biology - unit 3 aos 2
1/46
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
47 Terms
3A - enzymes
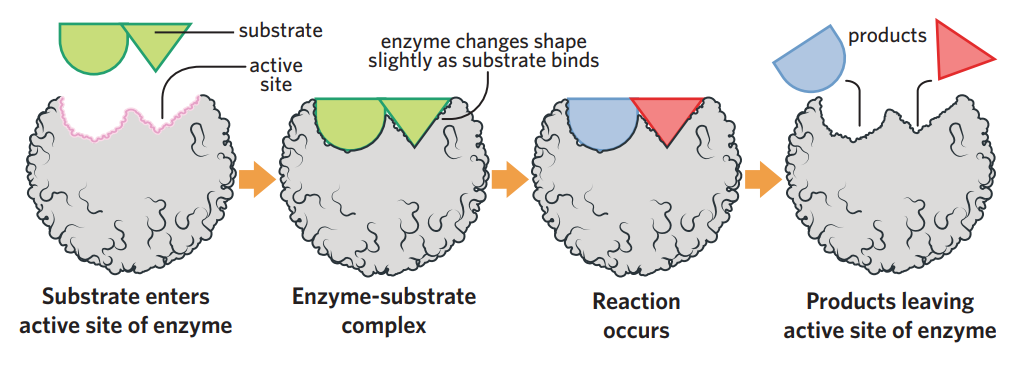
enzymes - proteins usually ending in ‘ase’ that catalyse (speed up) biochemical reactions by lowering the activation energy required required to initiate the reaction
substrate (reactant) binds to active site on enzyme (which has 3d complementary shape), making enzyme-substrate complex, then reaction occurs, and products are released
substrate specificity - each enzyme only binds to 1 substrate → only catalyse 1 reaction
coenzyme
ATP, NADPH/NADP+, NAD+/NADH, FAD/FADH2
Enzymes catalyse most of the reactions in photosynthesis. For example, in the lightdependent stage, the enzyme ATP synthase catalyses the reaction ADP + Pi → ATP
Having enzymes regulate each step in photosynthesis ensures reactions are sped up and controlled, so plants can metabolise efficiently.
3B - factors that affect enzymes
temperature
pH
concentration
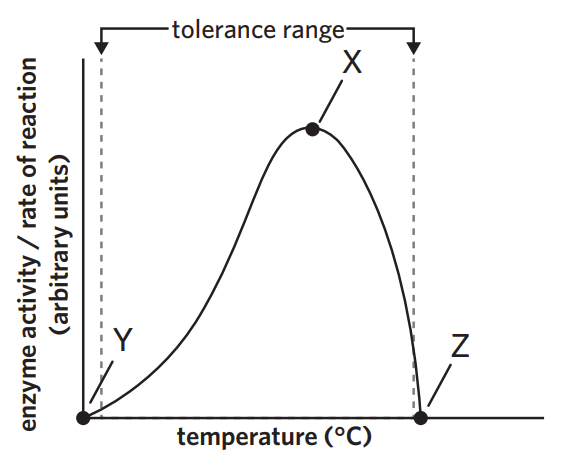
3B - temperature
assuming pH is constant
optimal temp - maximum rate of substrate entering the active site and products leaving
under optimal temp - particles move slower. substrate enters active site slower + products leave slower
above optimal temp -
way too above optimal temp - enzyme denatures. active site changes shape. substarte cannot bind.
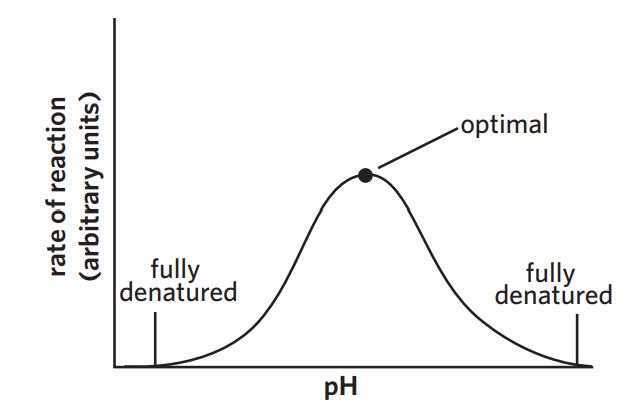
3B - pH
assuming temperature is constant
denatures if not optimal pH
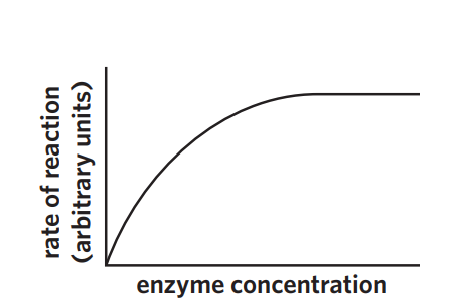
3B - concentration
saturation point -
excess enzyme
excess substrate
3B - enzyme inhibition
enzyme inhibitors - molecules that bind to an enzyme, preventing it from performing it’s function.
competitive inhibitors
chemical that binds to active site of enzyme (meaning it has a simialr shape to its’ substrate), and directly blocks the substrate. substrate + inhibitor are ‘competing’.
if there are competitive inhibitors, you can increase the substrate concentration, in order to increase the chance of substrate binding rather than inhibitor
non-competitive inhibitors
binds to the enzyme somewhere other than the active site, called an allosteric site. this changes the shape of the active site → substrate can’t bind
changing substrate concentration will not change non-competitive inhibition, because they bind to different locations, and aren’t bothered by an increase of something that doesn’t go to the same site.
irreversible - render an enzyme disfunctional even after enzyme detaches
biochemical pathway
biochemical pathway - a series of biochemical reactions, where the outputs of one reaction are the inputs of another reaction, and so on. ex, photosynthesis, cellular respiration
5A - photosynthesis
photosynthesis - the process in which plants (photoautotrophs) convert light energy into chemical energy (stored as glucose), that they can use for their own energy. occurs in two stages - light dependent and light independent.
equation:
6CO2 + 12H2O →(light) → C6H12O6 + 6O2 + 6H2O
6CO2 + 6H2O →(light) → C6H12O6 + 6O2
primary product is glucose - plants can use glucose immediately as an input for cellular respiration to create energy, or they can store it as starch, or use it to form more complex molecules.
5A - plant structures involved in C3 photosynthesis
plants mainly perform photosynthesis at the leaves. they typically have a large surface area to maximise the amount of light hitting the surface. there are many cell types in leaves that perform different functions:
mesophyll cells - contain lots of chloroplasts → main cell type in leaves that photosynthesise.
sheath bundle cells?
stomata - tiny pores on the surface of leaves that open and close to regulate gaseous exchange. when open, carbon dioxide in the atmosphere diffuses into the leaf, and oxygen and water are lost, which the plant must regulate by closing the stomata.
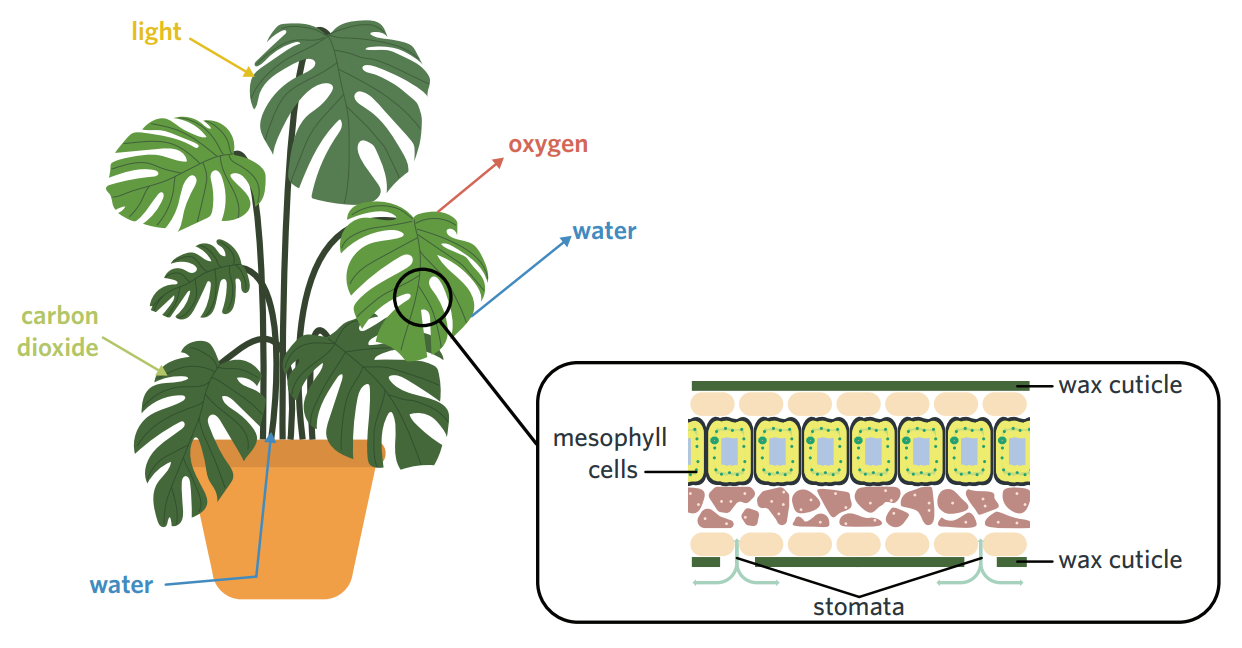
5A - chloroplasts
chloroplast - site of both stages of photosynthesis in plant cells
thylakoids - flattened membrane-bound sacs, arranged into grana. thylakoid membrane contains chlorophyll + proteins, + is site of LDS. fluid space inside thylakoid is lumen.
