Berzins lecture 1
0.0(0)
Studied by 2 peopleCard Sorting
1/51
Earn XP
Description and Tags
Last updated 3:54 AM on 10/17/22
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
52 Terms
1
New cards
metal
electropositive element
2
New cards
general properties of metals
-luster
- good thermal and electrical conductors
-high fracture toughness
- stronger, more ductile, and denser than nonmetals
- good thermal and electrical conductors
-high fracture toughness
- stronger, more ductile, and denser than nonmetals
3
New cards
alloy
A mixture of two or more metals (sometimes with nonmetals)
4
New cards
classification of metals
elemental vs alloy
cast vs wrought
noble vs non-noble
application and composition
cast vs wrought
noble vs non-noble
application and composition
5
New cards
bonding and structure of metals
metallic bonding, crystalline
6
New cards
crystal systems of metals
simple cubic
face centered cubic
body centered cubic
hexagonal close-packed
etc
face centered cubic
body centered cubic
hexagonal close-packed
etc
7
New cards
noble metals in dentistry
Palladium
Ruthenium
Osmium
Gold
Rhodium
Iridium
Platinum
Ruthenium
Osmium
Gold
Rhodium
Iridium
Platinum
8
New cards
3 major noble metals
gold, palladium, platinum
9
New cards
metals used as grain refiners
ruthenium and iridium
10
New cards
pure base metal vs pure noble metal in mouth
base metals have a greater tendency to corrode in the mouth compared to noble metals
11
New cards
high noble alloy
gold > 40% wt
noble metal content > 60wt%
noble metal content > 60wt%
12
New cards
Titanium and Titanium Alloys
titanium > 85%wt
13
New cards
noble alloys
noble metal content > 25% wt
14
New cards
predominantly base alloys
noble metal content
15
New cards
ADA type I alloy
hardness: soft
tensile yield strength:
tensile yield strength:
16
New cards
ADA type II alloy
hardness: medium
tensile yield strength: 140-200
elongation (%): 18
tensile yield strength: 140-200
elongation (%): 18
17
New cards
ADA type III alloy
hardness: hard
tensile yield strength:201-340
elongation (%): 12
tensile yield strength:201-340
elongation (%): 12
18
New cards
ADA type IV alloy
hardness: extra-hard
tensile yield strength: >340
elongation (%): 10
tensile yield strength: >340
elongation (%): 10
19
New cards
ADA alloy classification by physical properties
from type I to IV, hardness and tensile yield strength increases, elongation decreases
20
New cards
application of ADA alloy type I
sustainable stress: low, no occlusion
use: inlays
use: inlays
21
New cards
application of ADA alloy type II
sustainable stress: moderate, light occlusion
use: inlays and onlays
use: inlays and onlays
22
New cards
application of ADA alloy type III
sustainable stress: high, full occlusion
use: crowns, short-span FPD
use: crowns, short-span FPD
23
New cards
application of ADA alloy type IV
sustainable stress: very high
use: thin veneers, long-span FPD, RPD
use: thin veneers, long-span FPD, RPD
24
New cards
homogenous nucleation
solid forms from the liquid, sometimes requires 'supercooling'
25
New cards
solidification of metals
pure metals have a melting point, alloys have a melting range
26
New cards
grain boundaries
The boundaries between crystals (grains) in a polycrystalline material.
27
New cards
raid vs slow cooling of metals
rapid cooling --> more nuclei --> smaller grains
slow cooling --> fewer nuclei --> larger grains
slow cooling --> fewer nuclei --> larger grains
28
New cards
grain refiners
method used to reduce grain size
- add
- add
29
New cards
finer grain size (vs larger grains)
increased yield strength, composition uniformity, and corrosion resistance
30
New cards
dendrites
in pure metals, the dendrite formation is through thermal supercooling
- dendrite growth is along specific crystallographic directions
