OAT Boot Day 1
1/82
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
83 Terms
Definitions of Matter, Element, and Atom
Matter: Anything that takes up space and has mass
Element: A pure substance that has specific chemical and physical properties and can’t be broken down into a simpler substance
Atom: Smallest unit of matter that still retains the chemical properties of the element
Definitions of Molecules, Intramolecular Forces, Intermolecular Forces
Molecule: Two or more atoms joined together
Intramolecular forces: Attractive forces that act on atoms within a molecule
Intermolecular forces: Attractive forces that exist between molecules
Affect physical properties of the resulting substance
Definitions of Molecules, Intramolecular Forces, Intermolecular Forces
Monomers: Single molecules with the capability of polymerizing
Polymers: Substances made of many monomers linked together
Polymerization: The continuous bonding of one monomer to another, forming a polymer
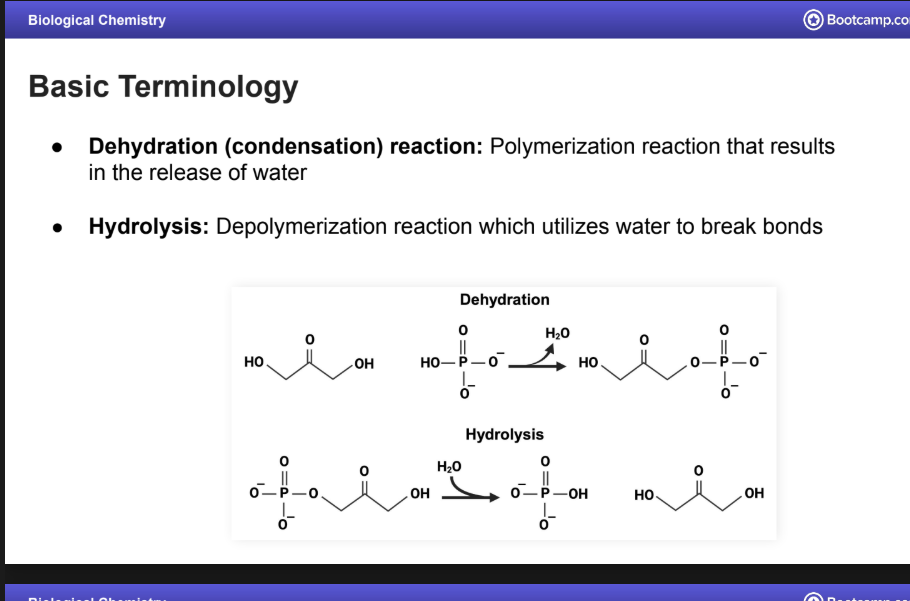
Dehydration Reaction
A.K.A a condensation reaction:
Polymerization reaction that results in the release of water
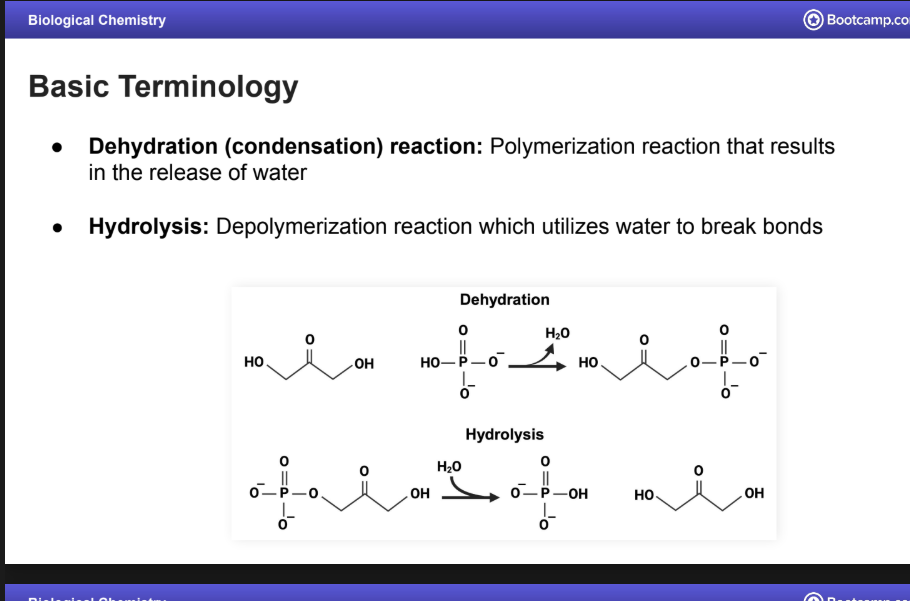
Hydrolysis:
Depolymerization reaction which utilizes water to break bonds
Carbs:
What are they made of
What are they used for functionally
● Carbohydrates: Molecules used for both fuel and structural support
● Contain: Carbon, hydrogen, and oxygen
● Types: Monosaccharides, disaccharides, polysaccharides
Monosacharides
Example
What are they
Monosaccharides: Carbohydrate monomers
● Common examples:
● Ribose: Five carbon monosaccharide
● Fructose: Six carbon monosaccharide
● Glucose: Six carbon monosaccharide
Disaccharides
Example molecule
What are they
What kind of bond do they have
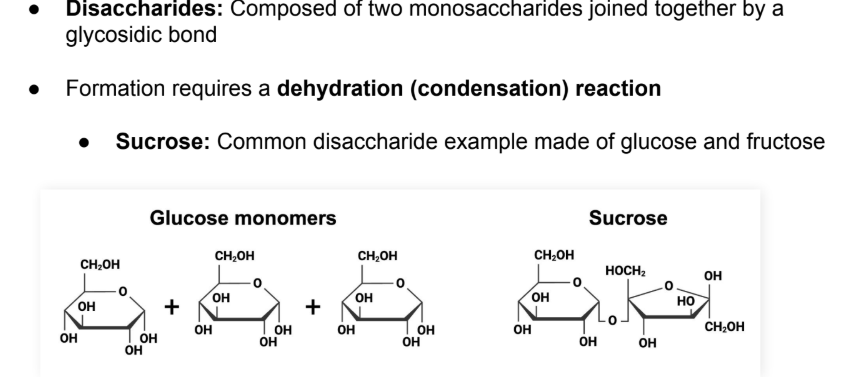
Disaccharides: Composed of two monosaccharides joined together by a glycosidic bond
Formation requires a dehydration (condensation) reaction
Sucrose: Common disaccharide example made of glucose and fructose
Polysaccharides
What are they
Examples for plants and animals
● Polysaccharides: Contain multiple monosaccharides held together in a long polymer by glycosidic bonds
Starch
Glycogen:
Celulose
Starch
Energy storage polysaccharide of glucose monomers held together and used by animals
Can be hydrolyzed, releasing free glucose monomers which can be utilized for energy
Glycogen
Energy storage polysaccharide of glucose monomers held together and used by animals
Can be hydrolyzed, releasing free glucose monomers which can be utilized for energy
Cellulose
Cellulose: Structural support polysaccharides made of many glucose monomers
Important component of plant cell walls
What are proteins made of?