granum (pl. grana) - a stack of thylakoids
chlorophyll - green pigment in thylakoid. absorbs red + blue wavelengths. responsible for green colour of thylakoids + → whole chloroplast
stroma - fluid between grana. contains enzymes for LIS + chloroplast’s DNA, which is stored in a circular chromosome (similar to bacteria → endosymbiotic theory)
membrane
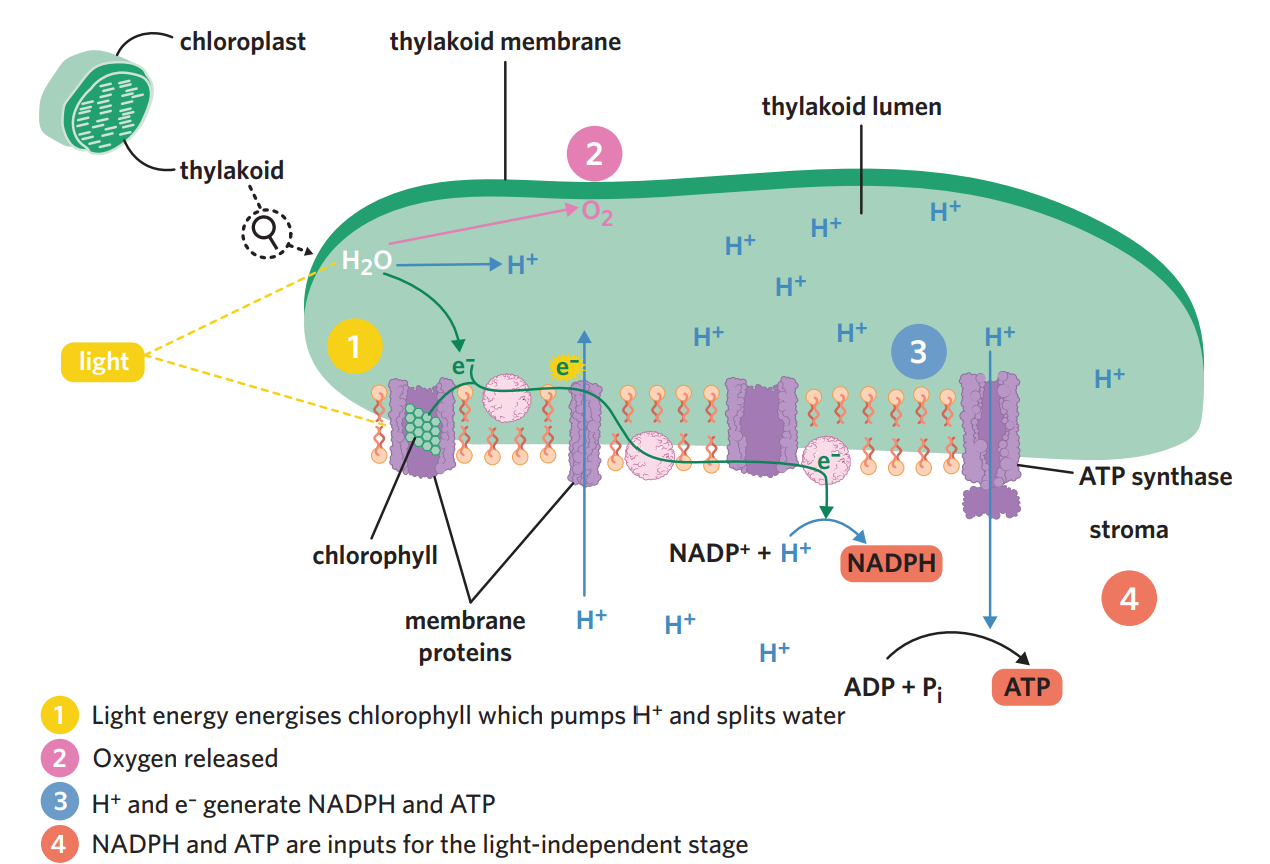
5A - light dependent stage
light dependent stage - light energy splits water molecules into hydrogen and oxygen. also generates high energy coenzymes NADPH and ATP. occurs on the thylakoid membranes of chloroplasts, and only when light is present.
process
on the thylakoid membrane, light energy excites electrons in chlorophyll. these electrons leave chlorophyll + move along proteins in the membrane, which powers the pumping of H+ ions into the thylakoid lumen. water donates electrons to chlorophyll to replace the electrons that left, causing water to split into oxygen and two H+(photolysis).
oxygen is released out of the chlorophyll → either diffuses out of stomata into environment or used as input for aerobic cellular respiration
H+ + e- from water combine with NADP+, which is reduced, generating the high energy coenzyme NADPH. Also, there is now a high concentration of H+ inside the lumen and a low conc outside in the stroma → concentration gradient which H+ ions want to balance → H+ ions leave the lumen through the protein ATP synthase. as H+ flows out, their movement provides energy, which converts ADP + Pi into ATP.
NADPH + ATP coenzymes are then used in light independent stage
inputs
12 H2O
12 NADP+
outputs
6O2
12 NADPH
18 ATP
5A - cycle of coenzymes from LDS to LIS
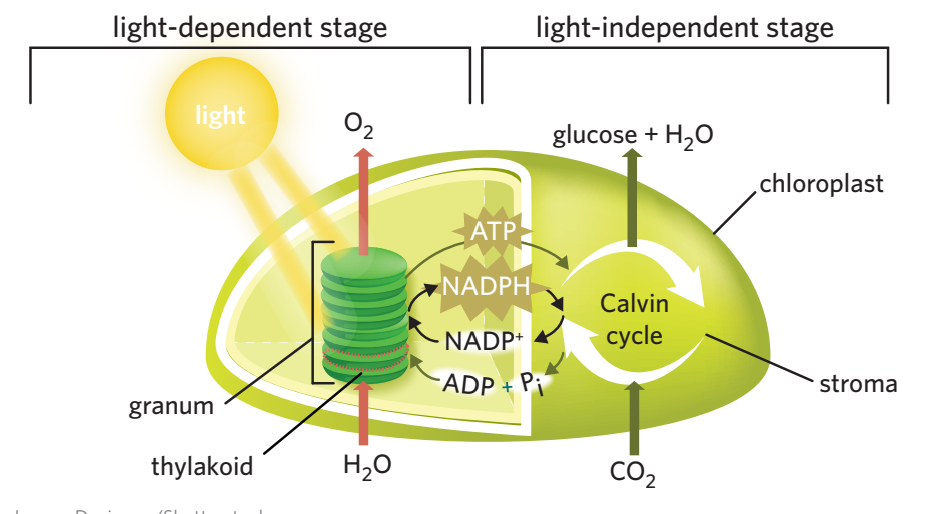
5B - light independent stage
light independent stage - using NADPH + ATP produced in LD stage + carbon dioxide, glucose is produced through a cycle of reactions (calvin cycle) occurs in the stroma of chloroplasts, and doesn’t require light.
