Bootcamp FL 1 - OC
1/29
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms
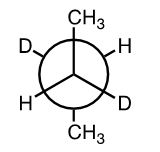
Which statement best describes the conformation of the following molecule?
A. Most stable, staggered, and gauche
B. Least stable, eclipsed, and syn
C. Most stable, eclipsed, and syn
D. Least stable, staggered, and anti
E. Most stable, staggered, and anti
E
The molecule below is a Newman projection, looking down from C2 to C3 in this deuterated butane:
All groups are spaced out as far as possible, meaning that the conformation is staggered.
The two bulkiest substituents (methyls) are 180 degrees from each other, meaning that the conformation is anti.
The staggered anti conformation is more stable than the staggered gauche and eclipsed conformations.
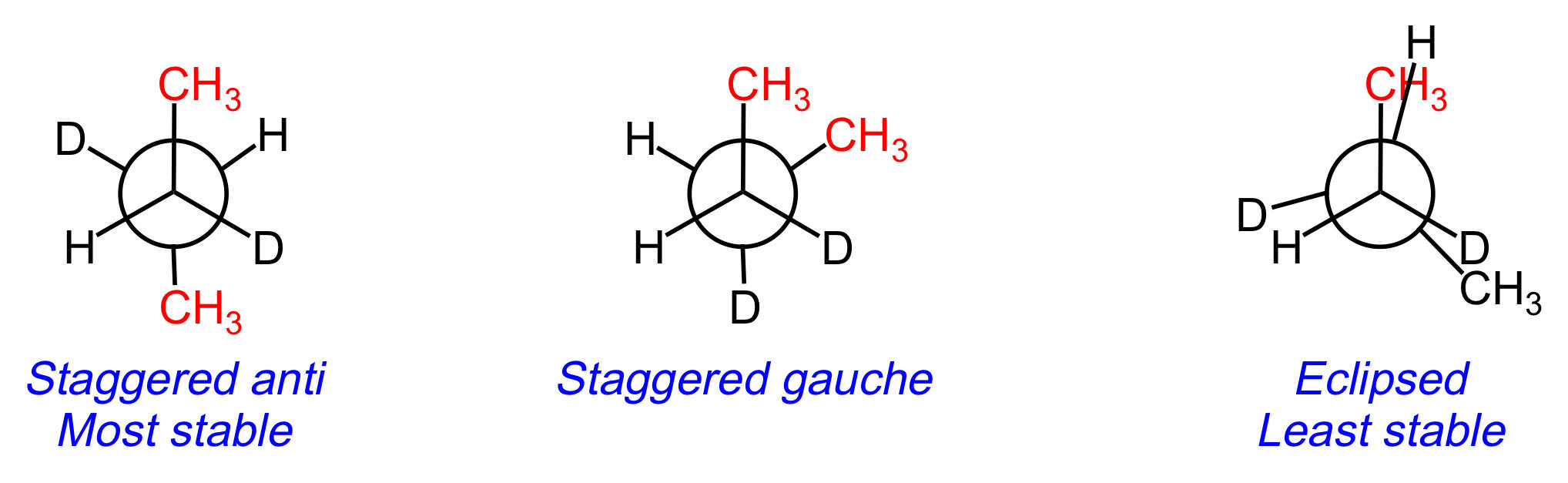
Key Takeaway:
The most stable conformation for a Newman projection is staggered anti.
Which of the following is an intermediate in the bromination of toluene?
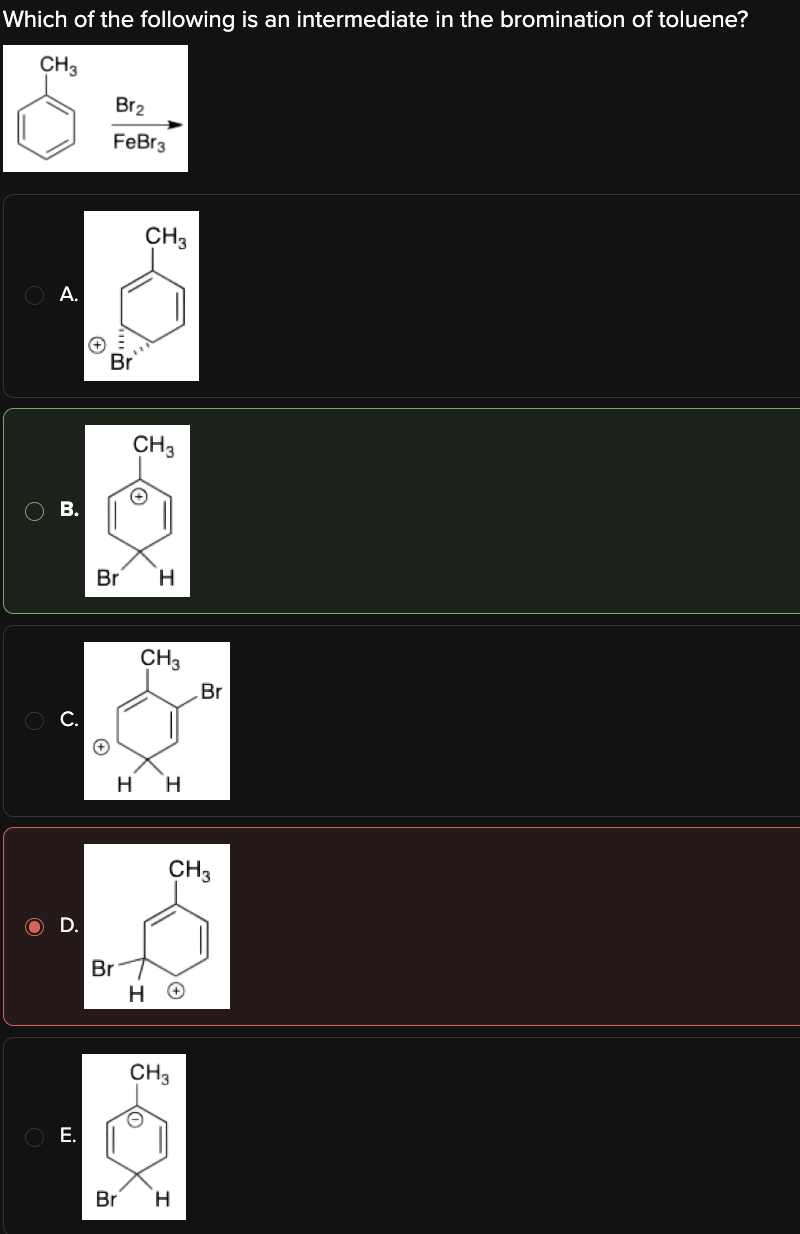
B
The bromination of toluene is an electrophilic aromatic substitution reaction.
The methyl group on toluene is a strong ortho/para director, so that the electron-rich aromatic ring attacks the electrophile (Br) in the para position.
The resulting carbocation intermediate is unstable due to the positive charge and the temporary loss of aromaticity. However, it is partially stabilized by resonance, which allows the positive charge to be distributed:
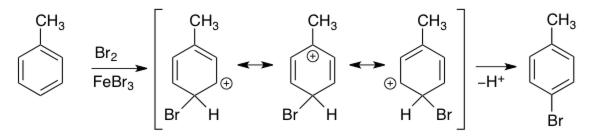
Key Takeaway:
In electrophilic aromatic substitution reactions, alkyl groups are strong ortho/para directors.
Which of the following structures both has a molecular formula of C5H10O and corresponds to the following infrared spectrum?
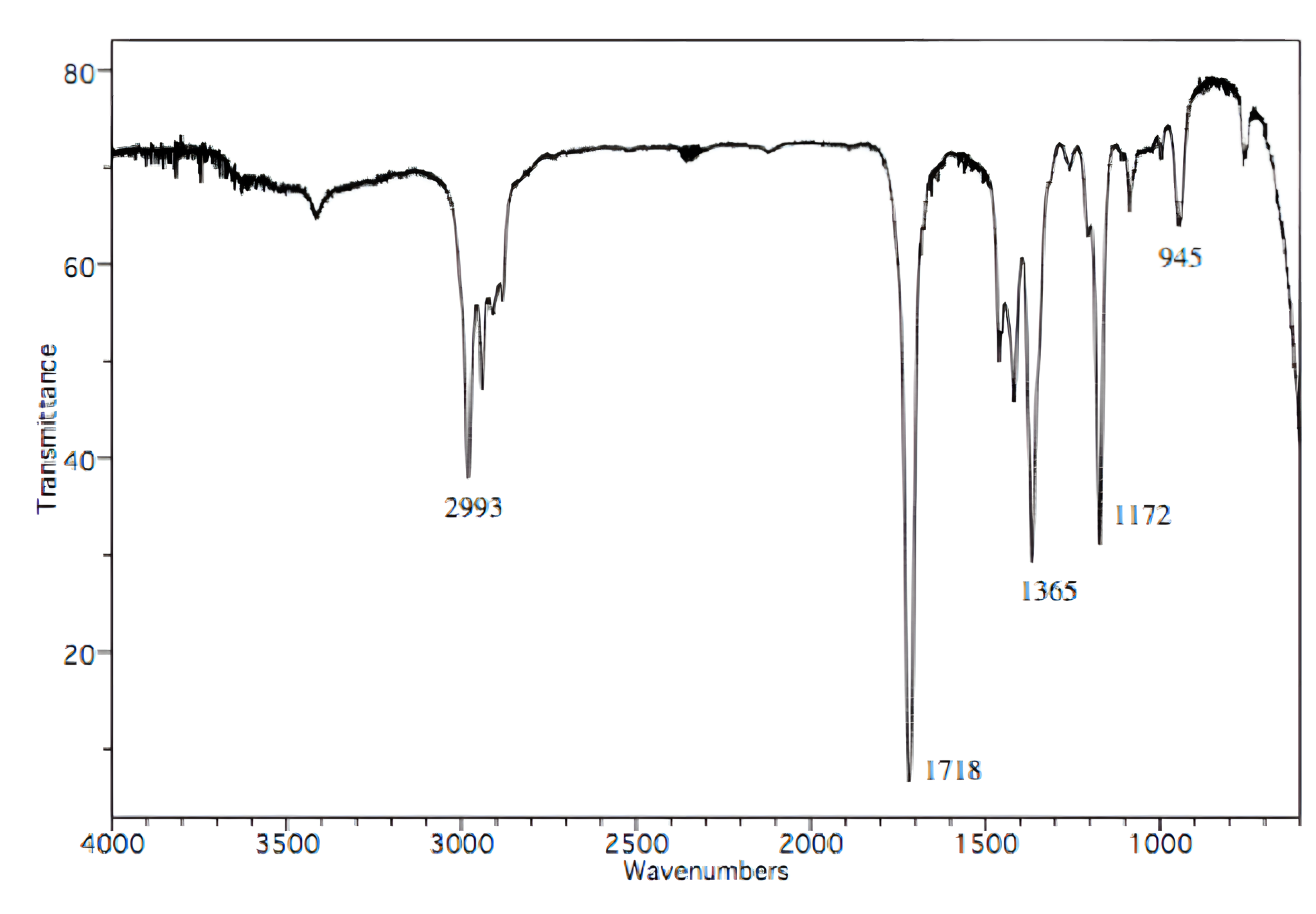
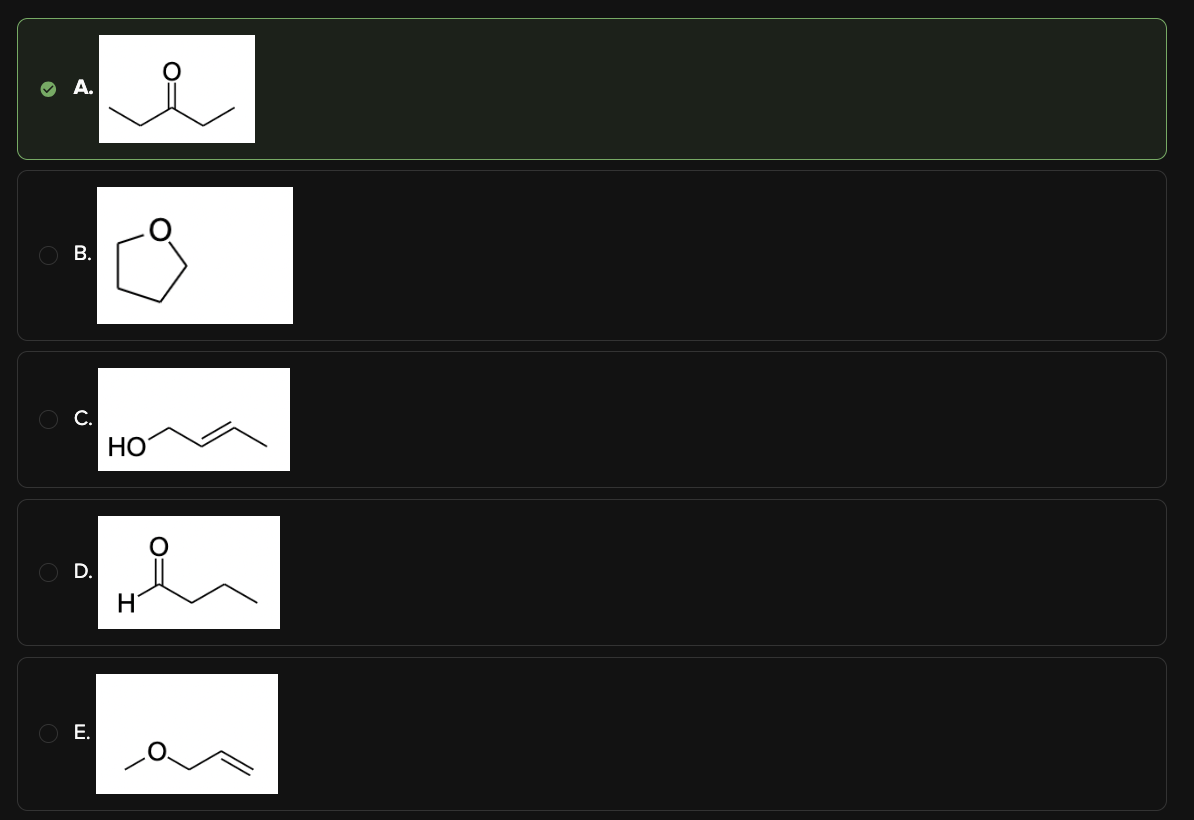
A

Infrared (IR) spectroscopy is a technique used to determine a molecule's structure by analyzing how its chemical bonds absorb infrared light. Each type of bond produces unique peaks on the IR spectrum.
To begin, assess which functional groups are present in the molecule based on the given IR spectrum. A carbonyl group (C=O) produces a large, sharp peak within the 1700 ± 50 cm-1 range, indicating the compound contains a carbonyl group.
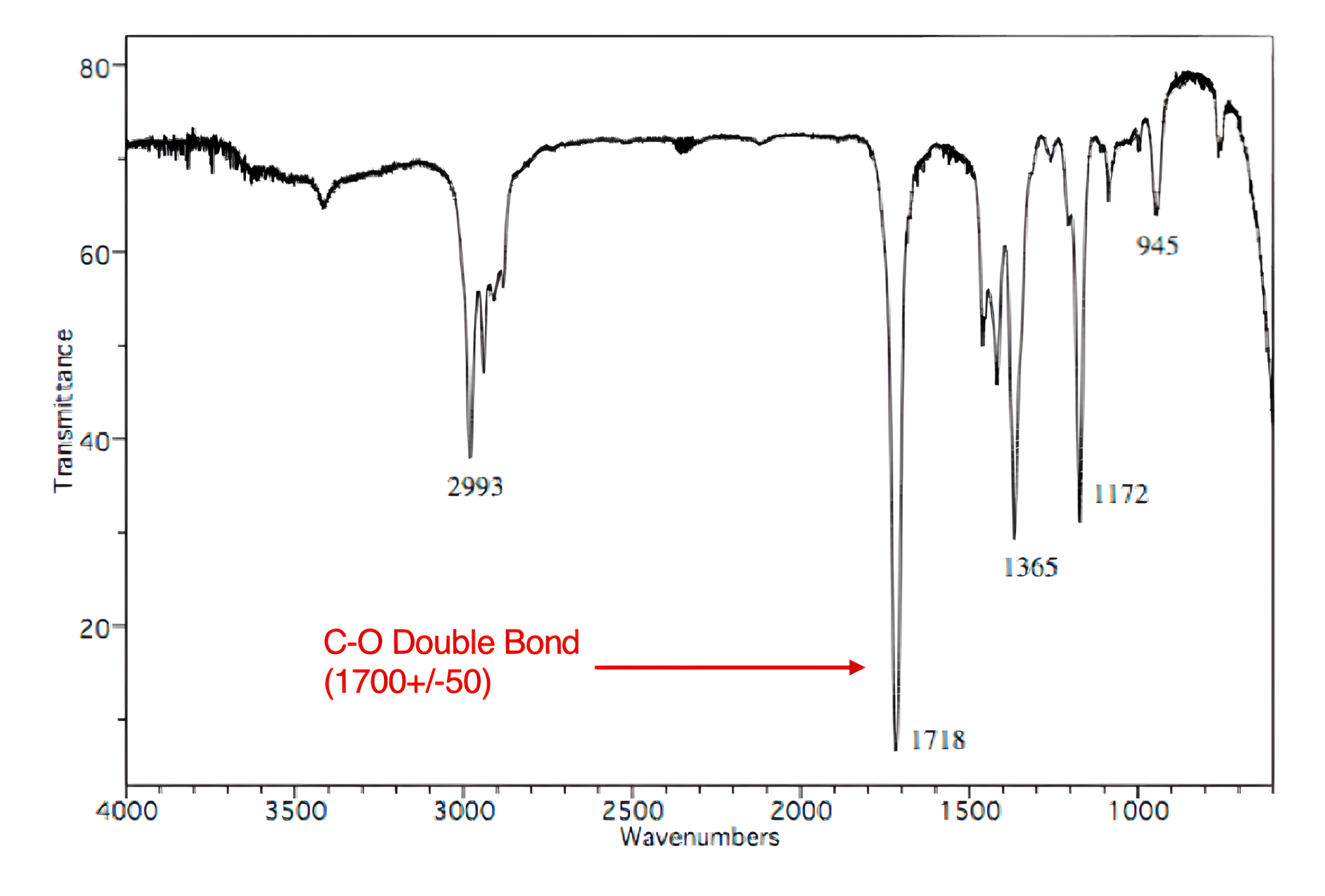
Choices [A] and [D] are the only options that include a carbonyl group.
The molecular formula of the compound is C5H10O. Among the remaining options, only choice [A] matches this formula.
Key Takeaway:
Carbonyl groups (C=O) produce large, sharp peaks within the 1700 ± 50 cm-1 range on an IR spectrum.

Draw this table
Recall table
What is the best reagent for the following reaction?
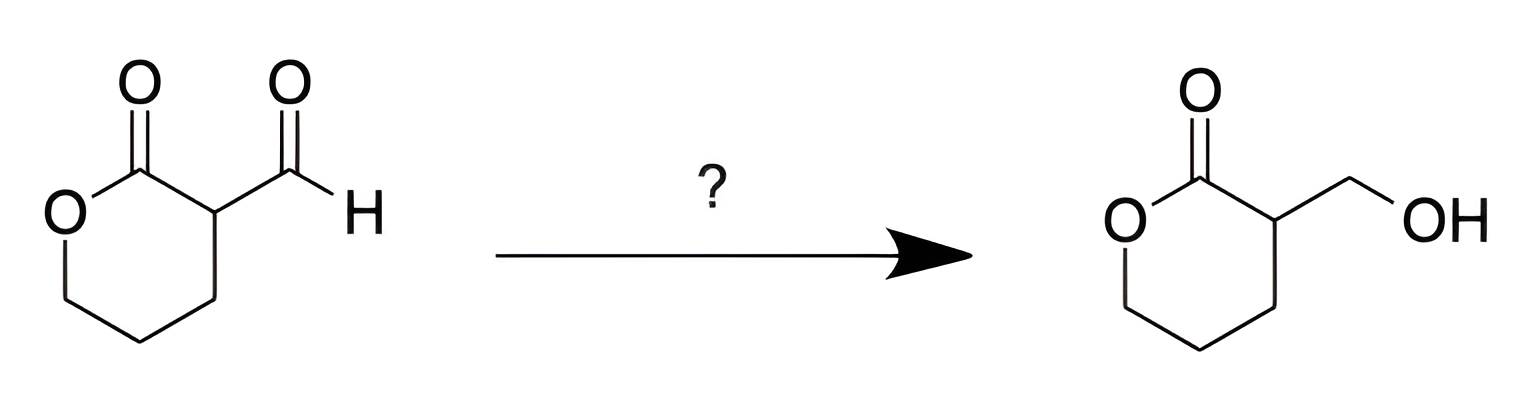
A. H2CrO4
B. LiAlH4
C. DIBAL-H, -78ºC / H2O
D. H2 / Ni
E. NaBH4
E
We are asked to determine the reagent that will convert an aldehyde into a 1° alcohol.