How much is there?
What does the structure look like.
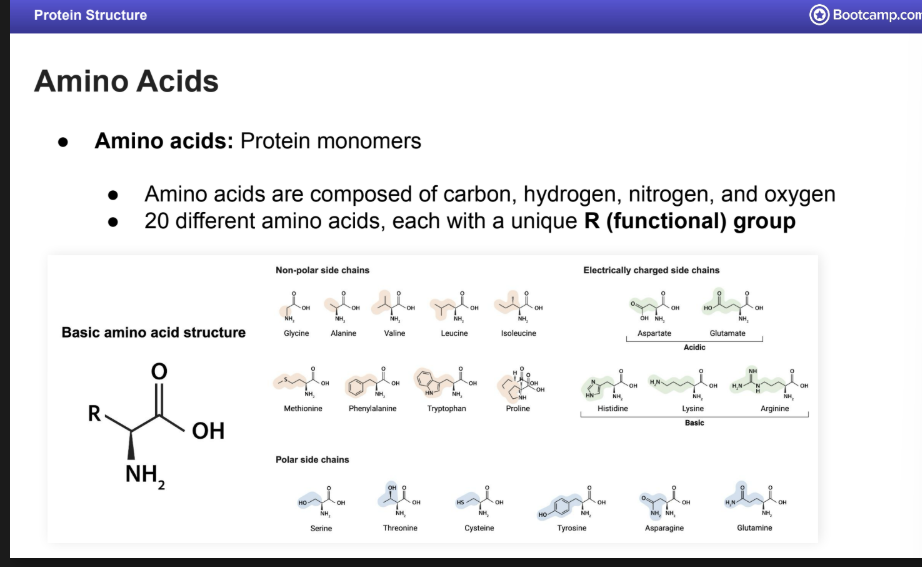
Amino Acids
There are 20 AA
They consist of an R group, nitrogen, hydrogen, and oxygen, and carbon.
Polypeptides:
What are they
What do they do
What kind of bonds do they have
How are these bonds created?
Polypeptides (proteins): Strands of many amino acids held together by peptide bonds
Diverse array of structures and functions
Peptide bonds: Between the carboxyl group of one amino acid and the amino group of another
created by dehydration (condensation) reactions; broken by hydrolysis reactions
Primary Protein Structure
Primary structure: The simple amino acid sequence resulting from mRNA translation
Secondary Protein Structure
Structures that show up
What bonds are present
Folding patterns of the primary amino acid sequence, including α-helices and β-pleated sheets
Determined by intermolecular forces between polypeptide backbones excluding R group
SPECIFICALLY, the C=O in the backbone causes these structural changes.
Tertiary Protein Structure
Bonds present
What interactions
Tertiary structure : Three-dimensional structure resulting from interactions between R groups
Hydrophobic interactions: Congregation or interaction of nonpolar molecules. Can occur between nonpolar R groups.
Disulfide bonds: Covalent bonding between two sulfur atoms
Additional tertiary interactions: Hydrogen bonding and ionic bonding between R groups are also possible
Quatenary Protein Structure
Multiple polypeptide chains come together to form one protein
Quaternary = tertiary + tertiary +....
