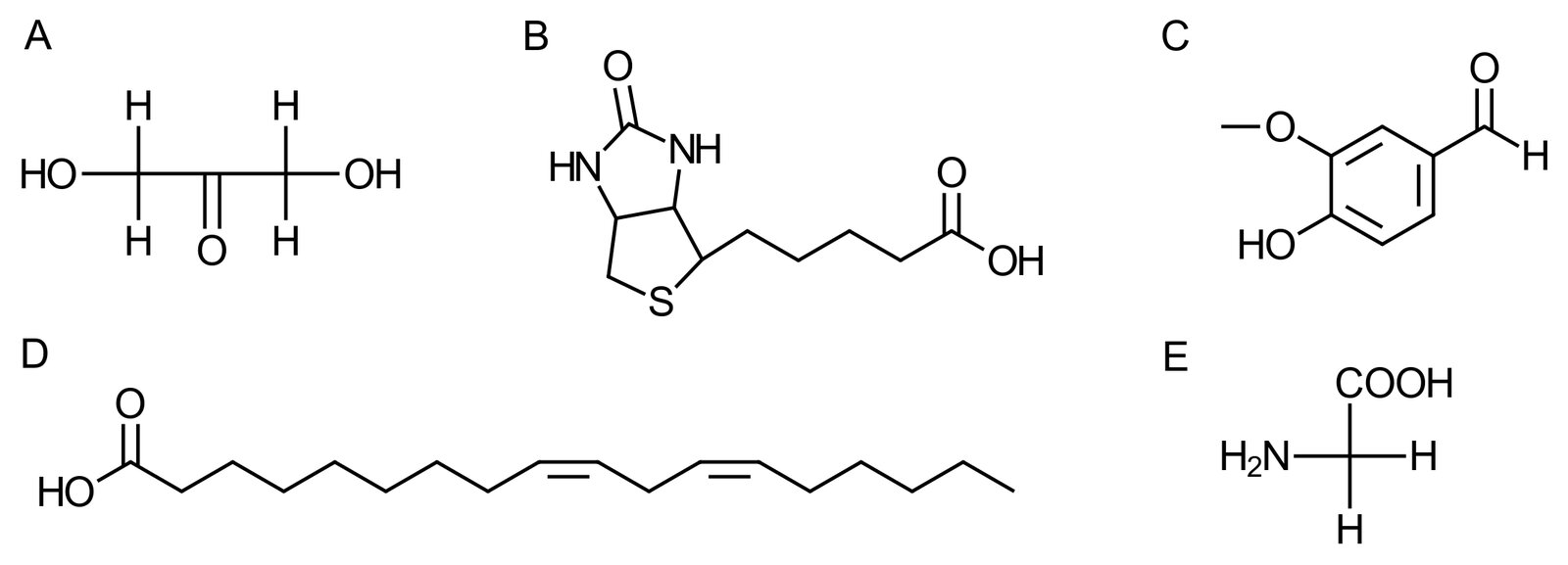
70. Which of the molecules shown in the figure has a chiral center and is therefore an enantiomer?
A. A
B. B
C. C
D. D
E. E
B. B
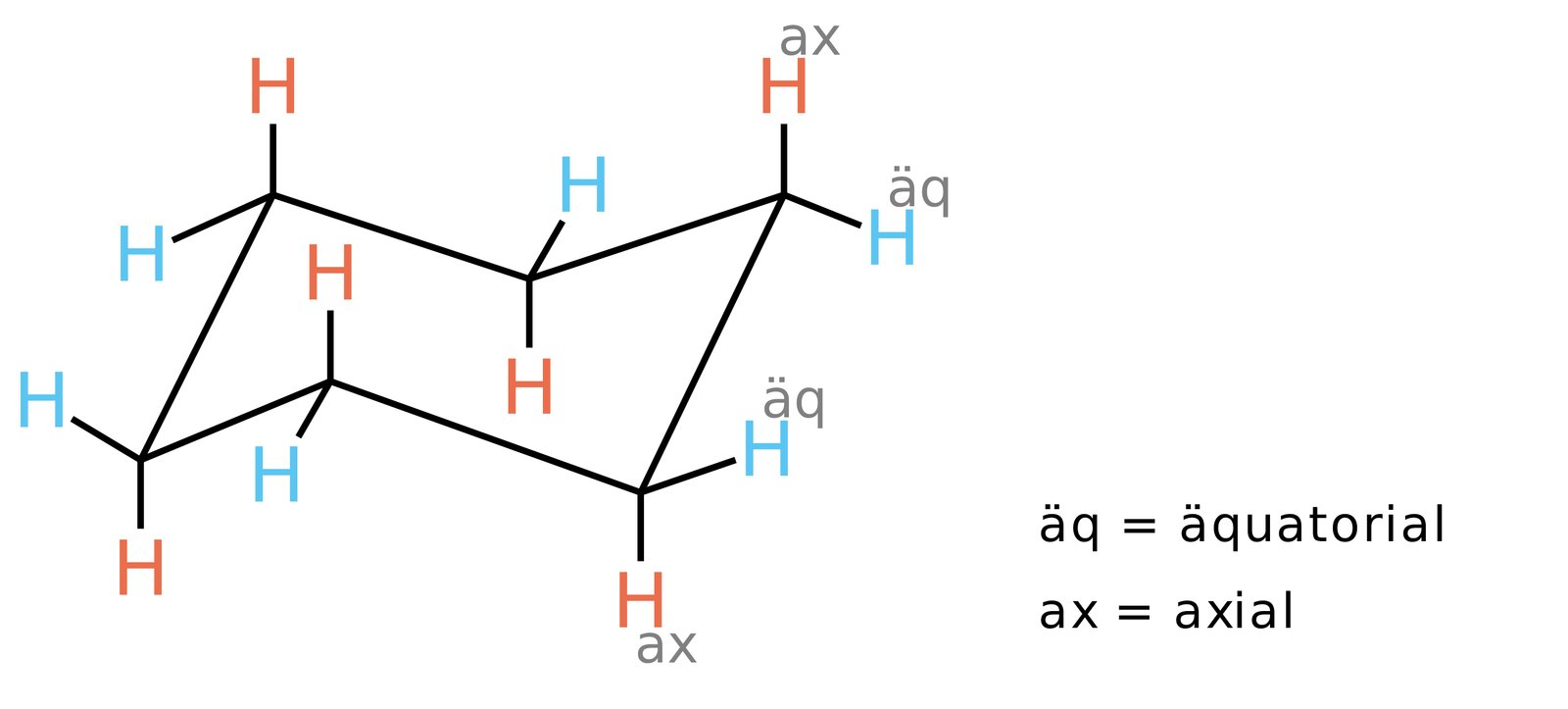
71. Cyclohexane is a component of the antifibrinolytic agent tranexamic acid, which is regularly used for bleeding prophylaxis.
Which of the following statements about the cyclic hydrocarbon cyclohexane is correct?
A. It is a polar molecule.
B. The chair conformation is its most stable spatial form.
C. Its molecular formula is C₆H₁₄.
D. It is gaseous at room temperature.
E. It is difficult to ignite.
B. The chair conformation is its most stable spatial form.
72. The neurotransmitter serotonin and its derivative, the hormone melatonin, contain an indole ring.
Which amino acid provides this heterocycle for the biosynthesis of these signaling molecules?
A. Proline
B. Threonine
C. Tyrosine
D. Histidine
E. Tryptophan
E. Tryptophan
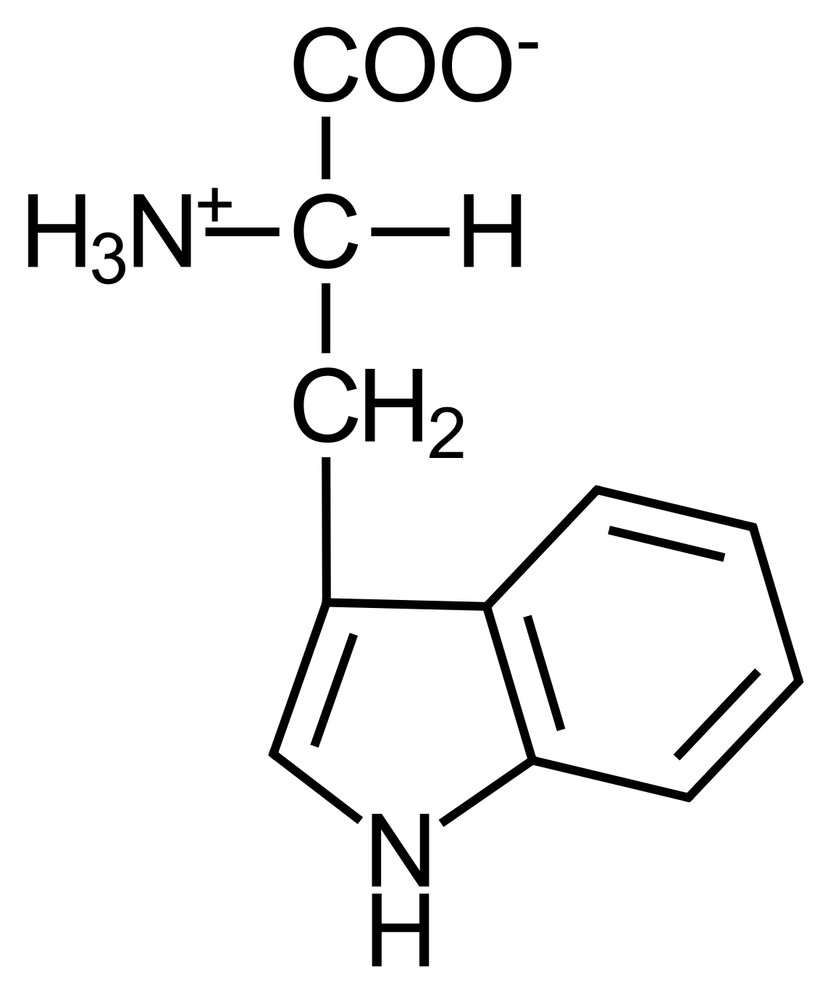
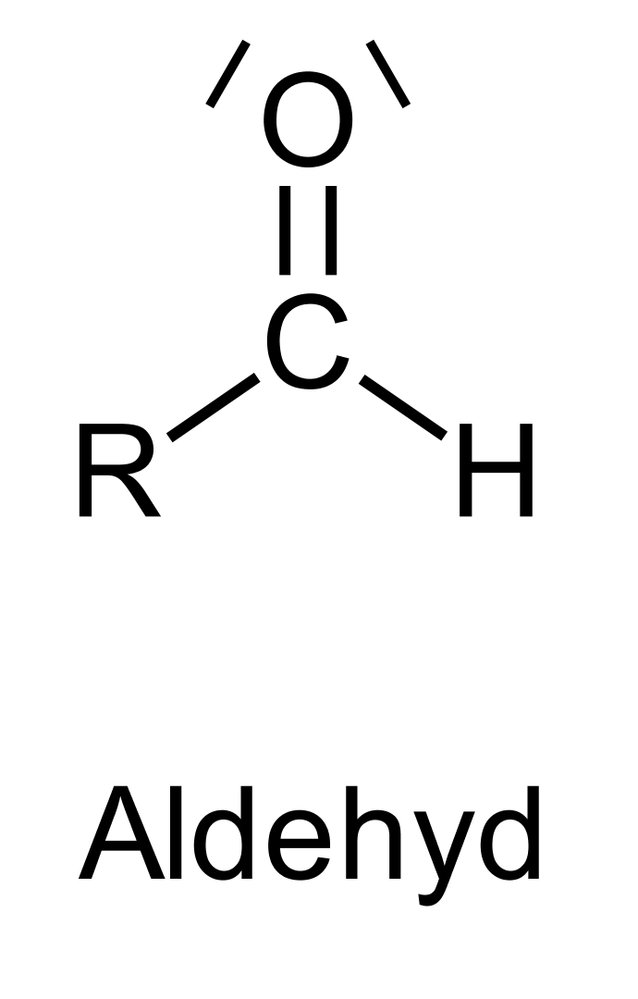
73. The functional groups of hydrocarbons are essential for their classification into compound classes.
Which functional group is characteristic of the class of aldehydes?
