Oceans
1/80
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
81 Terms
lattice enthalpy
energy change when one mole of solid is formed by the coming together of separate gaseous ions
All lattice enthalpies are
negative
What amount of energy do you need to put in to break down a lattice?
-∆LE H
Lattice enthalpies become more negative
ionic charge increases
ionic radii decreases
Lattice enthalpy is more negative
Why do ions with higher charge densities have more negative lattice enthalpies?
higher charges attract more strongly
closer ions attract more strongly
ions with smaller radii can come closer together and attract more strongly
Stronger attraction
more negative lattice enthalpy
Why are water molecules polar and behave as a dipole?
covalent bonds are polar
bent shape means molecule is polar
Hydration
polar water molecules move to surround ions
negative end of dipole faces positive ions
positive end of dipole faces negative ions
If water dipoles bond weakly, the ion is
not extensively hydrated
If water dipoles bond strongly, the ion is
extensively hydrated
Higher charge density means for a hydrated ion
more water molecules attracted
bigger hydrated ion
Why may hydration overcome lattice enthalpies?
Strong ion–dipole attractions
Small, highly charged ions
Ions with high charge density attract water molecules very strongly → more -hydration enthalpyenergy released during hydration is greater than the energy needed to break the lattice
→ overall energy change is negative
→ dissolution is energetically favorable
Breaking hydrogen bonds in water requires
energy
Enthalpy change hydration
enthalpy change for formation of a solution of ions from one mole of gaseous ions
Enthalpy change of hydration depends on
concentration
Why do we use very dilute solutions for enthalpy change of hydration?
assume interactions between ions are negligible
Most exothermic enthalpy change of hydration
greatest charge
smallest radii
Enthalpy change of solution
enthalpy change when one mole of solute dissolves to form a very dilute solution
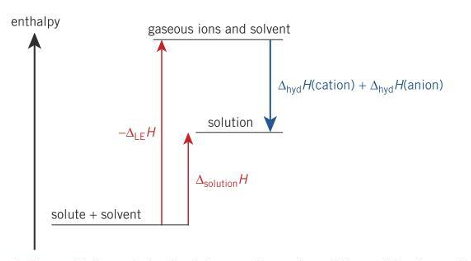
Equation of enthalpy change solution
∆solutionH= ∆hydH(cation) + ∆hyd(anion) - ∆LEH
When does a solution normally dissolve?
∆solH is negative
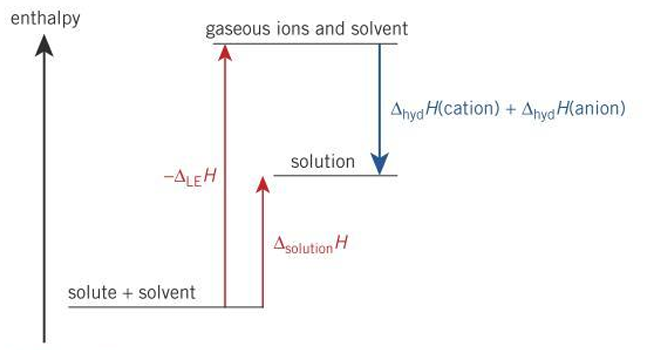
when does a solute not dissolve?
∆solH is large and positive
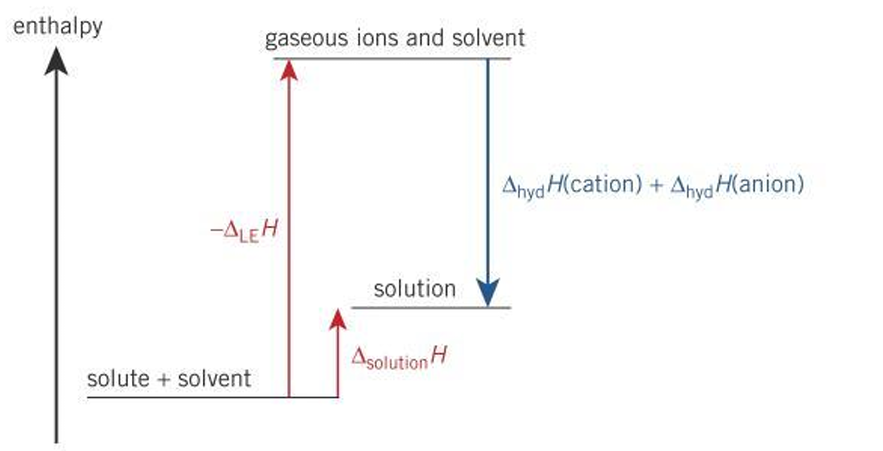
Why does an ionic substance dissolve when reaction appears energetically unfavourable?