process
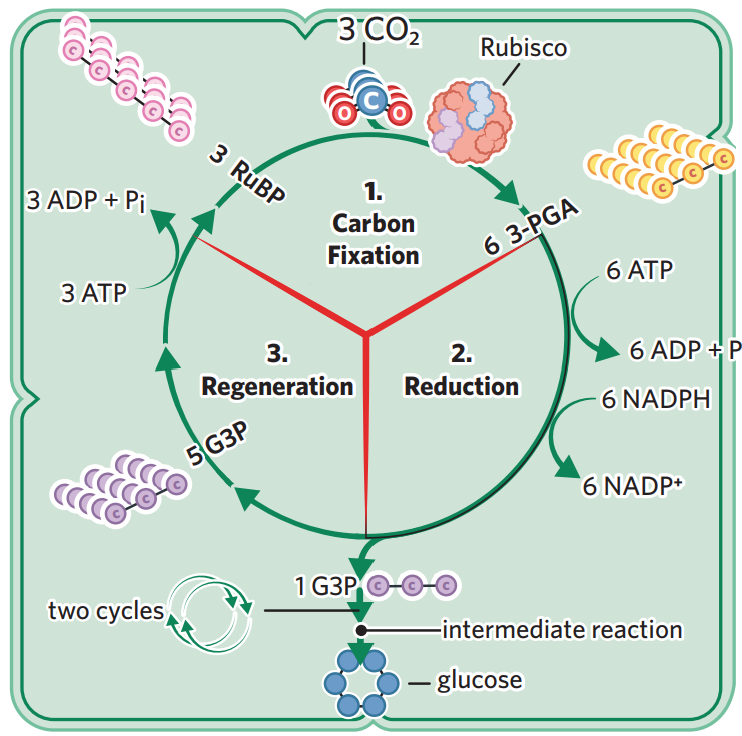
carbon fixation - inorganic (doesn’t contain a C-H bond) CO2 molecules (collected from the stomata in leaves) enter the stroma, and are ‘fixed’ into organic molecules. rubisco enzyme converts CO2 and RuBP (5C) into 3PGA (3C)
3 x CO2 + 3 x RuBP → 6 × 3PGA
reduction - NADPH donates electrons and H+ to 3PGA, reducing it. ATP break down into ADP + Pi, donating energy to 3PGA. through these processes, the 3PGA are then converted into G3P (3C), a higher energy molecule which can be converted into glucose
6 × 3PGA → 6 x G3P
regeneration - although 6 G3P are produced, 5 are recycled to regenerate RuBP, meaning each cycle produces 1 net G3P. however glucose requires 2 x G3P, so the cycle repeats twice for each 1 glucose made.
2 x G3P → 1 glucose
some of the oxygen molecules leftover from the breaking of CO2 at the beginning of the cycle combine with hydrogen ions from NADPH to create the output water.
inputs
6CO2
12 NADPH
18 ATP
outputs
C6H12O6
12 NADP+
18 ADP + Pi
6 H2O
5B - rubisco
rubisco - rubisco is a key enzyme in the LIS of photosynthesis. during the first stage of the calvin cycle (carbon fixation), it binds to CO2, and fixes carbon from it. however, sometimes it uses O2 as a substrate rather than CO2. this is a major flaw of rubisco, as when this happens, CO2 cannot bind, and a different reaction called photorespiration occurs instead
CO2 cannot bind - when O2 binds to rubisco, it acts as a competitive inhibitor, by binding to the active site which CO2 would’ve binded to.
photorespiration - photorespiration is also a wasteful process as it uses energy produced in photosynthesis
= by binding O2 and undertaking photorespiration, CO2 loses an opportunity to undergo LIS → less glucose produced
Rubisco has a lower affinity to bind to O2 over CO2 when there is low O2 concentration or when temperatures are normal or cool
5B - adaptations to photorespiration
there are 3 different types of plants when it comes to adapting to minimise photorespiration:
C3 (no adaptation)
C4 - LIS seperated spatially
CAM - seperated temperally
in hot + dry environments, stomata close, which prevents CO2 from coming in and prevents O2 from leaving → lower conc CO2 and higher conc O2 → likely for photorespiration to occur → have adaptations
to minimise photorespiration + increase photosynthesis, rubisco should be exposed to high concentration of CO2 and low concentration of O2.
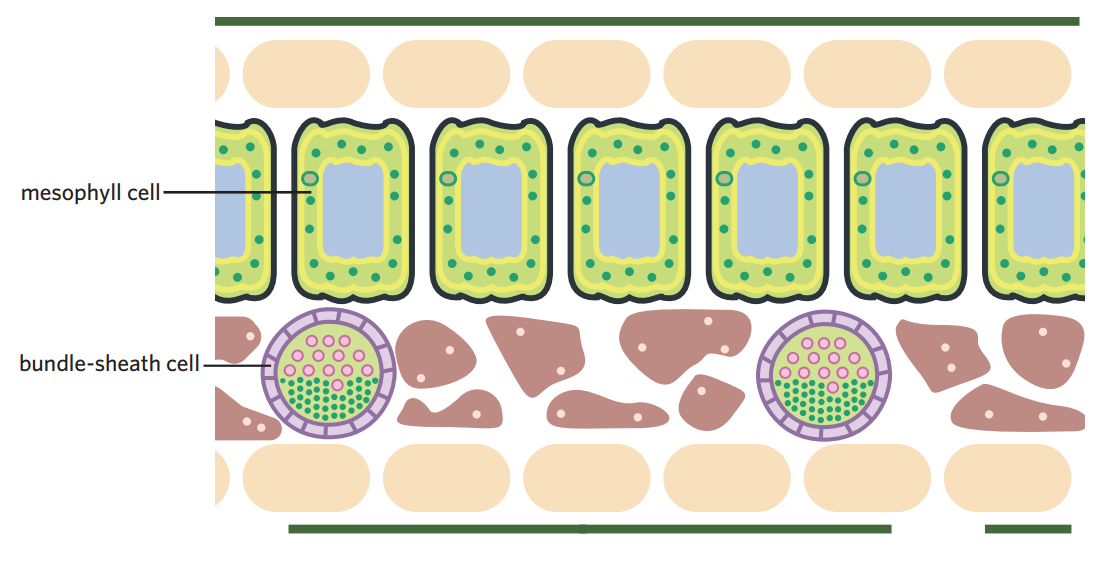
5B - C3 plants
C3 plants contain no mechanism to minimise photorespiration.
photosynthesis occurs within a single mesophyll cell. first stage of LIS, carbon fixation, involves rubisco fixing carbon from CO2 into 3-PGA, which is 3C (hence C3 plants)
C3 plants are vulnerable to photorespiration in hot dry environments - when C3 plants are in hot, dry environments, stomata may close to reduce water loss. however, this means oxygen produced in LDS cannot escape → builds up, increasing photorespiration.
examples - 85% of plants, incl rice, wheat, trees, + most fruits, vegetables, nuts’
better suited to cooler temepratures, lower light intensity
5B - C4 plants
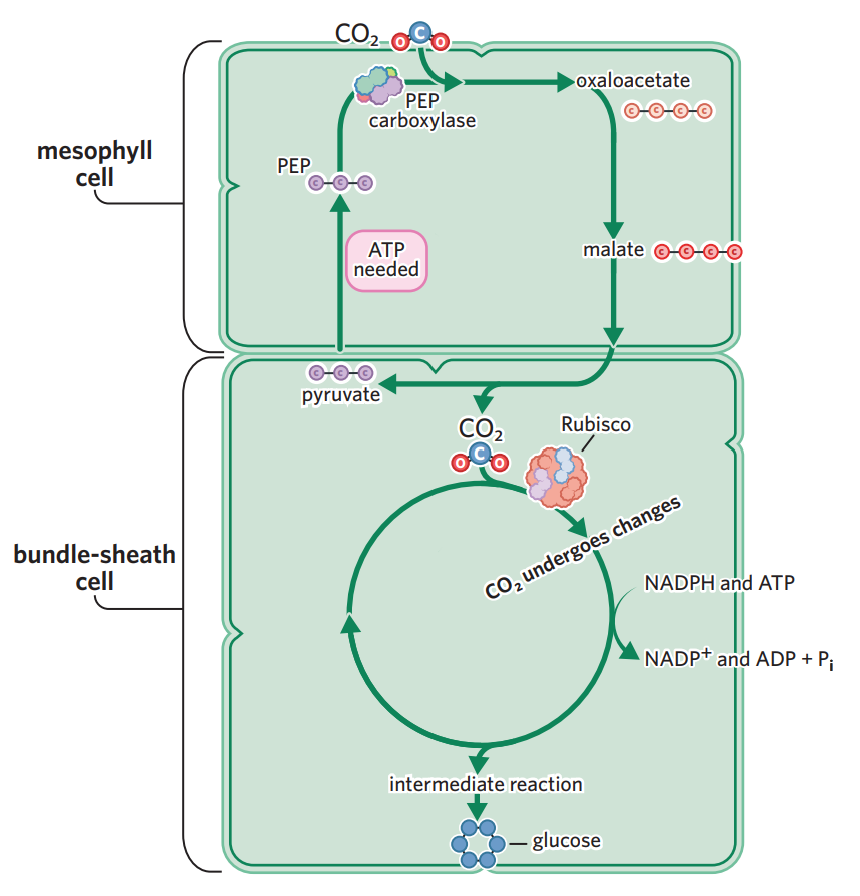
LDS is exactly the same as C3 plants. however, LIS/calvin cycle is seperated into two different cells - a mesophyll cell and a bundle sheath cell.
in mesophyll cell, CO2 enters. the enzyme PEP carboxylase (not rubisco, bc PEP carboxylase has no affinity to bind to O2) adds the carbon from CO2 to a PEP molecule (3C), fixing it into oxaloacetate (4C).
oxaloacetate (4C) is converted into malate (also 4C, but capable of being transported to bundle sheath cells). → malate transported to bundle sheath cells, creating high concentration of CO2 (in form of malate), meaning photorespiration is minimised.
in bundle sheath cell, rubisco breaks down malate + releases CO2 → this CO2 then enters calvin cycle like C3 plants.
Pyruvate formed from the breakdown of malate is transported back to the mesophyll cell and converted to another molecule, PEP, with the help of ATP
PEP is then ready to contribute to the fixation of CO2 and production of oxaloacetate and the cycle continues all over again.
example - maize, sugarcane
higher efficiency in hot, high light intensity environments. however, more ATP is required to convert pyruvate to PEP for the initial carbon fixation → C4 plants use more energy than C3 plants in photosynthesis.
requires more ATP to cycle PEP
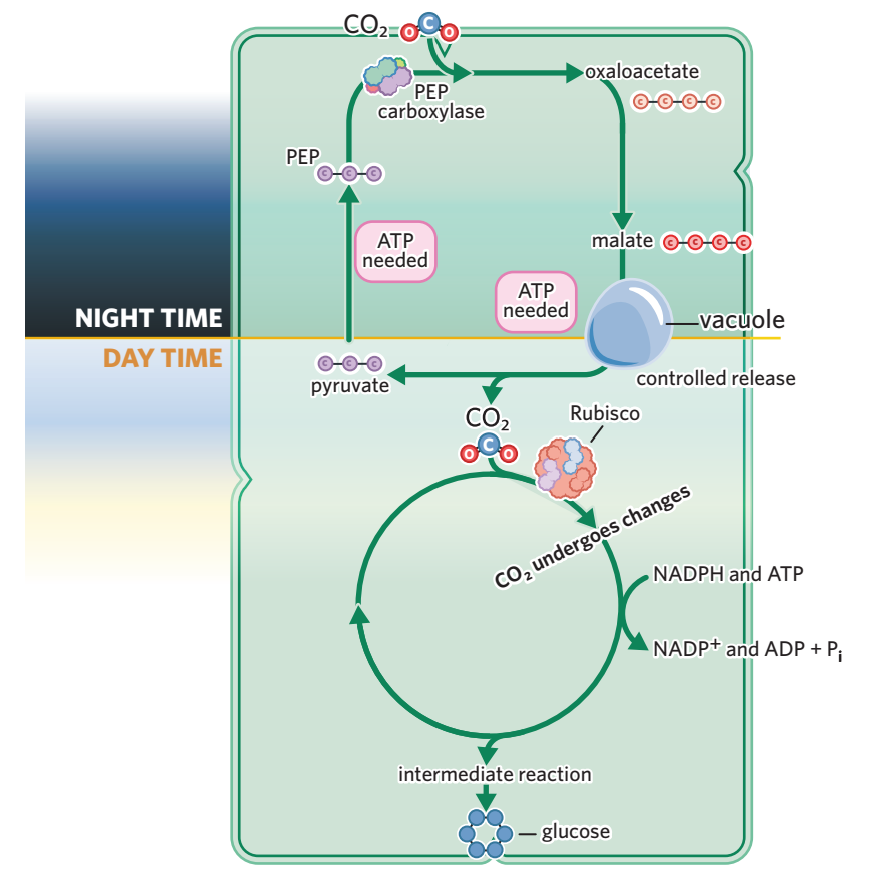
5B - CAM plants
light independent stage is seperated over time/temperally to minimise photorespiration.
Separate CO₂ fixation by time (night vs. day) to conserve water.
Night: Stomata open, CO₂ is fixed into a 4-carbon acid (malate) and stored.
Day: Stomata close, and the stored malate releases CO₂ for the Calvin cycle.→ controlled release
Advantage: Survive in arid environments by minimizing water loss.
example - cacti, pineapples, succulents
requires more ATP to cycle PEP
5C - factors affecting rate of photosynthesis
light intensity + wavelength
temperature + pH
carbon dioxide concentration
water availability
enzyme inhibition
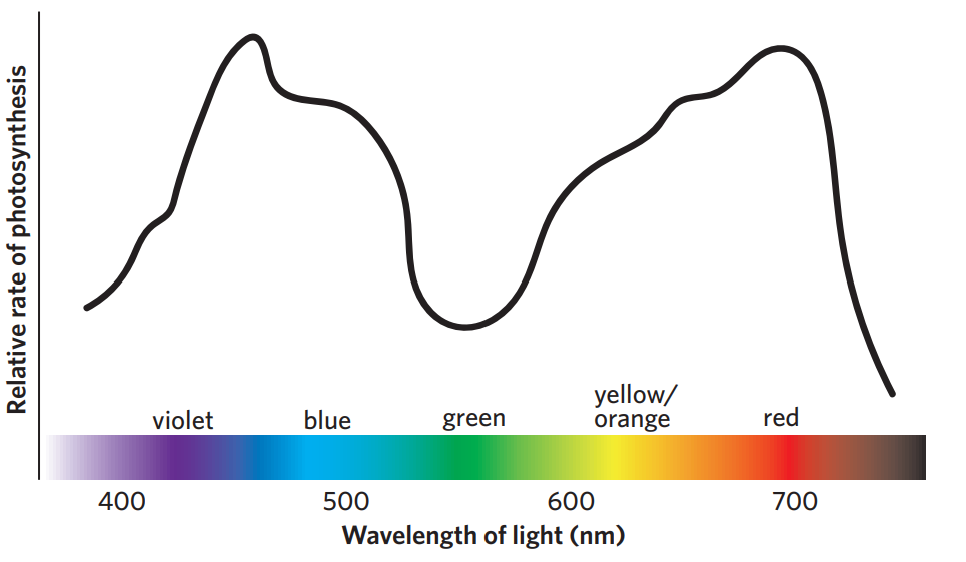
light wavelength
the greatest rate of photosynthesis occurs in violet or red light
lowest at green wavelength, because green is reflected (explains why chlorophyll is green)
5D - agricultural applications of CRISPR-Cas9
5
C3 - plants with no evolved adaptation to minimise photorespiration
C4 - plants that minimise photorespiration by separating initial carbon fixation and the remainder of the Calvin cycle over space
CAM plants - plants that minimise photorespiration by separating initial carbon fixation and the remainder of the Calvin cycle over time
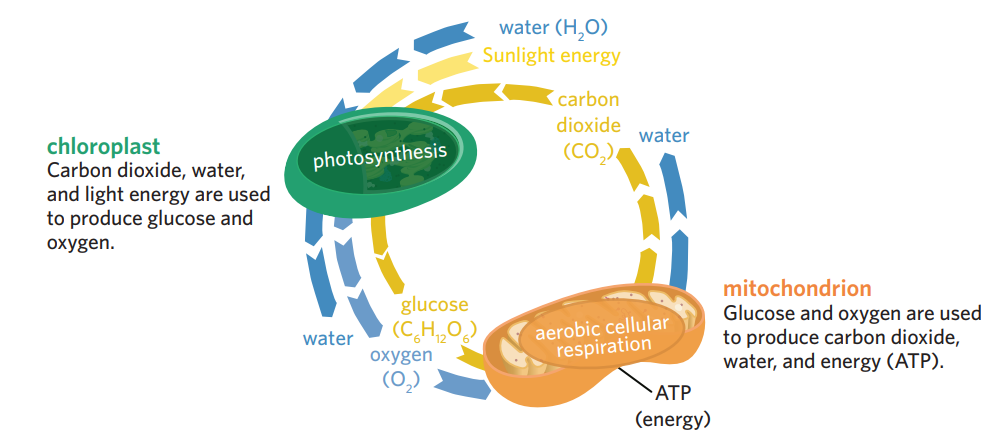
photosynthesis and cellular respiration
photosynthesis and cellular respiration are related, as each process can ‘recycle’ the outputs of the other reaction, using them as inputs
however, they are very different biochemical pathways, with very different structures and enzymes are involved
plants + algae can undergo both photosynthesis and cellular respiration → don’t need to source all their photosynthesis and cellular respiration inputs from the environment.