A. R–OH
B. R–COOR′
C. R–CO–R′
D. R–CHO
E. R–COOH
D. R–CHO
74. What is formed when the carbonyl group of a ketone reacts with an alcohol?
A. Hemiketal
B. Aldol
C. Ketal
D. Enol
E. Hemiacetal
A. Hemiketal
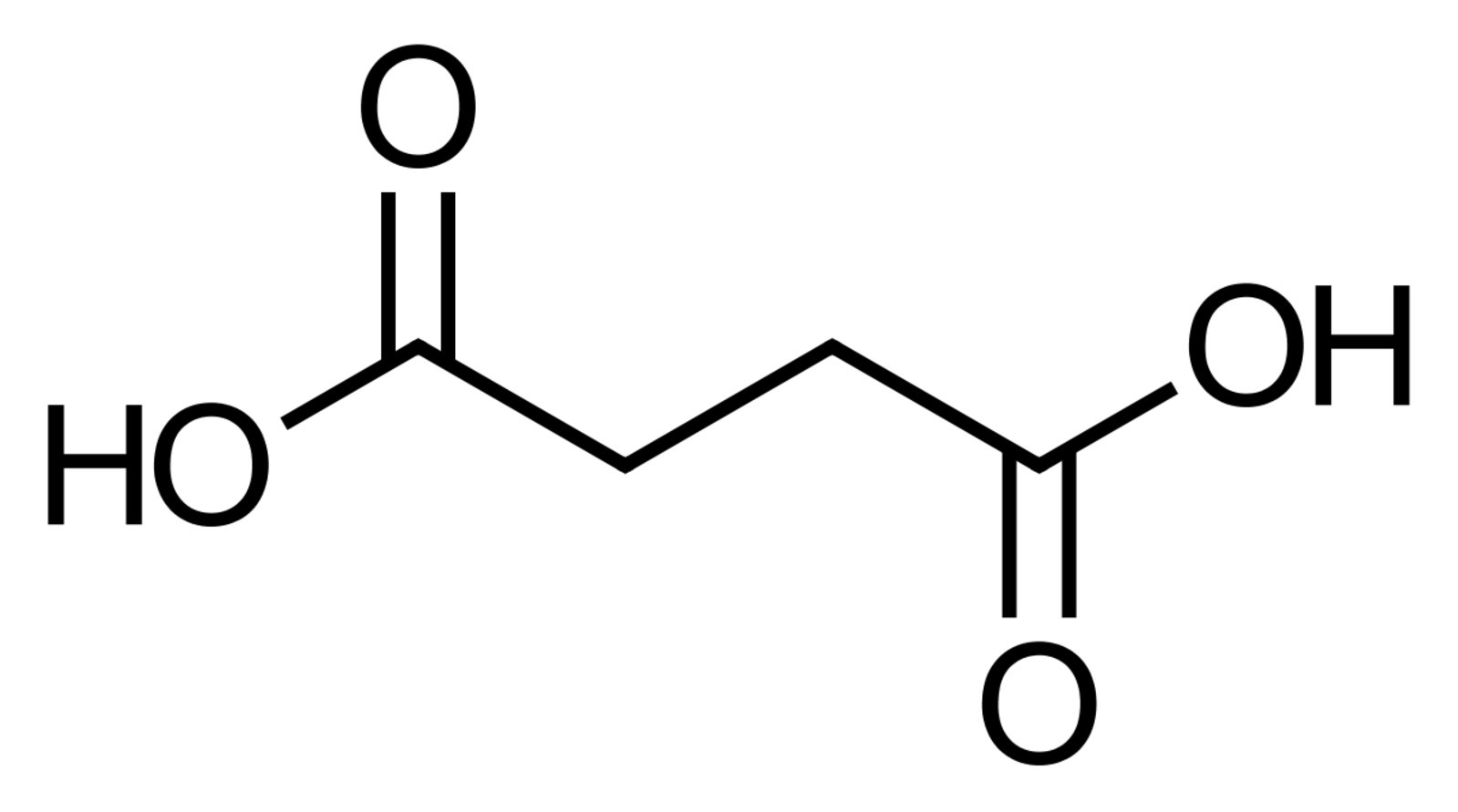
75. Succinic acid and its derivatives play a central role in cellular metabolism, for example in the citric acid cycle.