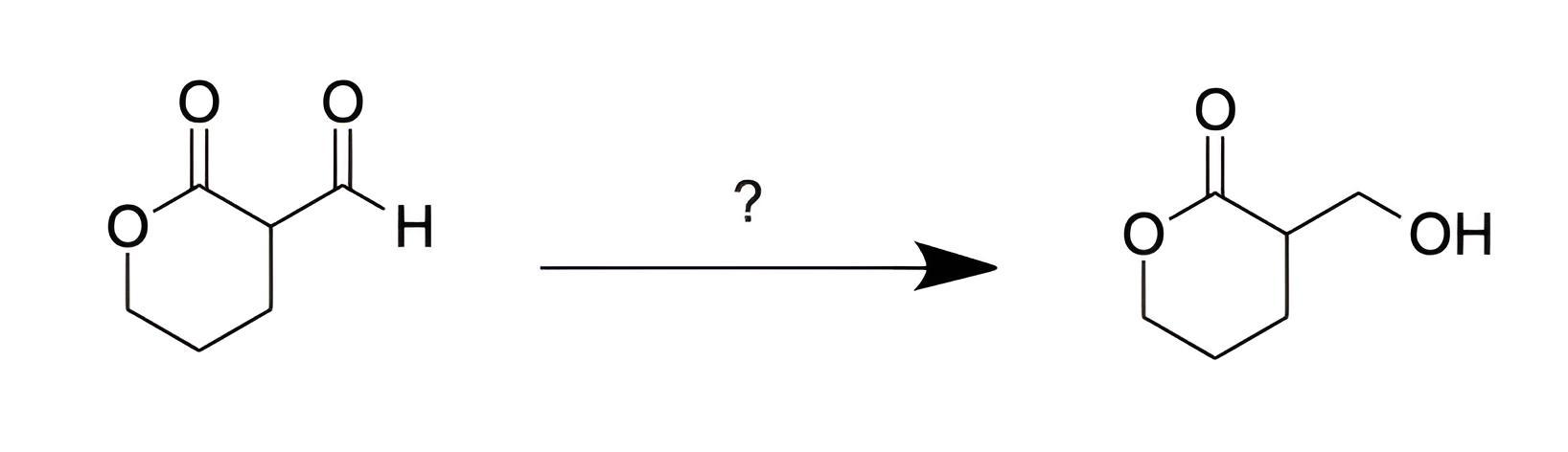
For the purposes of DAT, there are several important hydride reductions to be familiar with:
NaBH4 reduces aldehydes, ketones, acid chlorides, and acid anhydrides.
LiAlH4 reduces all acyl functional groups.
DIBAL-H, -78 °C / H2O reduces esters to aldehydes.
According to the list above, both NaBH4 and LiAlH4 reduce aldehydes to 1° alcohols. Eliminate Choices [A], [C], and [D].
It is important to note that an ester is present in both the reactant and product. Therefore, we are looking for a reducing reagent that will reduce aldehydes to 1° alcohols, but will not reduce esters.
NaBH4 is not powerful enough to reduce esters, but LiAlH4 will reduce esters to 1° alcohols. Choice [E] is the answer.
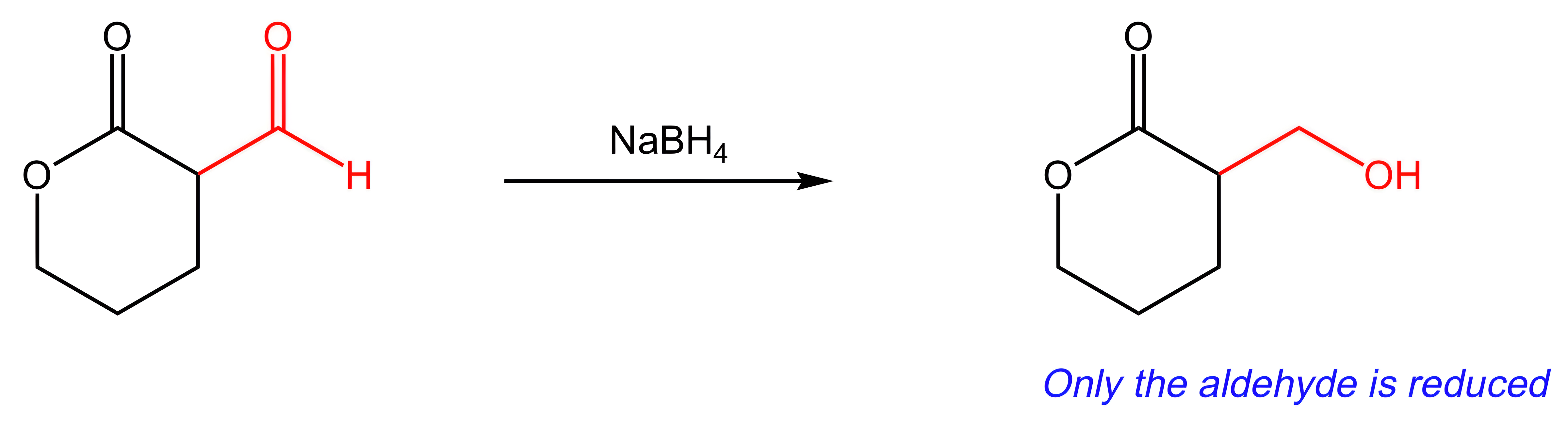
A. H2CrO4
H2CrO4 is an oxidizing agent. Because the reaction shown in this question is a reduction, oxidizing reagents will not be useful.
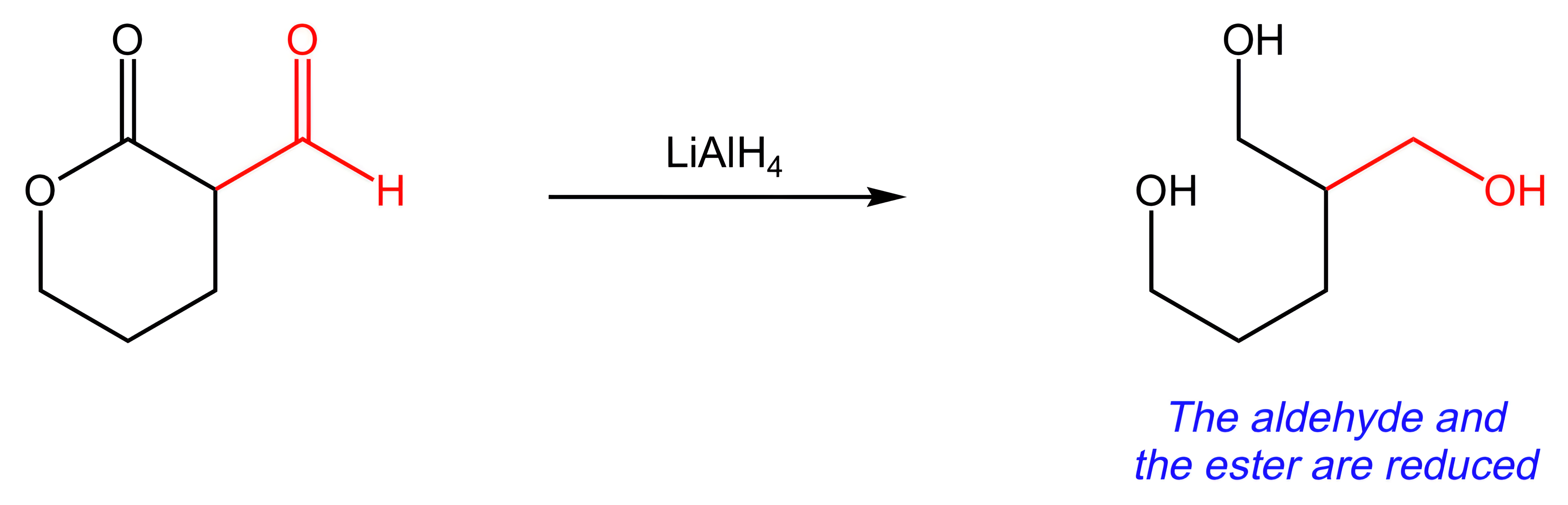
B. LiAlH4
LiAlH4 would reduce both the aldehyde and the ester to 1° alcohols.
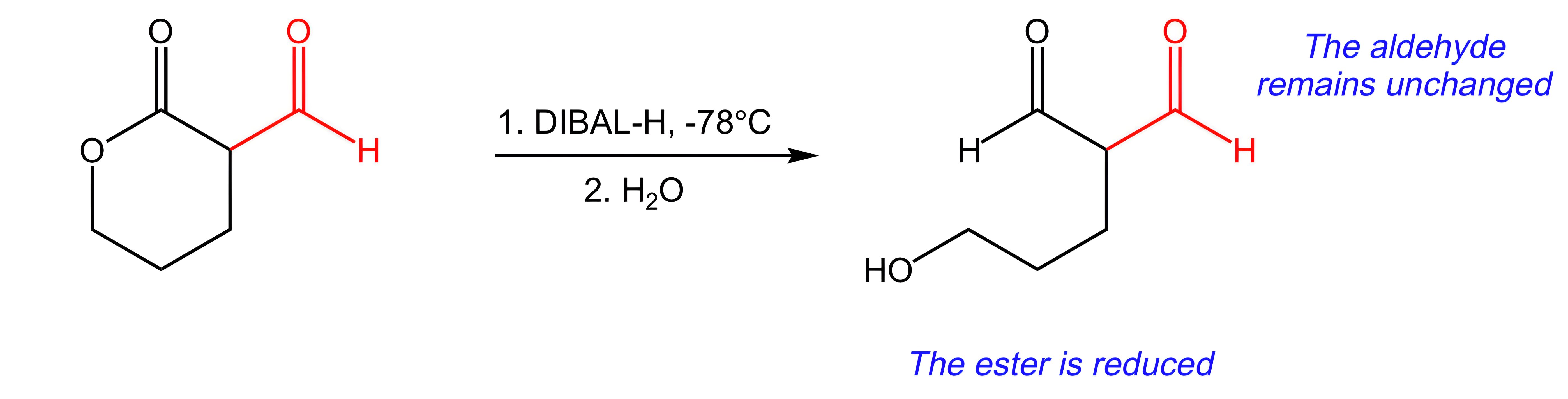
C. DIBAL-H, -78 °C / H2O
DIBAL-H, -78 °C / H2O would reduce the ester to an aldehyde, and the aldehyde would remain unchanged.
D. H2/Ni
H2/Ni participates in the catalytic hydrogenation of alkenes and alkynes to alkanes.
Key Takeaway:
NaBH4 and LiAlH4 reduce aldehydes, acid chlorides, and acid anhydrides to 1° alcohols, and ketones to 2° alcohols.
NaBH4 is not powerful enough to reduce carboxylic acids or esters. LiAlH4 reduces carboxylic acids and esters to 1° alcohols.
What is the major product of the following reaction?
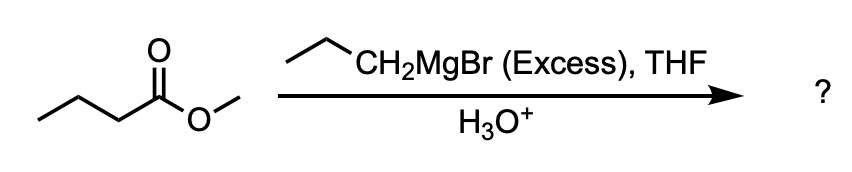
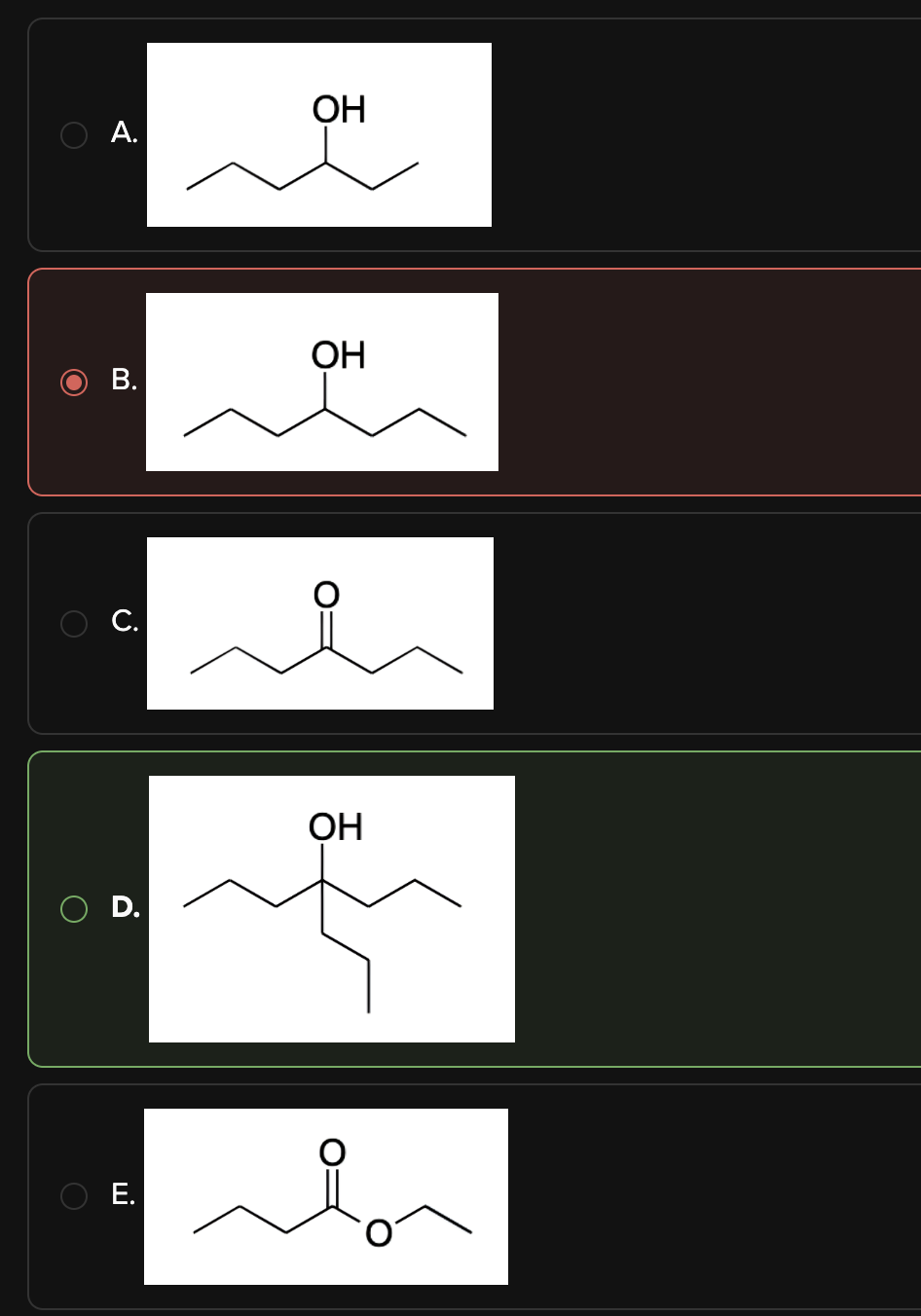
D
We are asked to determine the product of the reaction between methyl butanoate and excess Grignard reagent.
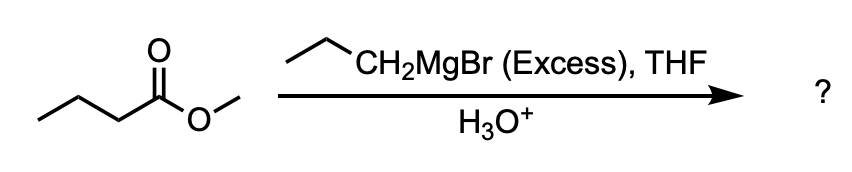
This reaction is addition of a Grignard reagent, and it proceeds via the steps listed below. It is important to note that since there is excess Grignard reagent present, multiple alkyl additions will occur.
The Grignard reagent adds to the carbonyl C, pushing the electrons up onto the carbonyl O.
The C=O bond is re-formed, and the leaving group is eliminated.
A second addition of the Grignard reagent to the carbonyl C occurs, pushing the electrons up onto the carbonyl O.
Acid quench converts the resulting O- into OH, forming the final product.
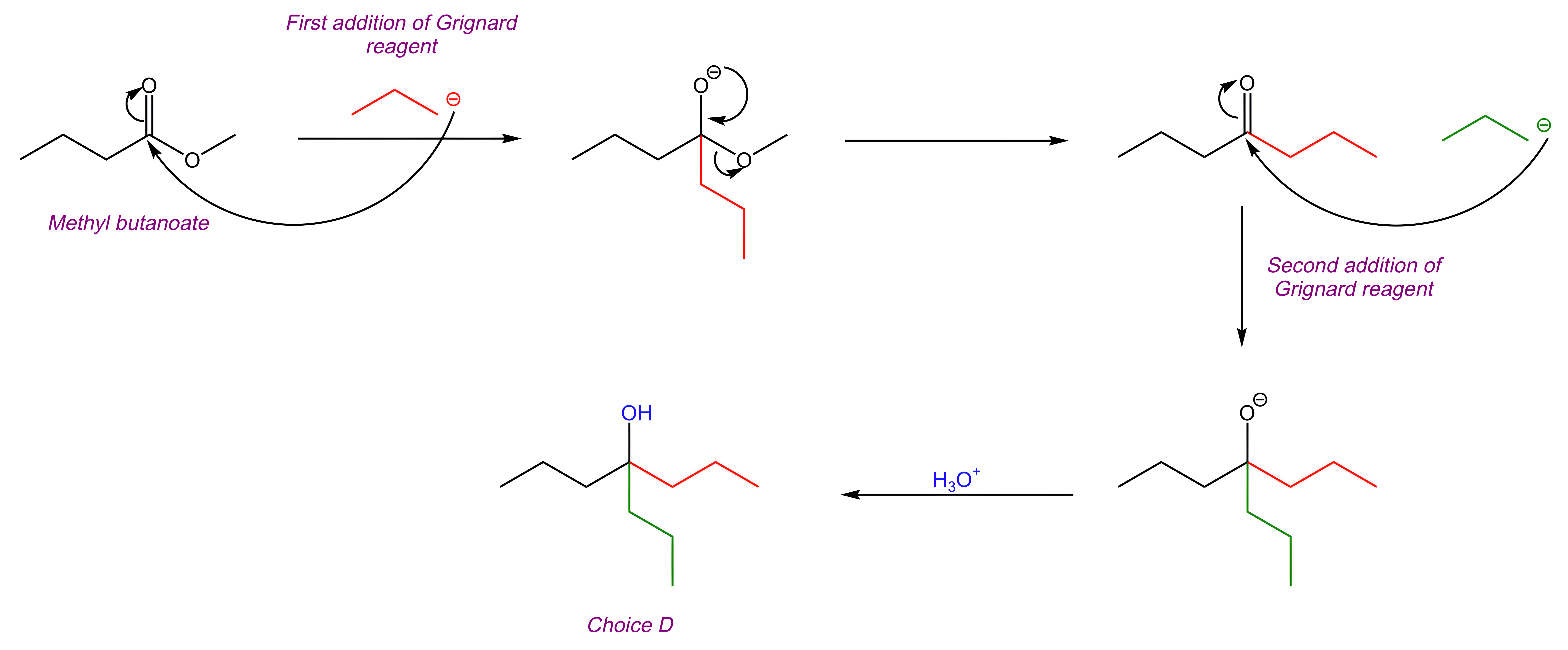
Key Takeaway:
In addition of a Grignard reagent, the Grignard reagent adds to the carbonyl, and then the leaving group is eliminated.
If excess Grignard reagent is present, multiple alkyl additions will occur.
The following molecule contains which functional groups?
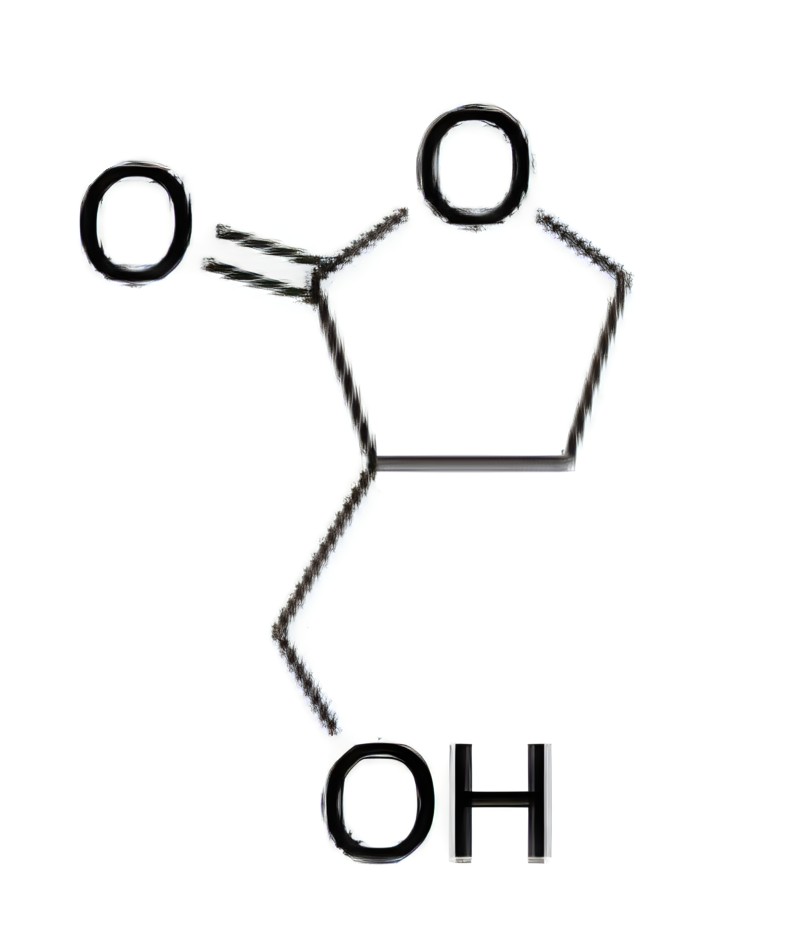
A. Ether and alcohol
B. Ester and alcohol
C. Anhydride and ether
D. Carboxylic acid
E. Ketone and alcohol
B
The molecule below contains a cyclic ester and an alcohol:
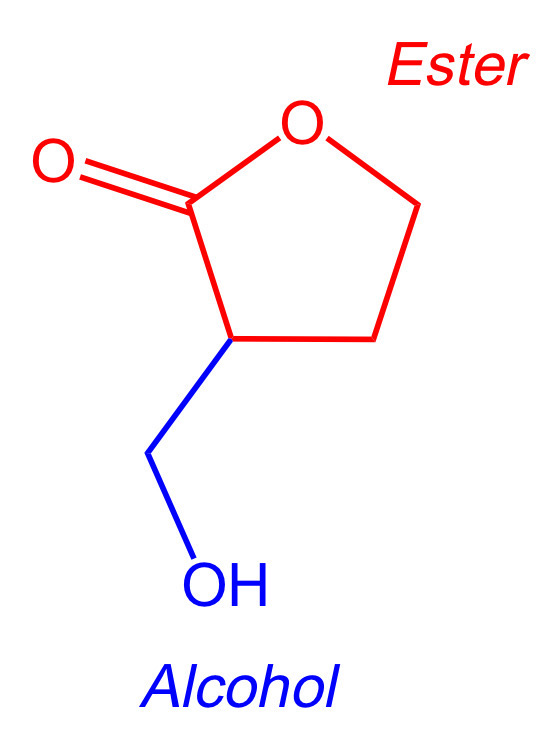
Key Takeaway:
Memorize the general structures and names of functional groups that commonly appear on the DAT.