✅6A - cellular respiration
cellular respiration - all cells undergo cellular respiration, which generates ATP from glucose. ATP is a high energy molecule that all organisms need to grow, produce enzymes and many other things.
there are 2 biochemical pathways in which ATP is produced from cellular respiration. main difference is presence or absence of oxygen.
aerobic cellular respiration - more efficient (produces 30 or 32 ATP)
anaerobic fermentation - faster than aerobic respiration, but less efficient (only produces 2 ATP) + produces harmful by-products
glucose carries too much energy to be useful in most biochemical reactions → cellular respiration breaks down glucose into smaller packages, ATP. DRAW DIAGRAM when the third phosphate is broken off, it releases lots of usable energy
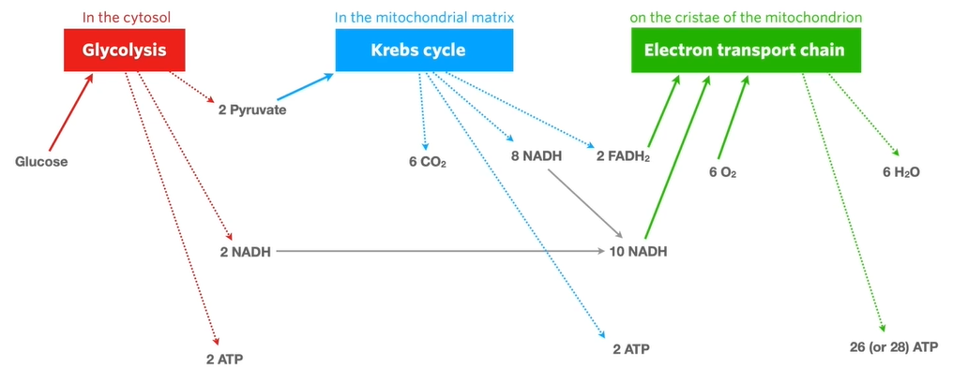
✅6A - aerobic cellular respiration
aerobic cellular respiration - a form of cellular respiration that occurs when oxygen is present. it is series of enzyme-controlled reactions in which glucose reacts with oxygen to produce carbon dioxide, water and ATP
overall equation: C6H12O6 + 6O2 → 6CO2 + 6H2O
however, above is a simplified version. there are many enzyme-controlled reactions involved that occur over 3 stages.
glycolysis
krebs cycle
electron transport chain
ATP yield per glucose molecule
glycolysis - 2
krebs cycle - 2
ETC - 26 or 28
= 30 or 32 ATP in total
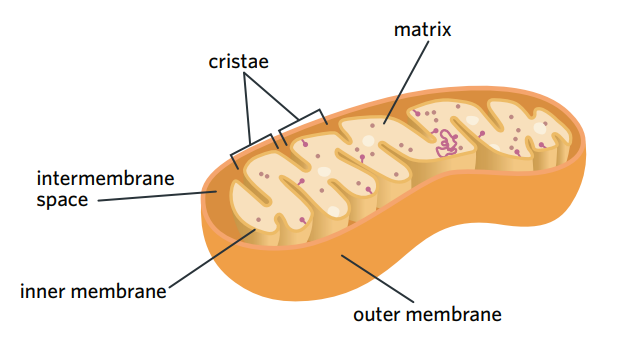
✅6A - mitochondria
the mitochondria is crucial to aerobic respiration as it is the site of the 2nd (krebs cycle) and 3rd (ETC) stages
structure
intermembrane space - the narrow, small space between the inner and outer membrane
inner mitochondrial membrane - membrane made of phospholipid bilayer which is the site of ETC.
cristae - the inner membrane folds into peaks and ridges called cristae
mitochondrial matrix - the space inside the inner membrane, filled with a dense fluid containing many enzymes and solutes. site of krebs cycle.
contains it’s own genome of DNA
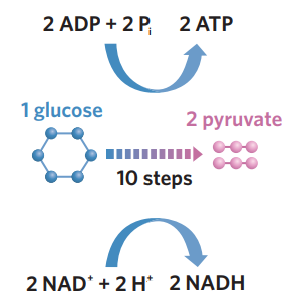
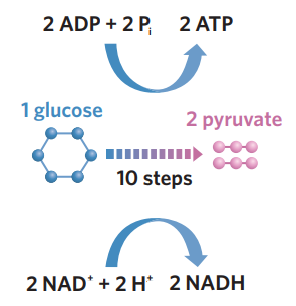
✅6A - glycolysis
glycolysis - in the cytosol, 1 glucose (6C) is broken down into 2 pyruvate molecules (3C). during this complicated process, glucose is partially oxidised (loses electrons + H+) + loses energy. NAD+ is reduced (accepts these electrons + some H+), producing 2NADH, and 2 ADP + Pi capture the energy to produce 2 ATP. pyruvate + NADH are then transported and used in other stages to generate more ATP.
1 glucose (C6H12O6) → 2 pyruvate
pyruvate molecules produced then transported to mitochondria matrix for krebs cycle, where they are further broken down and modified.
2NAD+ + 2H+ + 4e- → 2NADH
hydrogen and electrons inputs come from breakdown of glucose
NADH produced is then transported to the mitochondria for ETC
2 ADP + 2Pi → 2 ATP.
ATP produced can then be used for energy.
inputs + outputs (draw as table)
inputs
1 glucose
NAD+
2 ADP + 2 Pi
outputs
2 pyruvate
NADH
2 ATP
✅6A - link reaction
link reaction: pyruvate → acetyl-CoA + CO2 + NADH
pyruvate produced in glycolysis is transported to the mitochondria matrix. it then combines with coenzyme A (CoA) to form acetyl-CoA (2C). during this link reaction, pyruvate is further oxidised (loses electrons), and in order to capture this energy, coenzyme NAD+ is reduced (accepts these electrons), producing NADH
pyruvate has to be converted immediately because it’s toxic!!
this link reaction also releases CO2 and produces NADH that is later used in the ETC
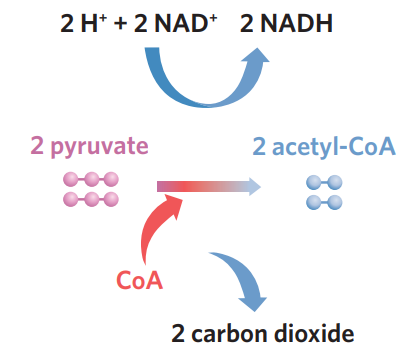
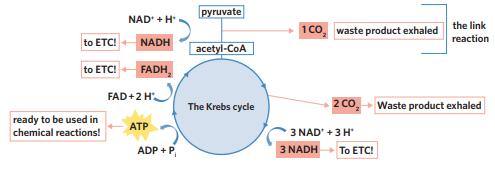
✅6A - krebs cycle
krebs cycle - generates lots of high-energy electron and proton carriers, NADH and FADH2, which can be used in the electron transport chain. Carbon dioxide is released, and small amounts of ATP are also produced.