Which of the following classes of hydrocarbon compounds does succinic acid most likely belong to?
A. Monocarboxylic acids
B. Esters
C. Dicarboxylic acids
D. Carboxylic acid anhydrides
E. Carboxylic acid amides
C. Dicarboxylic acids
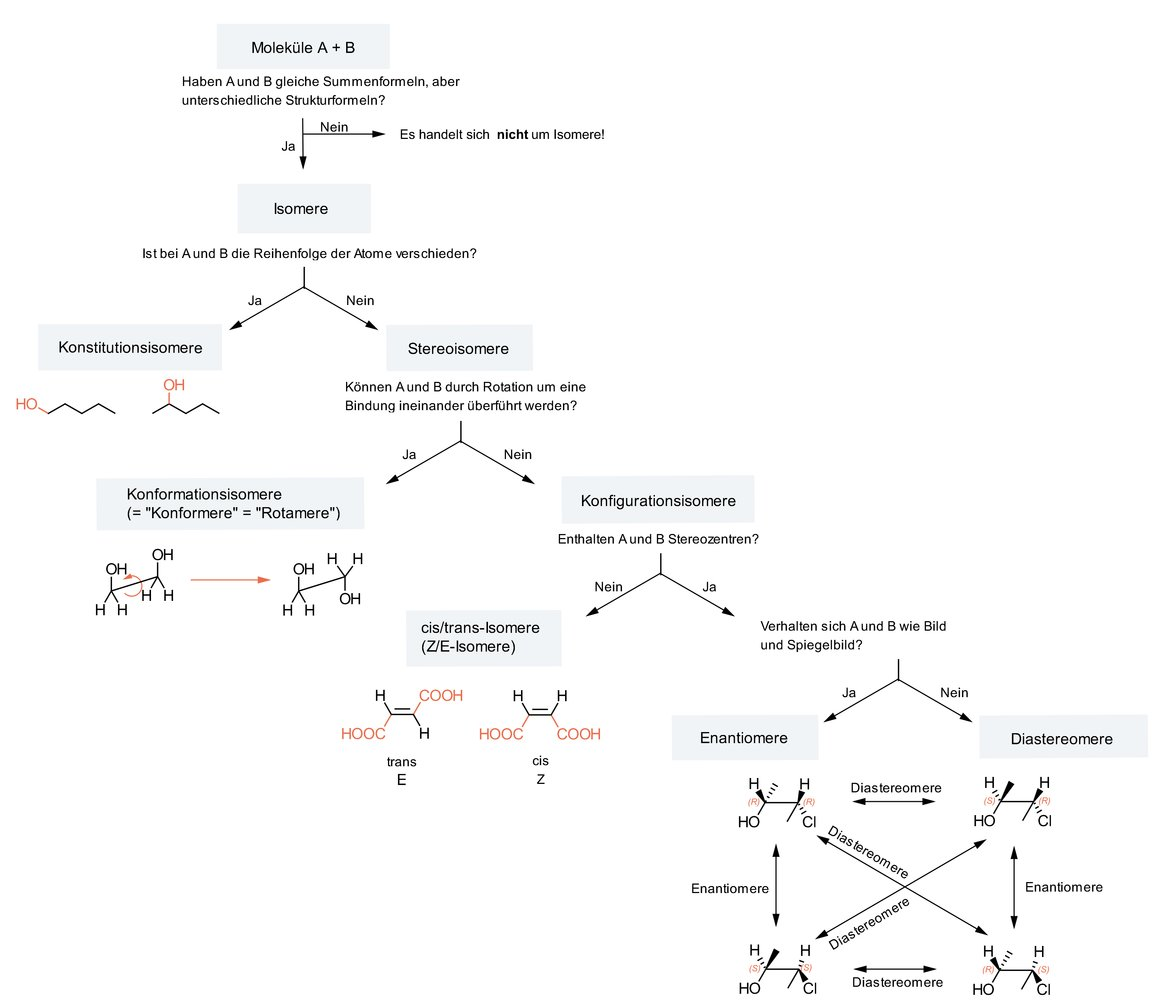
76. Which of the following statements about cis/trans isomers is correct?
