Essential OCHEM 2 Final Terms & Definitions for Chemistry Exam
1/51
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
52 Terms
what is limiting reactant
calculate theoretical yield. LR will be the smaller number
grams to moles
grams of substance / MW
which of the following are not essential from Diels-Alder reaction
electrophile
which of the following cannot be purified by recrystallization
liquid or gas compound
which of the following is true about reduction reaction
loss of oxygen and addition of hydrogen atoms
what satisfies the Diels-Alder mechanism
4+2 addition
which of the following is commonly used as an oxidizing agent for carbonyl group
LiAlH4 and NaBH4
which of the following statements is true abut selecting the solvent for recrystallization
solvents BP must be lower than the samples MP
witting reaction is useful for the following synthesis
alkene
which of the following will you use to deprotanate the phosphonium ion
NaOH
What is the side product for Wittig reaction
triphenylphosphine oxide
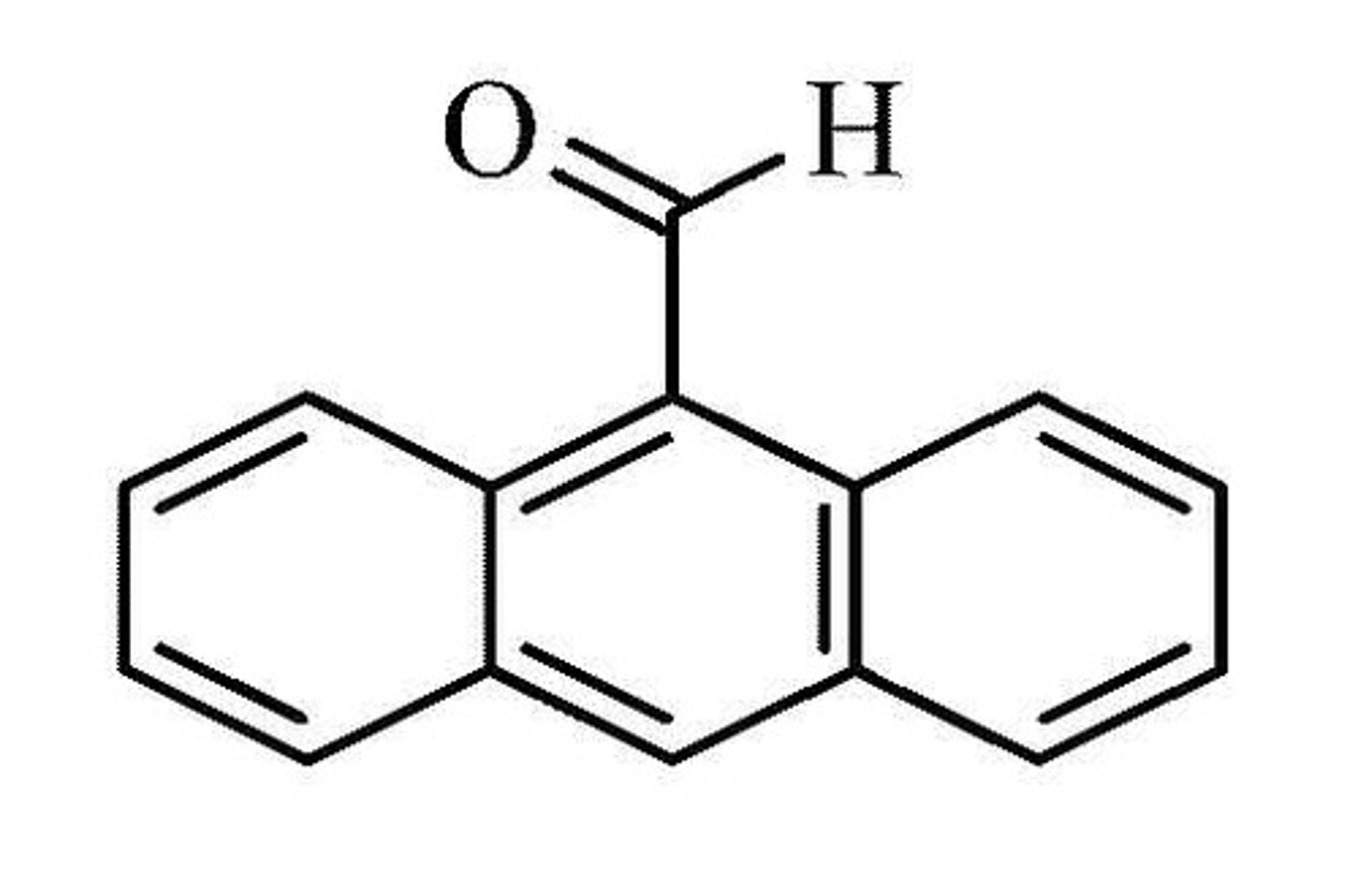
which of the following is 9-anthraldehyde
which of the following reagent will you use for 'Lucas Test'
ZnCl2
in oxidation test which is the reagent you use
CrO3
which of the following will oxidize in 'Jones' test
2-methylcyclohexanol
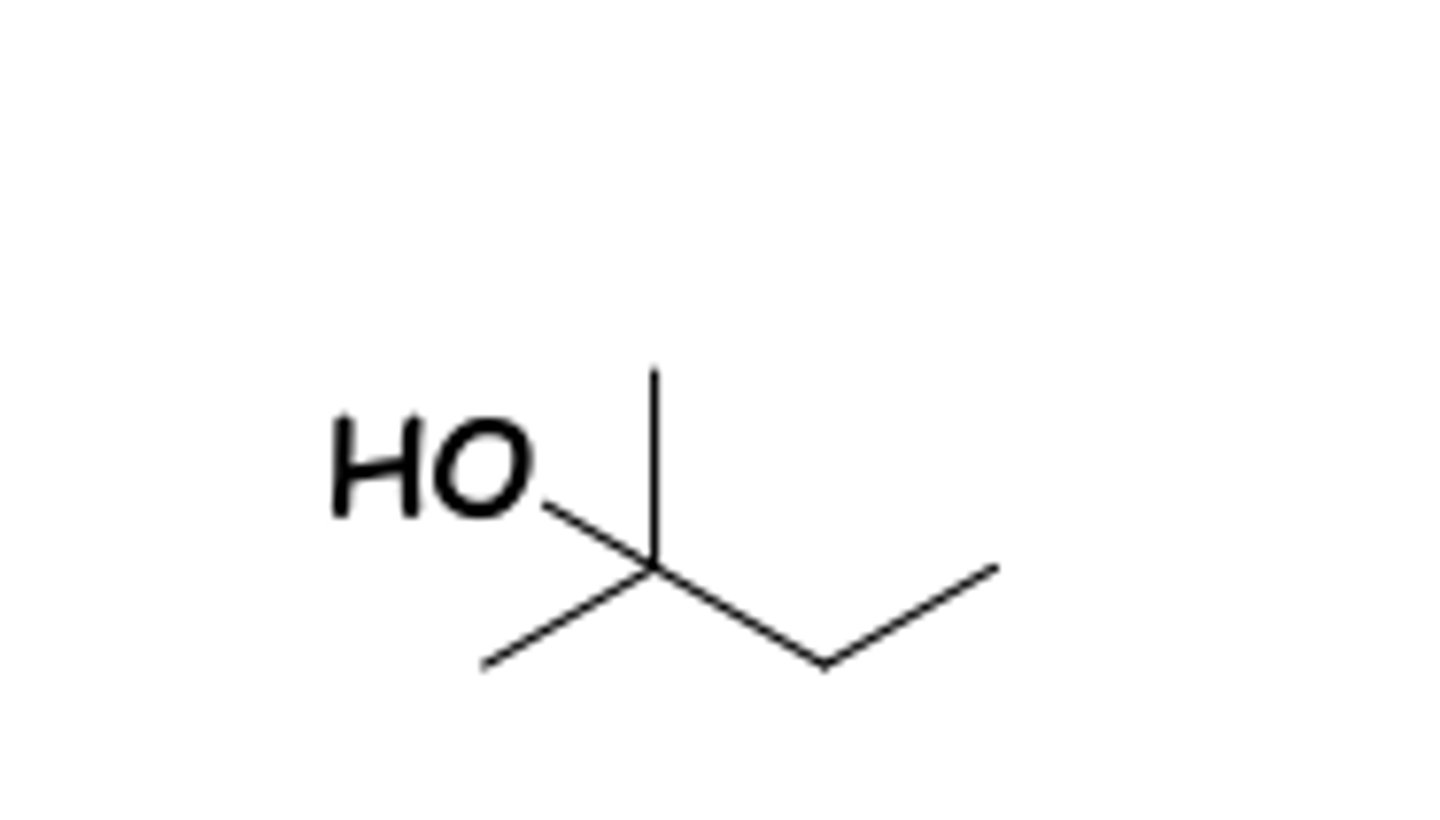
what is the structure of t-amyl alcohol
which of the following will you use for solubility test
NaOH and NaHCO3
in Lucas test what will give immediate positive results
t-amyl alcohol
which of the following is correct of aldol condensation bond formation
carbon-carbon
which of the following is the correct statement about aldol reaction
exothermic
which of the following method will you utilize to analyze the final product
melting point
Aldol products can be formed by _____ conditions
acidic and basic
which test can be used to identify a carbonyl compound
tollens test
which will give a positive result in schiffs test
aldehydes
chromic acid test is negative for
tertiary alcohols
which of the following compounds will give a positive result in iodoform test
acetophenone