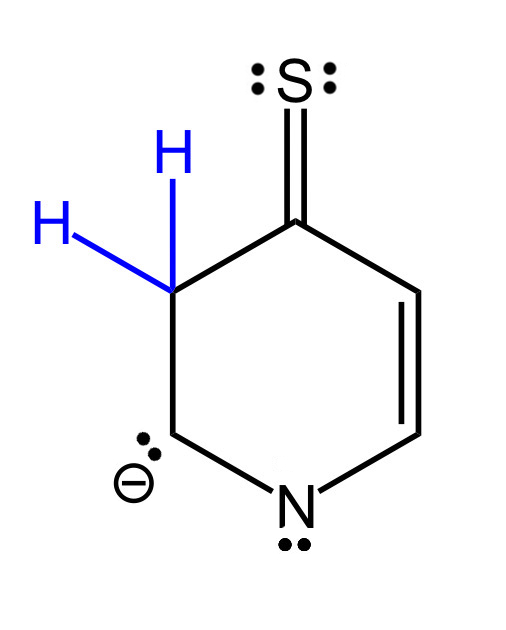
Which structure below best represents a reasonable resonance structure of the following molecule?
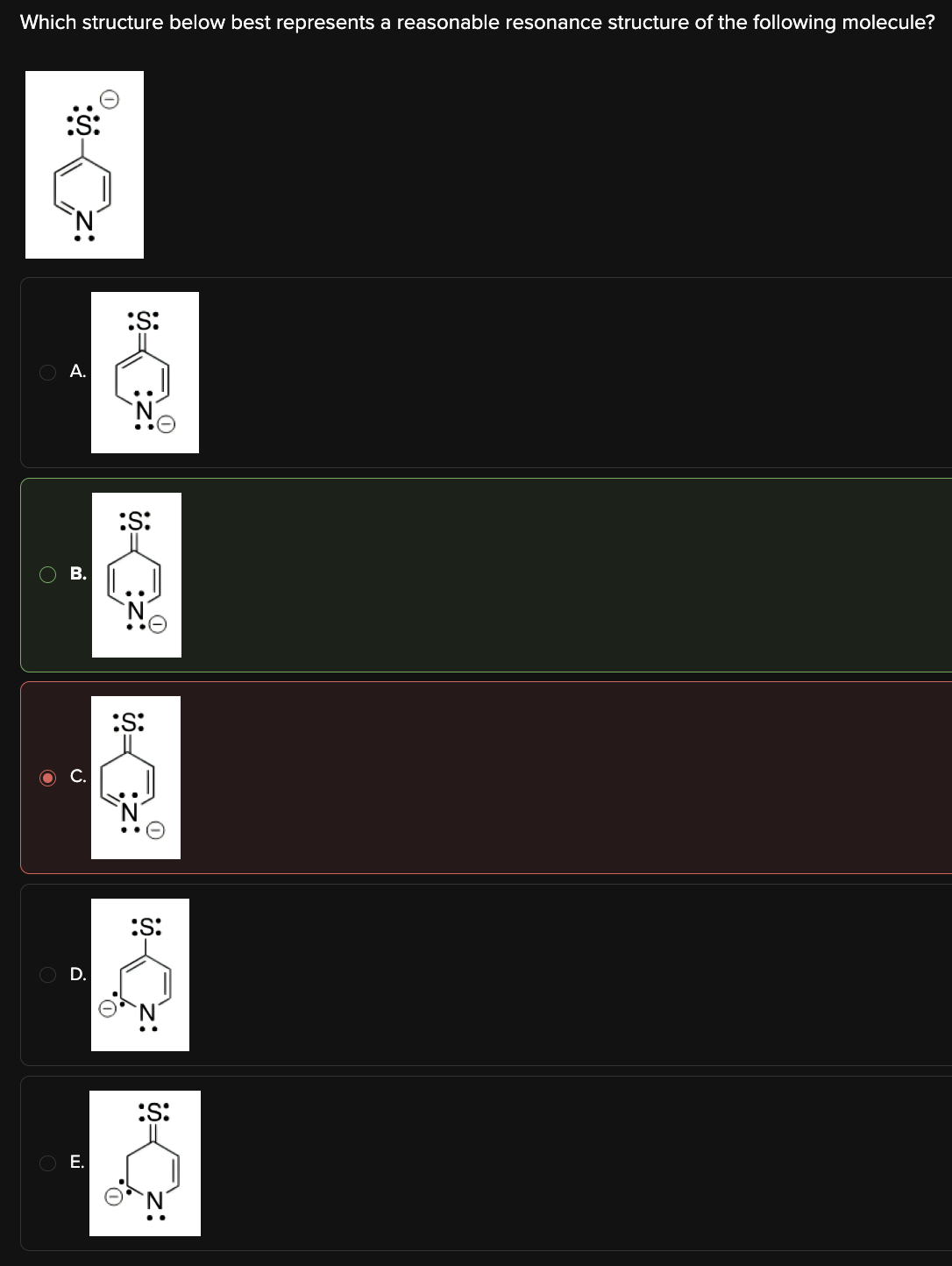
B
Resonance structures involve the movement of electrons through p-orbitals.
Any structure that violates the octet rule is not a reasonable resonance structure. Looking at Choice [B], we can see that each atom has a full octet; it is the only resonance structure listed that does not violate the octet rule.
[Choice A]:
This structure has a carbon bonded to 5 atoms, thereby violating the octet rule.
[Choice C]:
This structure has a nitrogen with 2 lone pairs and 3 bonds, thereby violating the octet rule.
[Choice D]:
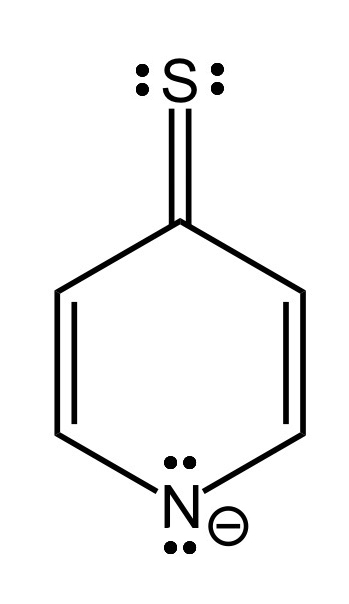
This structure has a sulfur with only 2 lone pairs and a single bond, thereby violating the octet rule.
[Choice E]:
This structure has an extra hydrogen, as well as an illogical electronic structure.
Key Takeaway:
When drawing resonance structures, remember the following rules:
Only pi electrons, lone-pair electrons, or negative charges can move.
You can move electrons toward or into an atom that does not have a full octet.
You can only move electrons into an atom that already has a full octet if you have other electrons push out the opposite side.
Which of the following has the lowest pKa?
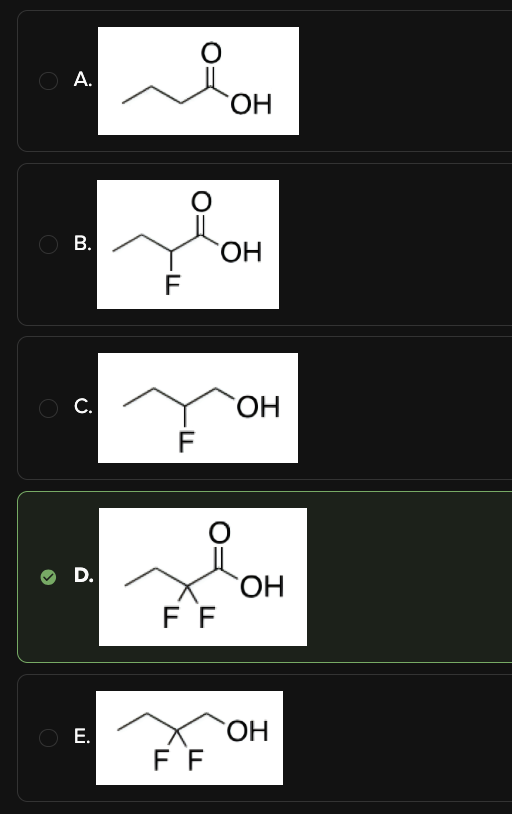
D
First, recall that thelower thepKa, thestronger the acid.
To determine the strongest acid, we use the acid ranking mnemonic CARDIO:
Charge
Atom
Resonance
Dipole Induction
Orbitals
First, let's look at charge. None of the molecules have a charge, so no choices can be eliminated.
Next, let's look at atom. All of the molecules have the acidic hydrogen bonded to oxygen, so no choices can be eliminated.
Let's look at resonance. The conjugate bases of Choices [A], [B], and [D] have a lone pair on oxygen that can resonance delocalize into the carbonyl carbon. The more stable the conjugate base, the stronger the acid. Eliminate Choices [C] and [E].
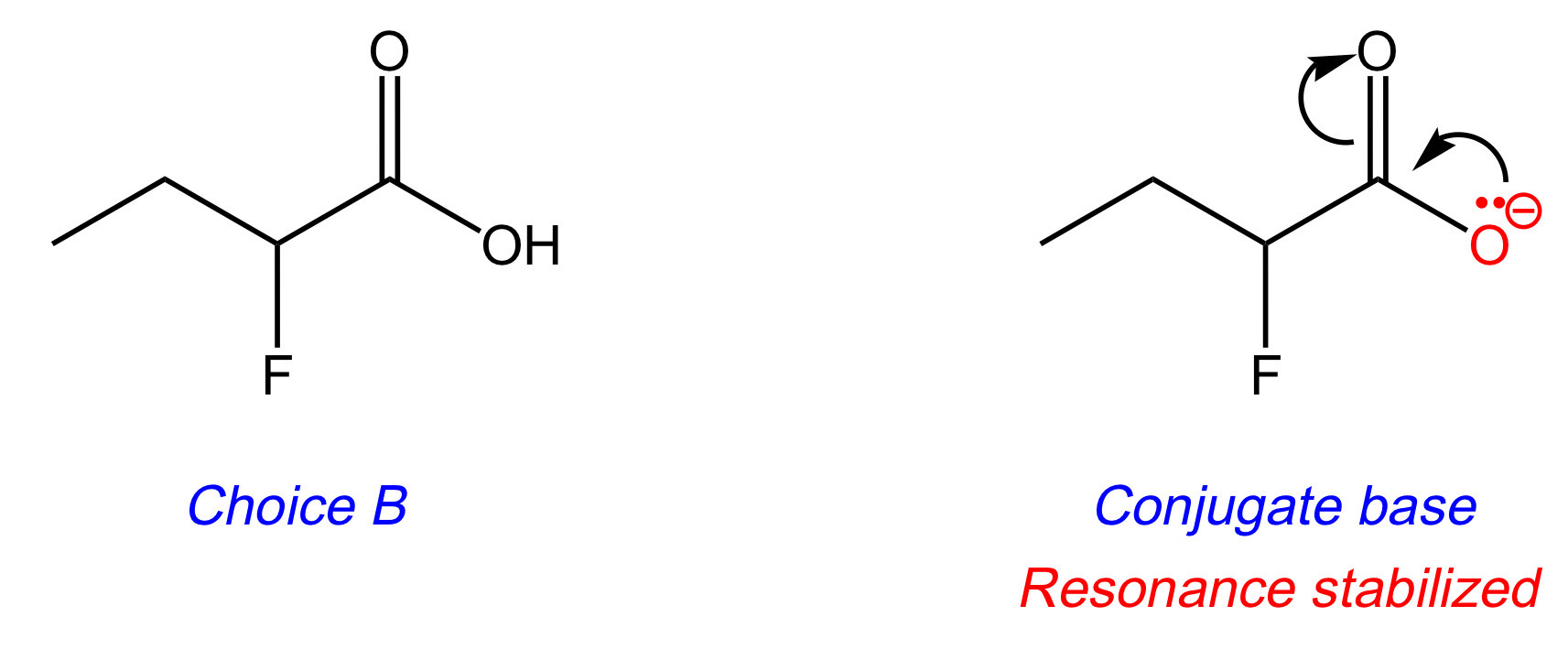
DAT Pro-Tip: Choices [C] and [E] are alcohols. Remember that alcohols have higher pKa's (i.e. are less acidic) than carboxylic acids due to less resonance stabilization.
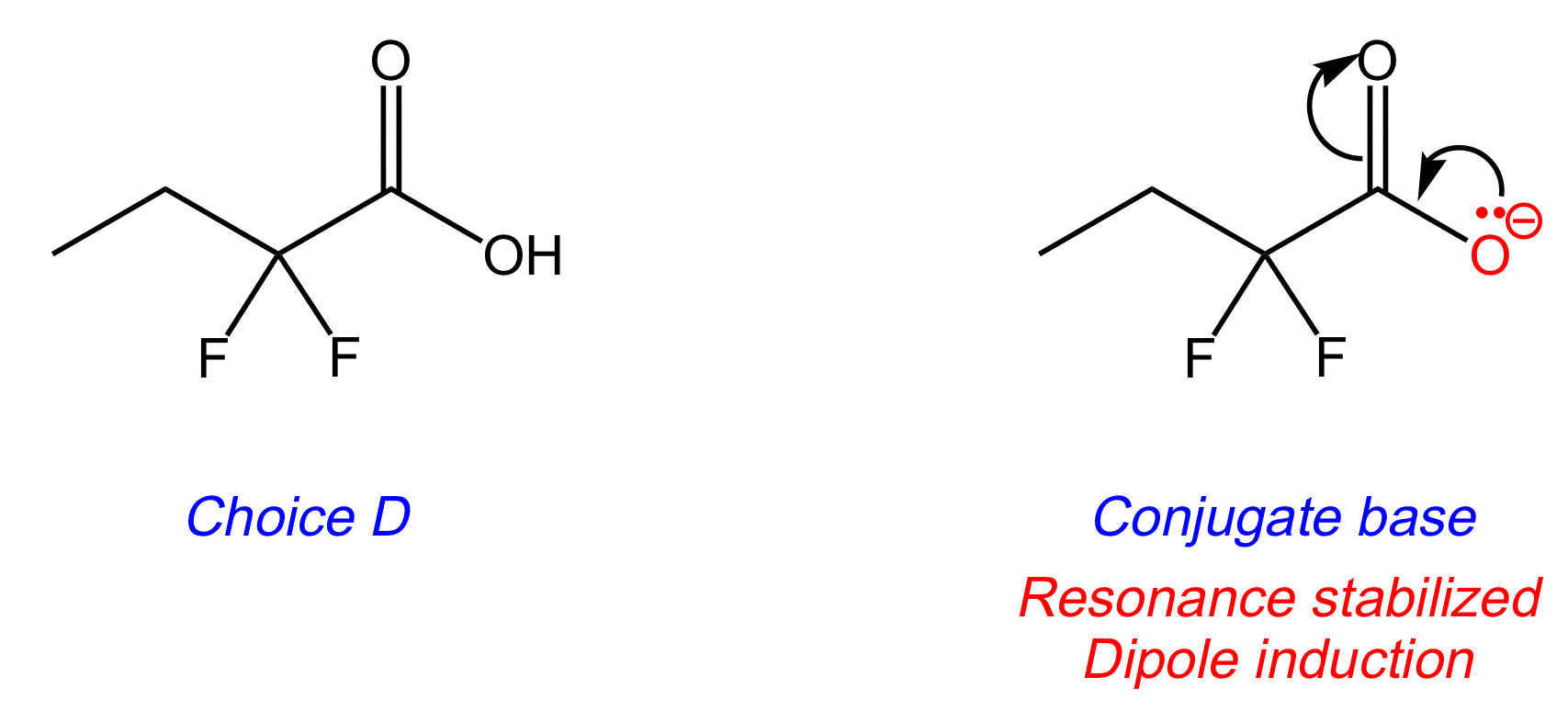
The key difference between Choices [A], [B], and [D] is the number of fluorines attached to carbon-2. Fluorine is an electron-withdrawing group, and thus makes the acidic hydrogen more acidic.
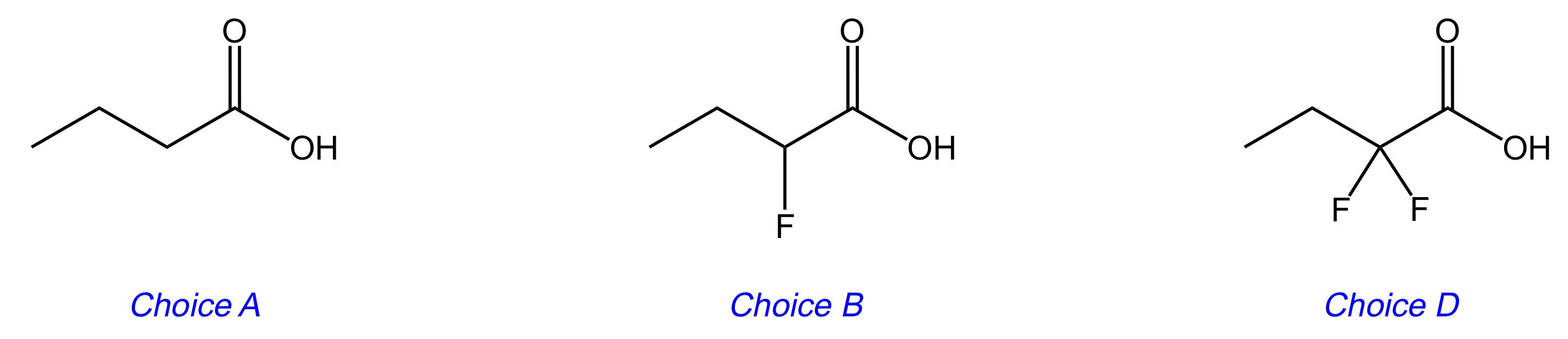
Looking at the conjugate base of Choice [D], the two fluorines inductively stabilize the negative charge through their electron-withdrawing properties. This spreads the negative charge throughout the molecule. The more stable the conjugate base, the stronger the acid. Choice [D] is the answer.
Key Takeaway:
To determine the strongest acid, use the acid ranking mnemonic CARDIO.
When bonded to non-aromatic rings, electron-withdrawing groups (e.g. F, Cl, Br, I, N, O) increase acidity.
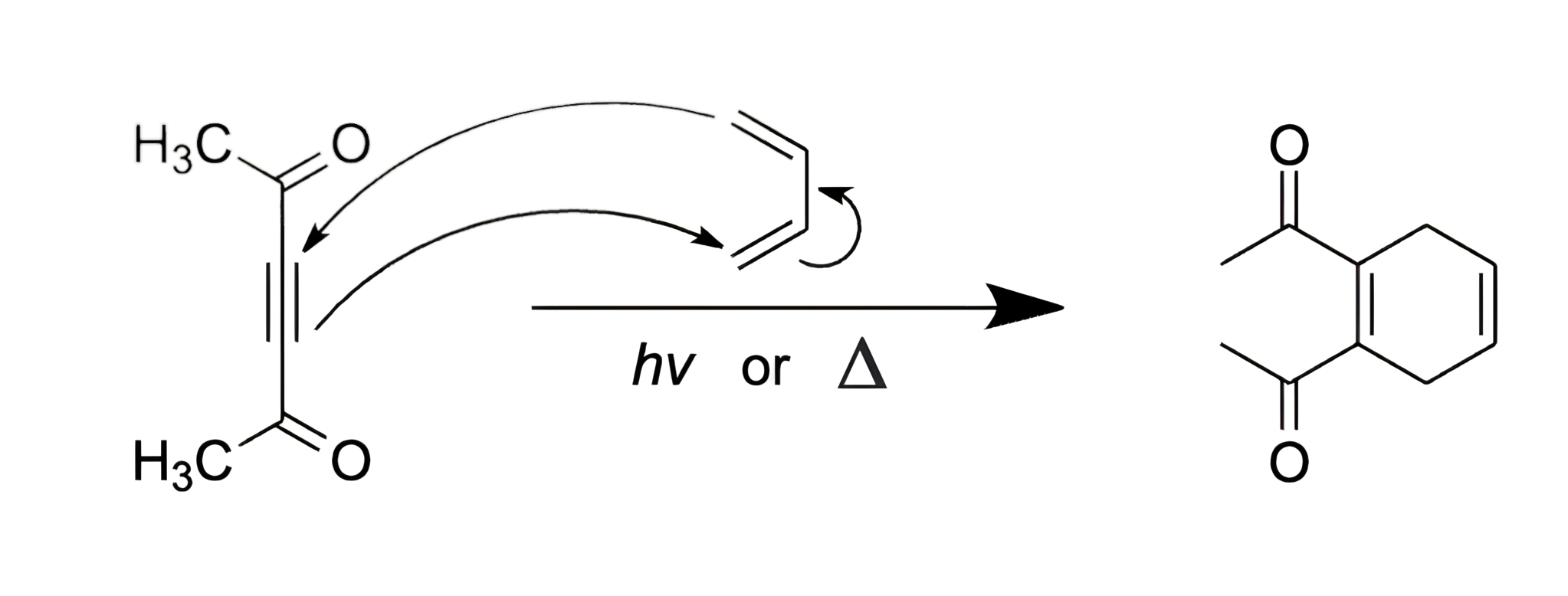
Identify the product of the following reaction:
D
In this question, a conjugated diene is reacted with an alkyne dienophile and UV light or heat. We are asked to determine the products of the reaction.
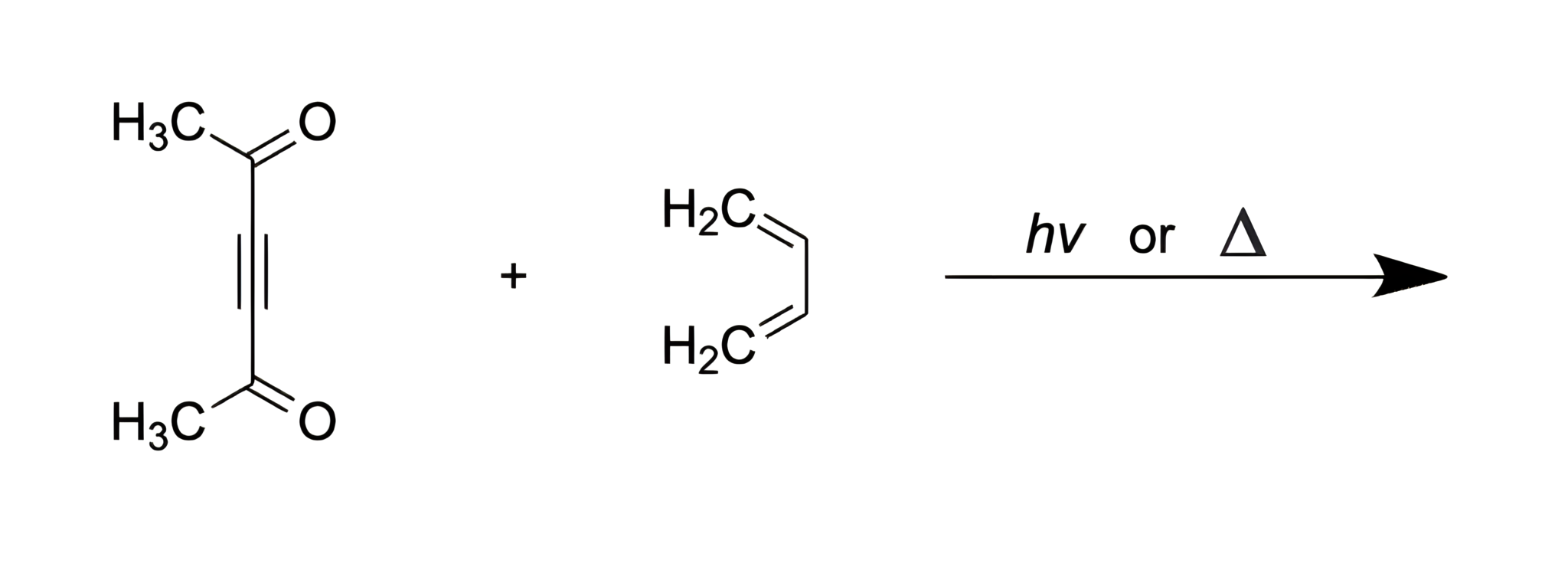
Diels-Alder reactions are performed by reacting conjugated dienes with alkene or alkyne dienophiles and UV light or heat. When alkyne dienophiles are used, the reaction results in the formation of a substituted cyclohexene derivative.
The mechanism for the Diels-Alder reaction shown is as follows:
Thus, choice [D] is the major product of the reaction shown above.
Key Takeaway:
Diels-Alder reactions result in the formation of a substituted cyclohexene derivative.
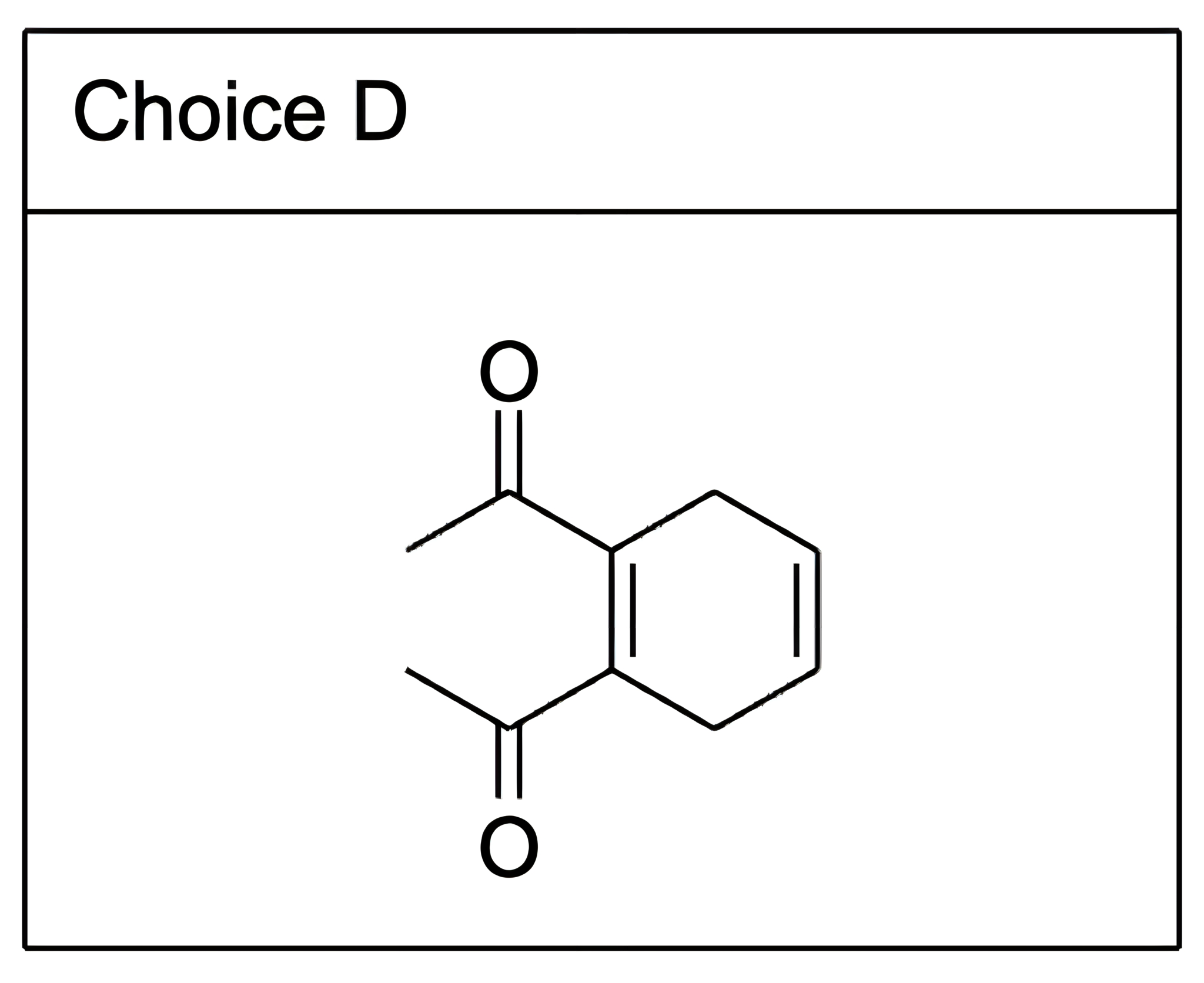
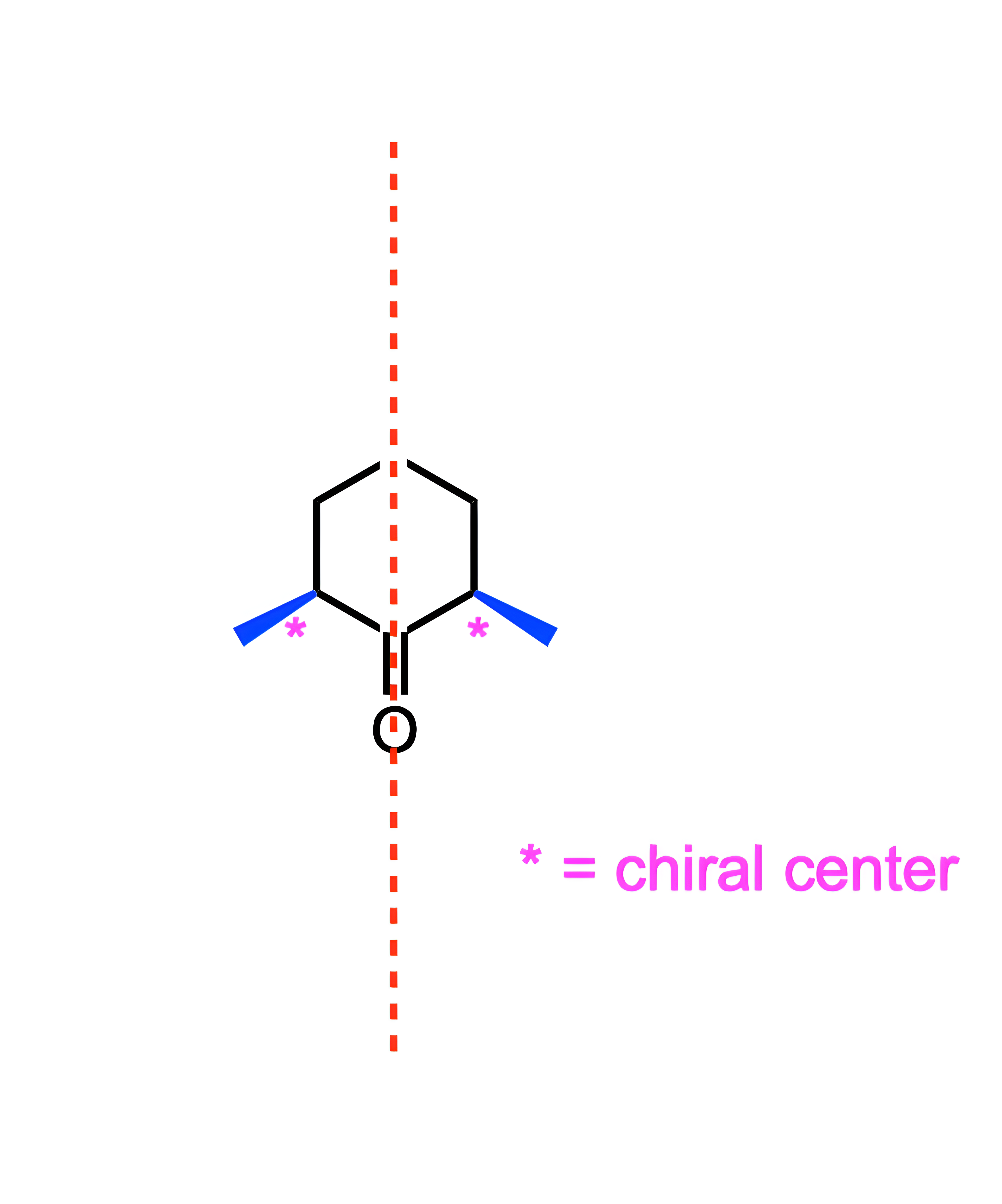
Which of the following is a meso compound?
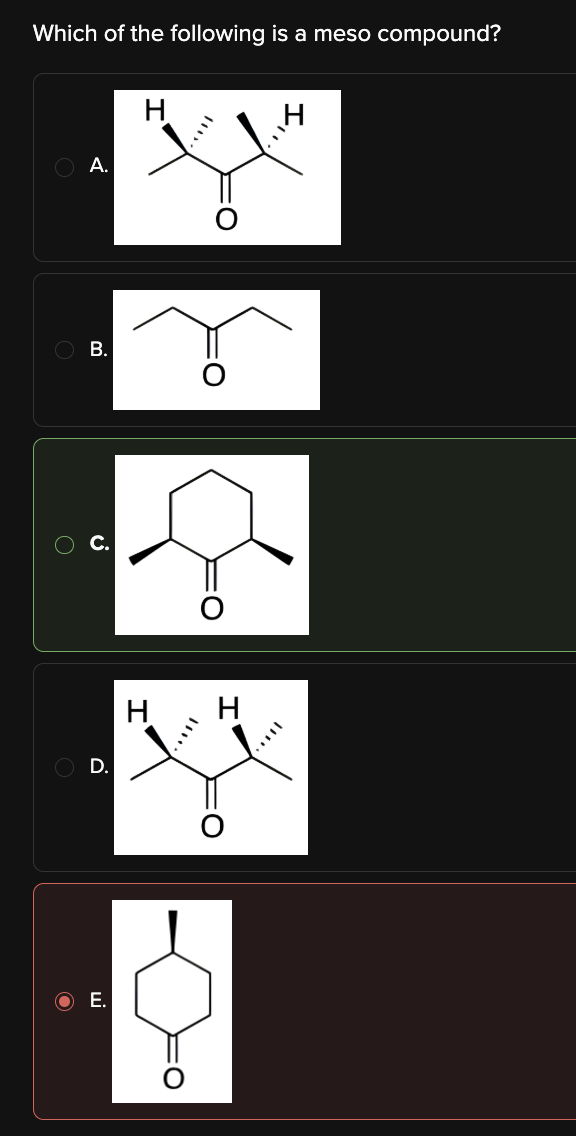
C
Meso compounds are molecules that have two or more stereocenters and can be bisected by a line of symmetry.
Choice [C] is the only compound that meets this criteria.
In contrast, Choices [A], [B], [D], and [E] do not have stereocenters.
Key Takeaway:
Meso compounds are molecules that have two or more stereocenters and can be bisected by a line of symmetry.
What are the organic products of the following reaction?
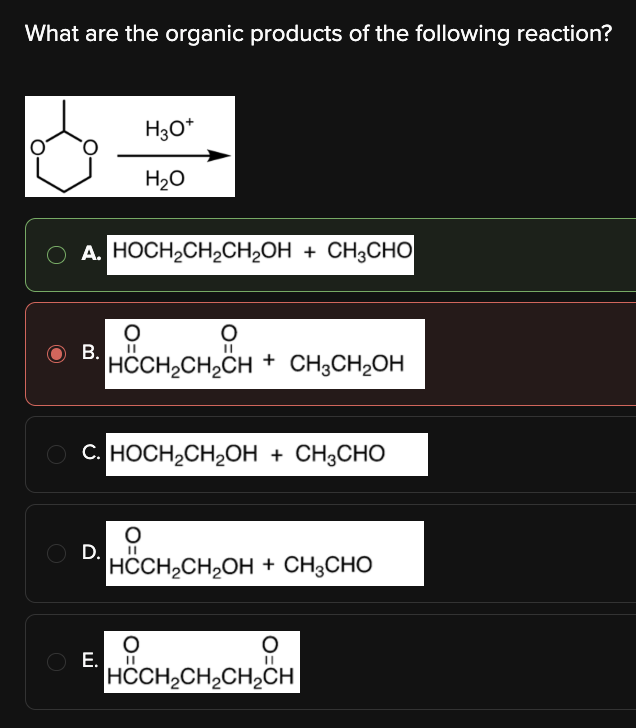
A
The key to answering this question is to realize that the starting molecule is an acetal (i.e. a carbon bonded to two OR groups).
When acetals are treated with acid and water:
The acetal carbon becomes the carbonyl carbon of an aldehyde.
The two OR groups are released as alcohols.
In the reaction below, note that the two OR groups are connected; this means that the OR groups will be released as a diol, rather than as two separate alcohols.
A simple way to determine the products is to envision the aldehyde and alcohols that are required to form the acetal, as shown below:
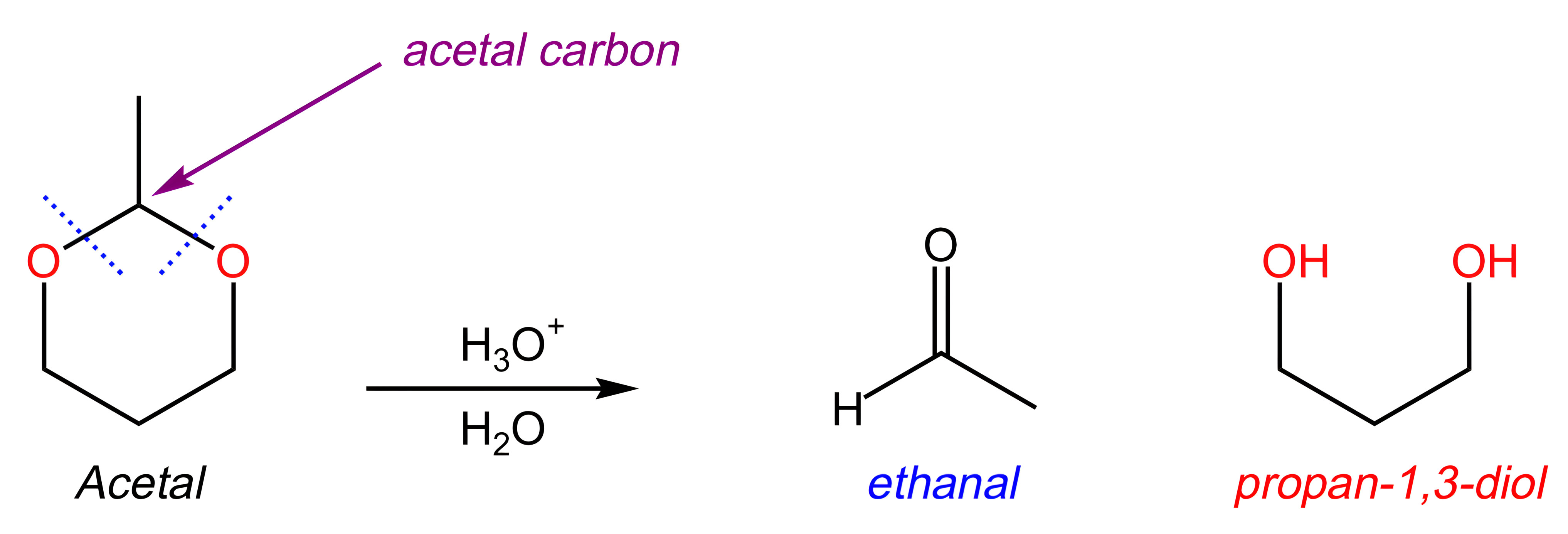
Key Takeaway:
When acetals are treated with acid and water, the acetal carbon becomes the carbonyl carbon of an aldehyde; and the two OR groups are released as alcohols. If the two OR groups are connected, they will be released as a diol.
Which set of reagents would convert the following starting material into the indicated product?
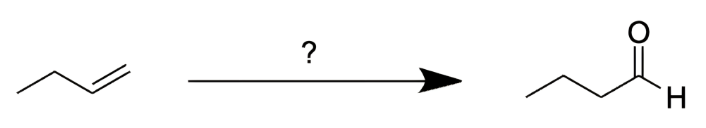
A. 1. PCC 2. H2SO4
B. 1. H3O+ 2. PCC
C. 1. BH3, THF 2. H2O2, OH- 3. PCC
D. 1. H3O+ 2. CrO3, H2SO4
E. 1. BH3, THF 2. H2O2, OH- 3. CrO3, H2SO4
C
In this question, we are asked to determine which of the reagents listed above will yield butanal from but-1-ene. The best way to approach to this question is to determine the product of each reaction pathway.
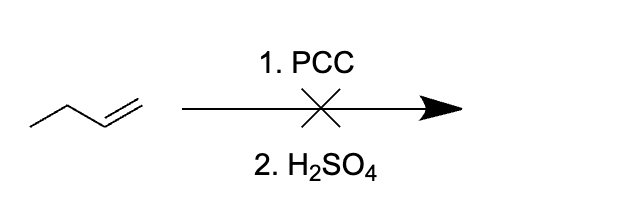
Choice [A]: The reagents do not yield a productive reaction.
Oxidation using 1. PCC 2. H2SO4 can only be performed on primary alcohols to produce aldehydes.
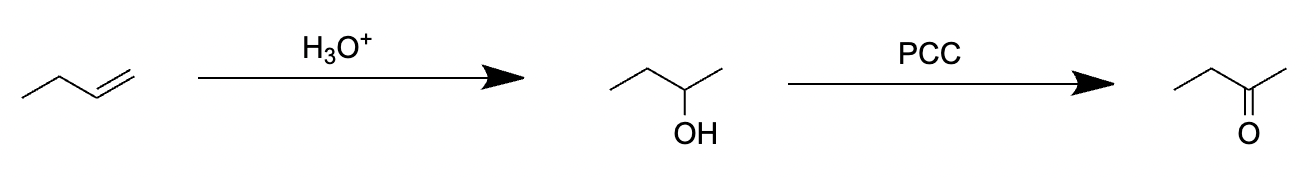
Choice [B]: The reagents produce a ketone.
The pathway for the acid-catalyzed hydration of but-1-ene and oxidation using PCC is as follows:
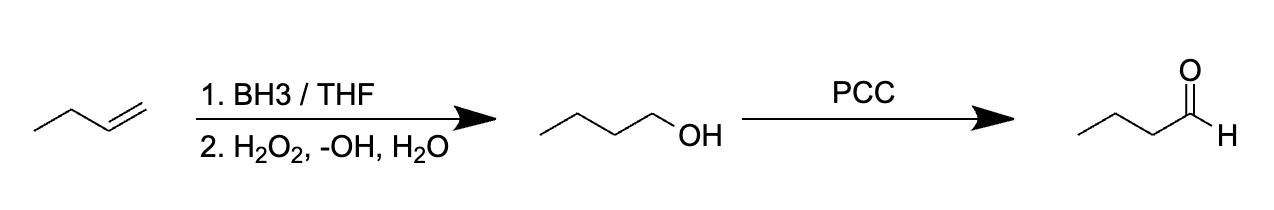
Choice [C]: The reagents convert alkenes to aldehydes.
The pathway for the oxidation of but-1-ene to butanal using 1. BH3, THF 2. H2O2, -OH 3. PCC is as follows:
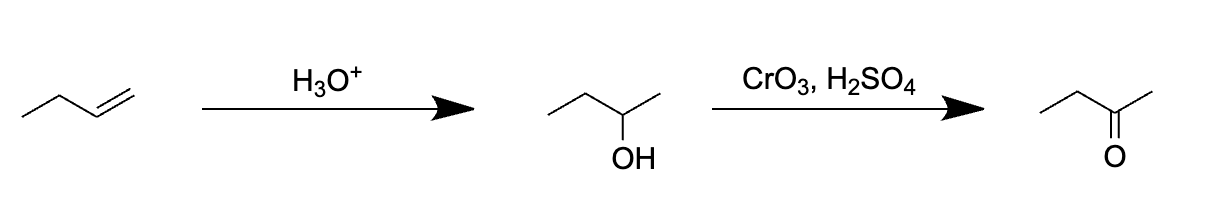
Choice [D]: The reagents convert alkenes to ketones.
