Addition reactions of alkenes (3.3.4.2)
1/6
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
7 Terms
Why are alkenes susceptible to attack by electrophiles?
The carbon-carbon double bond has a high electron density.
This causes non-polar molecules to experience an induced dipole. The δ+ atom then accepts a pair of electrons and acts as the electrophile
In polar molecules, there is a permanent dipole, so the electropositive atom accepts a pair of electrons and acts as the electrophile
Electrophilic addition is the addition of an electrophile to a double bond
What is the reaction between alkenes and hydrogen and what are the conditions?
Hydrogenation:
Alkene + hydrogen → alkane
Conditions:
Nickel catalyst
High temperature
What is the test for unsaturation using bromine water and what is the mechanism for this reaction?
Test for unsaturated compound:
Bubble compound into bromine water (Br2(aq))
Unsaturated compound present if solution turns from orange → colourless
Reaction:
Alkene + halogen → haloalkane
Mechanism:
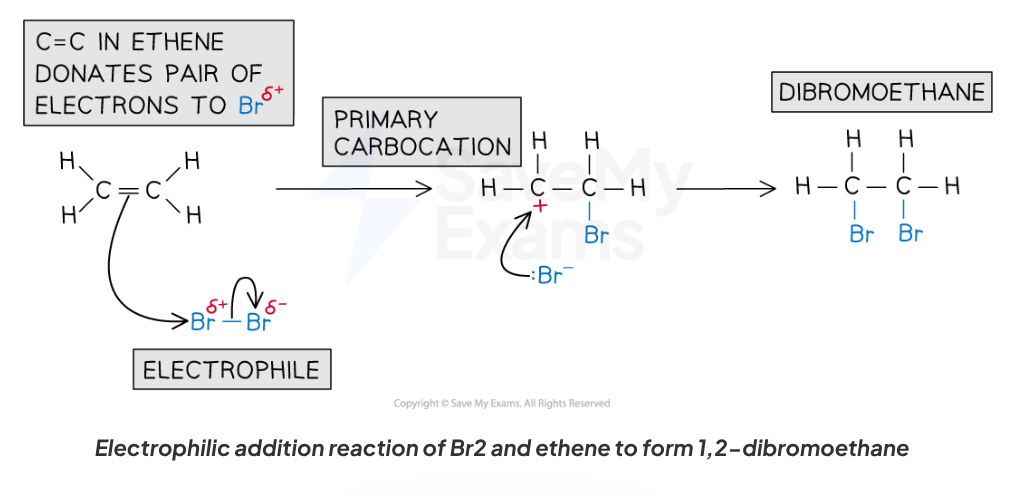
UV light isn’t needed
What is the mechanism of the reaction between alkenes and water and what are the conditions?
Hydration:
Alkene + water → alcohol
Conditions:
Strong acid catalyst (H2SO4)
High temperatures
Mechanism:
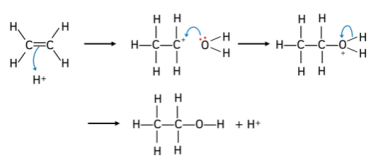
What is the mechanism of the reaction between alkenes and hydrogen halides and what are the conditions?
Reaction:
Alkene + hydrogen halide → haloalkane
Conditions:
Room temperature
Mechanism:
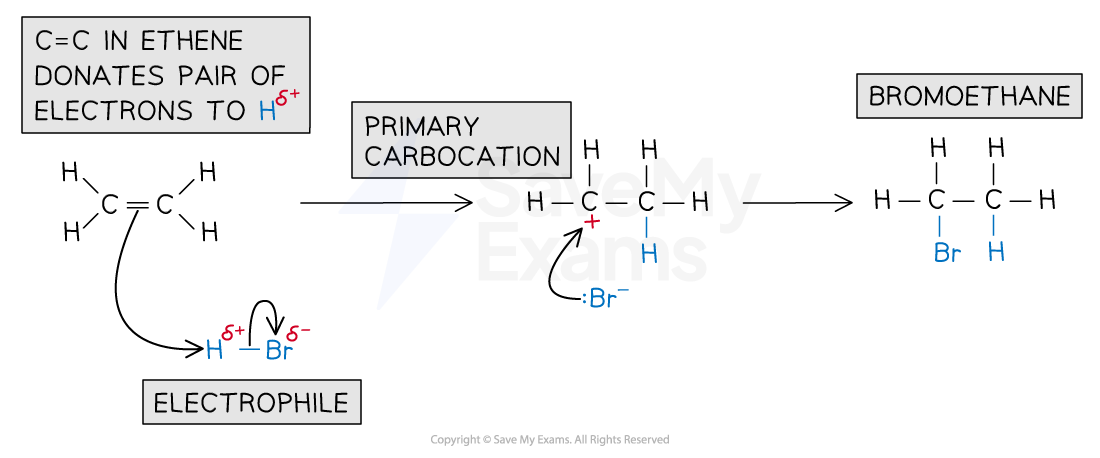
What is Markovnikov’s rule and what is the reason for it?
Markovnikov’s rule - when a hydrogen halide reacts with an asymmetric alkene, the hydrogen of the hydrogen halide bonds to the carbon of the alken with the greatest number of hydrogens and the fewest carbons bonded to it.

This is because in the intermediate step of the electrophilic addition, a more stable carbocation is formed which forms faster.
Carbocation stability:
Tertiary > secondary > primary
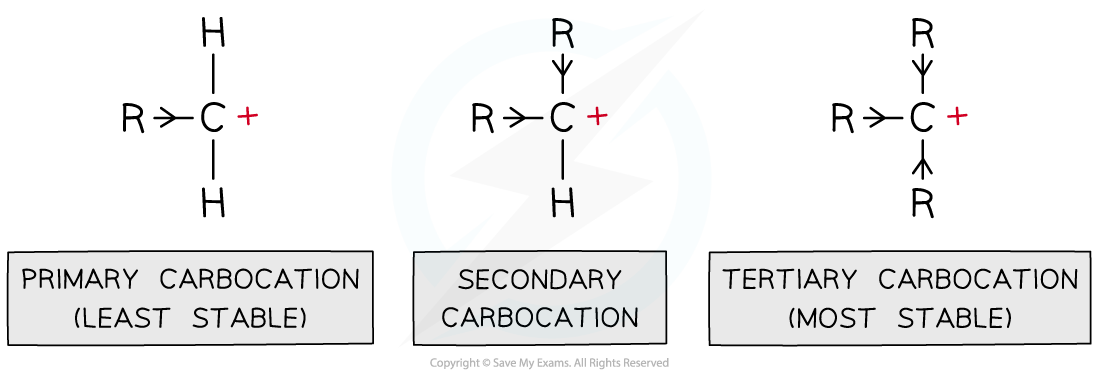
It is also because there are more electron releasing alkyl groups attached to the carbocation so there is a greater inductive effect (the alkyl groups push electrons away from themselves to the carbocation, causing it to become less positively charged so it is more energetically stable).
What is the mechanism of the reaction between alkenes and sulfuric acid and what are the conditions?
Reaction:
Alkene + sulfuric acid →alkyl hydrogensulfate
Conditions:
Cold, concentrated sulfuric acid
Mechanism:
