Organic Chemistry Ch 13
1/86
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
87 Terms
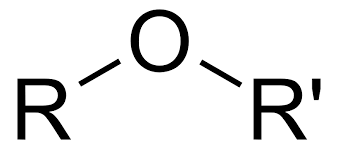
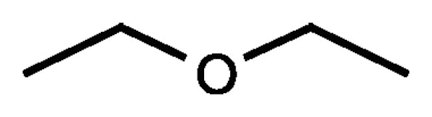
ethers
ethers common name
name each r group invidually, put alphabetically, add ‘ether’ at end
ethers IUPAC name
large r group is parent group ending with ‘ane’, other r group named with -oxy at end
important ether properties
low boiling point (no hydrogen bonding within ether molecule)
fairly unreactive
oxygen is sp3 hybridized
naming crown ethers
number total atoms - crown - number oxygen atoms
Li crown ether
12-crown-4
Na crown ether
15-crown-5
K crown ether
18-crown-6
polyethers
antibiotics that behave similarly to crown ethers by holding metal cations, AKA ionophores
crown ethers used for substitution rxn what it looks like
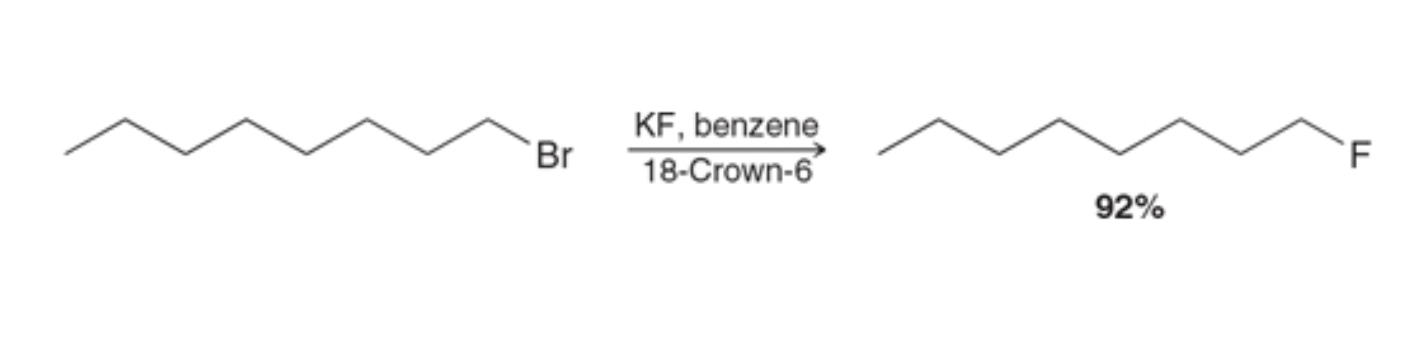
crown ethers substitution reaction reagents
XF, benzene & crown ether that correlates to ‘X’
3 methods of preparing ethers
acid-catalyzed dehydration of ethanol
williamson ether reaction
alkoxymercuration-demercuration
acid catalyzed dehydration of ethanol what it looks like
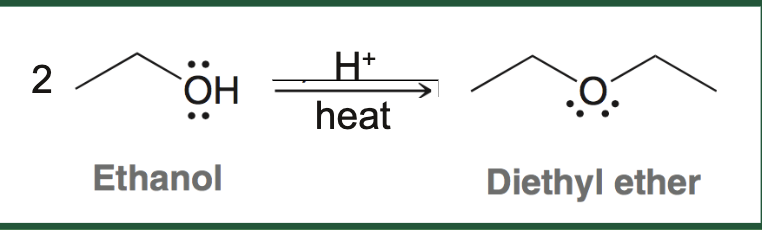
acid catalyzed dehydration of ethanol reagents
hydrogen, more ethanol (starting material), heat
acid catalyzed dehydration of ethanol product
diethyl ether, other symmetric/simple ethers
williamson ether reaction what it looks like
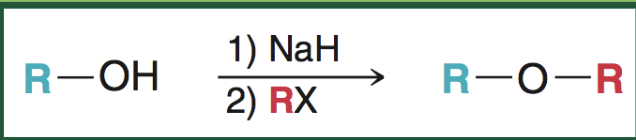
williamson ether reaction reagents
1) NaH
2) RX
williamson ether reaction starting material & ending product
start with R-OH
end with asymmetric ether R-O-R’
when does williamson ether reaction occur
with primary unhindered alkyl halides as R-X
if pathway needs R-X to be secondary or tertiary, a different pathway will be used
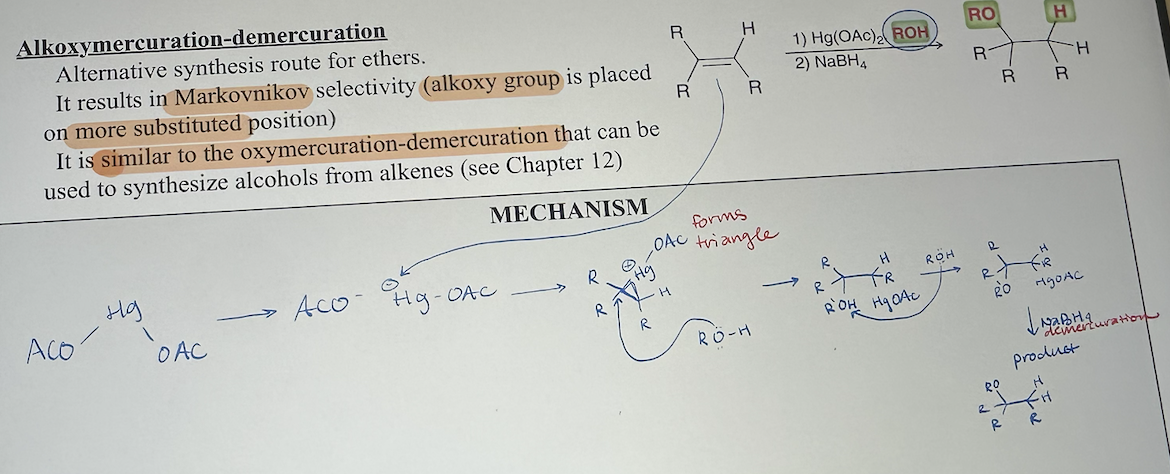
alkoxymercuration-demercuration what it looks like
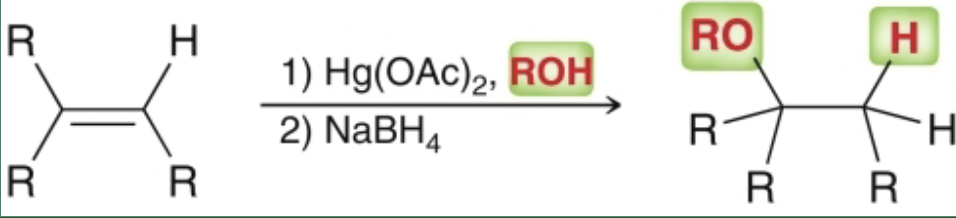
alkoxymercuration-demercuration reagents
1) Hg(OAc)2, ROH
2) NaBH4
alkoxymercuration-demercuration starting material & product
start with alkene
end with markovnikov addition of RO & H, alkoxy group goes on more substituted carbon
2 reactions of ethers
acid cleavage
autooxidation
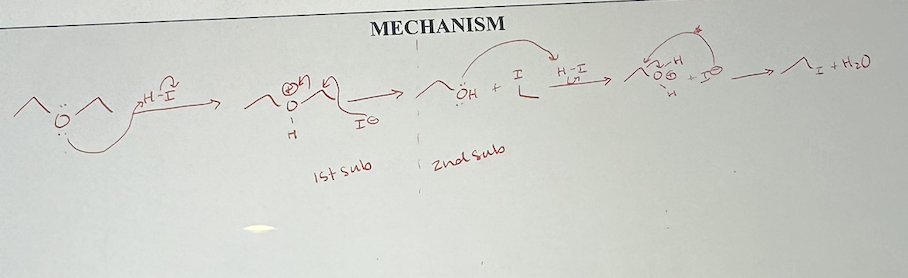
acid cleavage of ethers what it looks like
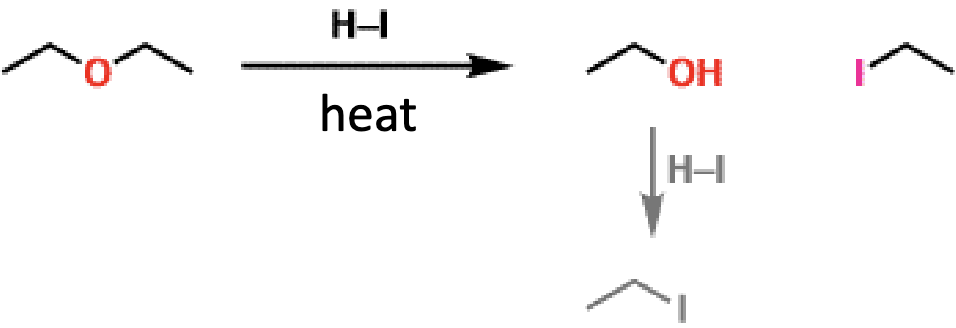
acid cleavage of ethers reagents
HBr or HI
acid cleavage of ethers how many substitutions
two (end with 2 alkyl halides) unless R group is aryl or vinyl, then only one (end with alcohol and alkyl halide)
autooxidation of ethers what it looks like
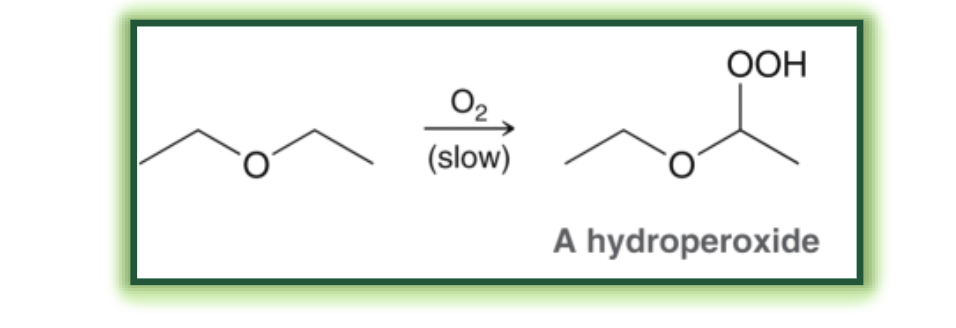
autooxidation of ethers reagents
O2
starting materials and products of autooxidation of ethers
start with ethers, end with hydroperoxides (explosive)
radical mechanism of autooxidation of ethers

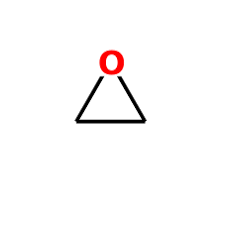
oxirane
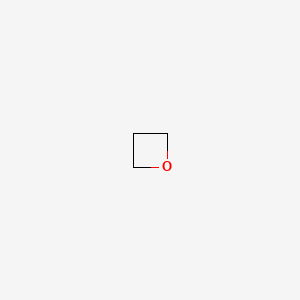
oxetane
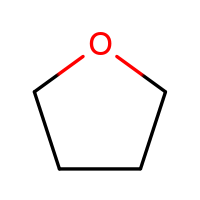
oxolane
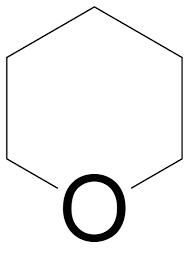
oxane
epoxides’ number of r groups
up to 4; more r groups is more reactive (more ring strain)
one way to name epoxides (O as side group)
treat oxygen as a side group
end with -epoxy after parent chain (oxane, etc)
put C numbers of 2 carbons connected to oxygen before parent chain name
one way to name epoxides (epoxide as parent chain)
oxirane, etc used as parent chain
oxygen is numbered first, number to get side groups at lowest number
name side groups, put before parent chain alphabetically as usual
3 ways to prepare epoxides
using peroxy acid
using halohydrins
sharpless epoxidation
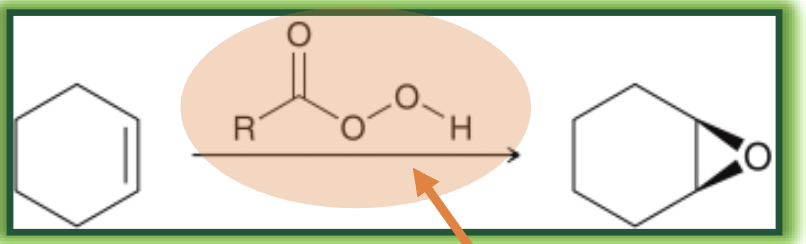
epoxidation with peroxy acid what it looks like
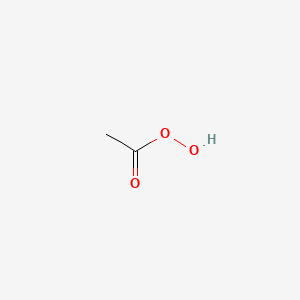
peroxyacetic acid structure
epoxidation with peroxy acid reagents
mCPBA or peroxyacetic acid
epoxidation with peroxy acid starting material and product
start with alkene
end with epoxide
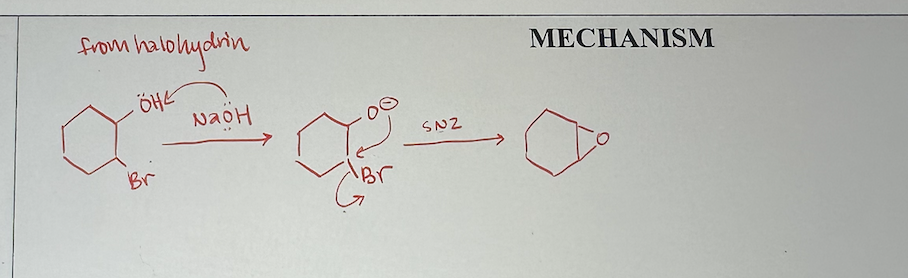
epoxidation with halohydrins what it looks like