The pathway for the oxidation but-1-ene to butanone using 1. H3O+ 2. CrO3, H2SO4 is as follows:
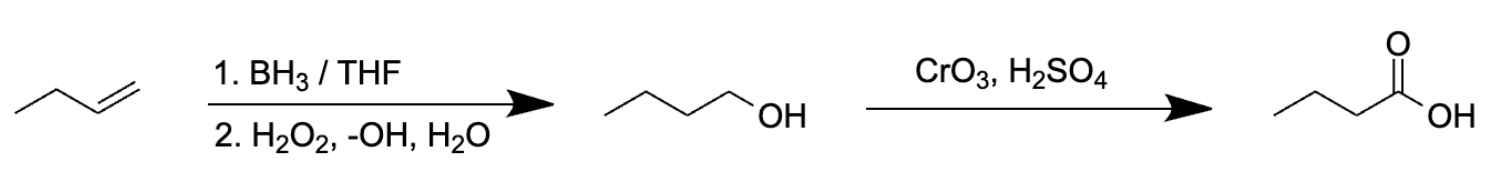
Choice [E]: The reagents convert alkenes to carboxylic acids.
The pathway for the oxidation of but-1-ene to butanoic acid using 1. BH3, THF 2. H2O2, -OH 3. CrO3, H2SO4 is as follows:
Thus, as can be seen above, choice [C] is the reaction pathway that produces butanal from but-1-ene.
Key Takeaway:
Reacting an alkene with 1. BH3, THF 2. H2O2, -OH 3. PCC converts alkenes into aldehydes.
Predict the product of the following reaction:
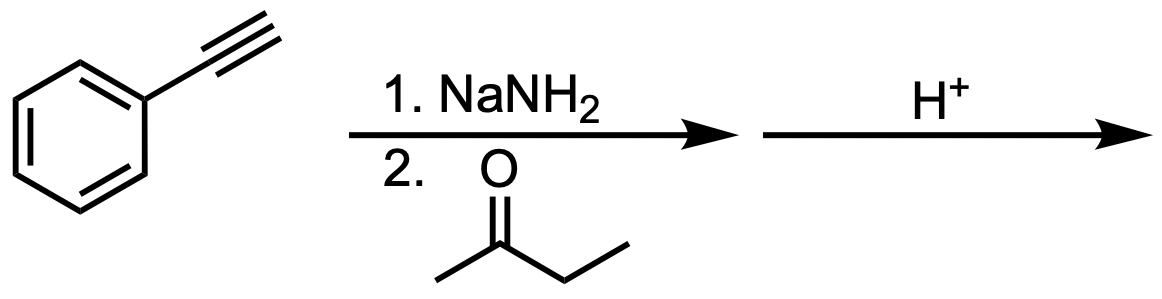
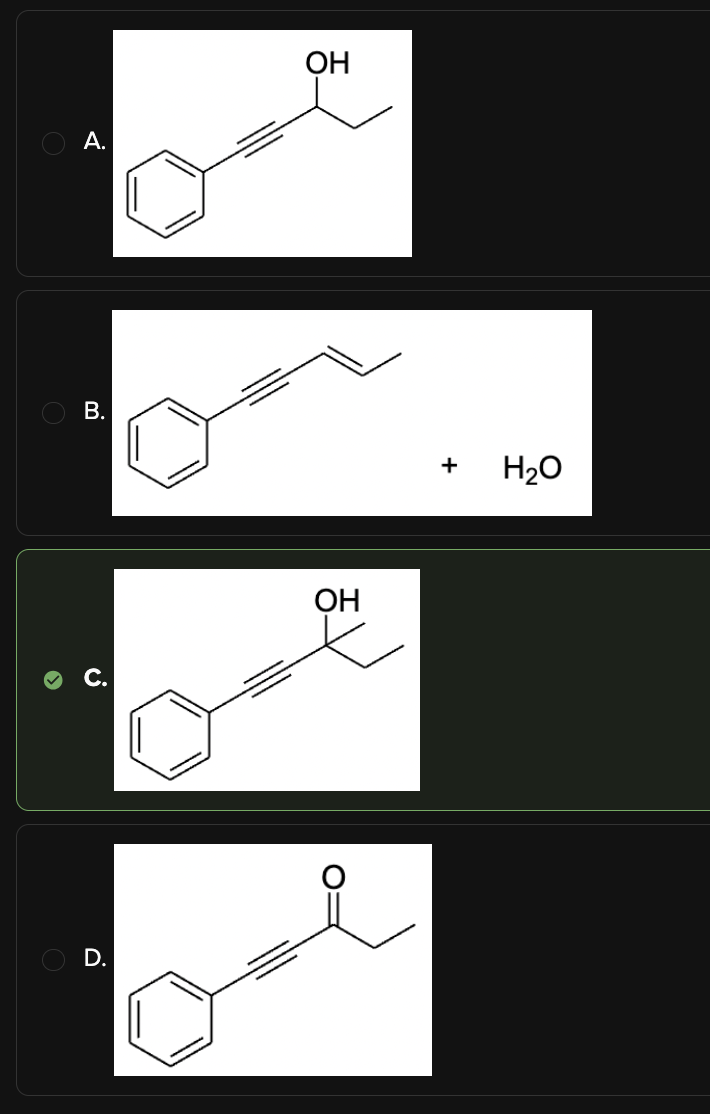
C
In this reaction, phenylethyne reacts with NaNH2, a strong base, which prepares the alkyne for nucleophilic attack on a ketone. After an acid workup, the final product is an alcohol. This reaction proceeds via the following mechanism:
The terminal alkyne is deprotonated by NaNH2, generating a negatively charged acetylide ion.
The acetylide ion acts as a nucleophile and attacks the electrophilic carbon of the ketone, forming a new carbon-carbon bond.
Acid workup protonates the oxygen, yielding the final alcohol product.
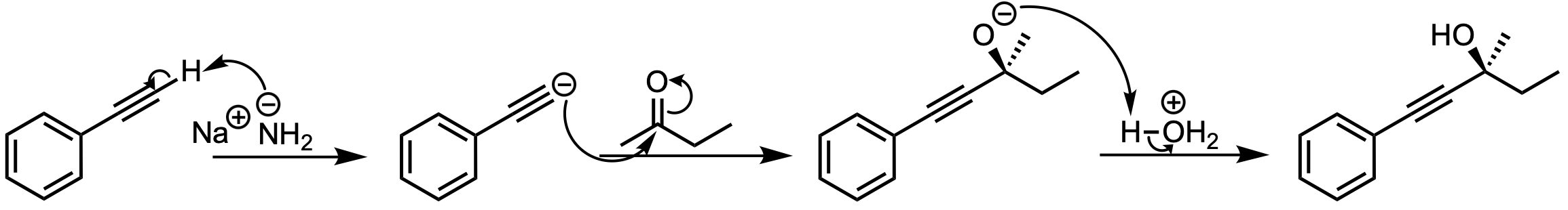
Key Takeaway:
Deprotonated terminal alkynes act as nucleophiles that can add to ketones, forming alcohols after acid workup.
Which of the following statements best describes the stereochemistry at the reactive site of an SN2 reaction?
A. The reaction is stereoselective and results in retetnion of configuration
B. The reaction is stereospecific and results in an inversion of configuration
C. The reaction is not stereospecific and results in a racemic mixture of products
D. The reaction is stereoselective and results in a racemic mixture of products
E. The stereoselectivity depends on the nature of the susbtrate and cannot be predicted
B
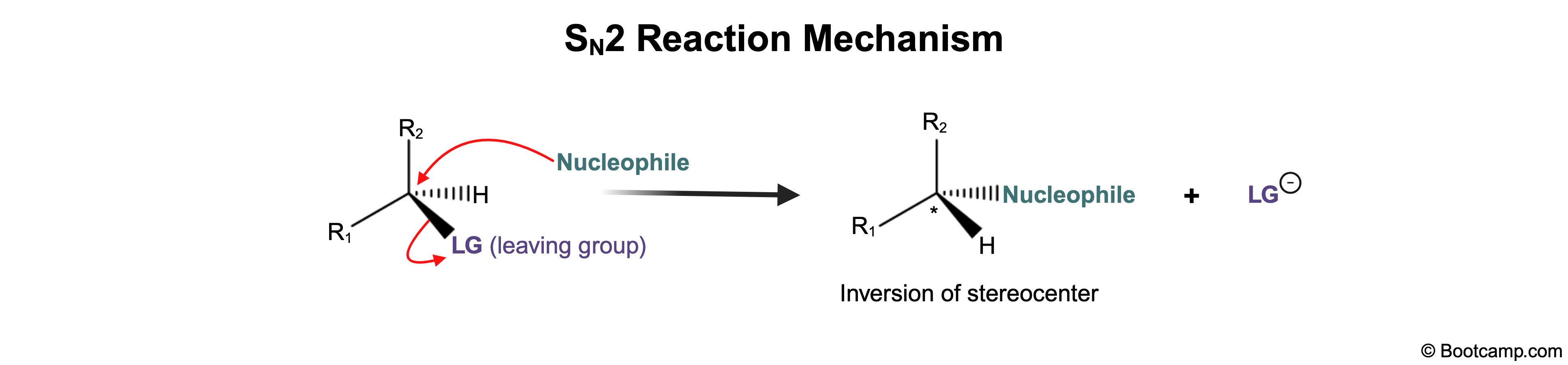
SN2reactions arestereospecific; that is to say, they produce one product with aspecific stereochemistry.
This stereospecificity results from the pentavalent transition state that occurs during the backside attack of the nucleophile. After the leaving group leaves, the product has an inversion of configuration.
Key Takeaway:
SN2 reactions are stereospecific and result in an inversion of configuration.
Determine the major product of the following reaction:
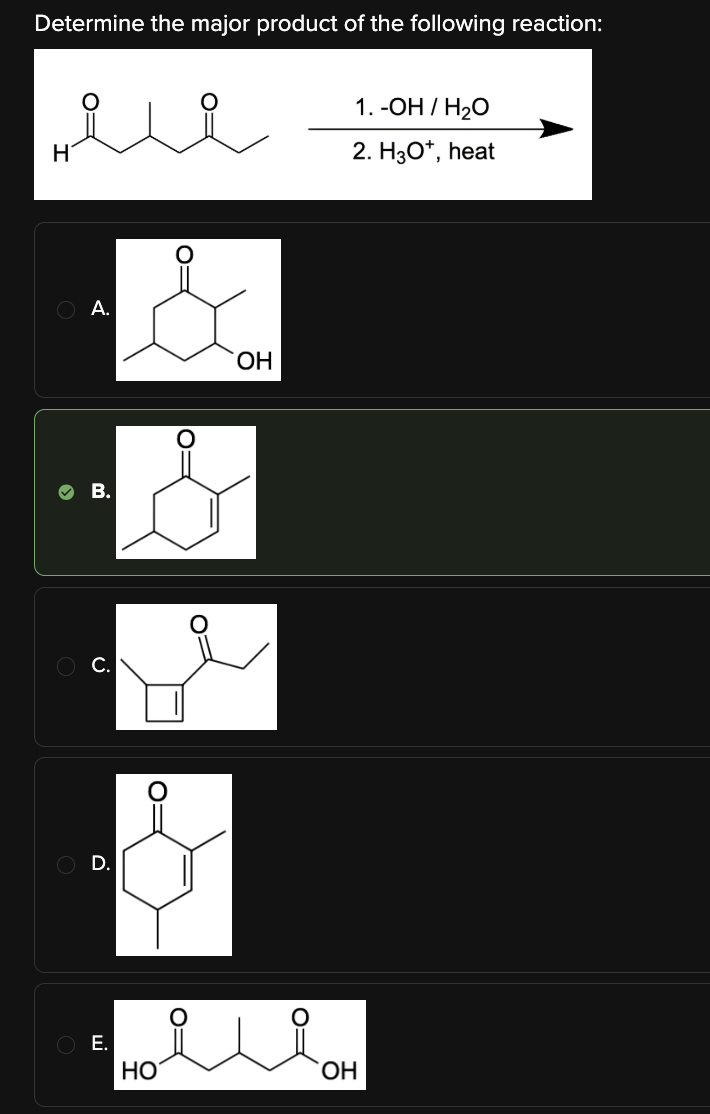
B
We are asked to determine the product of a mixed aldol condensation between an aldehyde and a ketone in 3-methyl-5-oxoheptanal.
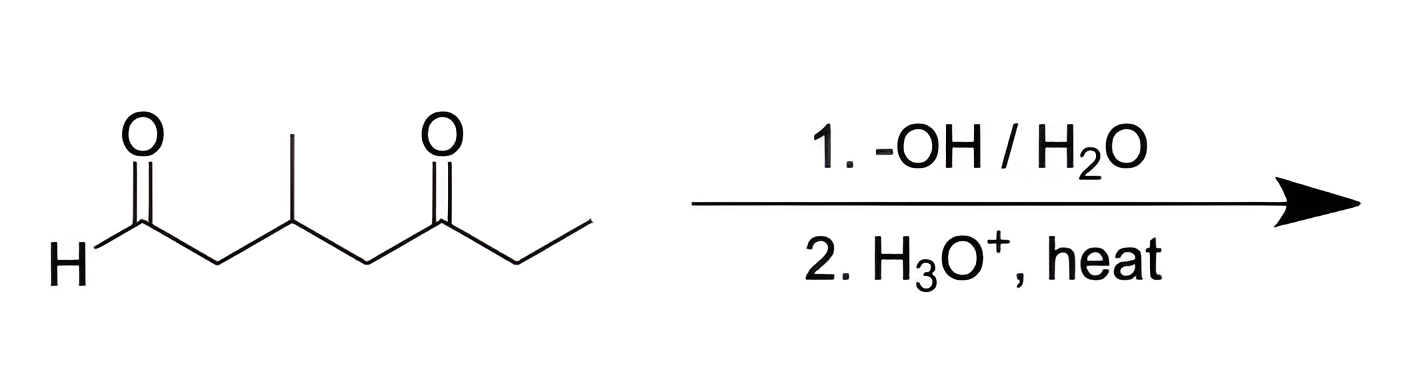
In this intramolecular mixed aldol condensation, the aldehyde and ketone react with -OH to form a β-hydroxy ketone, which is subsequently dehydrated to produce an α,β-unsaturated ketone. This reaction results in the cyclization of the starting molecule.
The reaction proceeds as follows:
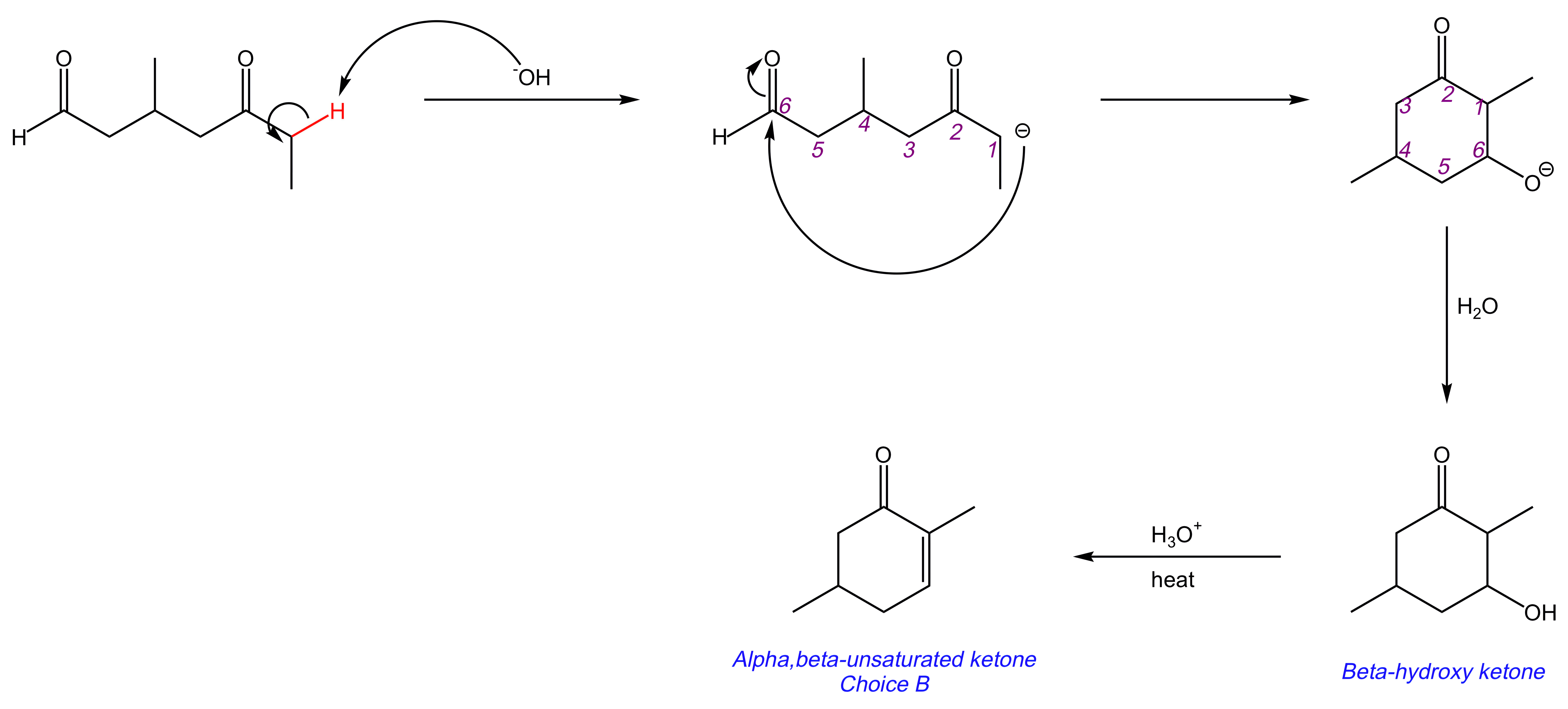
Key Takeaway:
Intramolecular mixed aldol condensation results in the cyclization of the starting molecule.
What is the major product of the following reaction?
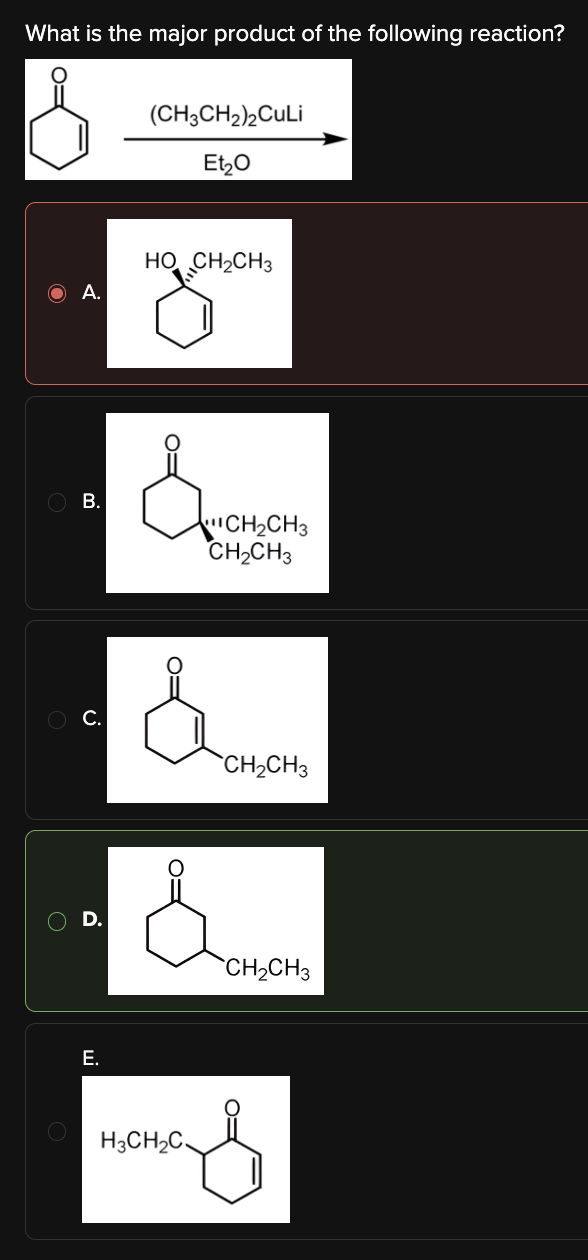
D
In the reaction below, an α,β-unsaturated ketone reacts with an organocuprate (R2CuLi).
Unlike Grignard reagents, organocuprates prefer to perform Michael (1,4) additions when reacting with α,β-unsaturated ketones.
Therefore, the reaction proceeds as follows:
The organocuprate (which can be viewed as CH3CH2-) attacks the alkene C located furthest away from the carbonyl C, pushing the electrons up onto the adjacent alkene C.
The resulting negative charge gets protonated during the quench step, forming product [D].
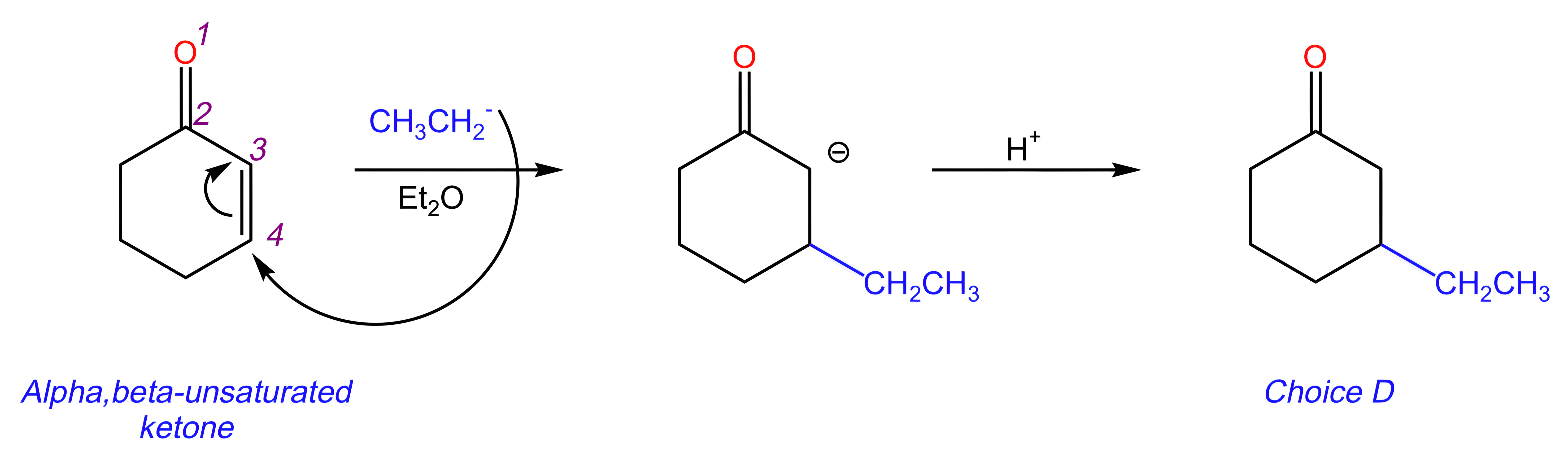
DAT Pro-Tip: Although organocuprates have the general formula R2CuLi, the R group will only add once when they react with α,β-unsaturated ketones.
