Cell and Molec Final
1/48
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
49 Terms
Describe the process of turning on a G-Protein Coupled Receptor
A signaling molecule binds to the inactive receptor and changes its conformation
This removes GDP from the G protein’s alpha subunit, allowing GTP to bind, activating the complex.
This releases the beta and gamma subunit from the alpha subunit, where any subunit can cause effector activation.
This allows the target protein to bind to the alpha subunit, causing an effect
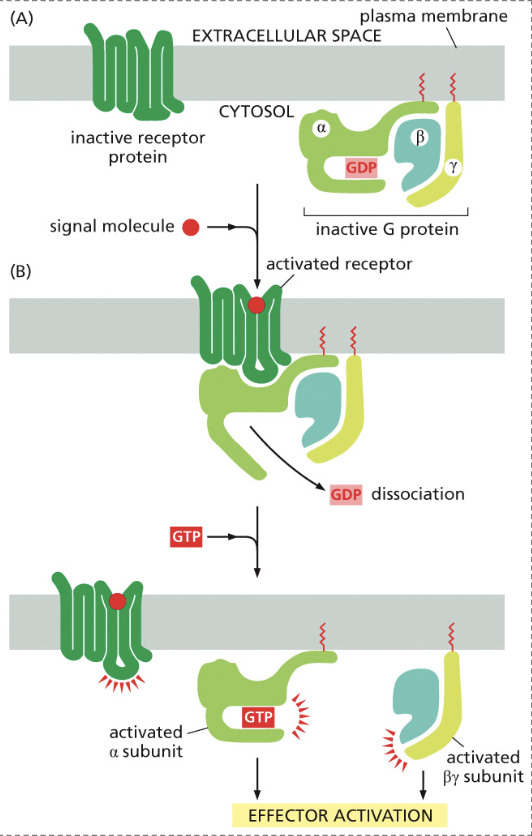
How is the G-Coupled Receptor Complex turned off?
By hydrolyzing the bound GTP to GDP, the subunit inactivates and causes it to dissociate from the target protein. From there, the alpha and beta subunits reassemble to reform an inactive G protein
When GTP is bound is the complex on or off?
It is on
Explain the steps of how epinephrine decreases glycogen levels
Epinephrine binds to the G-protein-coupled receptor and activates the alpha subunit of the G protein.
Once the alpha binds, adenylyl cyclase is activated.
This enzyme takes ATP to cyclic AMP(cAMP) to activate PKA (protein kinase A).
PKA then phosphorylates the next protein in the pathway, where effectors either activate glycogen phosphorylase or inactivate glycogen synthase to decrease glycogen.
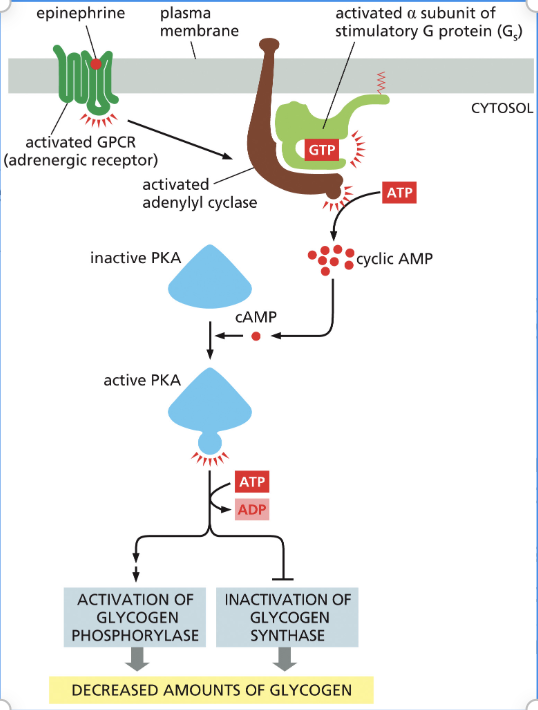
When GDP is bound is the complex on or off?