∆solH is slightly positive
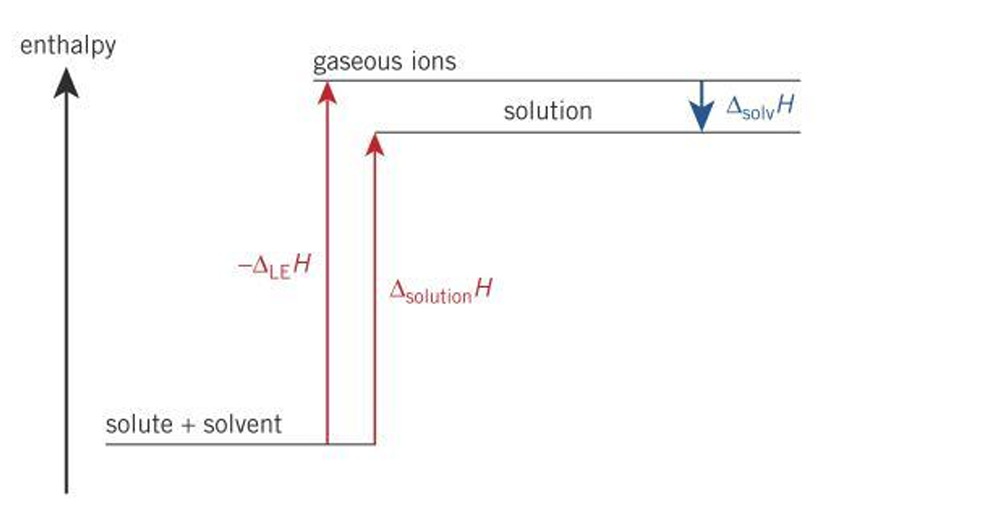
Enthalpy diagram for an ionic substance in a non-polar solvent
What happens to energy absorbed by greenhouse gas molecules?
Increase vibrational energy of molecules so bonds vibrate more and transfer kinetic energy to other molecules by collision, increasing temperature.
Some radiation is re-emitted to Earth and space
IR window
wavelengths of IR water vapour does not absorb so escape into space
Carbon dioxide _____________ wavelengths in the IR window
absorbs
The higher the temperature, the _________ the level of water vapour.
higher
Bronsted Lowry theory of acids and bases
Acids are H⁺/ proton donors
Bases are H⁺/ proton acceptors
conjugate base
remains after acid donates H⁺
conjugate acid
forms when base accepts H⁺
Conjugate acid base of HCl (aq)
Cl⁻
Conjugate acid of NH3
NH4+
Water acting as a base with HCl
HCl (aq) + H₂O (l) → H₃O⁺ (aq) + Cl⁻ (aq)
Water acting as an acid with NH3
H₂O + NH₃ → OH⁻ + NH₄⁺
pH
-log [H⁺]
low pH
high [H⁺]
Strong acids
strong tendency to donate H⁺
dissociate completely in aqueous solution
Reaction strong acid HA with water
HA (aq) + H₂O (l) → H₃O⁺ (aq) + A⁻ (aq)
Examples strong acids
HCl
H₂SO₄
weak acid
weak tendency to donate H⁺
does not dissociate completely in water
Dissociation weak acid HA
HA (aq) ⇌ H⁺ (aq) + A⁻ (aq)
What does the equilibrium sign show?
a significant amount of HA molecule is present
The further the equilibrium lies to the right-hand side, the ________ the acid
stronger
How to calculate pH of strong acid?