Key Takeaway:
Organocuprates (R2CuLi) preferentially attack the β-carbon of α,β-unsaturated ketones.
What is the product of the following reaction?
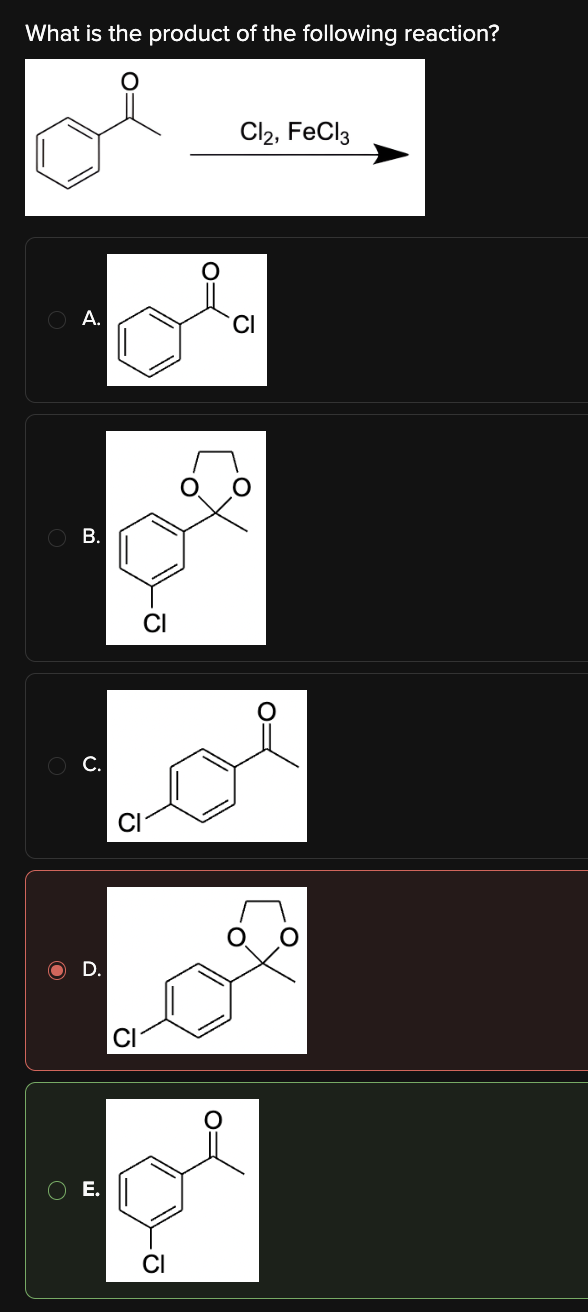
E
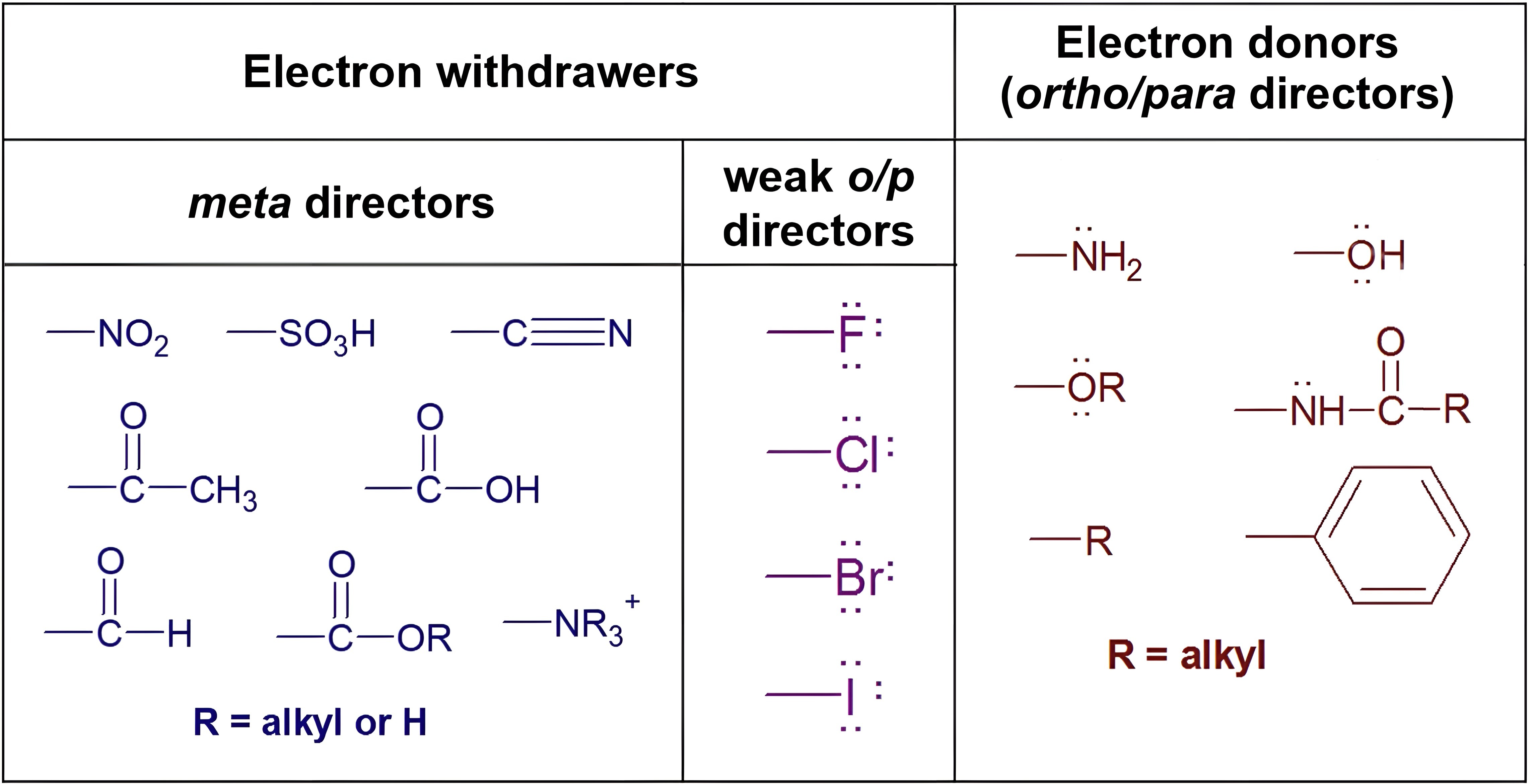
Benzene undergoes chlorination upon reaction with Cl2 in the presence of FeCl3 via electrophilic aromatic substitution. The regioselectivity of substitution depends on existing substituents. Electron-withdrawing groups are meta-directors, while electron-donating groups are ortho/para-directors.
The acetyl group is an electron-withdrawing group, making it a meta-director. Thus, the reaction mechanism proceeds as follows:
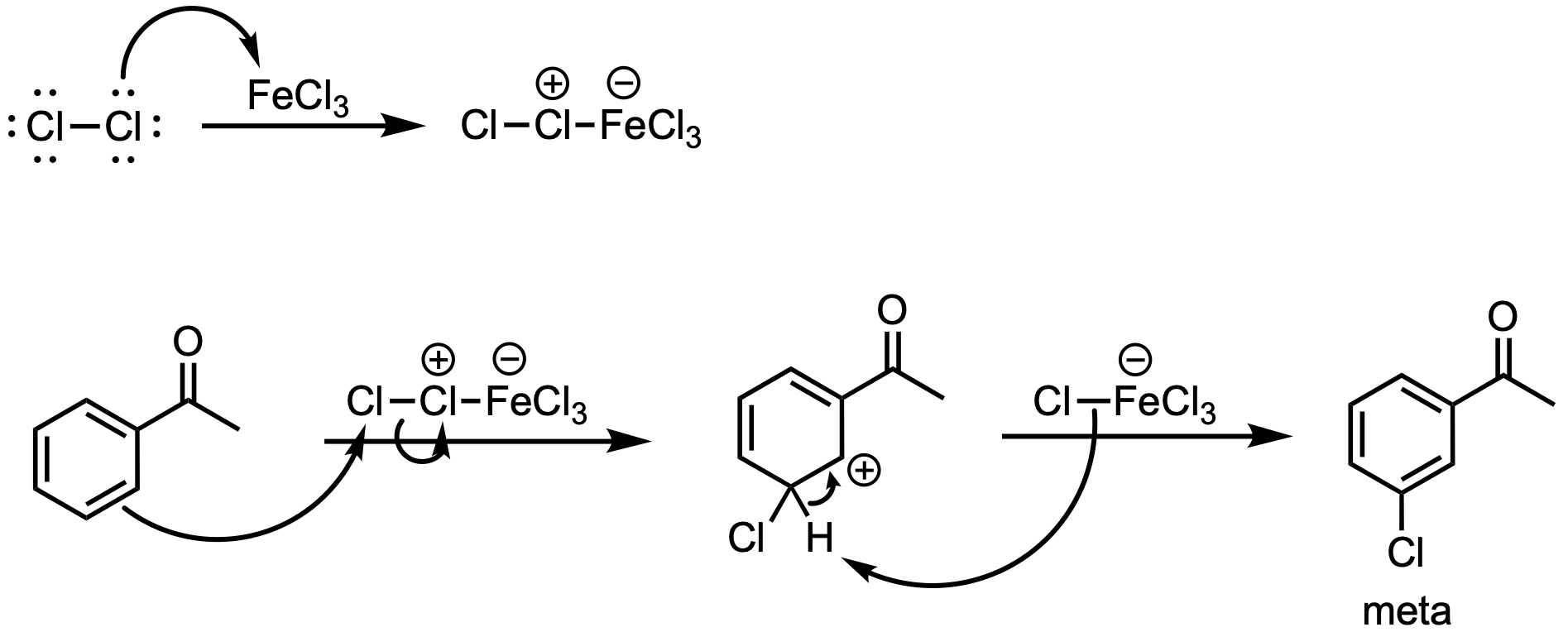
Key Takeaway:
Benzene undergoes chlorination upon reaction with Cl2 in the presence of FeCl3 via electrophilic aromatic substitution.
The regioselectivity of substitution is determined by existing substituents.
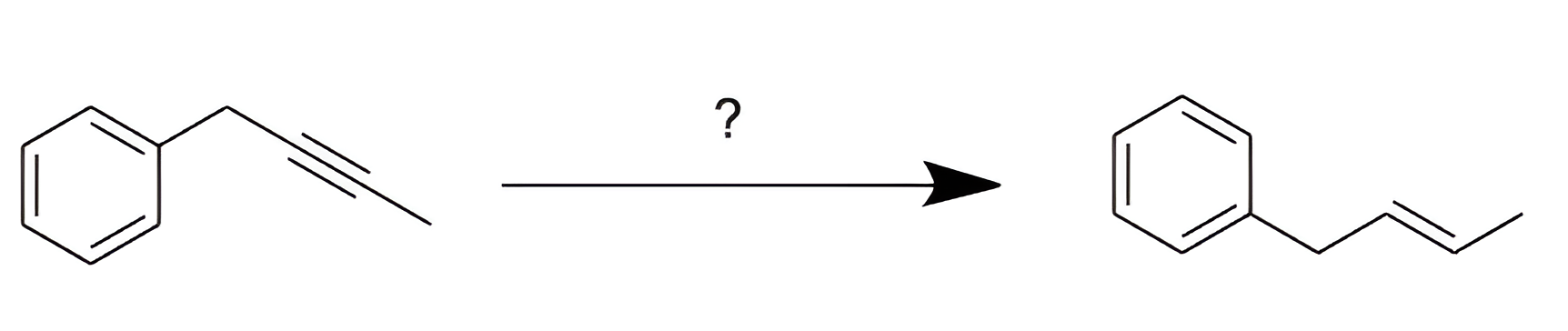
Which set of reagents will accomplish the following chemical transformation?
A. (1) NaNH2, (2) CH3Br, (3) H3O+
B. H2, Lindlar's catalyst
C. Li, NH3 (l) at -78ºC
D. (1) LiAlH4, (2) H3O+
E. HgSO4, H2SO4
C
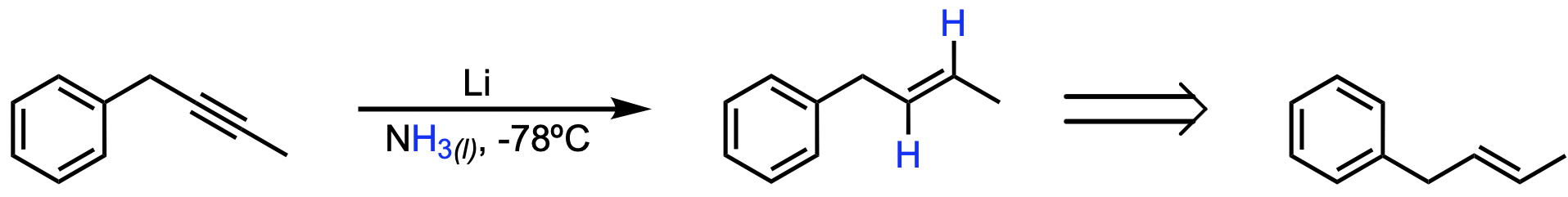
In this reaction, an alkyne is selectively reduced to a trans alkene. This transformation is accomplished using lithium metal (Li) in liquid NH3 at -78ºC. These conditions result in a partial reduction of the alkyne through anti addition, where hydrogen atoms are added to opposite sides of the triple bond.
A. (1) NaNH2, (2) CH3Br, (3) H3O+
This set of reagents is used for the alkylation of terminal alkynes. NaNH2 deprotonates the terminal hydrogen to form an acetylide anion, which then undergoes SN2 with CH3Br, adding a methyl group. These reagents do not react with internal alkynes.
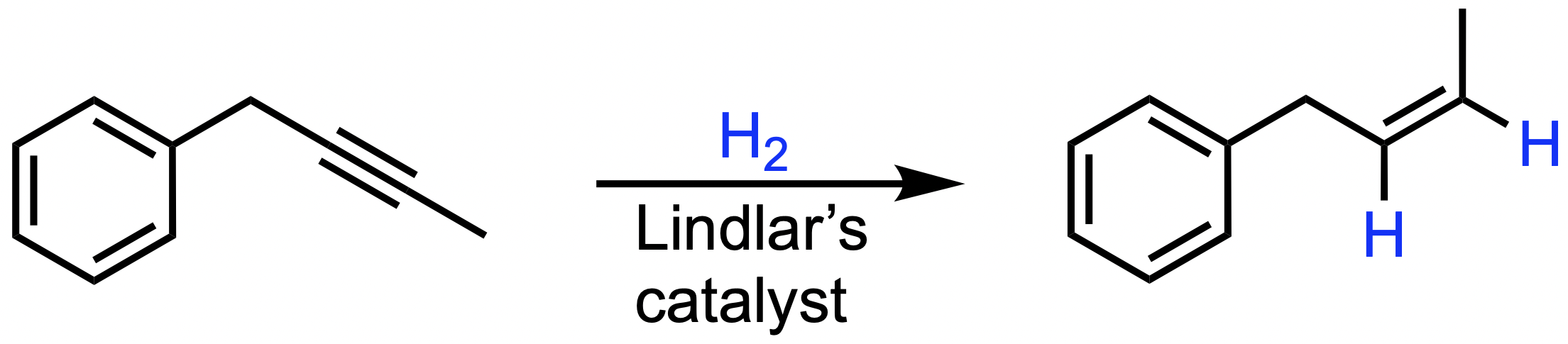
B. H2, Lindlar's catalyst
These reagents lead to the selective reduction of alkynes to cis alkenes via syn addition.
D. (1) LiAlH4, (2) H3O+
This set of reagents is used to reduce carbonyl groups and esters to alcohols. These reagents do not react with alkynes.
E. HgSO4, H2SO4
These reagents are used to hydrate alkynes, resulting in the formation of enols that tautomerize to ketones.

Key Takeaway:
Treating an alkyne with Li and NH3(l) at -78ºC results in the formation of a trans alkene.
What is the major product of the Markovnikov addition of HBr to the following molecule?
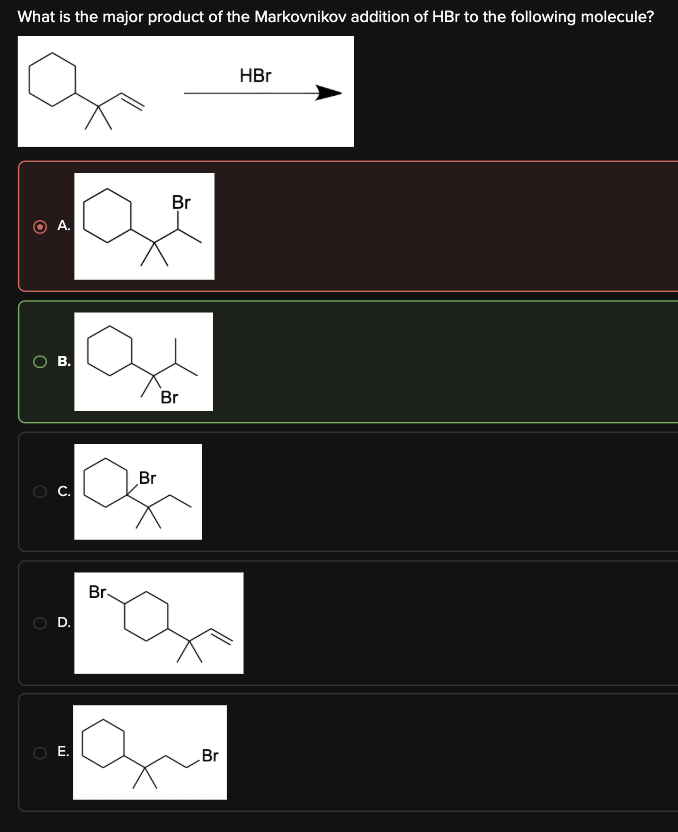
B
In this reaction, an alkene undergoes hydrohalogenation with HBr. The alkene initially forms a carbocation intermediate following Markovnikov's rule. However, instead of remaining in its original position, the carbocation rearranges itself to form a more stable intermediate. This reaction proceeds via the following mechanism:
The π electrons of the alkene double bond attack the H atom of HBr, forming a secondary carbocation intermediate.
The carbocation undergoes a 1,2-methyl shift to form a more stable tertiary carbocation intermediate.
The Br- ion attacks the carbocation, yielding the final product.
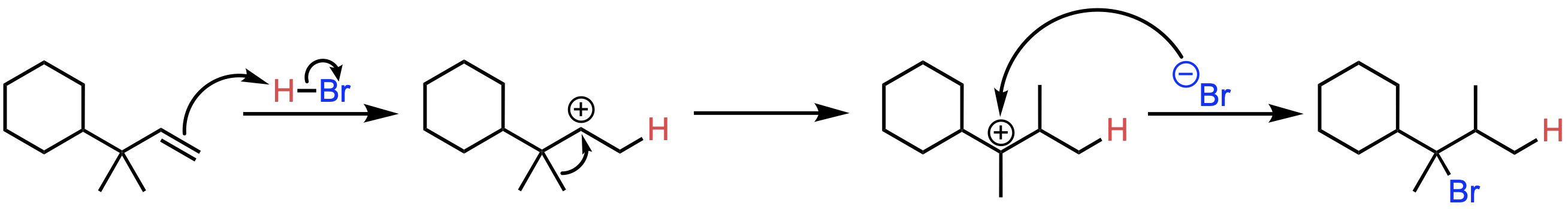
Key Takeaway:
In hydrohalogenation reactions, a carbocation may rearrange itself if a more stable intermediate can form.
Which of the following is the LEAST acidic?
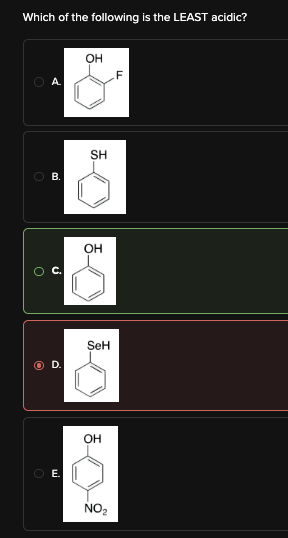
C
To determine the strongest acid, we use the acid ranking mnemonic CARDIO:
Charge
Atom
Resonance
Dipole Induction
Orbitals
The acidity of a compound increases as the compound's positive charge increases. Since none of the molecules in the answer choices have a charge, we cannot use this parameter to compare acidity.
Next, let's look at atom:
Choices [A], [C], and [E] have the acidic hydrogen bonded to oxygen.
Choice [B] has the acidic hydrogen bonded to sulfur.
Choice [D] has the acidic hydrogen bonded to selenium.
The acidity of a compound increases as the atom that the acidic hydrogen is bound to increases in size. In order of decreasing size: selenium > sulfur > oxygen. Since Choices [A], [C], and [E] have the acidic hydrogen bonded to the smallest atom (oxygen), they will be the least acidic acids.