occurs in the matrix of the mitochondria. loads co-enzymes
after link reaction, acetyl-coA can now enter the Krebs cycle in the mitochondrial matrix. coenzymes break down acetyl-CoA, extracting its energy, and then CoA is recycled back for use in the link reaction. loaded coenzymes NADH and FADH2 are high energy electron and proton carriers, which are then used in ETC. co2 is also released and 2 ATP
inputs of krebs cycle
2 acetyl-CoA (derived from pyruvate)
unloaded coenzymes
2 ADP + 2Pi
NAD+ + H+
FAD + H+
outputs of krebs cycle
CO2
loaded coenzymes (→ inner membrane for ETC)
2 ATP
NADH
FADH2
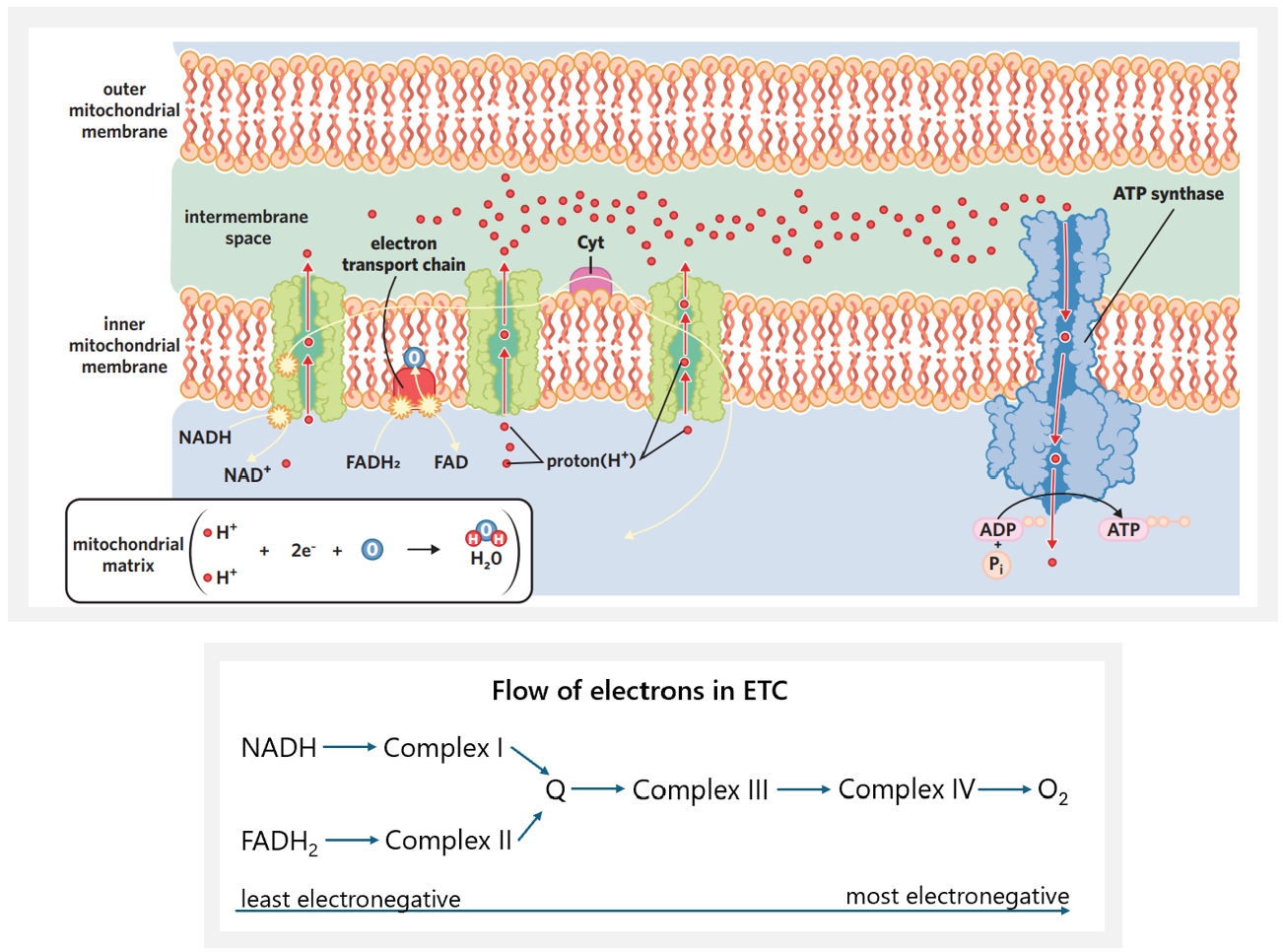
✅6A - electron transport chain
electron transport chain - using NADH and FADH2 produced in the Krebs cycle, electrons are passed between enzymes in the inner membrane/cristae and H+ is pumped from the mitochondrial matrix into the intermembrane space. this sets up a H+ gradient that ATP synthase uses to make ATP. oxygen is used when it combines with the electrons and hydrogen ions, forming the by-product water.
coenzymes unload - NADH and FADH2 unload H+ ions and electrons at the first and second protein complexes of the ETC on the inner membrane. after unloading H+ + e-, the coenzymes stay in the matrix and are recycled into the Krebs cycle, where they can become loaded again.
NADH → NAD+ + H+ + 2e-
NADH from Krebs cycle (8 NADH per glucose) + glycolysis (2 NADH per glucose) unloads H+ + e- at protein complex I. it is oxidised to NAD+ → recycled into Krebs cycle.
FADH2 → FAD + 2H+ + 2e-
FADH2 from Krebs cycle (2 FADH2 per glucose) unloads H+ + e- at protein complex II. it is oxidised to FAD → recycled into Krebs cycle.
electron movement - when electrons are unloaded into the inner membrane, they enter at protein complex I and II, and are transferred by a mobile electron carrier (nonpolar phospholipid bilayer repels charged things → electrons can’t move freely through membrane) to protein complex III. they then move to complex IV. (before they are accepted by oxygen). the movement of electrons through the ETC (see diagram) is due to oxygen present in the matrix. oxygen is very electronegative, meaning it has a high electron-pulling power. oxygen pulls electrons through the ETC. this movement of electrons powers H+ to be actively transported to the intermembrane space from transmembrane protein complexes I, III and IV. this means if oxygen runs out → electrons no longer pulled + H+ no longer pumped → ETC stops + less ATP is produced.
concentration gradient - this leads to a buildup of H+ in the intermembrane space. because this space is narrow and small, concentration of H+ quickly increases → creates a steep concentration gradient
ATP synthase - to move down their concentration gradient, H+ must travel through ATP synthase → causes ATP synthase to spin like a turbine, generating kinetic energy which powers the reaction that synthesises ATP from ADP + Pi. this produces 26 or 28 ATP per glucose molecule.
water formation - in order for the ETC to function, electrons must go somewhere, and H+/protons flow back to the matrix through ATP synthase. high concentrations of unbound electrons + protons are dangerous for cells, so to prevent a buildup in the matrix, oxygen accepts electrons at complex IV, acting as a terminal acceptor, and then combines with H+ to form water, a harmless by-product.
½ O2 + 2H+ + 2e- → H2O
inputs
26 or 28 ADP + Pi
NADH
FADH2
O2
outputs
26 or 28 ATP (some cells have the capacity to make 28 bc more efficient, some can only make 26)
NAD+ + H+
FAD + H+
H2O (H+ from NAD+ and FAD)
6B - anaerobic fermentation
anaeorbic fermentation - cellular respiration involving the breakdown of glucose when oxygen is not present. both stages occur in the cytosol. faster than aerobic respiration, but less efficient + produces harmful by-products - these can be disposed of, but accumulation is inevitable → anaerobic fermentation cannot continue indefinitely
in anaerobic conditions, glycolysis still occurs, but electrons + H+ in the ETC can’t bond with oxygen to form water → coenzymes can no longer unload + be recycled into the Krebs cycle. = the oxygen dependent pathways of the Krebs cycle + ETC are disrupted + this key source of ATP is lost. as a result, these two stages happen:
glycolysis
fermentation
ATP yield - 2 ATP per glucose molecule (all produced during glycolysis)
6B - glycolysis
glycolysis in anaerobic conditions is identical to in aerobic conditions
glycolysis - in the cytosol, 1 glucose (6C) is broken down into 2 pyruvate molecules (3C). during this complicated process, glucose is partially oxidised (loses electrons + H+) + loses energy. NAD+ is reduced (accepts these electrons + some H+), producing NADH, and ADP + Pi capture the energy to produce ATP.
1 glucose (C6H12O6) → 2 pyruvate
pyruvate molecules produced then transported to x
2NAD+ + 2H+ + 4e- → 2NADH
hydrogen and electrons inputs come from breakdown of glucose
NADH produced must be converted back to NAD+ to be recycled as an input for glycolysis → transported to x for fermentationb
2 ADP + 2Pi → 2 ATP.
ATP produced can then be used for energy.
in anaerobic glycolysis less ATP is produced??
inputs + outputs
inputs
1 glucose
2 NAD+
2 ADP + 2 Pi
outputs
2 pyruvate
2 NADH
2 ATP
6B - fermentation
after anaerobic glycolysis, NAD+ must be regenerated from NADH for anaeorbic glycolysis. also, the very toxic molecule pyruvate is produced. through the process of fermentation, NAD+ is regenerated + recyced, and pyruvate is converted into less toxic by-products, all done without oxygen.
fermentation occurs in the cytosol (also where glycolysis occurs), which allows for efficient recycling of NADH and NAD+ as the coenzymes can freely diffuse in the cytosol without needing to be transported across a phospholipid membrane. fermentation differs between plants and animals:
yeasts + plants - alchoholic fermentation
animals - lactic acid fermentation
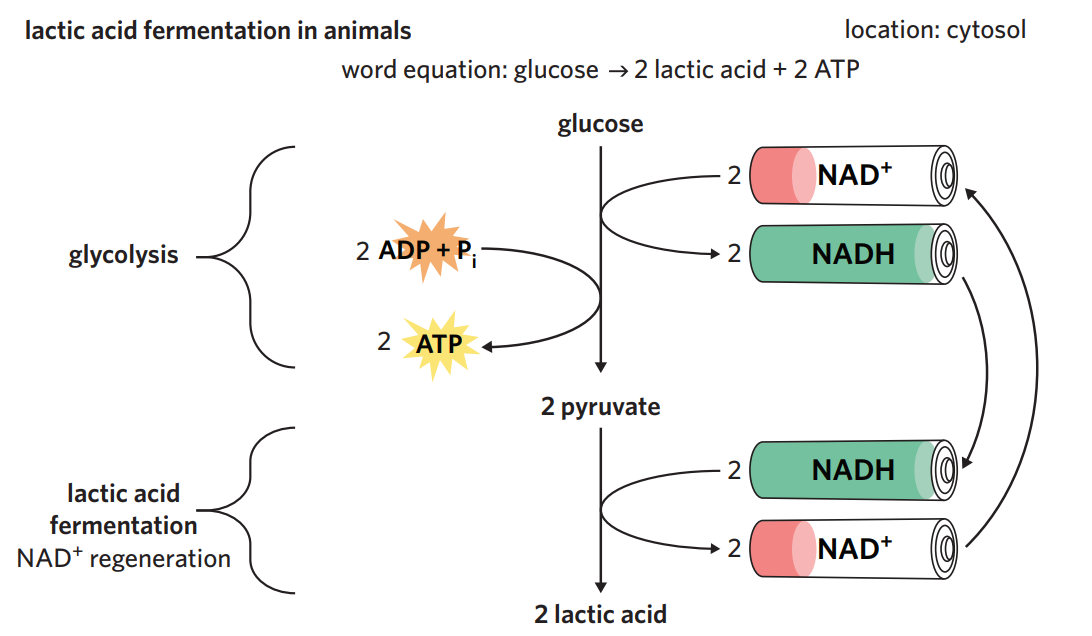
6B - lactic acid fermentation
in animals - lactic acid fermentation
when oxygen availability is insufficient, (ex. from working at high intensities), animals undertake lactic acid fermentation after glycolysis.