Conjugated Proteins Def
Examples
Composed of amino acids and non-proteins components
EX
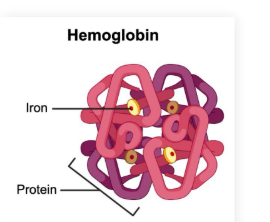
Metalloproteins
Glycoproteins
Metalloprotein
Def + Example
Metalloproteins: Proteins which contain a metal ion cofactor
Ex. hemoglobin contains an iron ion cofactor (Fe2+)
Glycoprotein
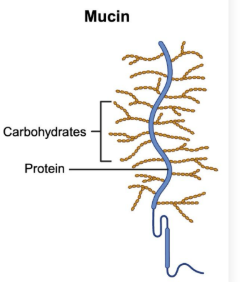
Def + Example
Glycoproteins: Proteins that contain a carbohydrate group
Ex. mucins can contain various carbohydrates
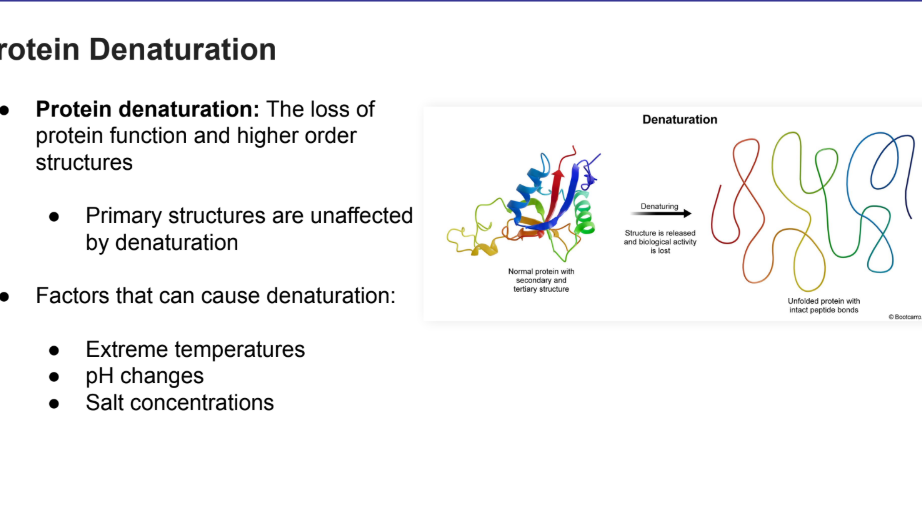
Protein Denaturation
Def + what causes it
The loss of protein function and higher order structures
Primary structures are unaffected by denaturation
Factors that can cause denaturation:
Extreme temperatures
pH changes
Salt concentrations
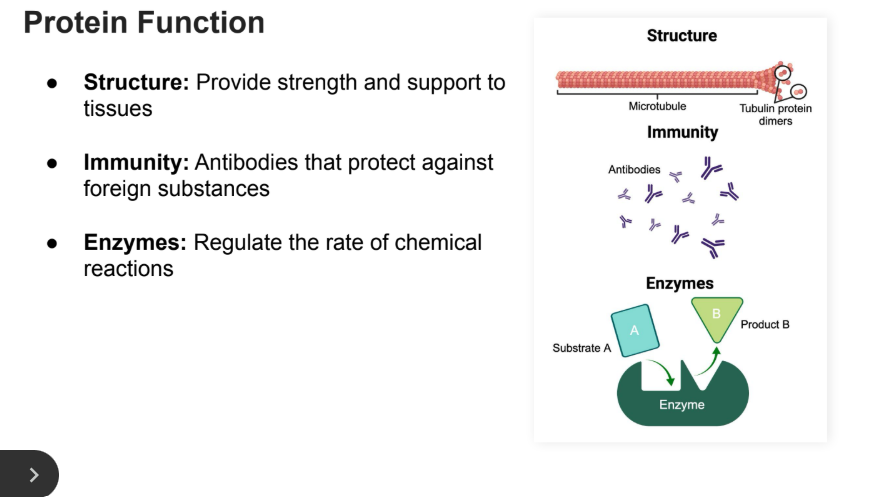
Protein Functions
Storage
Reservoir of Amino Acids
Hormones
Signaling Molecules that regulate physiological processes (they can be, not all hormones are these). EX: Insulin
Receptors
Some may be able to effect change in the intracellular enviroment
Structure: Provide strength and support to tissues
Immunity: Antibodies that protect against foreign substances
Enzymes: Regulate the rate of chemical reactions
What is a Catalyst
What does it do?
How does it affect Equilibrium Point, Spontaneity, Activation energy, or the Transition State.
Increases reaction rates by decreasing activation energy
Reduces the activation energy required to reach the transition state
Do not shift a chemical reaction’s equilibrium point or alter spontaneity
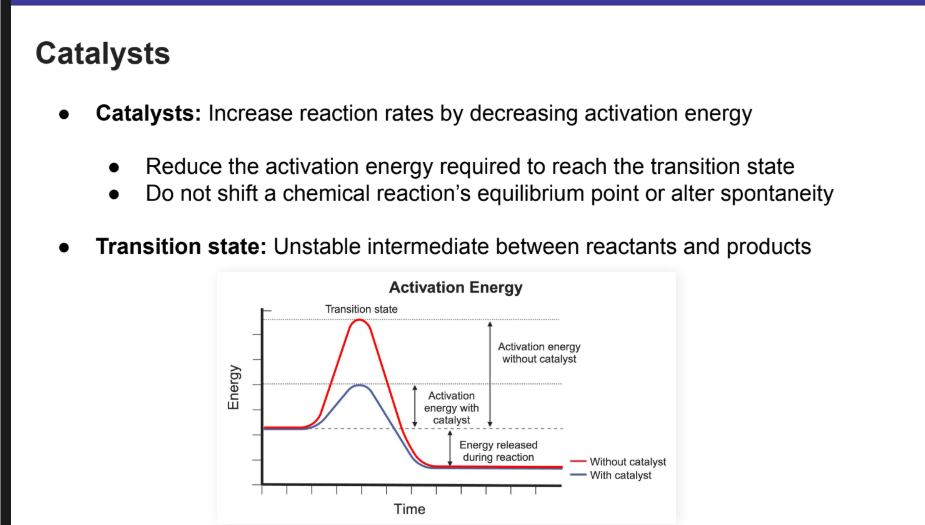
Transition State
Transition state: Unstable intermediate between reactants and products
Enzymes: Def, also what makes up the majority of enzymes
Biological catalysts, most are proteins
Active Sites:
Receptor regions on an enzyme that are specific for a substrate
Specificity constant:
Specificity constant: Measures enzyme binding efficiency for a specific substrate
Kcat/ KM (for past reference)
Explain the Induced Fit theory and the Lock and Key model
What do enzymes need to work?
Induced fit theory: Active sites mold to some degree to fit the substrate upon binding
“Lock and key” model: Outdated theory suggesting active sites are rigid and substrates are shaped perfectly to fit within them
Protein enzymes can be denatured and require optimal temperature and pH
Examples of Non-protein enzymes
Ribozymes
Cofactor
Coenzyme:
Holoenzyme
Apoenzyme
Prosthetic Groups
Ribozymes
RNA molecules that are capable of acting as enzymes
Cofactor
Non-protein molecule that helps enzymes function
● Coenzyme: Organic cofactor such as vitamins
● Inorganic cofactors include metal ions
Holoenzyme
Complex formed when an enzyme binds its cofactor.