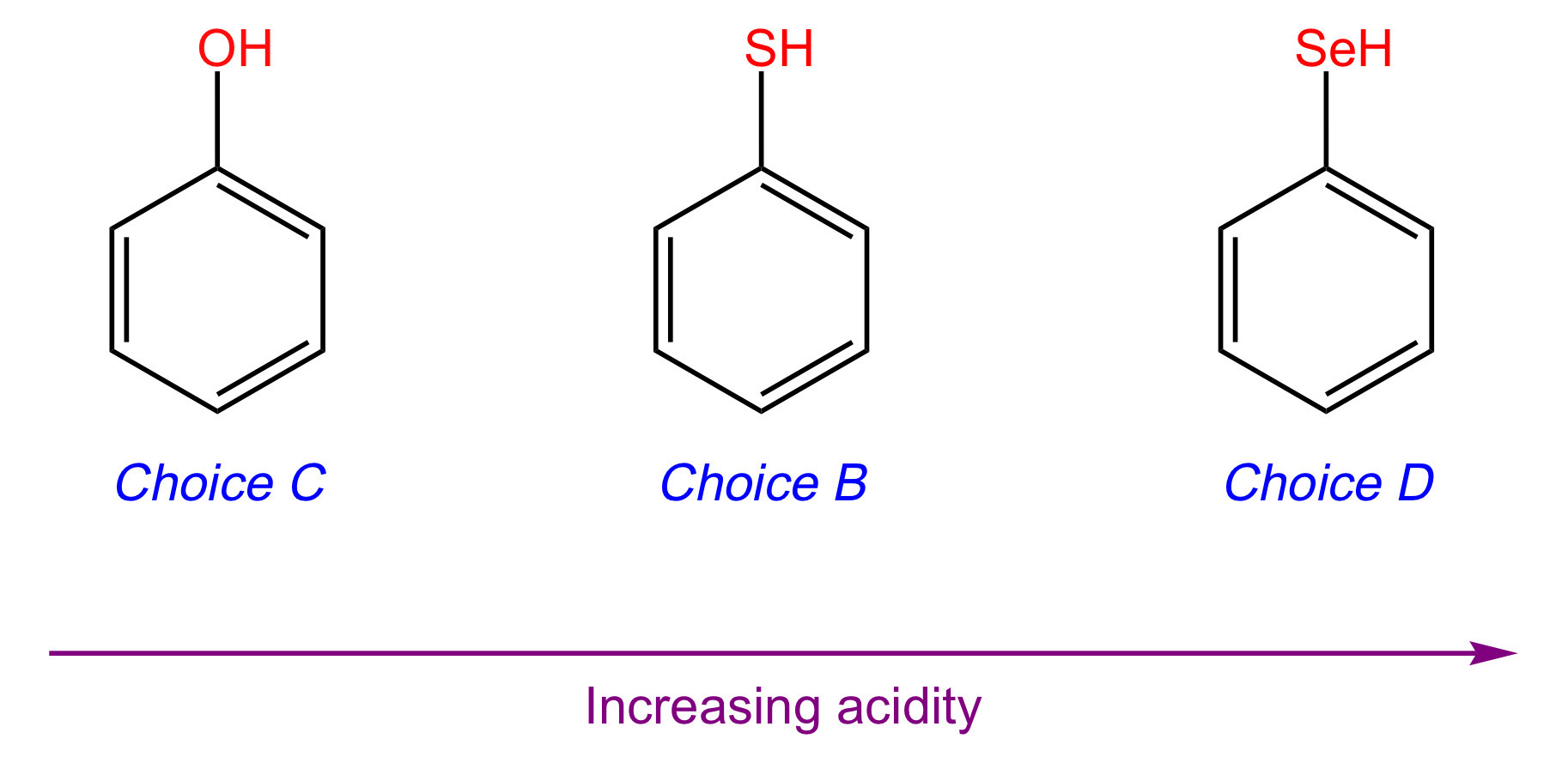
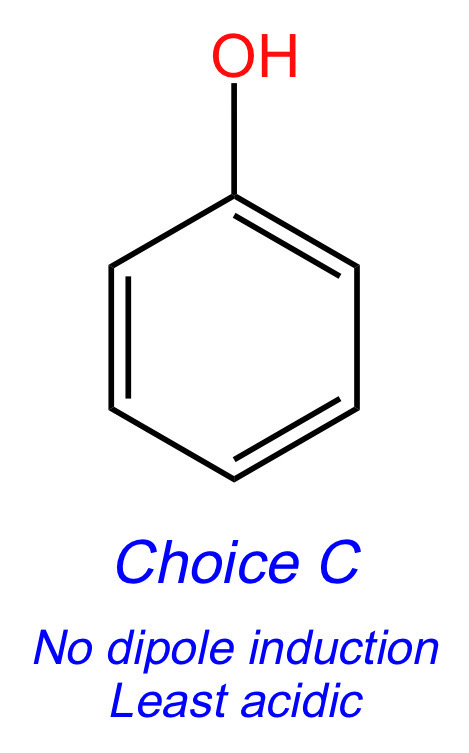
Choices [A], [C], and [E] all have resonance, so let's see if dipole induction will break the tie between these molecules. Choice [A] has F, and Choice [E] has NO2.
Both F and NO2 are electron-withdrawing groups that make the acidic hydrogen more acidic. Eliminate Choices [A] and [E]. Choice [C] is the least acidic acid.
Key Takeaway:
To determine the strongest acid, use the acid ranking mnemonic CARDIO.
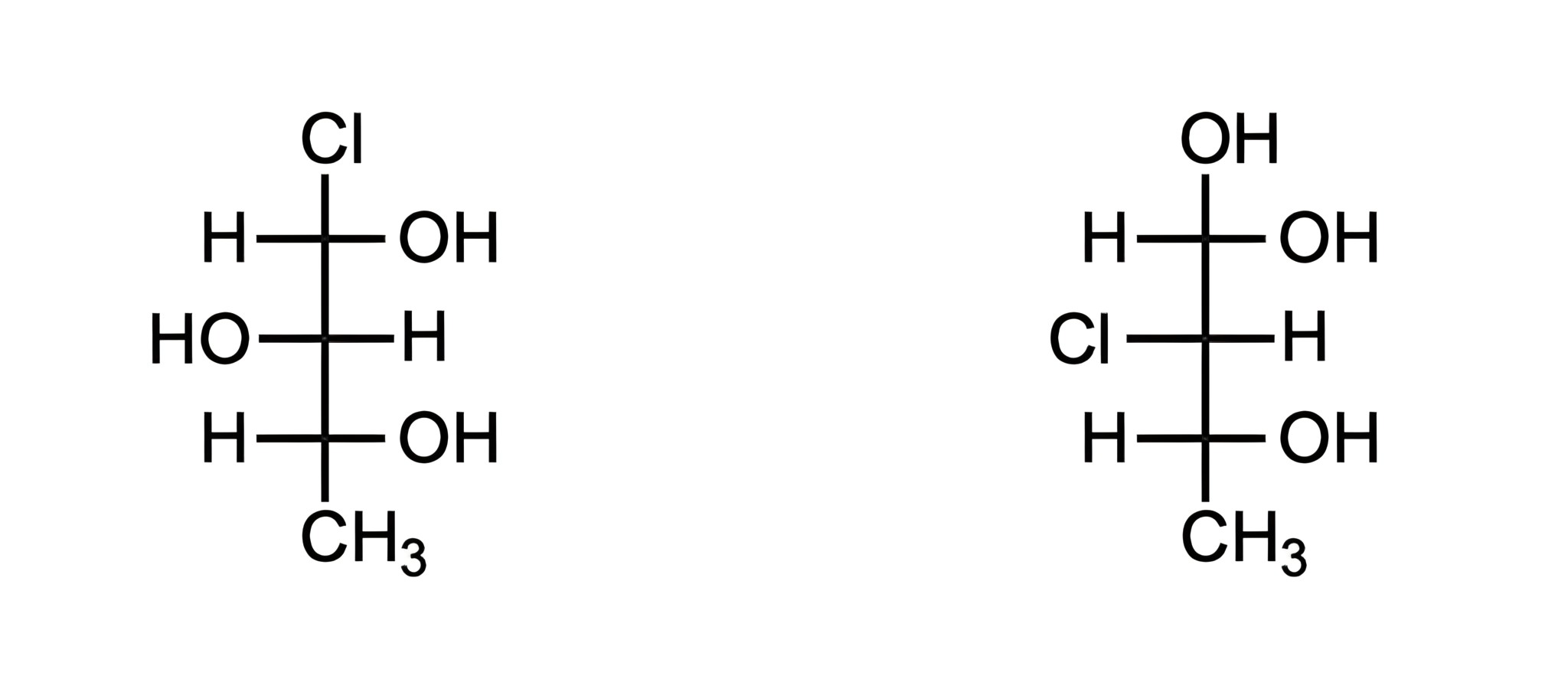
What is the relationship between the two Fischer projections?
A. Enantiomers
B. Diastereomers
C. Different molecules
D. Constitutional isomers
E. Identical
D
Looking at the two Fischer projections below, we can see that they have the same chemical formula (C4O3H9Cl), but that they differ in the arrangement of those atoms-making them isomers.

The key differences are:
On the left, Cl is located at carbon-1; whereas on the right, Cl is located at carbon-2.
On the left, OH is located at carbon-2; whereas on the right, OH is located at carbon-1.
Since these molecules have the same chemical formula, but they differ in the connectivity/location of those atoms, they are constitutional isomers.
DAT Pro-Tip: When comparing two molecules, use this decision tree to determine what kind of isomers they are.
Key Takeaway:
Constitutional isomers have the same chemical formula, but they differ in the connectivity/location of those atoms.
Which of the following would have the highest boiling point?
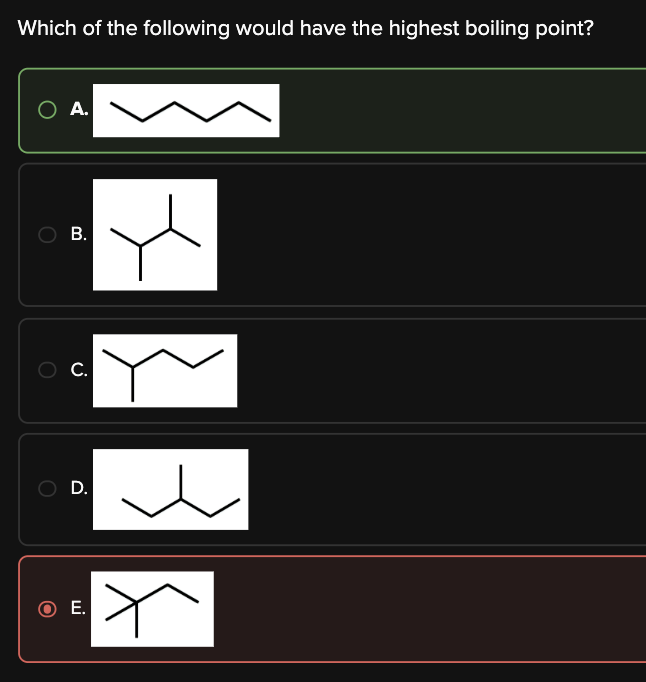
A
Usually, boiling point is determined by the strength of the intermolecular forces in the molecule. In order of decreasing strength of intermolecular forces: hydrogen bonding > dipole-dipole > dispersion forces.
However, all of the choices have the same chemical formula (C6H14) and mass. They differ in the amount of branching that they have.
Branching has the following effects on physical properties:
Branching increases the melting point.
Branching decreases the boiling point.
Thus, to determine which molecule has the highest boiling point, we must look for the molecule that has the least branching.
Choice [A] is a straight chain isomer (i.e. no branching), and will therefore have the highest boiling point.
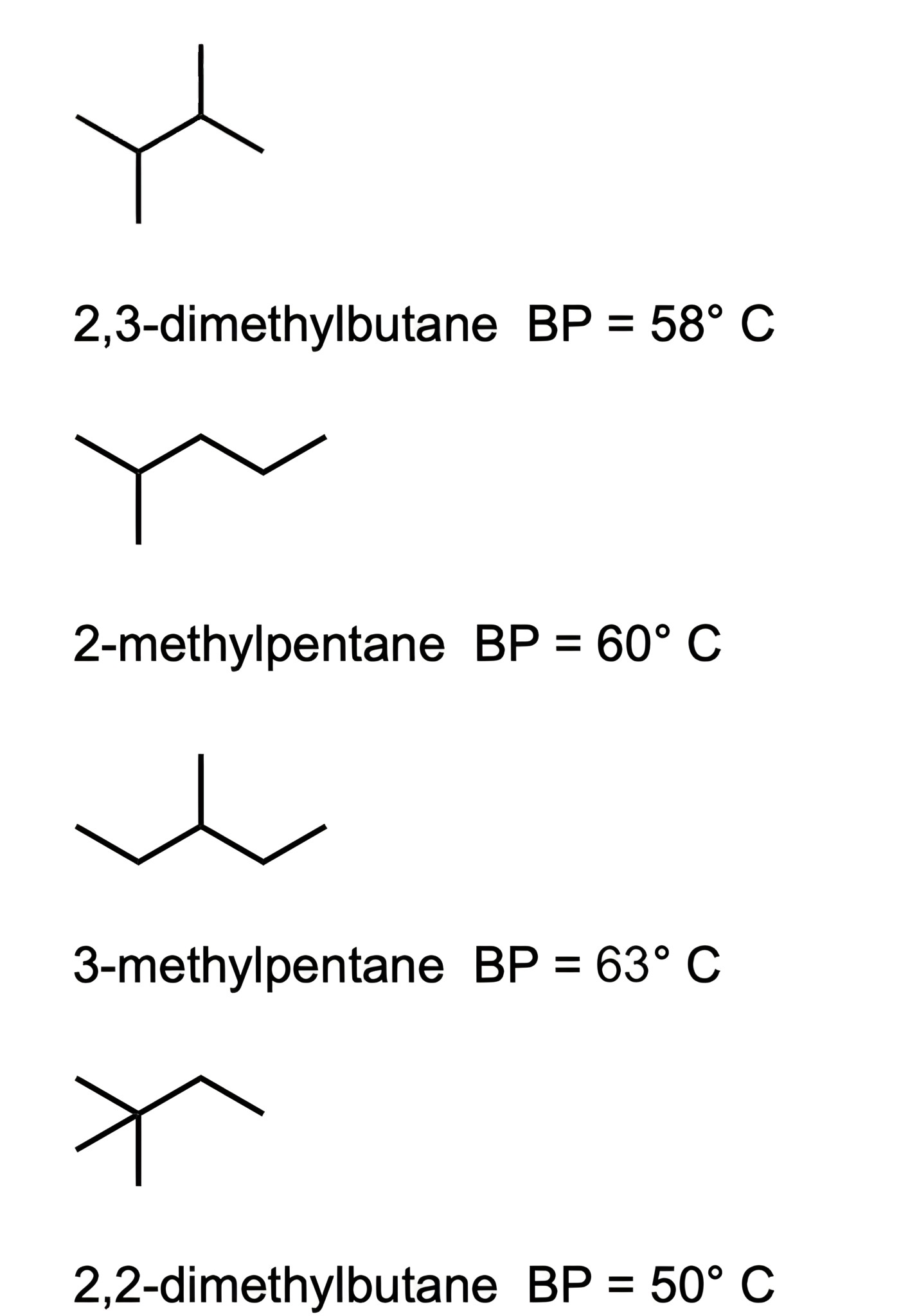
The remaining choices are all branched molecules, and will therefore have lower boiling points. Choice [E] is the most branched molecule, and will therefore have the lowest boiling point.
Key Takeaway:
Branching has the following effects on physical properties:
Branching increases the melting point.
Branching decreases the boiling point.
Which orbital does the lone pair on nitrogen reside in?
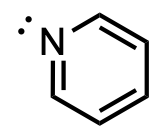
A. 1s
B. 2s
C. 2p
D. sp2
E. sp3
D
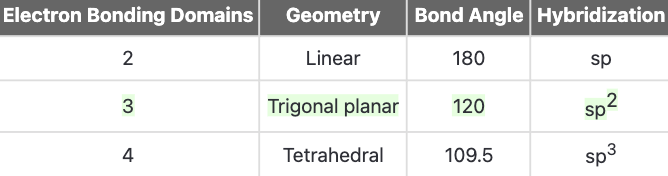
As the nitrogen has 3 electron domains, it is sp2-hybridized. Therefore, the lone pair electron domain will be sp2-hybridized. The lone pair does not participate in resonance as doing so would break aromaticity and lead to an unstable molecule.
A. 1s, B. 2s
The lone pair is one of the three electron domains, therefore it resides in an sp2 orbital.
C. 2p
The lone pair does not participate in resonance as it is not needed to complete aromaticity. In pyridine, there are 6 pi electrons already in the ring, fulfilling the 4n+2 rule. Therefore, the lone pair is not involved in pi bonding and is not in a 2p orbital.
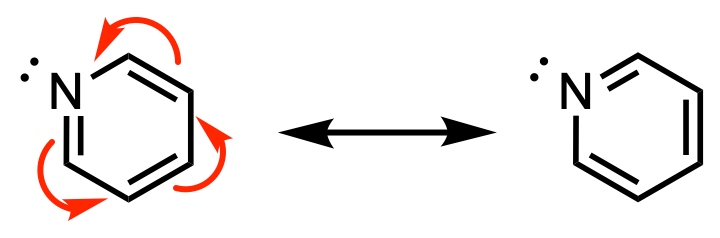
The lone pair on pyridine is does not participate in aromaticity, therefore it resides in an sp2 orbital.
In contrast, in pyrrole (shown below), the lone pair is required to participate in resonance to complete aromaticity. Without the lone pair, pyrrole only has 4 electrons in the ring. If the lone pair is involved in resonance to achieve aromaticity, the lone pair will be located in a 2p orbital. Thus in pyrrole, the lone pair is in a 2p orbital.
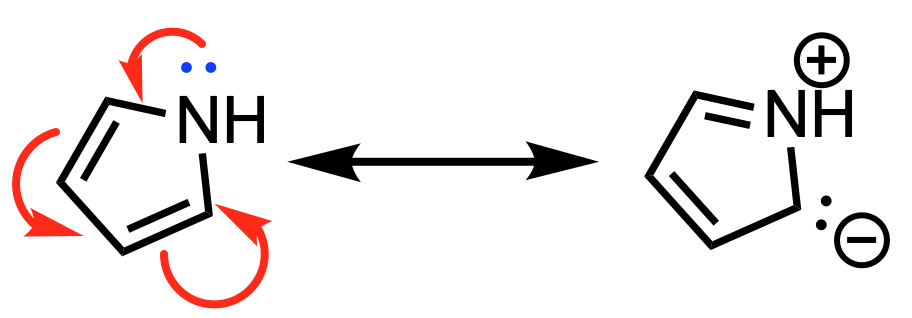
The lone pair in pyrrole participates in aromaticity, therefore it resides in a 2p orbital.
E. sp3
The nitrogen has 3 electron domains, so it is sp2-hybridized.
Key Takeaway:
If a lone pair is involved in pi bonding through resonance, it resides in a p orbital. If it does not participate in resonance, it resides in the atom's hybridized state.
Which of the following molecules would show only two singlets in an 1H NMR spectrum?
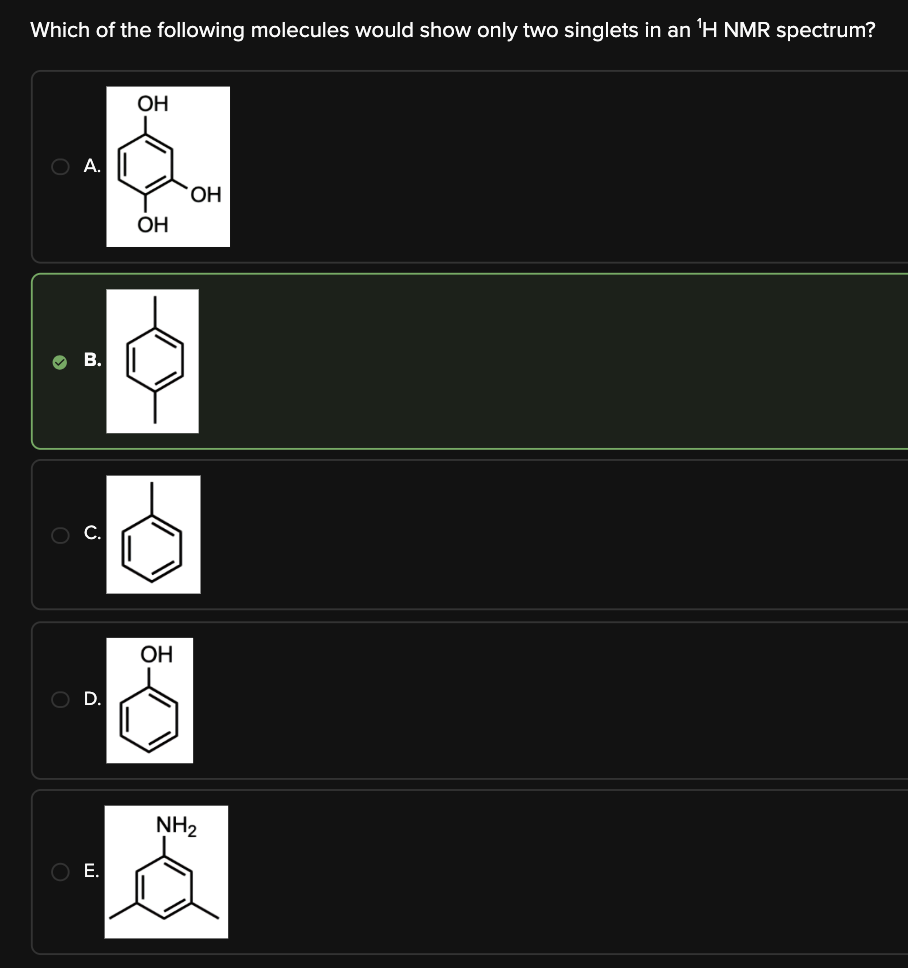
B
This question asks us to determine the molecule that would show only two singlets in an 1H NMR spectrum.
By finding the planes of symmetry in each molecule, we can determine the number of unique H's and how many signals they would show in an 1H NMR spectrum.