process - 2 x pyruvate (3C) produced from glycolysis is converted into 2 x lactic acid (3C). NAD+ is regenerated during the process + recycled back for use in glycolysis.
however, lactic acid lowers the pH of animals’ cells and blood, and can be toxic in high amounts → anaerobic respiration is not preferred; once oxygen is available, any remaining lactic acid is converted back to pyruvate so it can undergo aerobic respiration (converted into acetyl-CoA instead)
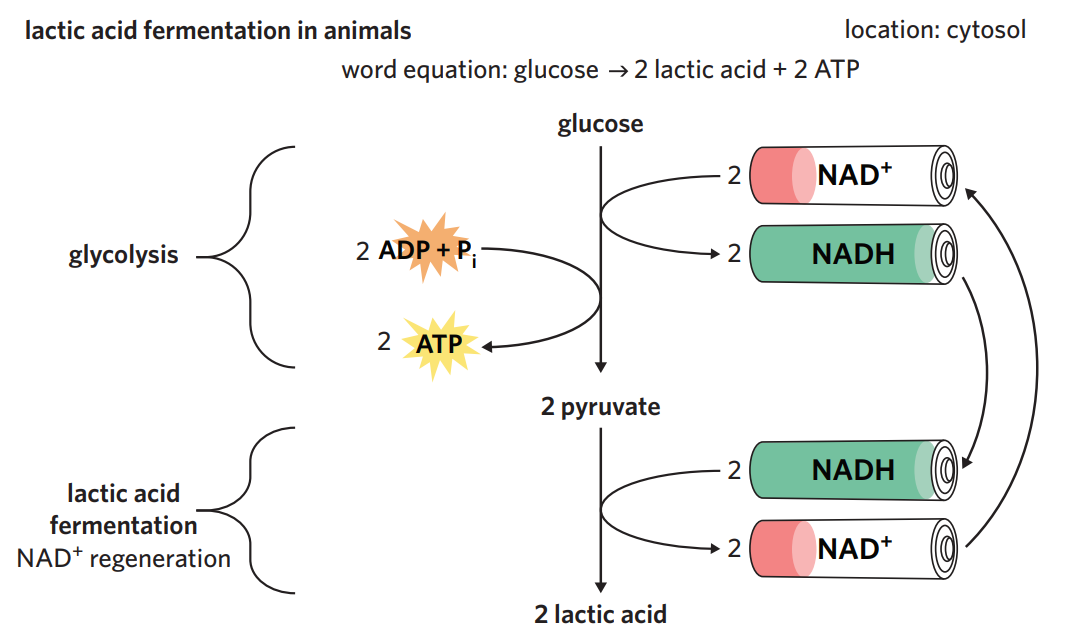
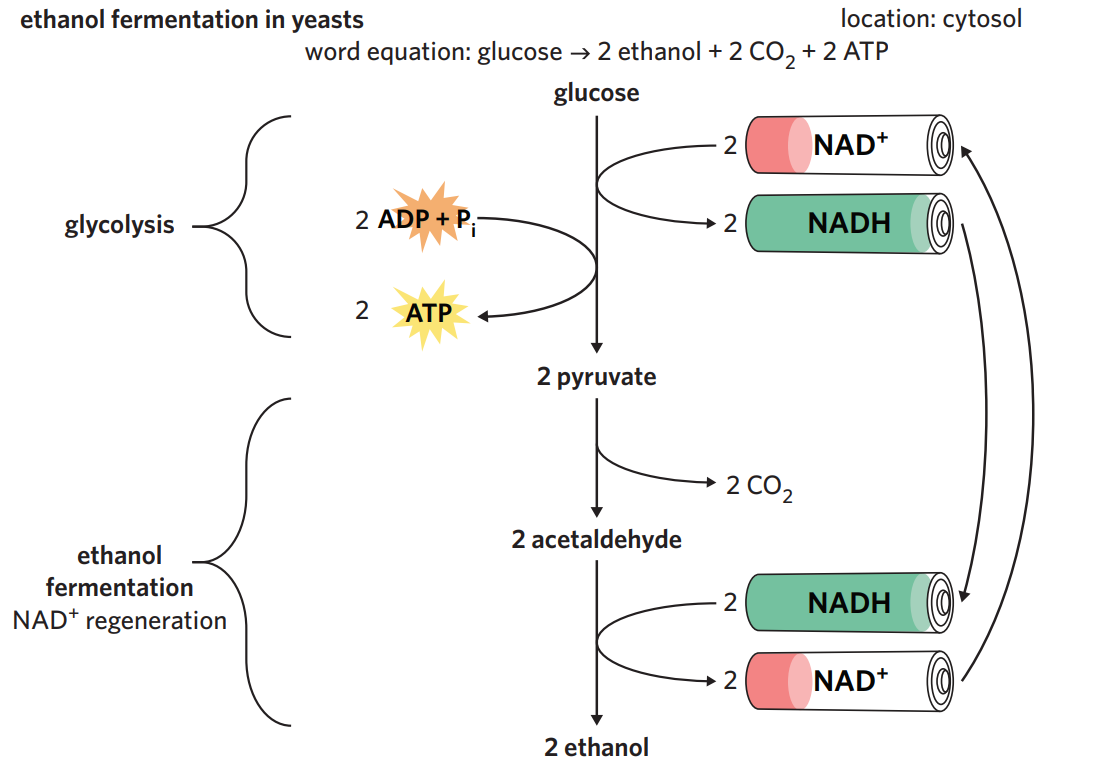
6B - alchoholic fermentation
in yeasts + plants - alchoholic fermentation
after glycolysis, yeasts + plants undergo alchoholic fermentation when oxygen isn’t present.
process - 2 x pyruvate (3C) is converted into 2 x ethanol (2C) + 2 x CO2. NAD+ is regenerated during the process + recycled back for use in glycolysis.
