Chemistry | Test 1 and Exam 1 - Definitions
1/44
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
45 Terms
Element
A pure substance made of only one type of atom
Metal
An element that is usually shiny, conducts electricity, and can be shaped.
Atom
Building blocks of all matter. The smallest unit of an elements that keeps its chemical properties.
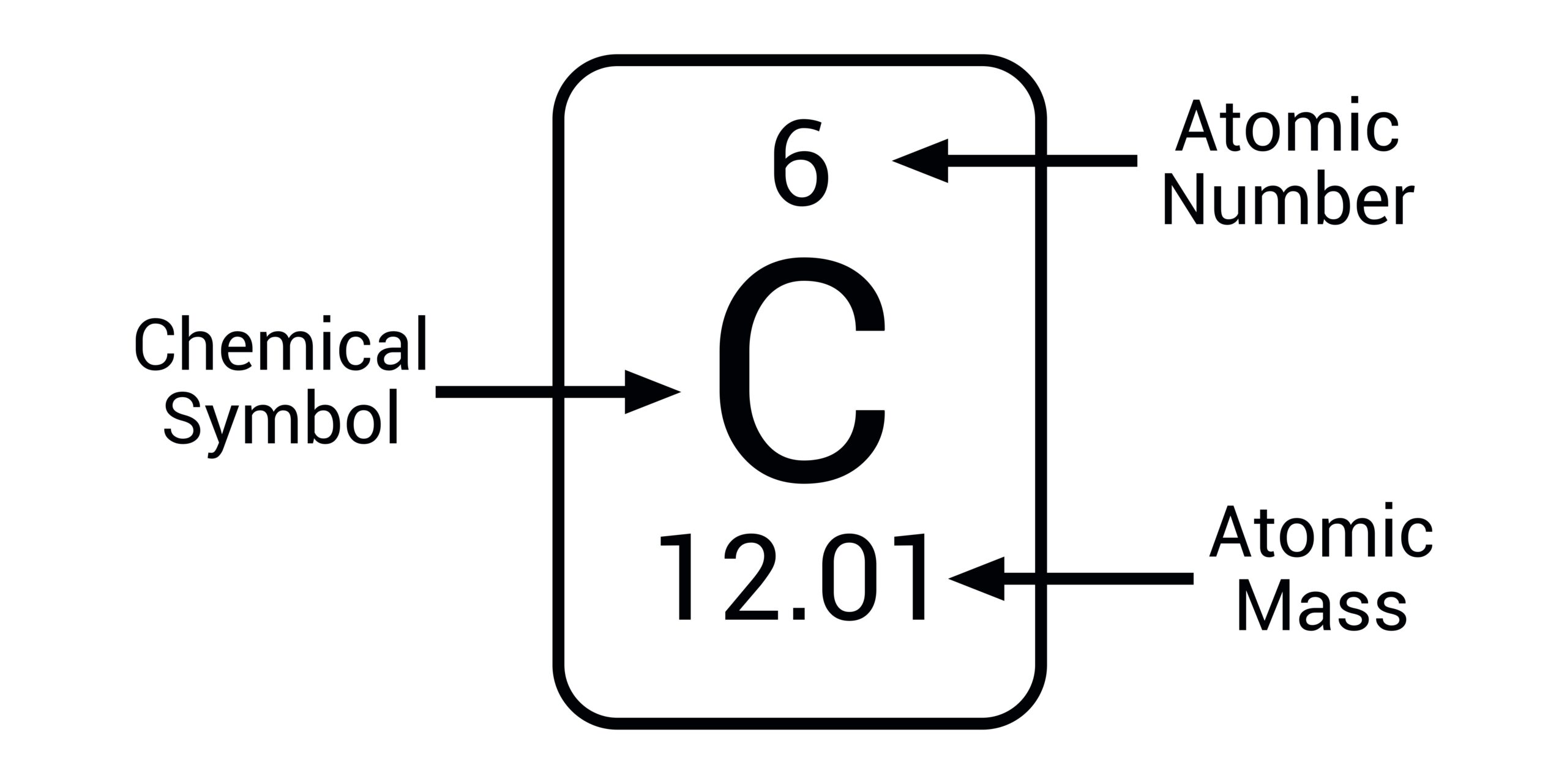
Atomic number
The number of protons in an atom: it identifies the element.
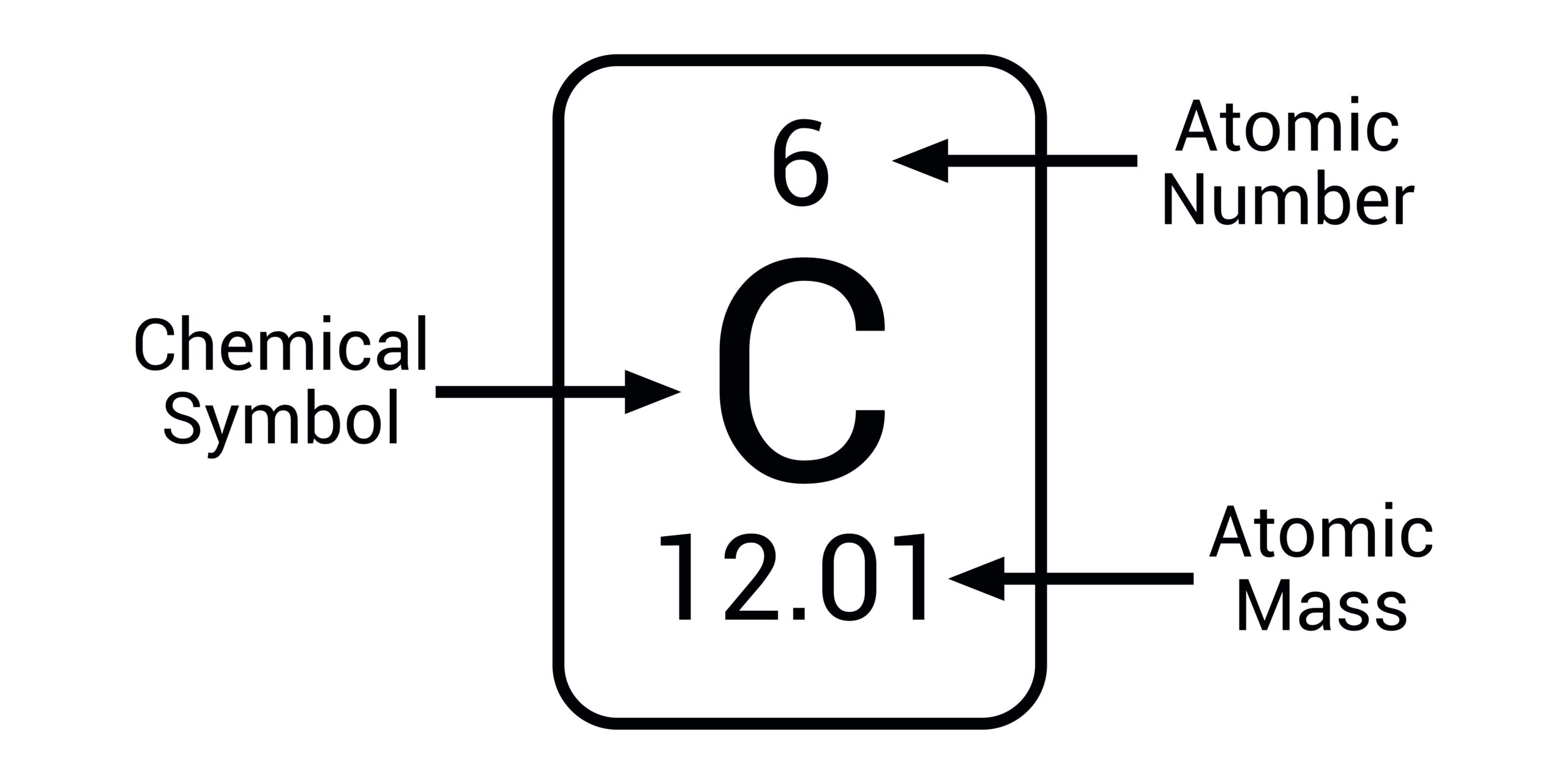
Mass number
The total number of protons and neutrons in an atom
Electron configuration
The arrangement of electrons in the shells of an atom
Electrically neutral
Having no overall charge because the number of protons equals the number of electrons
Ion
An atomic or group of atoms with an electrical charge due to gaining or losing electrons
Valency
The number of electrons an atom can gain, lose, or share when bonding to achieve a stable, full outer shell
Nucleus
The central part of an atom that contains protons and neutrons
Electron
A negatively charged particle that moves around the nucleus in shells
Proton
A positively charged particle found in the nucleus of an atom
Neutron
A particle with no charge found in the nucleus of an atom
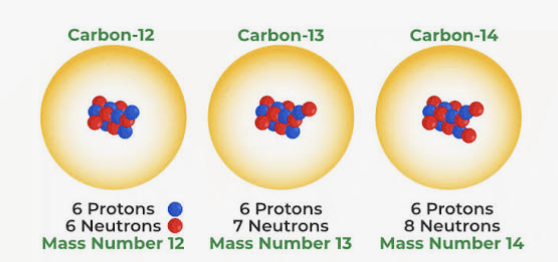
Isotopes
Atoms of the same element within the same number of protons but different number of neutrons
Period
A horizontal row in the periodic table
Group
A vertical column in the periodic table; elements in a group have similar properties
Electron shell
An energy level around the nucleus where electrons are found
Polyatomic
Made up of more than one atom
Monoatomic
Made up of a single atom
Compound
A substance formed when two or more different elements are chemically bonded
Ionic compound
A compound formed when electrons are transferred between atoms, creating ions
Covalent
A type of bonding where atoms share electrons
Molecule
two or more atoms joined together
the atoms can either be the same or different
Lattice
A repeating three-dimensional structure of ions in an ionic compound
Chemical property
A property that describes how a substance reacts to form new substances
Soluble
Able to dissolve in a particular solvent (usually water)
Insoluble
Not able to dissolve in a particular solvent