-log [HA= H⁺]
Assumptions for calculating pH of weak acids?
[H⁺] = [A⁻] (including H⁺ from water since very small amount)
[HA] in equilibrium = [HA] put into solution (very small fraction H⁺)
How to calculate pH of weak acid?
Acidity constant
-log[H⁺]
Acidity constant
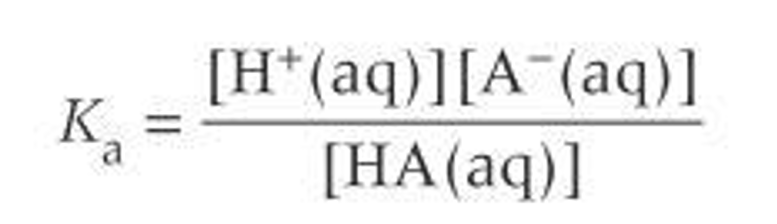
How to calculate pKa?
pKa= -log Ka
How to calculate pH of strong alkali?
Ionisation product of water
-log[H⁺]
Equation Kw water?
Kw = [H⁺] [OH⁻] / [alkali]
[alkali] = [OH⁻]
alkali in excess so ignore
Kw= [H⁺][OH⁻]
The larger the Ka value
the stronger the weak acid
The larger the pKa value
the weaker the weak acid
Buffer
resists changes in pH on addition small amounts of acids and alkalis
What are buffer solutions made from?
Weak acid + salt
Weak base + salt
Assumption about buffers made from weak acid and salt?
All A⁻ come from salt
Almost all HA put into buffer remain unchanged
What happens in a buffer when H+ are added?
A⁻ from salt react with H⁺ to form HA and water
removes excess H⁺ and pH re-established
What happens when OH- are added in a buffer?
H⁺ ions are removed to form water
H⁺ ions are regenerated by HA and pH is re-established
How does a solution of H+ and A- ions act as a buffer?
HA → H+ + A-
When H+ is added, equilibrium shifts to the left to form acid and remove excess H+
When OH- is added, equilibrium shifts to the right to replace H+
Both H+ and OH- are in excess
Ka buffer using assumptions
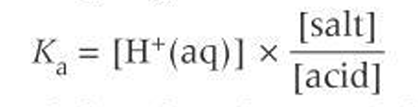
What does the [H+] in a buffer depend on?
value Ka
salt: acid
Why is pH of buffer not affected by dilution?
concentration of salt and acid are reduced equally
Ksp
Solubility product
What does Ksp represent?
conditions for equilibrium between sparingly soluble solid and saturated solution
How to calculate Ksp for reaction AB → A + B?
[A]a[B]b
Ion product, Q
state of the solution right now
How to find Q?
[conc at that moment]
Why does precipitate form when Q>Ksp?
ions present in higher concentration than the solution can hold,
extra solid precipitates out.
If value Q in excess of Ksp?
precipitate out
If Q smaller than Ksp?
no precipitate
If Q=Ksp?
saturated
Solubility
maximum amount of a substance that dissolves in water to form a saturated solution
mol/L
How to calculate solubility of HA → H+ + A-
[H+]=[A-]
Ksp= s x s = s²
s= Ksp1/2
Entropy
measure of number of ways of arranging molecules and distributing energy
When does a collection of molecules have higher entropy?
spread out more
energy is shared among more molecules
How to calculate total entropy change?
∆totalS= ∆sysS + ∆surrS
How to calculate enthalpy change of surroundings?
-∆H/ T
When is reaction feasible?
positive ∆totalS
What kind of value is ∆totalS for spontaneous reactions?
positive
What happens when ∆totalS is 0?
equilibrium is constant
Kc= 1
When are positive ∆totalS reactions not spontaneous?
large activation enthalpy