A. They belong to the group of conformational isomers.
B. They are characterized by having multiple chiral centers.
C. They can be interconverted by rotation around a C–C double bond.
D. In the cis isomer, the substituents are located on the same side.
E. They are related as object and mirror image.
D. In the cis isomer, the substituents are located on the same side.
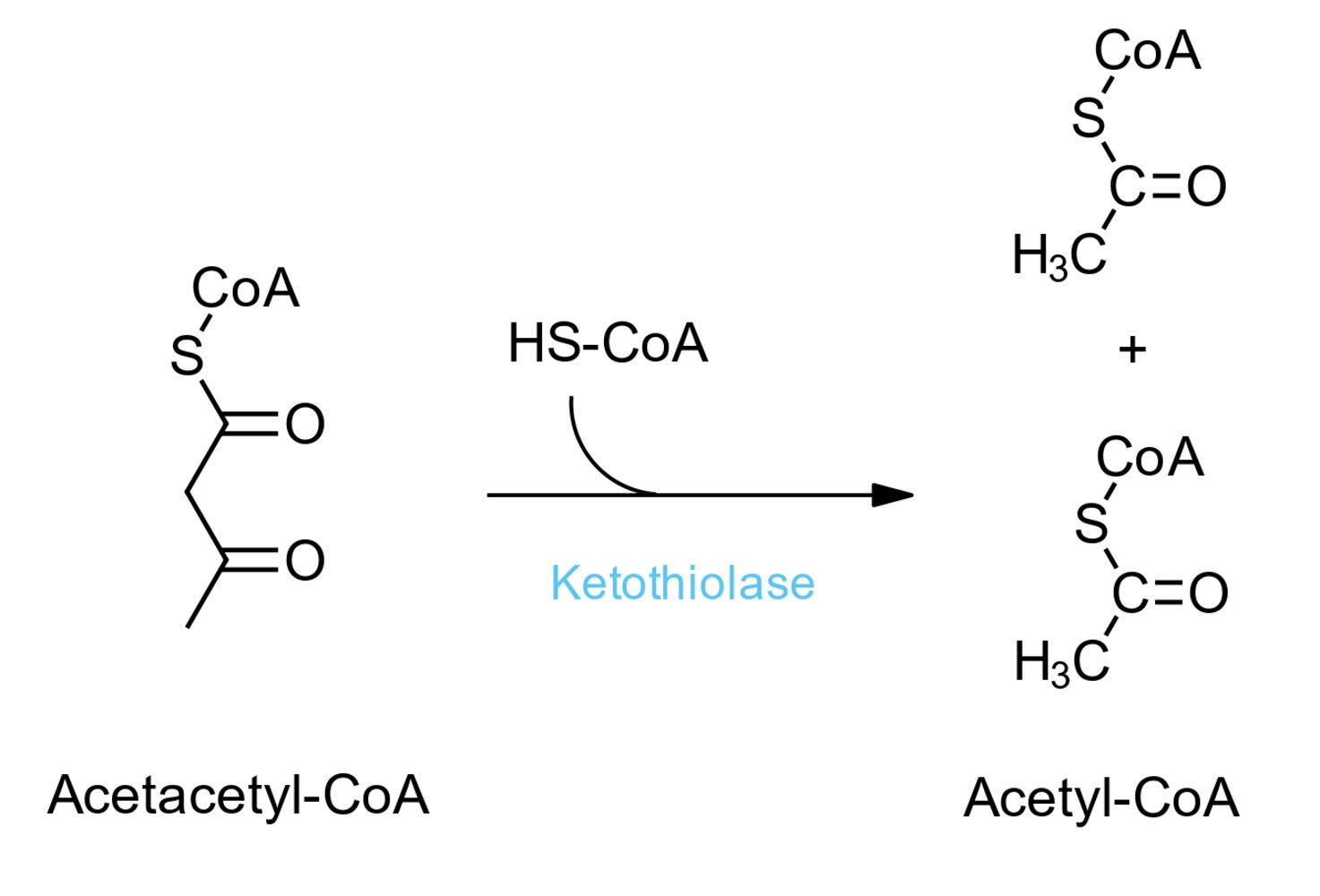
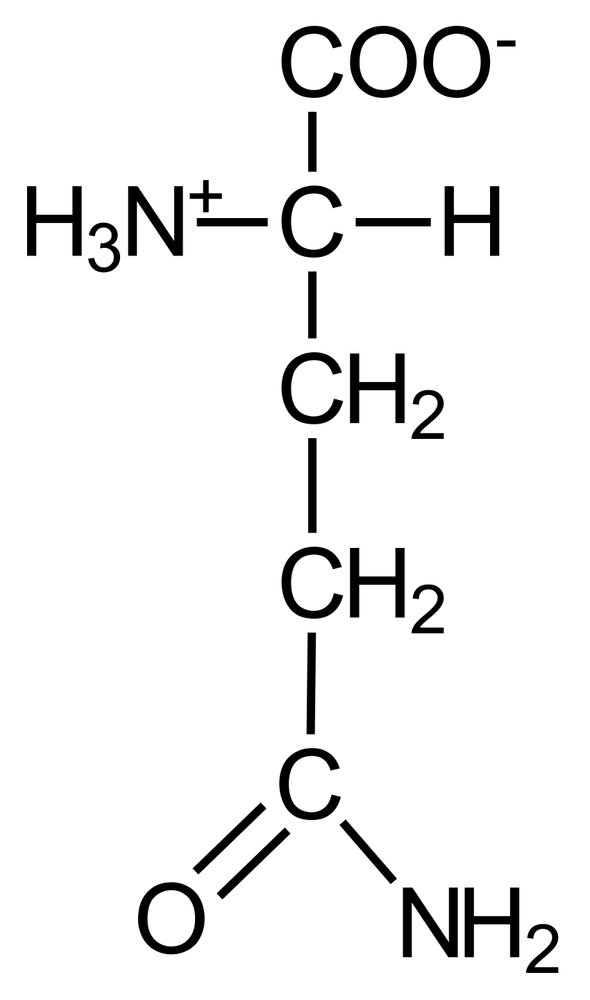
77. As part of ketone body degradation, thiolase catalyzes the reaction shown. Assume the following concentrations are measured:
Acetoacetyl-CoA: 0.01 mM
HS-CoA: 0.05 mM
Acetyl-CoA: 0.5 mM
What is the equilibrium constant for this reaction?