Precipitate
The insoluble solid substance that forms and separates from a liquid solution.
Precipitation reaction
A reaction where a solid precipitate forms from two aqueous solutions
pH
A scale from 0-14 that measures how acidic or basic a solution is
Acid
A substance that produces hydrogen ions (H+) in water
Base
A substance that produces hydroxide ions (OH-) in water or accepts hydrogen ions. Can neutralise an acid
Indicator
A substance that changes colour depending on the pH of the solution it is mixed with, allowing for the detection of acidic or base/alkaline conditions.
Litmus paper
An indicator that turns red in acids and blue in bases
Universal indicator
An indicator that shows a range of colours to indicate pH value - how acidic or basic a solution is on a scale from 0 to 14
Neutralisation
A chemical reaction where an acid and a base react to cancel each other out, produces salt and water
Salt
an ionic compound formed from the neutralisation reaction of an acid and a base.
Mole
A unit used to measure the amount of a substance
Molar mass
The mass of one mole of a substance, measure in g/mol
Mole ratio
The ratio of moles of substances in a balanced chemical equation
Collision theory
States that chemical reactions occur only when reactant particles (atoms, ions, or molecules) collide with sufficient activation energy to break bonds and in the correct orientation
Concentration
The amount of solute dissolved in a given volume of solution; higher concentration increases reaction rate
Surface area
The total area of exposed solid; larger surface area increases reaction rate
Temperature
A measure of how hot or cold something is; higher temperature increase reaction rate
Catalyst
A substance that speeds up the rate of a chemical reaction without being consumed in the process