which of the following tests can be used to distinguish aldehydes and ketones together from other carbonyl compounds
2,4-dinitrophenylhydrazine
which is considered a primary amine
2-aminobutane
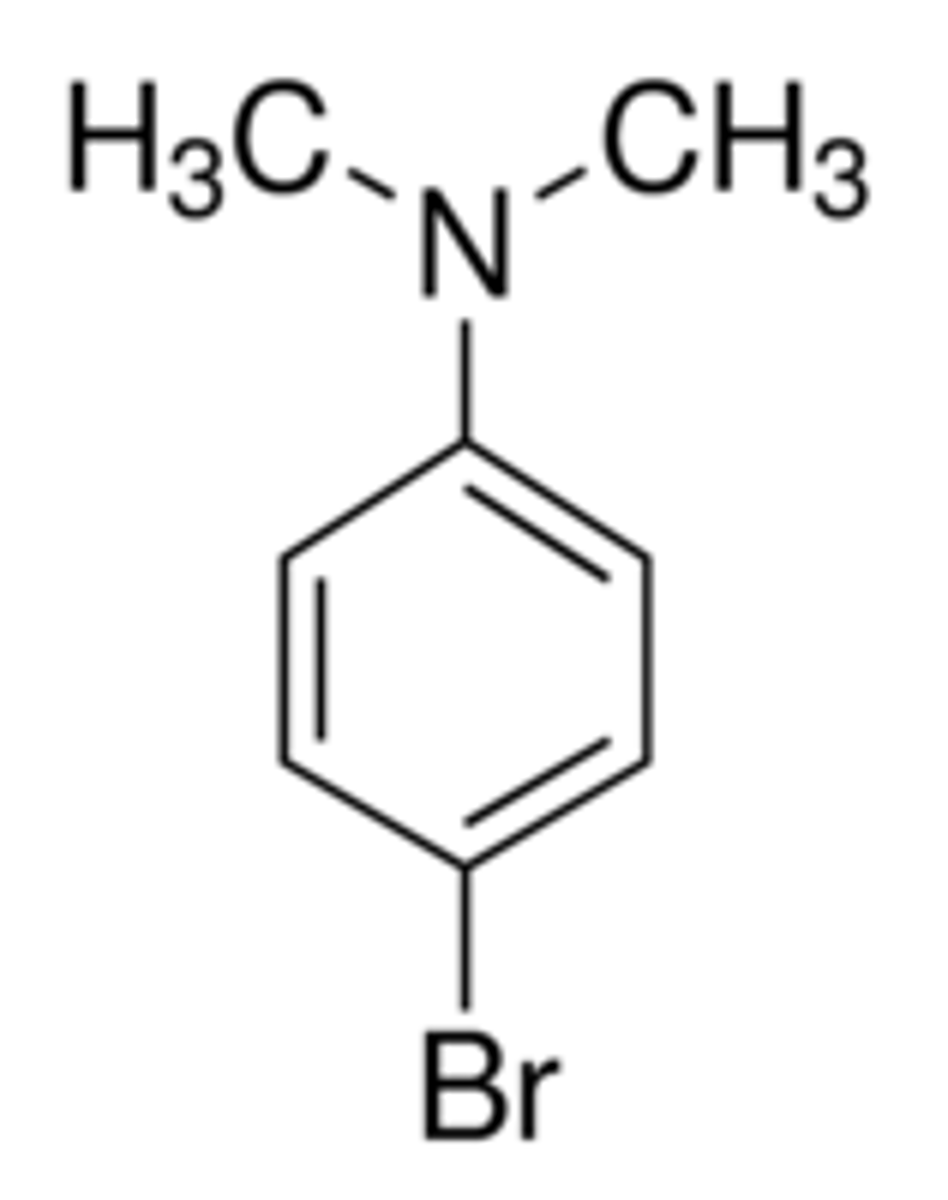
structure of N,N dimethylaniline
which of the following solvents was used in solubility amine
10% NaOH
what are main reagents for hinsberg test
benzenesulfonyl chloride
what would be the correct prediction if organic phase dissolves in 10% HCl in Heinsberg test
primary amines
what would be the correct prediction if organic phase does not dissolve in 10%HCl Heinsberg test
secondary amines
which is the best for separating crystals from solvents
vacuum filtration
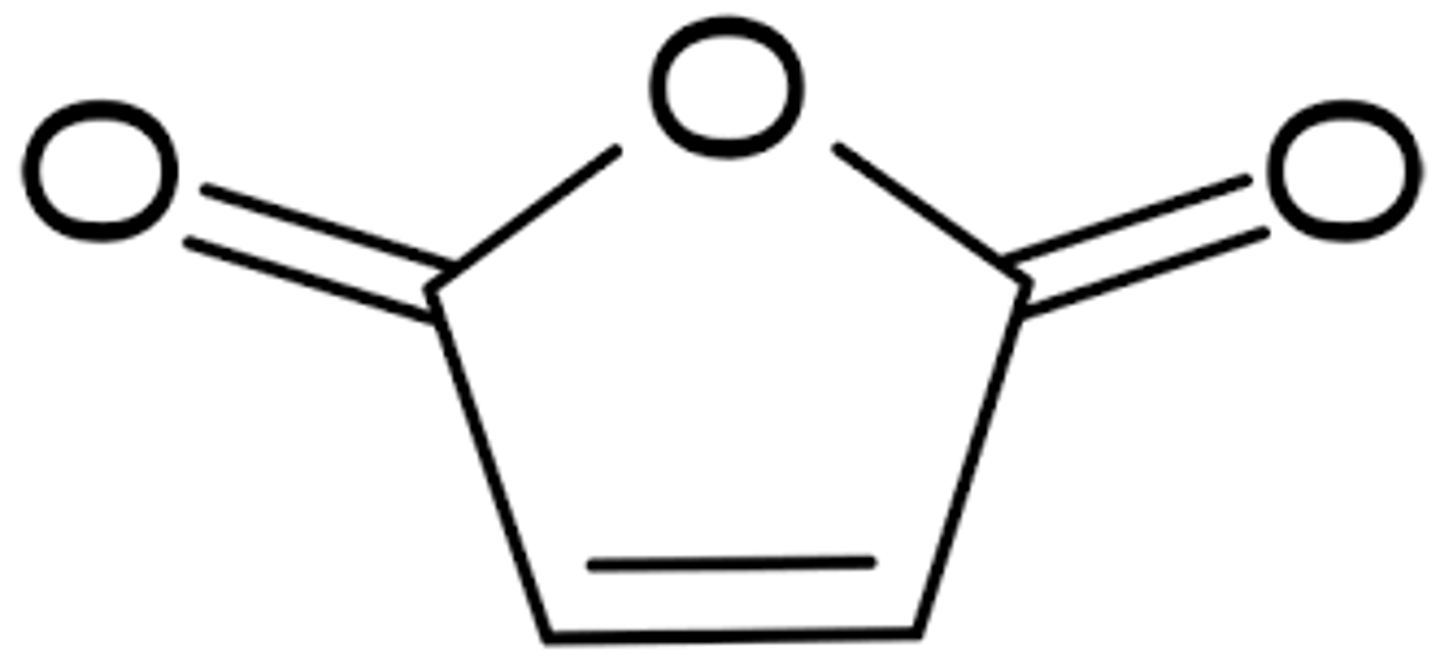
maleic acid
which of the following statements is true about nitration of methyl benzoate
product is o-nitro methyl benzoate and products Rf < starting Rf
which of the following solvent condition utilized for TLC during nitration experiment
Hexane-ethyl acetate
which of the following objectives for recrystallization
analysis, purification, confirmation
what is the role of sulfuric acid in nitration of methyl benzoate
quenching the m-nitro methyl benzoate
the overall reduction of carbonyl group to a hydroxyl group involves which of the following
addition of 2 H atoms
what is the purpose of using CaCl2 in 'epoxide' experiment
drying agent
aqueous solution of NaCl is known as
brine
reduction of benzil to hydrobenzoin reagent
NaBH4
which of the following is true about Diels-alder
cycloaddition
what is used in Diels- Alder reaction
maleic anhydride
what is true abut Diets-alder product
6 member ring
what is the name of nitration reaction
electrophilic aromatic substitution
which of the following is the limiting reagent in nitration
methyl benzoate
what is true about concentrated acids
strong oxidizing agent and corrosive
which of the following are the intermediate for the epoxide synthesis
halohydrin formation
direct synthesis of epoxide contain which of the following
SN2, halohydrin, mCPBA
which of the following will you utilize to synthesis of epoxide from carvone
H2O2, NaOH, methanol