Prosthetic groups:
Prosthetic groups: Cofactors that are tightly or covalently bound to their enzyme
Apoenzyme:
Enzyme without a bound cofactor
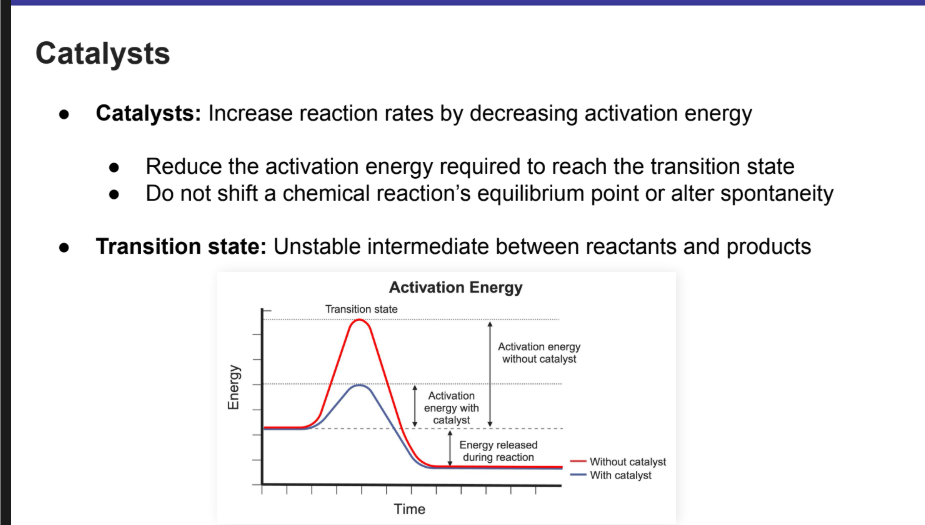
Mechanisms of Enzyme Catalysis
What do enzymes do to catalyze reactions?
What kind of interactions do they have with substrates?
Common ways in which enzymes can catalyze reactions:
Conformational changes to bring substrates closer together with correct orientation
Stabilization of the transition state
Having basic groups that can accept protons from a substrate
Having acidic groups that can donate protons to a substrate
Electrostatic interactions between the enzyme and the substrate
Draw out what the mechanism of enzyme catalysis looks like IG.
Phosphatase
They help with the Cleavage of a phosphate group off of substrate molecule
This is also knows as Dephosphorylation
Phosphorylase
Directly adds a phosphate group to a substrate molecule
Breaks substrate bonds in order to add the new phosphate group
Undergoes phosphorylation
Kinase
Indirectly adds a phosphate group to a substrate molecule
Transfers a phosphate group from an ATP molecule
No bonds are broken in order to add the new phosphate group
They are more like mediators, since they do not actually hold the enzymes themselves.
What factors influence Enzyme Activity?
Enzyme reaction rates can be affected if a molecule other than the desired, original substrate for a reaction binds either to the enzyme’s active or allosteric site
What is an active site, what is an allosteric site?
Active site: Receptor region that is specific for the desired reaction substrate
Allosteric site: Substrate binding location that is separate from the active site
Competitive Inhibitors
What do they do on the enzyme
Directly competes with reaction substrates for an enzyme’ active site
Competitive Inhibition
Effects
Competition between inhibitor and enzyme may cause reaction rate to decrease as inhibitor occupies the active site
Can be overcome by increasing substrate concentration
Enzyme Kinetics
What does the X axis mean
What does the Y axis mean
Enzyme kinetic plots can be used to visual the effects inhibitors have on enzymes
X-axis: Represents the concentration of substrate available
Y-axis: Represents the resulting reaction rate or velocity
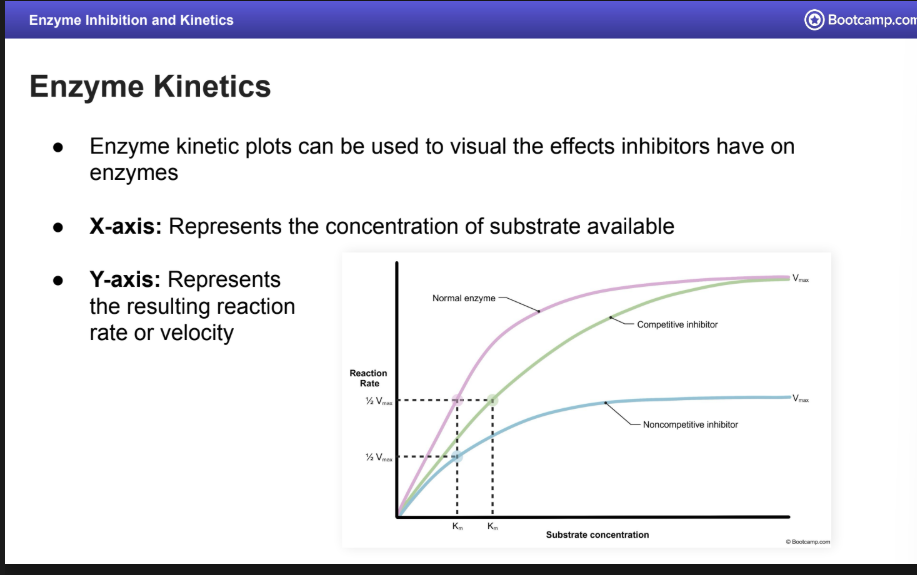
What is the Vmax and saturation mean on an Enzyme Kinetics Graph?
V max
Represents the maximum reaction velocity
Saturation: When all enzyme active site are occupied
Velocity will no longer increase and the graph will plateau
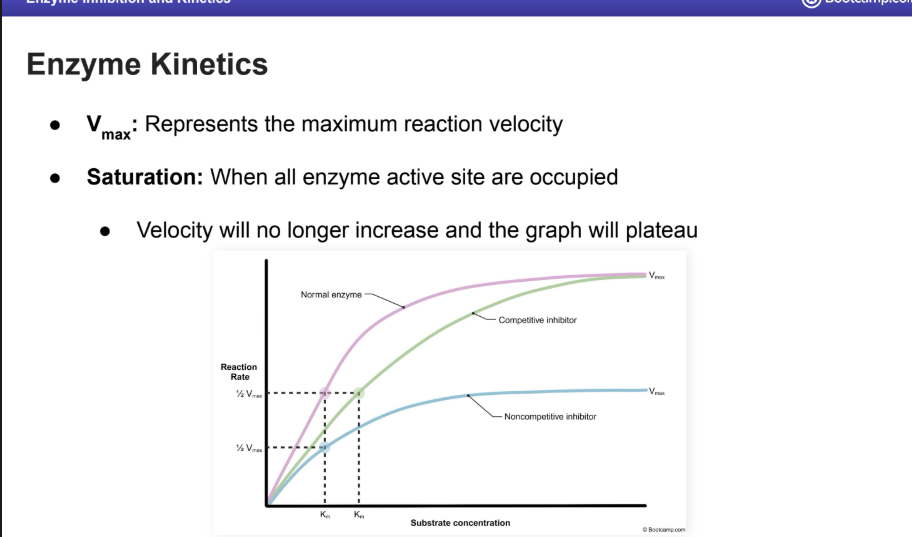
What is a Michaelis Constant?
AKA the KM
This is inversley related to enzyme substrate binding strenght.
HIGH KM means that more substrate is required to reach ½ Vmax
LOW KM means less substrate is required to reach ½ Vmax
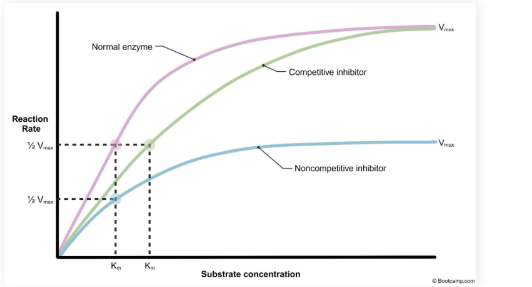
What occurs to the KM and Vmax when there is competitive inhibition.
KM is increased but V max does not change
Normal V max can still be reached with an increased substrate concentration
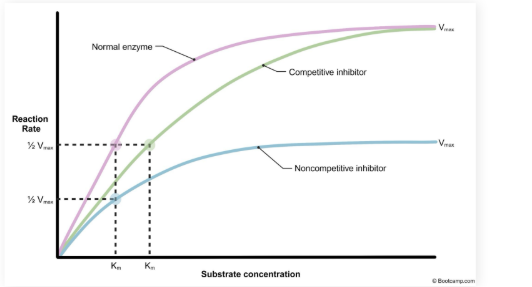
Noncompetitive inhibition changes to KM and VMAX
VMAX is decreased but KM does not change
Reduction in active enzymes available
Normal V max cannot be reached with increased substrate
Product Feedback Regulation (enzymes)
Describe how the product of a reaction affects the rate of the reaction itself
Negative Feedback Regulation (enzymes)
Reaction products will slow or inhibit the original reaction
Positive Feedback Regulation (enzymes)
Reaction products will activate or increase the original reaction
Lipids:
What are they made of?
Carbon, Hydrogen, Oxygen
What are fatty acid Tails
Long hydrocarbon tails with hydrophobic properties
Hydrophobic, Hydrophilic, Lipophillic, Amphipathic
Hydrophobic: Repels Water
Hydrophilic: Attracts Water
Lipophillic: Attracts Lipids
Amphipathic: Molecule with hydrophobic and hydrophilic parts
Another way to say this would be they have a hydrophilic and a lipophilic portion (ig)
Fatty Acids
Differences between Saturated and Unsaturated.
What are they?
What do they do to fluidity?
Saturated: Hydrocarbon chain without any double bonds
Single bonded chains packed tightly together
Unsaturated: Hydrocarbon chain with double bonds
Double bonding may produce a kink in the hydrocarbon chain
Unsaturated fatty acids pack less tightly together
Increase membrane fluidity
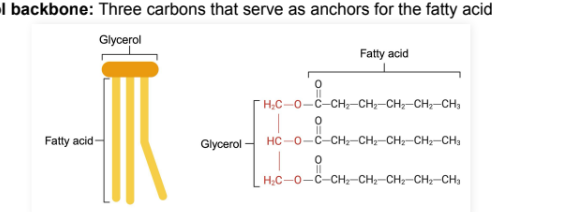
Triglycerides
Triglyceride (triacylglycerol):
Lipid type containing a glycerol backbone and three fatty acids
Most triglycerides are completely nonpolar and hydrophobic
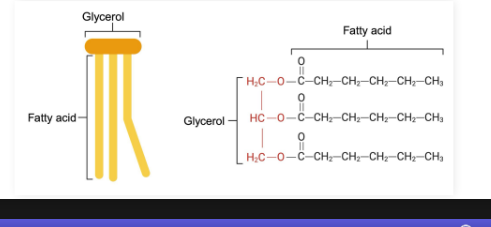
What is a glycerol backbone?
Three carbons that serve as anchors for the fatty acid chains
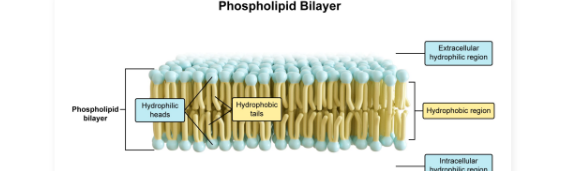
Phospholipids
Composed of a glycerol backbone, one phosphate group, and two fatty acid tails
Amphipathic: Phosphate “head group” is polar and hydrophilic while the fatty acids are nonpolar and hydrophobic
Spontaneously form bilayers in aqueous solutions
Factors that Affect Membrane Fluidity
Temperature
Cholesterol
Degrees of Saturation
How do these factors affect fluidity:
Temperature
Cholesterol
Degrees of Saturation
Temperature
Fluidity Increases with higher temperatures and decreases with lower temperatures
Cholesterol
Holds membranes together at high temperatures and keeps the membrane fluid at low temperatures
Degrees of Saturation
Fluidity increases when many kinked, unsaturated fatty acids are present in the membrane and decreases as saturated fatty acid concentrations rise.