It is off
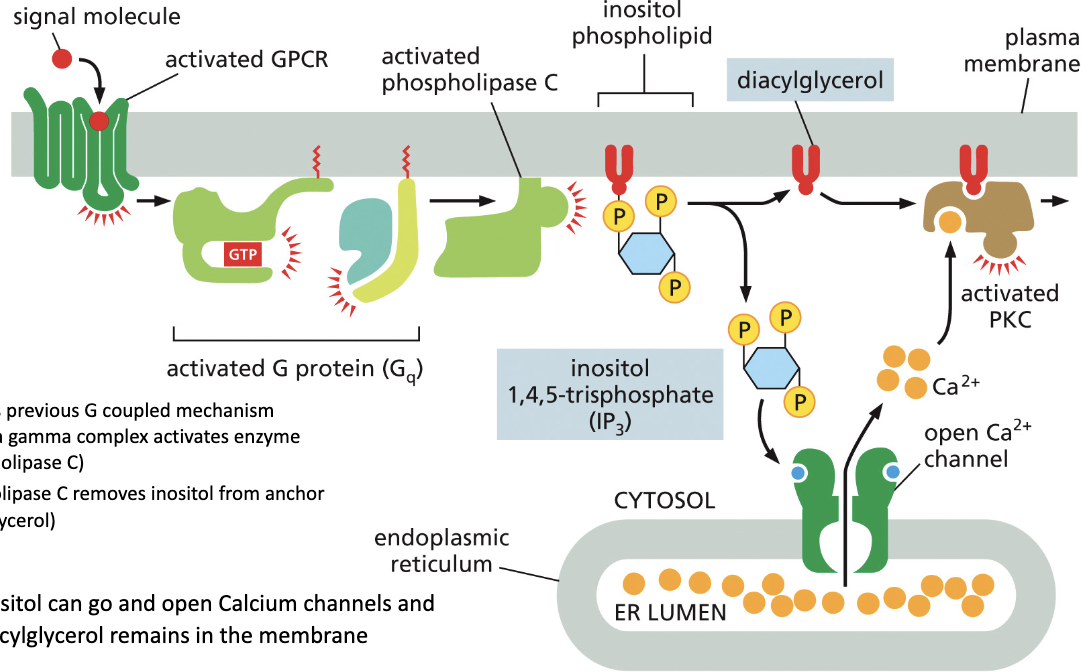
Describe the Phospholipase C pathway.
A signaling molecule binds to the inactive receptor and changes its conformation
This removes GDP from the G protein’s alpha subunit, allowing GTP to bind, activating phospholipase C.
Activated removes inositol from is anchor
Inositol then can go and open calcium ion channels while the anchor remains in the membrane.
The calcium the activates PKC on the original anchor activating effector proteins.
Can inactive PKA enter the nucleus?
No, the nuclear localization sequence present in PKA is only visible when activated by cAMP.
How doe cAMP result in gene expression?
By binding to inactive PKA, it activates it, allowing it to enter the nucleus and activate transcription regulators for gene expression.
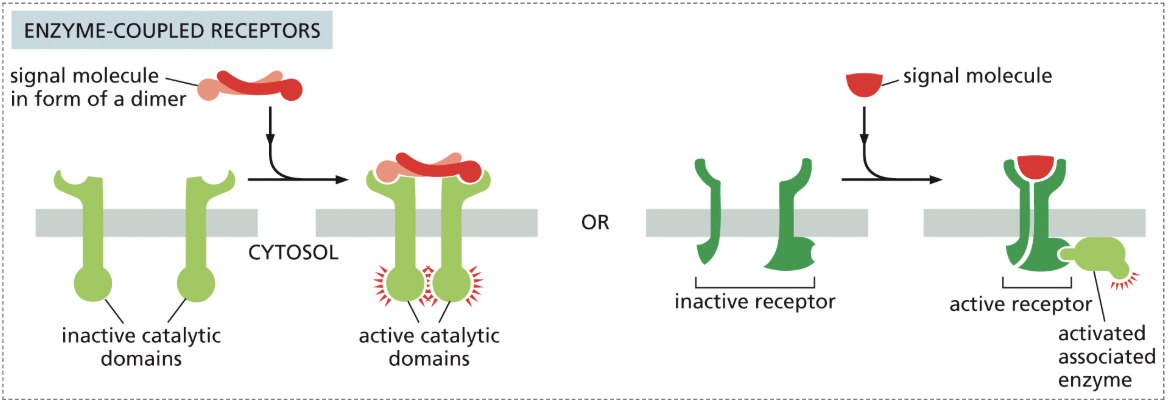
Describe the process of Receptor Tyrosine Kinase (RTKs)
An inactive RTK enzyme-coupled receptor has a large space between the two inactive monomers, inhibiting function
A signal molecule dimer binds, bringing both monomers close, causing dimerization
This activates the kinase portion of the receptor
Each part of the dimer then phosphorylates its partner on the other dimer (cross-phosphorylation)
Proteins recognize those sites, and they serve as docking stations for activated intracellular signaling proteins
These signal proteins activate downstream pathways
Describe the MAP pathway
An adaptor protein attached to a Ras-GEF docks on the activated RTK
An inactive Ras protein phosphorylates, removing GDP from its complex and binding GTP
This activated Ras protein activates MAP kinase kinase kinase
ATP is phosphorylated to ADP, which activates MAP kinase kinase
This step repeats to form an activated MAP kinase
MAP kinase then activates protein effectors for changes in protein activity or changes in gene expression
Describe the Pl-3-kinase-Akt pathway
Activated Pl-3-kinase binds to an active RTK
Inositol phospholipid is phosphorylated by the PI-3-kinase
This phosphorylates protein kinase 1, which activates Akt
Akt phosphorylates active BAD(pro-apoptotic protein), which is attached to an inactive Bcl2(anti-apoptotic protein)
When BAD is phosphorylated, it is inactivated and releases active Bcl2, which stimulates cell survival
What extracellular signal will act fastest
A protein that isn’t always used but is available when needed. This requires lots of energy but yields a faster response.
What kinds of molecules can bind to intracellular receptors?
Small hydrophobic signaling molecules
What is the range of paracrine signaling?
It is typically short range impacting nearby cells by traveling through the extracellular space.
What is the range of endocrine signaling?
It is a long range signal traveling through blood
What is the range of neuronal signaling?
A highly specific short range signal that has one neuron to a synapse of the nearby target cell
How intermediate filaments made?
A monomer associates with a second monomer and coils around each other to form a dimer
This formed dimer coils with another dimer to make a tetramer
It then organizes to be antiparallel
8 tetramers then laterally associate with another 8 tetramers to form a growing filament
What are the four main types of intermediate filaments and where are they found?
Keratin filaments: Epithelial cells
Vimentin filaments: Connective tissue, muscle cells
Neurofilaments: Nerve cells
Nuclear lamins: All animal cells
How does keratin mutation impact cell integrity?
Certain mutations will impact the strength of the monomer by inhibiting coiling.
What is plectin?
It is a protein that aids in the bundling of intermediate filaments and cross links microtubules to microfilaments to create stronger support
What is the nuclear lamina?
It is an intermediate filament that resides within the nucleus, allowing for the maintenance of the nuclear structure
Describe the structure of microtubules.
Tubulin dimers associate to form a microtubule subunit
Many subunits form protofilaments oriented with a plus end made of exposed beta tubulin and a minus end of exposed alpha tubulin
Multiple protofilaments connect to form a hollow tube, making the microtubule
What is the MTOC?
It is the microtubule-organizing center where microtubules are synthesized, with the minus end anchored in the centromere and the plus end extending out
Describe the process of microtubule growth.
GTP-tubulin dimers are added to the plus end of a microtubule
This addition proceeds faster than the hydrolysis of GTP by the dimers causing crowth
Describe the process of microtubule shrinkage.
GTP is hydrolyzed faster than the addition of new dimers
When tubulin binds to GDP, there is a weak affinity, so the protofilaments with GDP-tubulin begin to peel away, causing shrinkage
The GDP-tubulin is then released into the cytosol
What is the MTOC called in a non-dividing cell called, a dividing cell, and a ciliated cell?
Centrosome
Mitotic Spindle
Basal body
Describe how motor proteins transport cargo.
The two heads of the motor protein are attached to the microtubule
The tail domain either interacts directly with cargo by attaching to it or uses an adaptor protein to indirectly attach
ATP hydrolysis loosens the attachment of head one
ADP is released, causing ATP to bind, changing the conformation of head one, pulling head two forward
This movement only goes towards the minus end(negative motor dynein) or the plus end(positive motor kinesin) of the microtubule
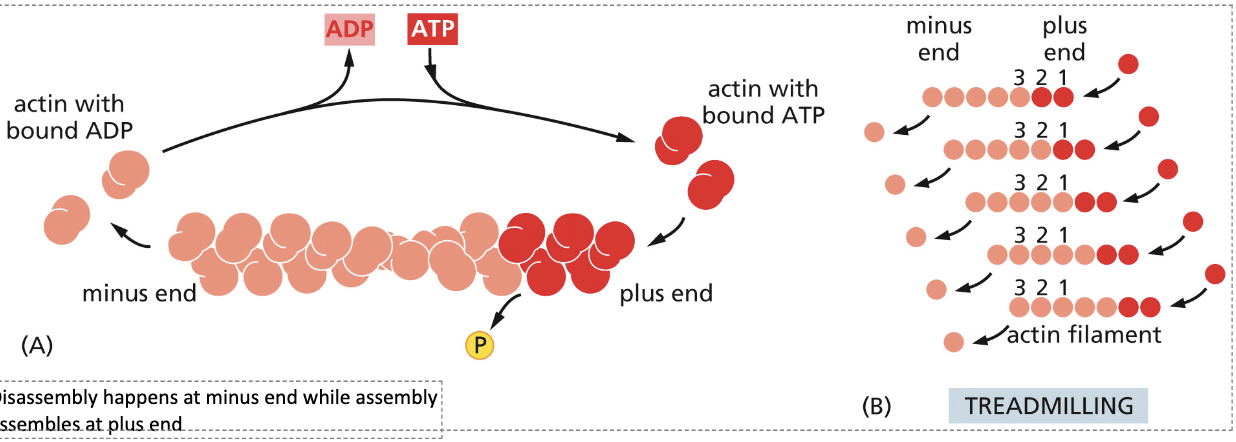
What is the structure of actin filaments?
Two sets of actin monomers twisted around each other with a plus and minus end.
Describe tread-milling.
The disassembly of actin bound with ADP on the minus end, while assembly occurs with actin bound to ATP on the plus end.
Describe the pathways of the actin binding motor protein myosin-I.
The head domain of myosin-I interacts with the actin filaments walking towards the plus end of the filament
The tail domain acts on cargo or whatever it is transporting as it treadmills over to the destination
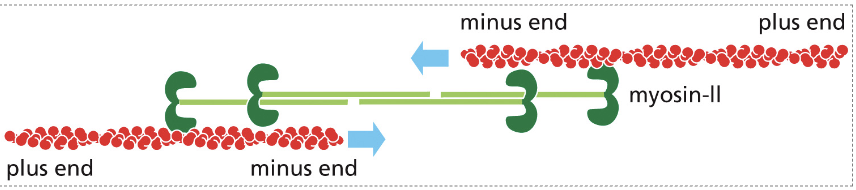
Describe how muscle contraction works.
The myosin-II head interacts with an actin filament while the tail interacts with other tail domains
This forms a double arrow line with plus ends on either side with two minus ends in the center
The plus ended motor contracts to slide towards the opposite plus end with the other plus end does the same thing
This creates a sliding movement to create contraction
What is the structure of a sarcomere?
It is consisted of a thick(myosin-II) and thin(actin) filaments with a z disc on either side of the overlap region.
How does calcium play a role in muscle contraction?
T-tubule becomes activated by neuronal trigger causing an action potential
Calcium is released from the sarcoplasmic reticulum into the cytosol via a voltage gated channel
This action caused myofibril contraction
Tropomyosin is pushed off the myosin binding site when calcium binds to troponin
This reveals the myosin-II binding site allowing myosin-II to bind moving it towards the plus end causing contraction
What are the phases of the cell cycle and what are their primary roles?
G1 Phase: Majority of cell growth, high metabolism, repairs
S Phase:
G2: Phase
M Phase:
What is the cell cycle control system?
A system of progression based on the accumulation of cyclins to regulate the activity of Cdks (cyclin-dependent protein kinase) to push cells into the next phase
How do Cdks influence G1 phase?
Mitogen binds to extracellular receptor to trigger intracellular signal
This activates G1-Cdk and G1/S Cdk to phosphorylate retinoblastoma (RB) (retinoblastoma keeps the cell off)
This inactivates it allowing the activation of a transcription regulator to transcribe genes for the entry into S phase
What happens in G1 phase if the DNA is damaged?
Damage activates signaling pathway to phosphorylate p53 keeping it from being degraded
The activated p53 binds to the regulatory region of p21 (a Cdk inhibitor)
This inactivation halts the cycle allowing for repair
How do Cdks influence the S phase
In G1 phase DNA helicase is recruited to bind to the origin recognition complex on a strand of DNA
S-Cdk then phosphorylates helicase to activate it where it begins to unwind the DNA
S-Cdk will also phosphorylate the ORC to turn it off so replication can’t start from the origin more than once
How are Cdks degraded?
Ubiquitylation of cyclin by APC/C will occur signaling the cyclin for destruction in the proteasome
What must occur for M-Cdk to be active?
Inhibitory kinase (Wee 1) adds inhibitor phosphates to inactivate Cdk in rest state
Cdc25 (phosphatase) will remove the inhibitor phosphates allowing for the activatation of M-Cdk
How does Cdk effect M-phase?
After the activation of M-Cdk, M-Cdk has a positive feedback on the activation of Cdc25 phosphatase.
What are the ways in which the complexes of the cell cycle prevent the recycling of the cells?
Elimination of all existing Cdks
Blocking the synthesis of new cyclins
Deployment of Cdk-cyclin inhibitors
What is G0?
The checkpoint phase before the start of S phase that determines whether or not to pause or continue depending on whether or not anything is wrong/need to be fixed