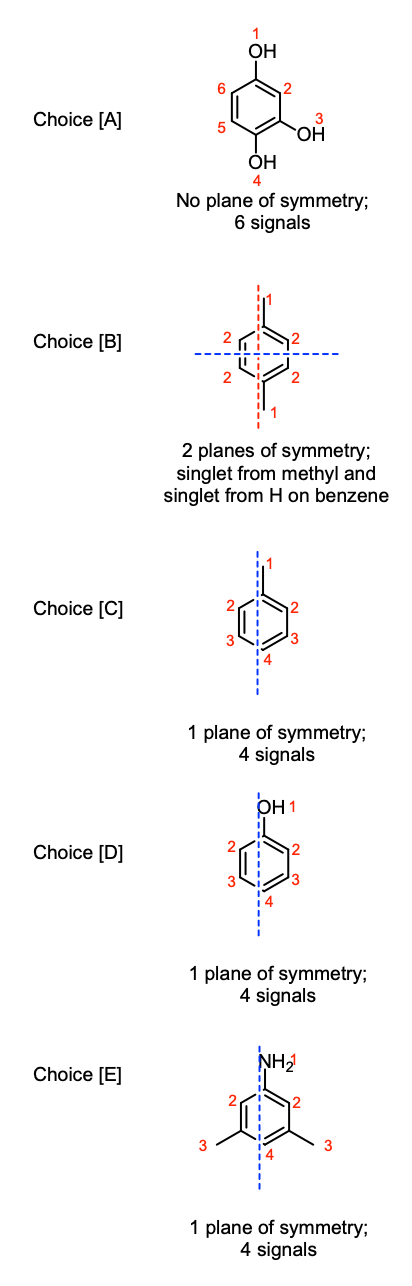
Let's look at Choice [B]. This molecule has two unique H's. We use the "n+1 rule" to determine its splitting pattern, where n is the number of hydrogens attached to adjacent atoms.
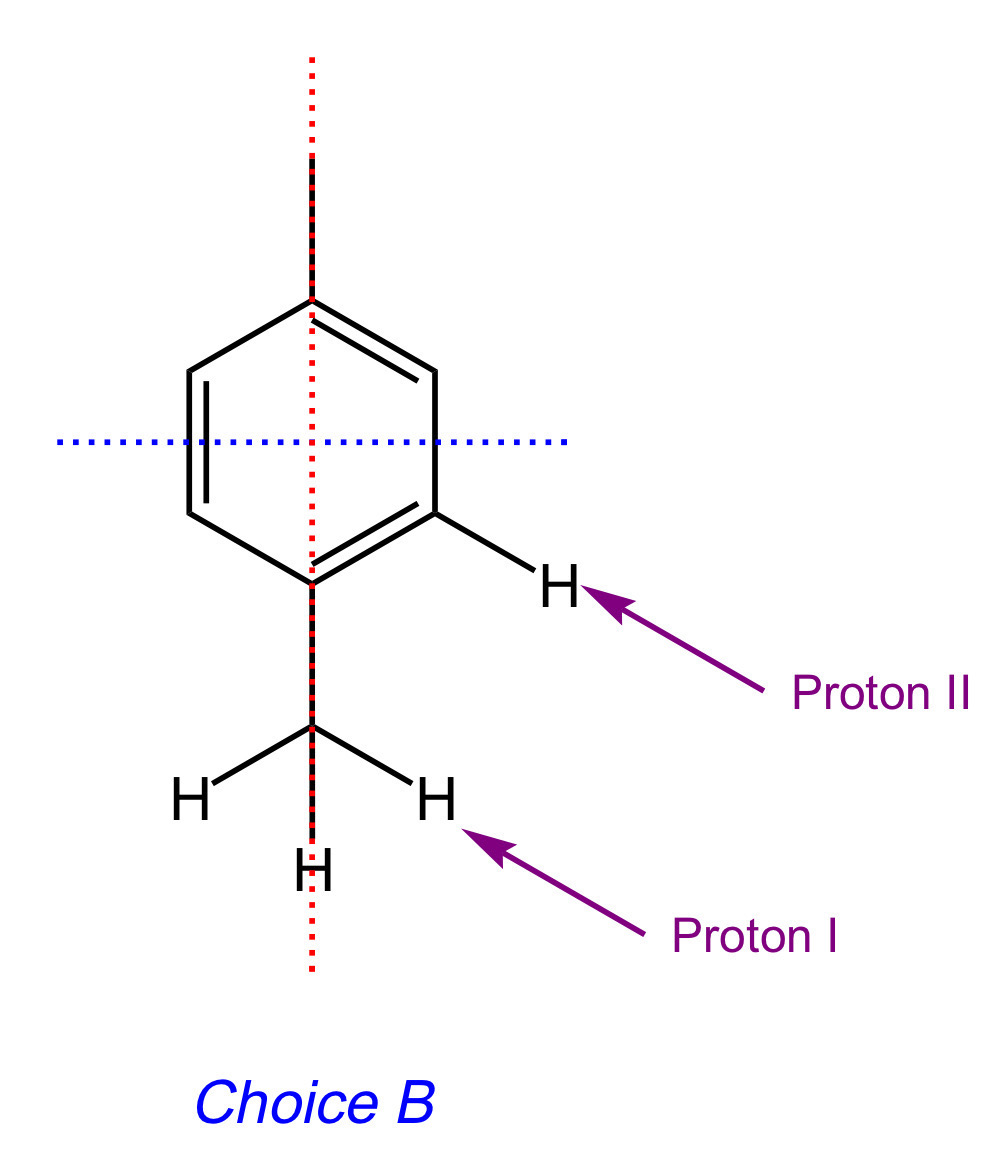
Looking at Proton I, we can see that there are zero non-equivalent hydrogens coming off of the adjacent carbon (i.e. "next door" to Proton I). Therefore, the signal for Proton I will be a singlet.
Looking at Proton II, we can see that there are zero non-equivalent hydrogens coming off of the adjacent carbon. Therefore, the signal for Proton II will be a singlet.
Below is the 1H NMR spectrum for Choice [B], known as p-xylene or 1,4-dimethylbenzene. The signal for Proton I will appear at 2 ppm, and the signal for Proton II will appear at 7 ppm.
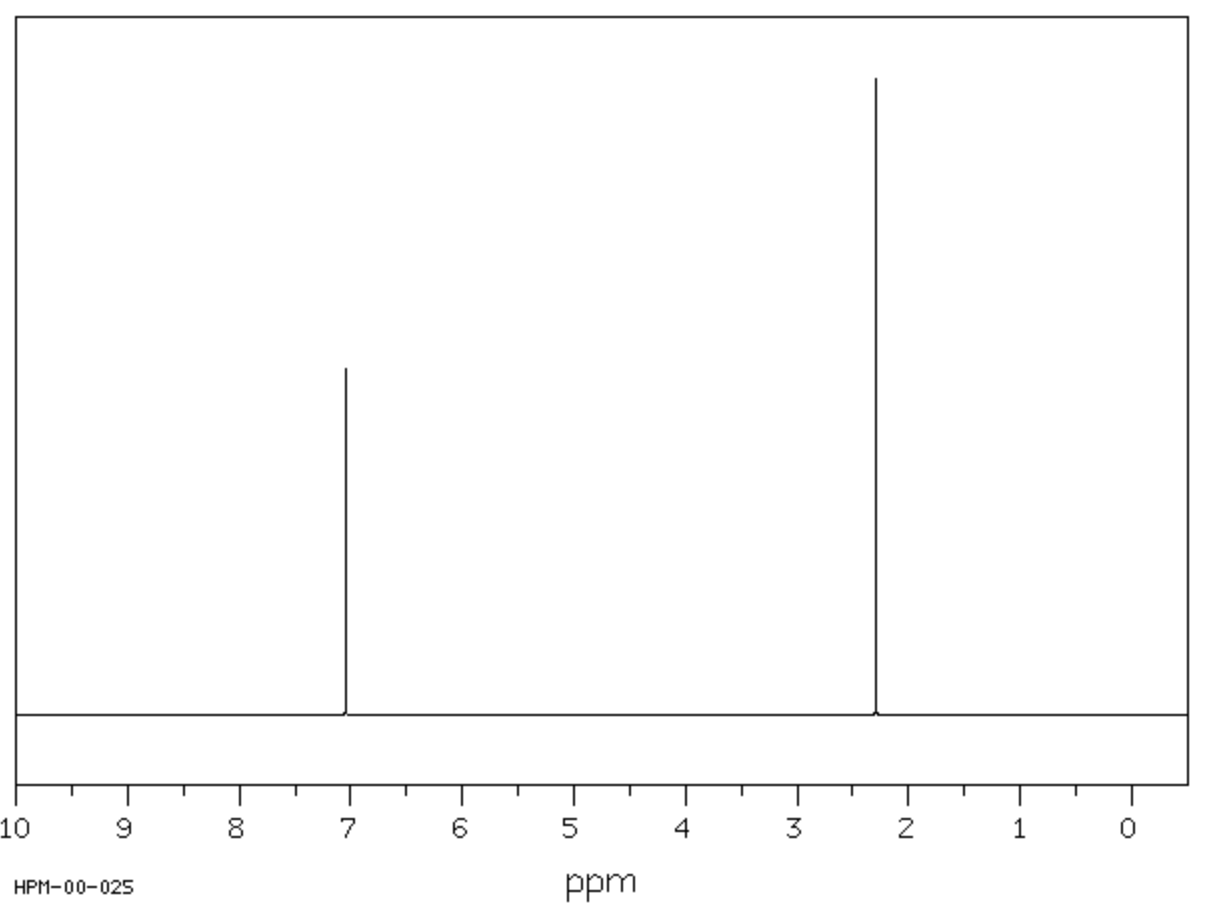
Key Takeaway:
The "n+1" rule is used to determine the splitting pattern of hydrogens on 1H-NMR, where n is the number of hydrogens on adjacent atoms with unique chemical environments.
Which of the following products will form the fastest in an elimination reaction?
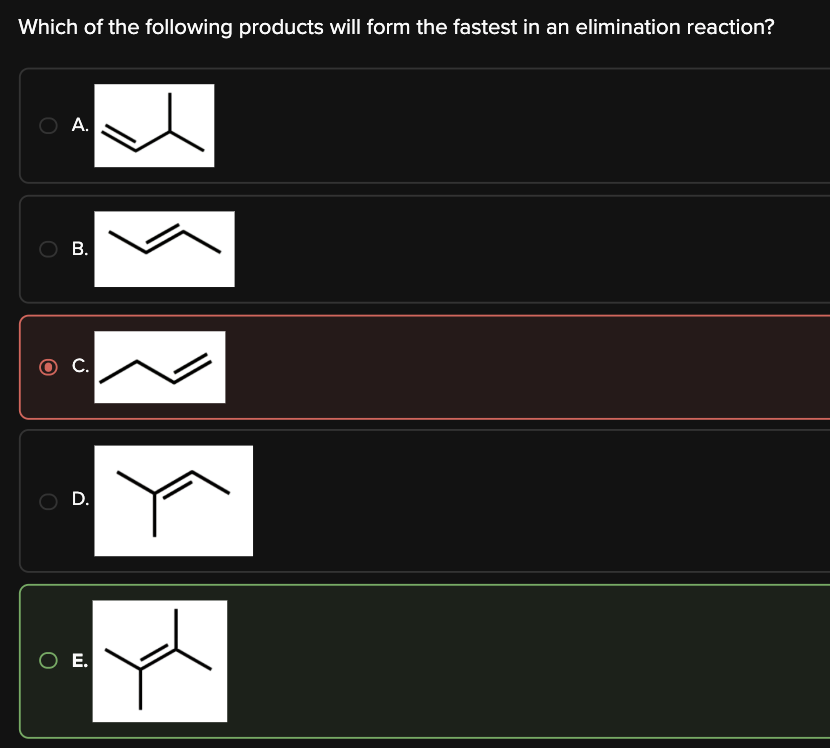
E
This question deals with elimination reactions, so let's quickly review E1 vs. E2:
E1 vs. E2 Reactions | ||
E2 | E1 | |
Base | strong | weak |
Electrophile | 3° > 2° > 1° | 3° > 2° (no 1° or methyl) |
Solvent | aprotic preferred | protic |
Leaving Group | I > Br > Cl > F | I > Br > Cl > F |
Rearrangement | Not possible | Possible (hydride/methyl shifts) |
Stereochemistry | Antiperiplanar | None |
Intermediate | None, uses high energy transition state | Carbocation Intermediate |
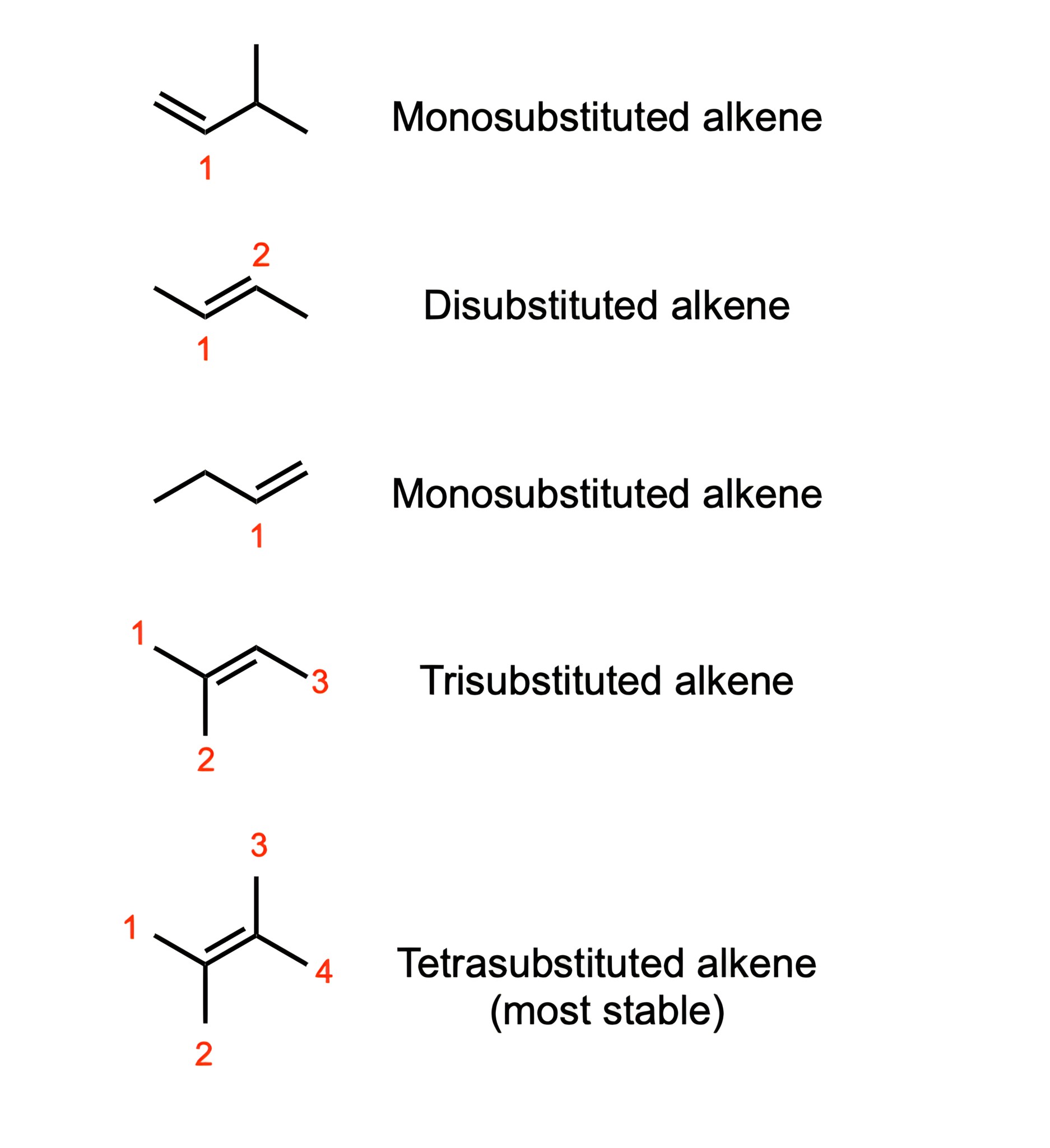
Both E1 and E2 reactions prefer 3° alkyl halides in order to form the most substituted alkene; this is known as Zaitsev's rule.
Since 3° alkyl halides are the most substituted, they will have the most stable transition state or carbocation intermediate, thereby allowing it to have the lowest activation energy possible. This results in the formation of the most substituted alkene.
Therefore, to determine which product will form the fastest in an elimination reaction, we must look for the most substituted alkene. Choice [E] is the answer.
Key Takeaway:
The more substituted the final alkene, the faster the E1/E2 reaction will be.
What is the major product of the following reaction?
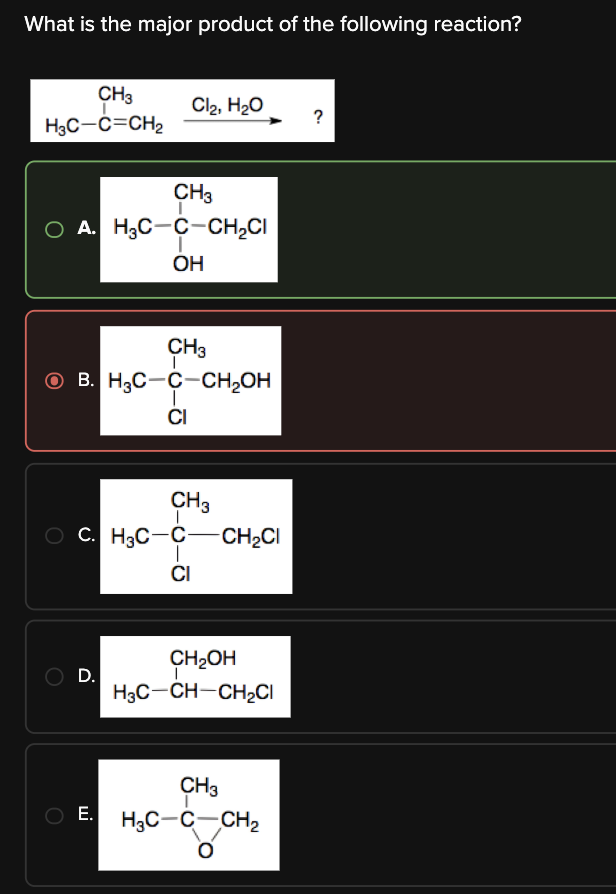
A
The reaction below results in the anti addition of Cl and OH to form a chloro alcohol.
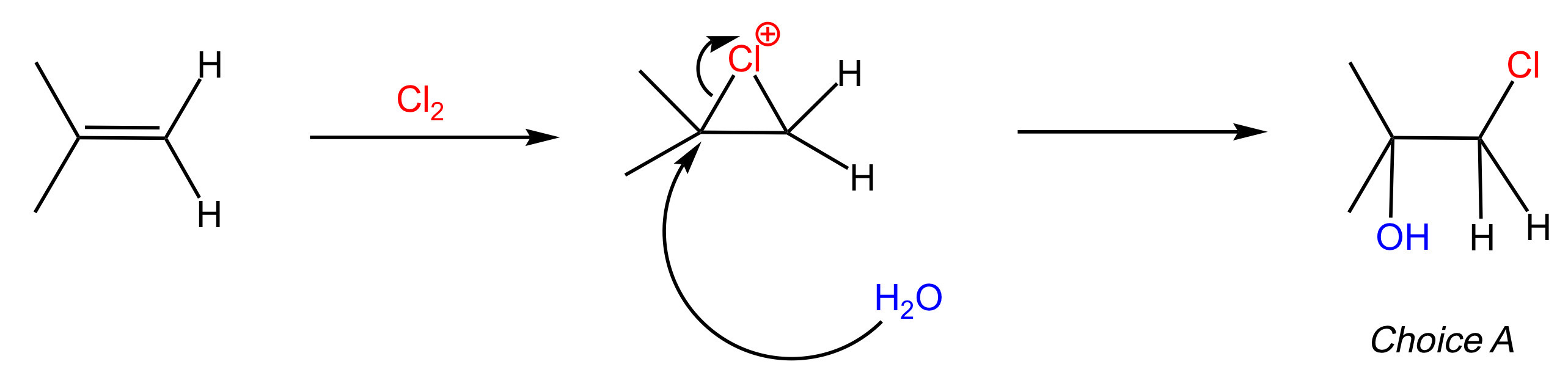
The reaction proceeds as follows:
The alkene reacts with Cl2 to form a chloronium intermediate.
H2O attacks the most substituted carbon of the chloronium intermediate, thereby opening the ring.
Deprotonation leads to the formation of the final product.
Key Takeaway:
The reaction between an alkene and Cl2/H2O results in the anti addition of Cl and OH, with OH added to the most substituted alkene carbon.
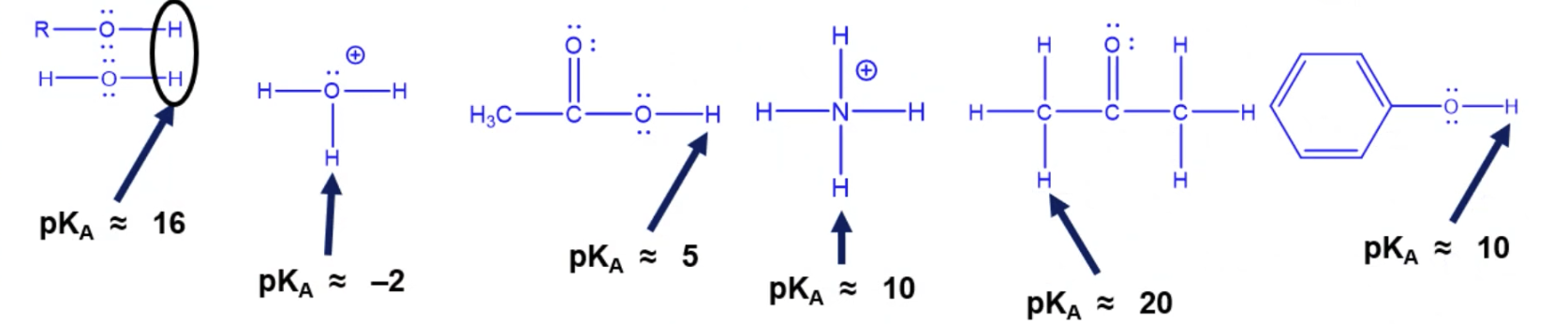
Memorize these pKa’s and structures

Memorize which is which
What are the three criteria a compound must meet to be aromatic?
1. Must be cyclic or polycyclic
2. All the atoms in the ring must be sp2 or sp hybridized
3. Huckel's rule: The number of pi e- in the ring must equal 4n + 2 (n = 0, 1, 2, etc)