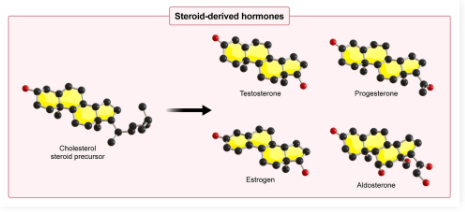
Cholesterol
DEF: Amphipathic lipid component of cell membranes
Precursor molecule to steroid hormones (4 hydrocarbon rings), vitamin D, and bile acid
Maintains membrane stability at both high and low temperatures
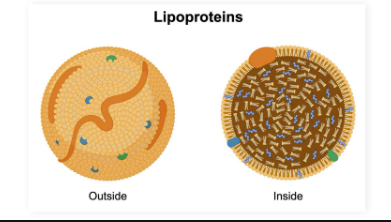
Lipoproteins
DEF + What are they used for
Lipoproteins: Round complex of lipids and proteins that can carry lipophilic (hydrophobic) molecules through the blood
Outer coat composed of phospholipids, cholesterols, and proteins
Can carry molecules like cholesterol and proteins
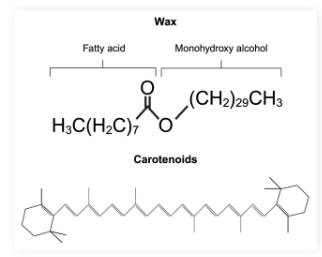
Waxes and Carotenoids defs
What are they made of, what do they do
Waxes: Simple lipids with long fatty acid chains connected to alcohols
Carotenoids: Lipid derivatives containing long carbon chains conjugated double bonds that function mainly as pigments
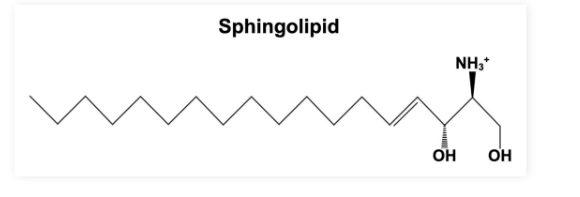
Sphingolipids
Sphingolipids: Lipids with a backbone containing aliphatic (non-aromatic) amino alcohols
Important functions in structural support, signal transduction, and cell recognitions
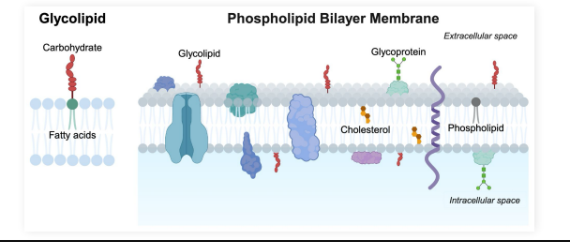
Glycolipids
Lipids found in plasma membranes with carbohydrate groups bound rather than a phosphate group
Amphipathic with a polar head and nonpolar tail
What are nucleic acids?
Nucleic acids: Diverse molecule class composed of sugar monomers
Functions including genetic information storage, transfer of other molecules, gene silencing, etc.
DNA and RNA are the two main types of nucleic acids
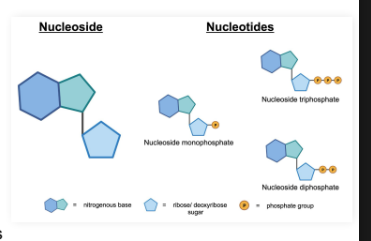
Nucleoside vs Nucleotide
Nucleoside: Made of 1 ribose or deoxyribose sugar and 1 nitrogenous base
Nucleotide: Made of 1 ribose or deoxyribose sugar, 1 nitrogenous base, and