ethanol is a toxic by-product and can’t be converted into anything useful → ethanol easily diffuses out of yeast cells, but can accumulate to toxic levels if in a confined environment
6C - factors affecting rate of cellular respiration
factors that affect rate of cellular respiration, which therefore affect rate of ATP production:
temperature + pH
glucose
oxygen
enzyme inhibition
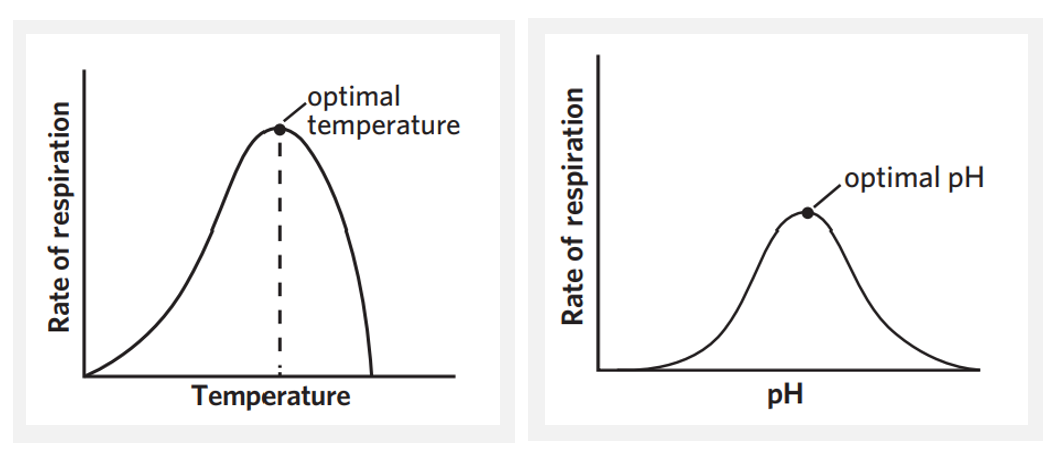
6C - temperature + pH
(CRR = cellular respiration rate)
enzymes are catalysts that speed up reactions and they are essential in cellular respiration → the factors that affect enzyme function also affect CRR
temperature
enzymes each have an optimal temperature at which their function is maximised. therefore, CRR is highest when the temperature aligns with the enzyme’s optimal temperature
below optimal temperature - enzymes + substrates have less kinetic energy → less successful collisions → slower CRR
above optimal temperature - enzymes begin to denature + CRR drops rapidly due to loss of enzyme function
pH
enzymes also have an optimal pH. in order to function at all, they must be pretty close to this pH, however it’s function is also maximised at the exact optimal pH → CRR is highest at enzymes’ optimal pH
above + below optimal pH - enzymes begin to denature
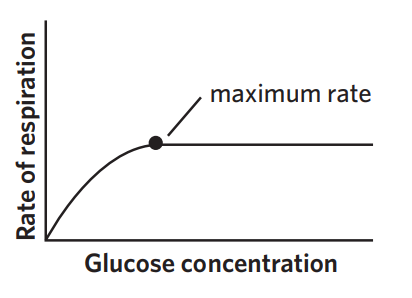
6C - glucose
glucose is the input for glycolysis (which happens in both aerobic + anaerobic respiration) → increase in glucose availability increases CRR
however, increasing glucose concentration doesn’t increase CRR indefinitely. at some point, CRR will plateu because enzymes involved have reached their saturation point at which they are operating at maximum capacity
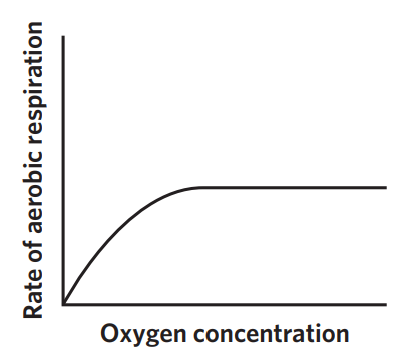
6C - oxygen
aerobic respiration requires oxygen for the ETC to function. when oxygen is not present, the cell switches to anaerobic respiration. as oxygen is increased, the CRR increases, but also only till enzymes are saturated
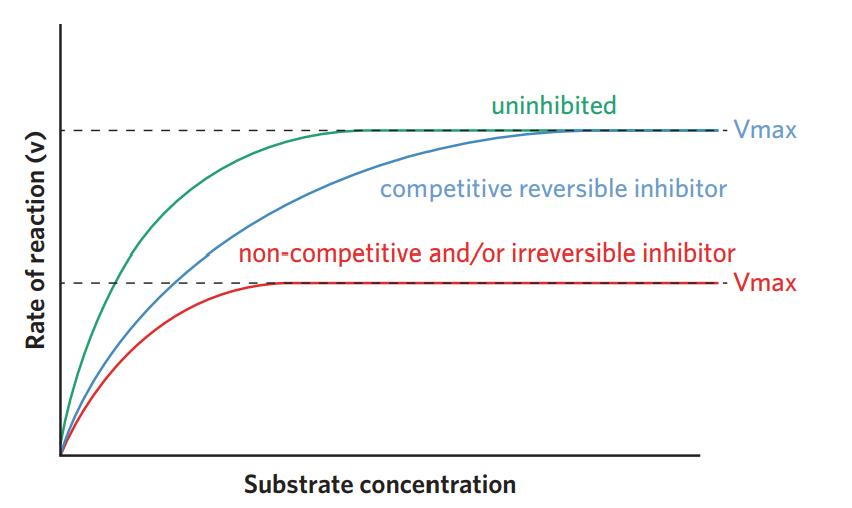
6C - enzyme inhibition
there are competitive and non-competitive enzyme inhibitors. there are also reversible and irreversible inhibitors (stop enzyme’s function even when inhibtors is displaced). all inhibitors can slow down CRR.
the effect of competitive inhibitors can be overcome if the substrate concentration is increased → displaces inhibitors. (only if reversible)
6D - biofuels NOT ON THE SAC
biofuels - fuels made from biomass (organic material from plant material and animal by-products)
biofuels are a renewable energy, meaning it can be replenished at the same or a faster rate at which it is consumed. this is because biomass can be sourced indefinitely.
biofuel production is also a carbon neutral process, as the CO2 released during production + combustion is equivalent to the CO2 that was originally captured from the atmosphere. → no net increase in CO2 in the atmosphere. CO2 in the atmosphere can then be recaptured, meaning that biofuel production simply utilises the carbon cycle.
plant material biomass - during photosynthesis, plants capture CO2 from the atmosphere, and retains this CO2 until it is released during biofuel production + combustion.
animal by-product biomass - animals eat plants (or other animals that ate plants). this means they consume carbon in plants that was captured from the atmosphere. this is just adding an extra step in the carbon cycle, as the CO2 released from animal by-product biofuel is equivalent to the CO2 that the original plants captured.
biofuels also ‘burn clean’, meaning they undergo complete combustion, and don’t release pollutants upon combustion.
6D - bioethanol
bioethanol - a biofuel which is produced from the anaerobic fermentation of glucose (from biomass) using yeast, which produces ethanol which can be harnessed to produce bioethanol
process
deconstruction
biomass is broken down to increase surface area in order to make the fermentation process more efficient. this can be done in many ways, such as the physical grinding of the biomass.
digestion by enzymes
the starch/cellulose from the broken down biomass is then exposed to enzymes + further broken down and converted into glucose
(starch if from first generation biomass, cellulose if from second generation biomass)
ethanol fermentation
glucose is put in solution with water and the microorganism yeast. yeast cells use glucose as an input, and thus yeast facilitates the anaerobic fermentation of glucose.
yeast prefers to undergo cellular respiration through the anaerobic pathway. also, the aqueous environment means that the oxygen (if any) is used up quickly → ensures glucose undergoes anaerobic fermentation (which produces ethanol) rather than aerobic respiration
= a large amount of ethanol is produced as a by-product of this anaerobic fermentation. this ethanol diffuses out of the yeast cells + is captured for use as a biofuel.
purification and dehydration
the ethanol is distilled, removing water, converting it into a usable biofuel. it is then purified + ready to be used as liquid fuel
6D - application of biofuels
biofuels can be used as a renewable alternative to fossil fuels in many ways.
transportation - biofuels can replace traditional fuels such as petrol and diesel, as they are able of combusting in most engines. ex. bioethanol is blended with gasoline to cut down on smog-causing emmissions, often 10% bioethanol, 90% gasoline, E10
more uses include cleaning, heating homes, backup energy generation
6D - first vs second generation biofuels
the ‘generation’ of a biofuel relates to the type of biomass that it is sourced from
first generation biofuels - produced directly from food (ex. corn, sugar-cane). starch is broken down.
→ competes with food crops → can impact food security
if more crops grown deliberately for biofuel → can involve land degradation + loss of biodiversity
easy conversion into biofuels
second generation biofuels - produced from cellulose-based non-food crops (ex. straw, waste paper) left over from other crops/processes. cellulose is broken down.
→ doesn’t compete with food crops
more difficult to break down + convert into biofuels
6D - implications + ethics of biofuels
strengths
climate impact - biofuels are carbon neutral, compared to fossil fuels, which produce lots of CO2 emissions → excess CO2 causes enhanced greenhouse effect → climate change + extreme weather events. replacing fossil fuels with biofuels mitigates enhanced greenhouse effect.
energy security - biofuels are a renewable energy, while fossil fuels are not (take millions of years to form). as our energy demands continue to increase, the finite supply of fossil fuels diminishes. replacing fossil fuels with biofuels decreases our reliance on non-renewable energy → energy security.
localised energy - biomass can be sourced + farmed all around the world, but fossil fuels cannot, and rely on international imports + exports. replacing fossil fuels with biofuels helps nations rely less on these imports + exports, and rather produce their own fuel, thus increasing energy independence + security of nations, decentralising control over fuel supplies, increasing job opportunities and reducing the risks associated with fossil fuel transport (such as oil spills).
weaknesses
food vs fuel - first generation biofuels compete with food crops, which can lead to food insecurity.
second order environmental impacts - 1st and 2nd gen biofuels require farmland, which can lead to deforestation, loss of biodiversity and land degradation. farming can also involve fertiliser, which can pollute waterways + release nitrous oxide
cost + difficulty of uptake - biofuels are more expensive to produce than fossil fuels, and there is also a lack of infrastructure to support biofuels → difficult to penetrate the market
experimental design
to create an experient:
hypothesis
independent variable
dependent variable
constant variables
control
expected results
repeatability - multiple trials under exactly same conditions ex. one person can do experiment multiple times
reproduceable - same thing but something has been slightly changed. ex. whole class can all do experiment
validity - show only one independent variable (with control)
precise - multiple trials
accuracy - true value to compare it to + it’s similar (most of the time don’t know)