A. 0.0005
B. 0.002
C. 500
D. 1,000
E. 2,000
C. 500
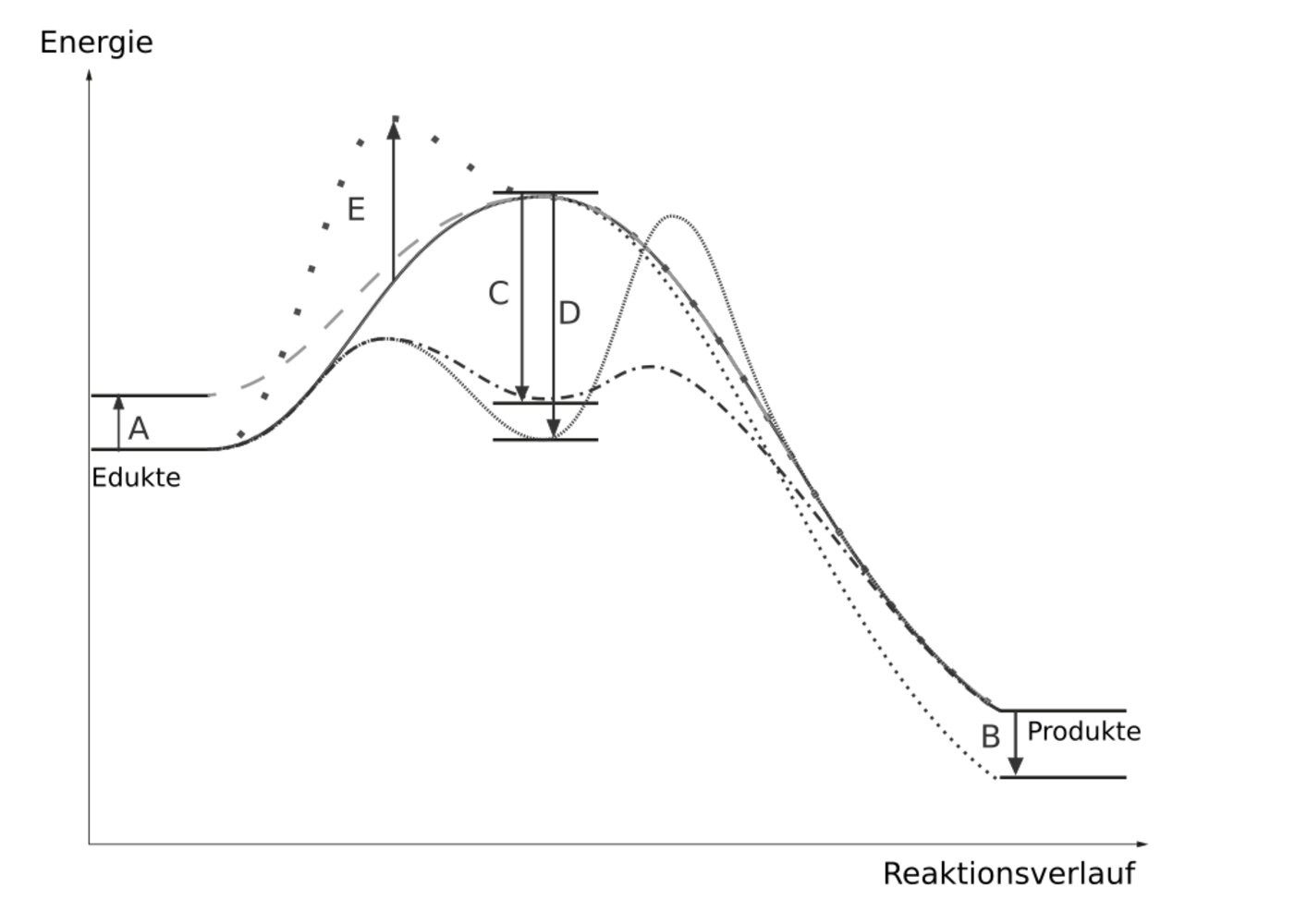
78. Which of the changes shown here best describes the mode of action of the most effective catalyst?
A. (A)
B. (B)
C. (C)
D. (D)
E. (E)
C. (C)