phosphate(s)
Name depends on the amount of phosphate groups
Ex. Nucleotide containing 3 phosphates is a nucleoside triphosphate
Mnemonic: Nucleosides are sugars with bases; nucleotides have phosphates
RNA
RNA (ribonucleic acid): Molecule containing ribose sugar nucleotides that is typically single stranded
Ribose sugar with hydroxyl groups (-OH) on both the 2’ and 3’ carbons
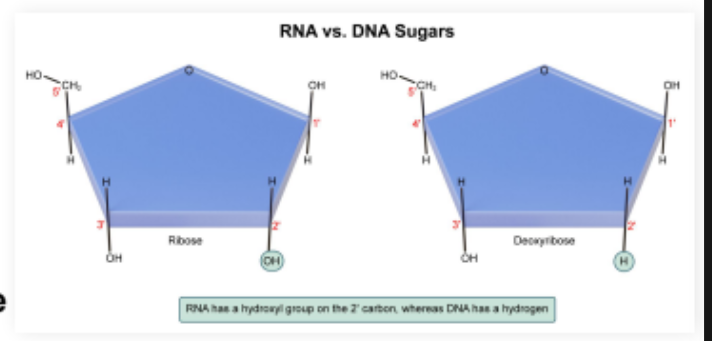
DNA
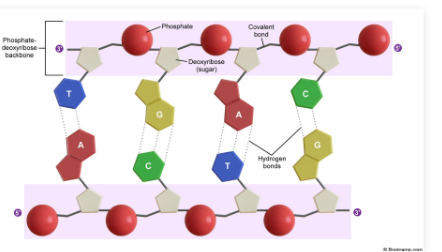
DNA (deoxyribonucleic acid): Antiparallel double helix molecule containing deoxyribose sugars
Deoxyribose sugars with a hydroxyl group on only the 3’ carbon, not the 2’
Two complementary strands wind around each other
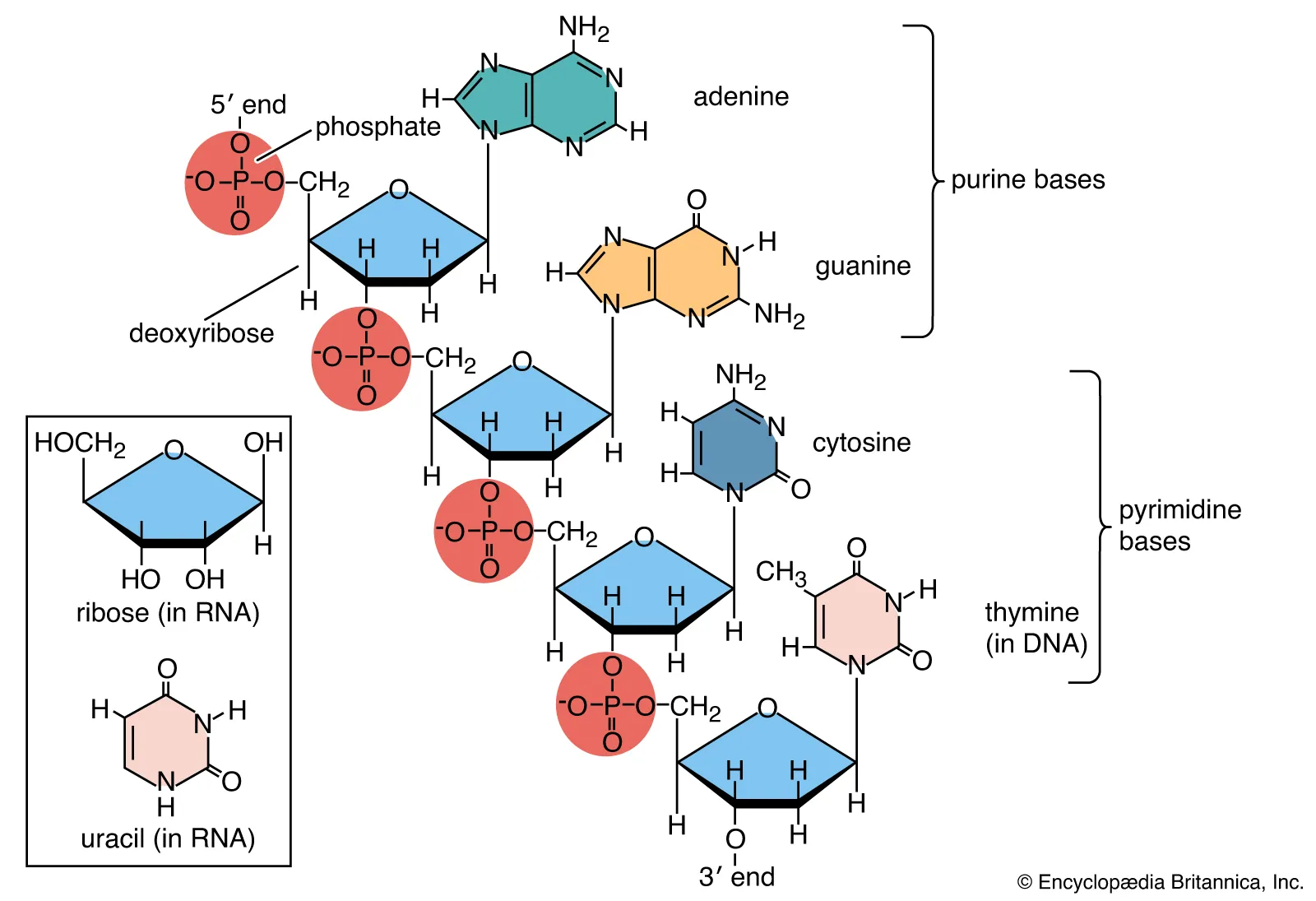
Sugar Phosphate Backbone of DNA
Sugar-phosphate backbone: Structural chain of alternating sugars and phosphates held together by phosphodiester bonds
Terminal ends will be one 3’ hydroxyl group and one 5’ phosphate group
Phosphodiester bonds: Condensation reaction used to link nucleotides together
Bond between the 5’ phosphate group of one nucleotide and the 3’ hydroxyl group of another nucleotide
OK, so here is a much easier explanation. At the ribose 3 prime end, there is a phosphate attatched. That phosphate connects to a 5,4 prime end of ribose (it really is just the 4’ end though). Then at the 1’ end, of the ribose, a nitrogenous base is added (like adenine, guanine, cytosine, etc)
Nucleic Acid polymerization
Nucleic acid polymerization: Nucleic acids elongate as nucleoside triphosphates (DUMB WAY OF SAYING IT IS A NUCLEOTIDE :p) are continually added to the free 3’ hydroxyl group of the molecule.
ESSENTIALLY, WE ADD STUFF TO THE 3 PRIME HYDROXYL END.