- dendrite growth is along specific crystallographic directions
31
New cards
solid solution
created with incorporation of foreign atoms (solute) into a crystal structure of the matrix atoms (solvent)
- may be a wide range of compositions
- may be a wide range of compositions
32
New cards
substitutional solid solution
B atoms replace where A atoms should be (disordered and ordered)

33
New cards
interstitial solid solution
B atoms fit in-between the A atoms

34
New cards
disordered vs ordered substitutional solid solution
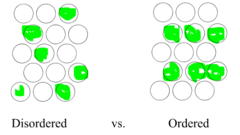
35
New cards
amount of solute 'in solution' depends on:
1. size
2. crystal structure
3. valence
4. chemical reactivity
2. crystal structure
3. valence
4. chemical reactivity
36
New cards
size of solute
- substitutional solubility decreases as size difference increases (
37
New cards
crystal structure and solubility
greater solubility if same (eg FCC)
38
New cards
valence and solubility
greater solubility if same
-eg. Mn2+ and Zr4+ are similar size but charge makes solution improbable
-eg. Mn2+ and Zr4+ are similar size but charge makes solution improbable
39
New cards
eutectic point
liquid solidifies into two solid phases
40
New cards
intermetallic compound
differ from solid solutions int that they exist as fixed anatomic ratios
eg. NiTi, Ti2Ni, TiNi3
eg. NiTi, Ti2Ni, TiNi3
41
New cards
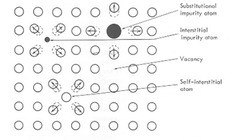
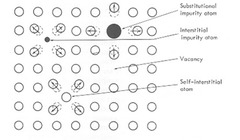
point defect
substitution or interstitial atoms, vacancies, self-interstitial
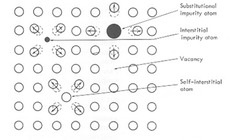
42
New cards
vacancy
vacant lattice site
43
New cards
self-interstitial
a matrix atom in an interstitial site
44
New cards
dislocations
line imperfections in crystalline solids
- atoms missing or extra-half-planes of atoms are formed
- decrease the strength
- atoms missing or extra-half-planes of atoms are formed
- decrease the strength
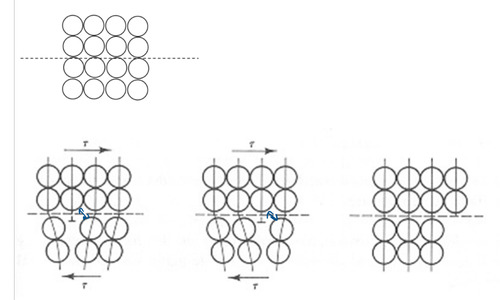
45
New cards
plastic deformation of metals
hinder dislocation movement --> increase strength
46
New cards
strengthening mechanisms of metals
- solid solution strengthening
- strain or work hardening
- precipitation hardening
- transformation strengthening
- strain or work hardening
- precipitation hardening
- transformation strengthening
47
New cards
solid solution strengthening of metals, general features
1. higher strength and hardness, but less ductile than either pure metal
2. melting range, melt below highest melting point of the pure metals
3. higher corrosion resistance than multi-phase alloy
2. melting range, melt below highest melting point of the pure metals
3. higher corrosion resistance than multi-phase alloy
48
New cards
Strain hardening (cold working)
strength and hardness increase, ductility decreases
-- with plastic deformation, it becomes more difficult to deform more
-- increase in number and interaction between dislocations
-- with plastic deformation, it becomes more difficult to deform more
-- increase in number and interaction between dislocations
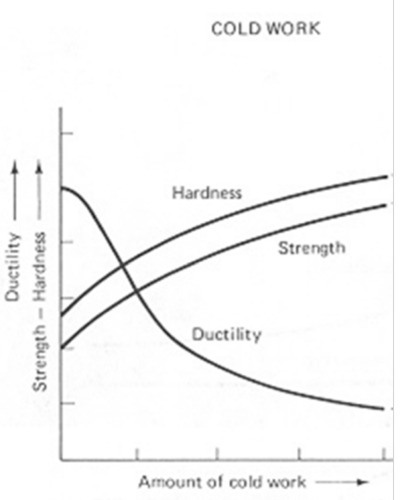
49
New cards
annealing
heat treatment used to soften metals and refine their grains
50
New cards
Recrystallization
above a certain temp new grains nucleate
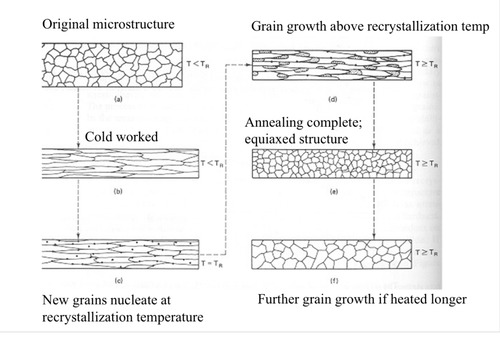
51
New cards
precipitation hardening of metals
solute atoms are not dissolved but form separate 2nd phase particles dispersed in the matrix
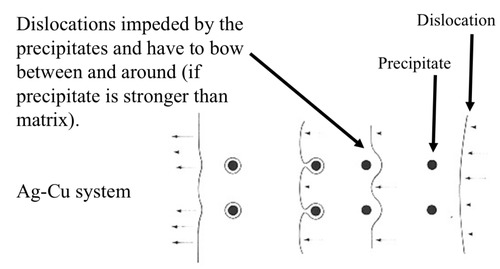
52
New cards
transformation strengthening of metals
change in the morphology of the lattice structure which occurs upon heating or cooling
- titanium: BCC to HCP
--- strained lattice retards dislocation movement
- titanium: BCC to HCP
--- strained lattice retards dislocation movement