mRNA
Single stranded molecule resulting from DNA transcription
Contains the information required to create proteins
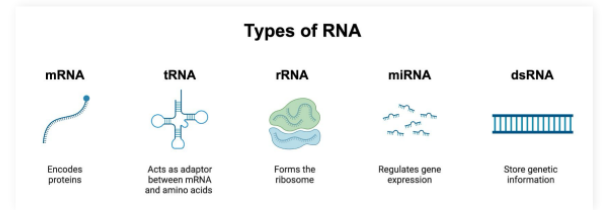
tRNA
tRNA (transfer RNA): Participates in protein synthesis by carrying amino acids to ribosome enzymes
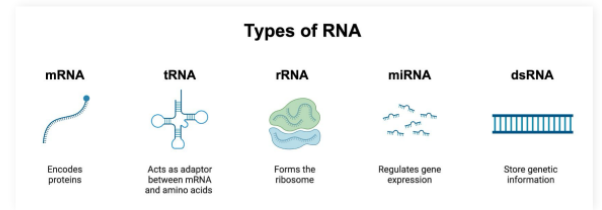
rRNA
ribosomal RNA: Participates in protein synthesis by complexing with proteins to make the ribosome enzyme that reads mRNA molecules
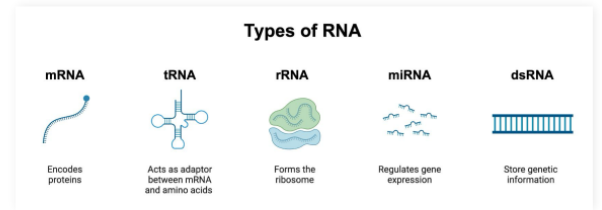
miRNA
microRNA: Capable of silencing gene expression
Base pair to mRNA molecules to prevent them from being read by ribosomes
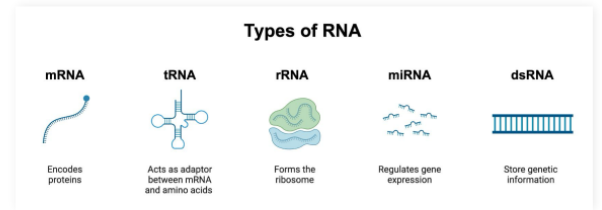
dsRNA
double stranded RNA:
Genetic information reservoir used by some viruses instead of DNA
Pro-Tip: dsRNA must have complementary base pairing and therefore will have equal amounts of A/U and G/C
What is a hypothesis and a theory
Hypothesis: A proposed explanation of a scientific phenomena that is based on prior knowledge but requires further testing and scrutiny
Theory: Explanation of a scientific phenomena that has been accepted due extensive testing yielding repetitive results
Primordial Earth Theory (11 points)
Biological Hypotheses and Theories | Biology
Theory explaining how early Earth provided the conditions needed for life
The Universe is approximately 13.8 billion years old
Primordial atmosphere: Comprised of inorganic compounds creating a reducing environment (low O2 gas)
Earth cooled and gases condensed, formed the primordial sea
Simple compounds evolved into more complex organic compounds
Organic monomers linked into polymers
Protobionts: Abiotic, lipid capsules that emerged as a precursor to cells
The first cell arrived on Earth 3.5 billion years ago
Heterotrophic, obligate anaerobic prokaryotes developed
Autotrophic prokaryotes, such as cyanobacteria capable of photosynthesis, formed
a. Led to oxygen production and accumulation, creating an oxidizing
Primitive eukaryotes emerged
Support for the endosymbiotic theory
More complex eukaryotes and multicellular organisms evolved
Modern Cell Theory: 7 points
Theory describing how cells govern high order iterations of life
All lifeforms have one or more cells
The cells is the basic structural, functional, and organizational unit of life
All cells come from other cells via cell division
Genetic information is stored and passed down through DNA
An organism’s activity is dependent on the total activity of its independent cells
Metabolism and biochemistry (energy flow) occur within cells
All cells have the same chemical composition within organisms of similar species
Central Dogma of genetics
Central dogma of genetics: Information is passed from DNA to RNA to proteins
Exceptions include reverse transcriptase and prions
PRIONS: Self replicating
REVERSE TRANSCRIPTASE: RNA to DNA
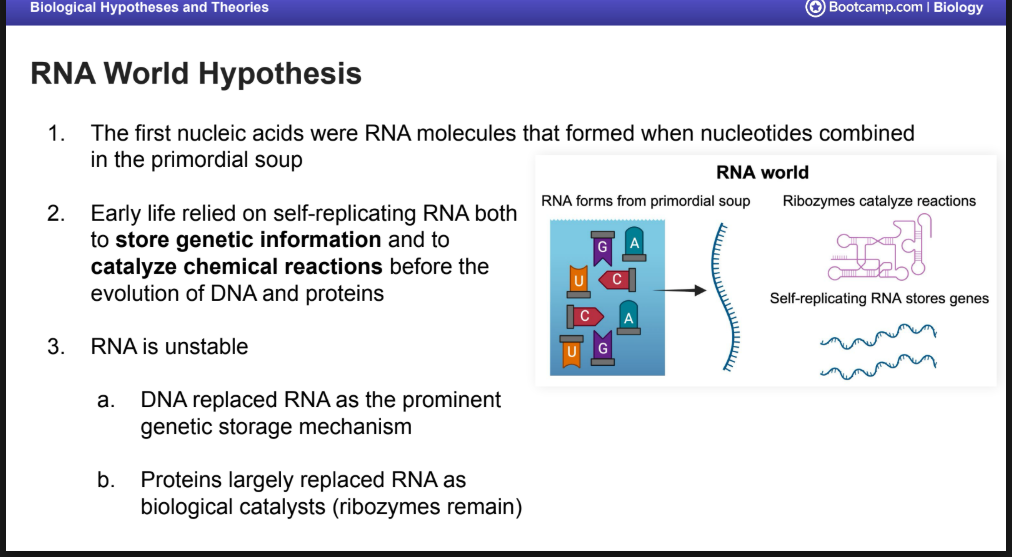
RNA world Hypthesis
1. The first nucleic acids were RNA molecules that formed when nucleotides combined in the primordial soup
Early life relied on self-replicating RNA both to store genetic information and to catalyze chemical reactions before the evolution of DNA and proteins
RNA is unstable
a. DNA replaced RNA as the prominent genetic storage mechanism
b. Proteins largely replaced RNA as biological catalysts (ribozymes remain)
Endosymbiotic Theory
Theory describing how eukaryotes came to house membrane-bound organelles
Eukaryotes developed when aerobic bacteria were internalized as mitochondria while the photosynthetic bacteria became chloroplasts
Evidence for this theory includes the similarities between mitochondria and chloroplasts
a. They are similar in size
b. They possess their own circular DNA
c. They have ribosomes with a large and small subunit
d. They reproduce independently of the host cell
e. They contain a double membrane