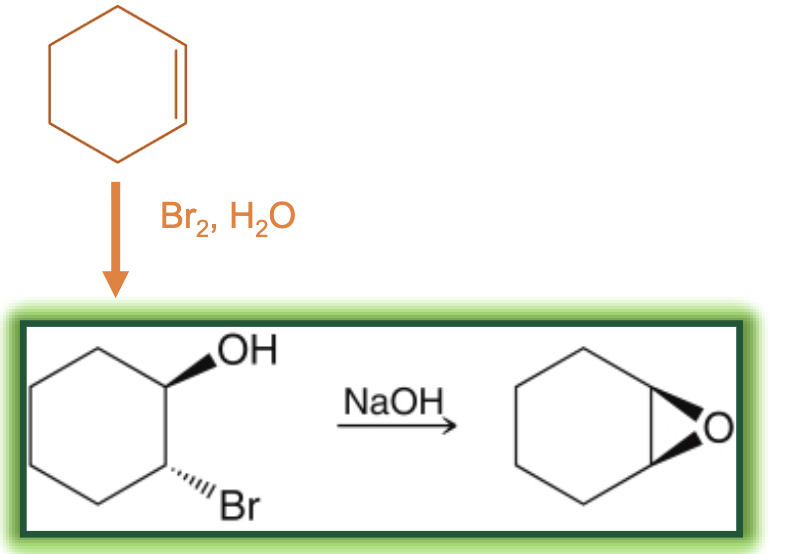
epoxidation with halohydrins reagents
1) Br2, H2O (alkene to halohydrin)
2) any strong base, like NaOH (halohydrin to epoxide)
epoxidation with halohydrins starting material and product
start with alkene, then create halohydrin (has OH and X attached), then product is epoxide
stereoselectivity of epoxidation using peroxy acid and halohydrins
forms a racemic mixture, is not enantioselective
sharpless epoxidation what it looks like
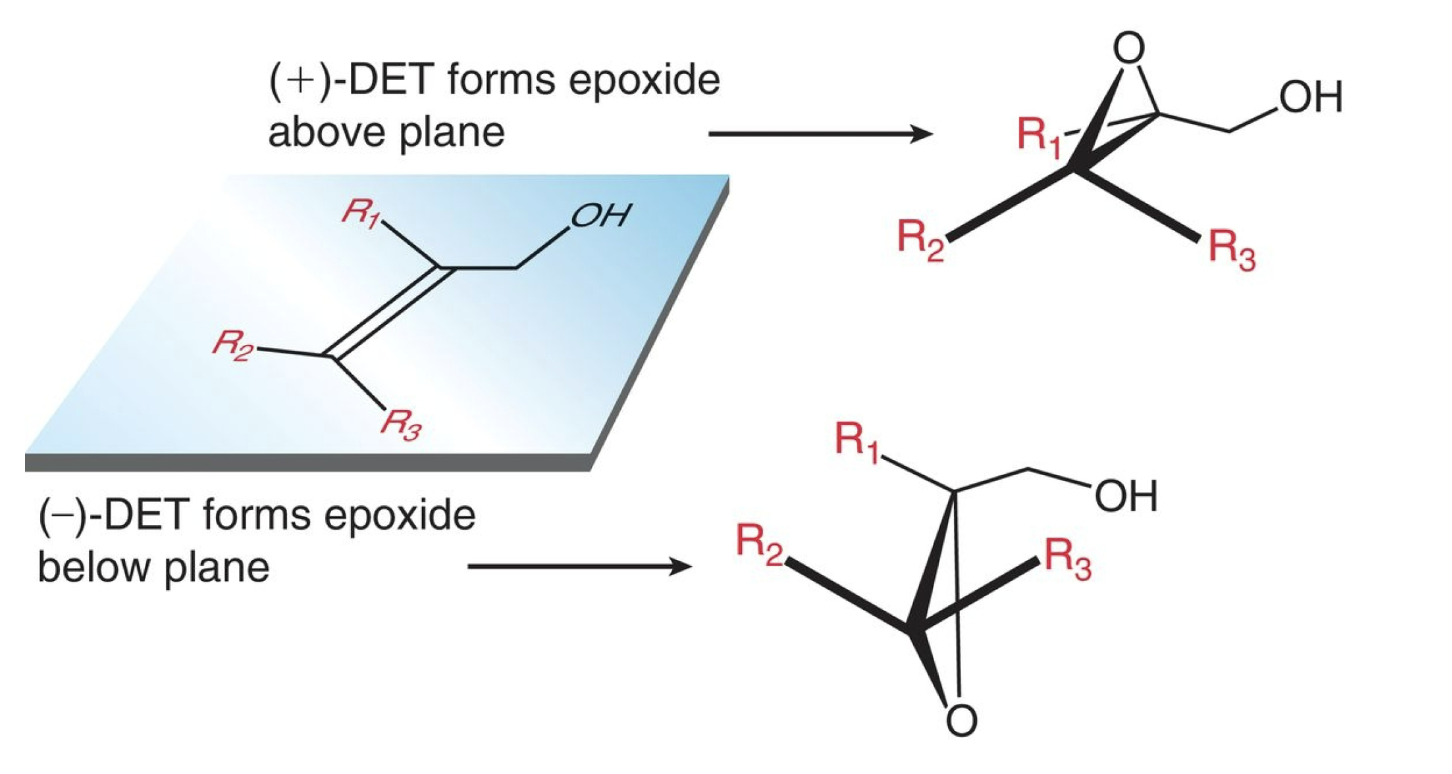
sharpless epoxidation reagents
(CH3)3COOH, Ti[OCH(CH3)2]4 & (+)-DET OR (-)-DET
enantioselectivity of sharpless epoxidation
(+)-DET used - epoxide oxygen/triangle will form above plane
(-)-DET used - epoxide oxygen/triangle will form below plane
sharpless epoxidation starting material and product
start with alkene
end with epoxide, above or below plan depending on whether (+/-)-DET is used
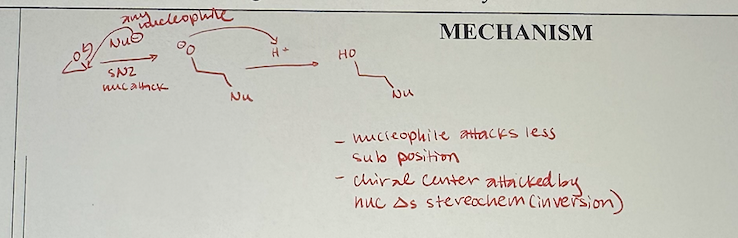
epoxide reactions with strong nucleophiles what they look like
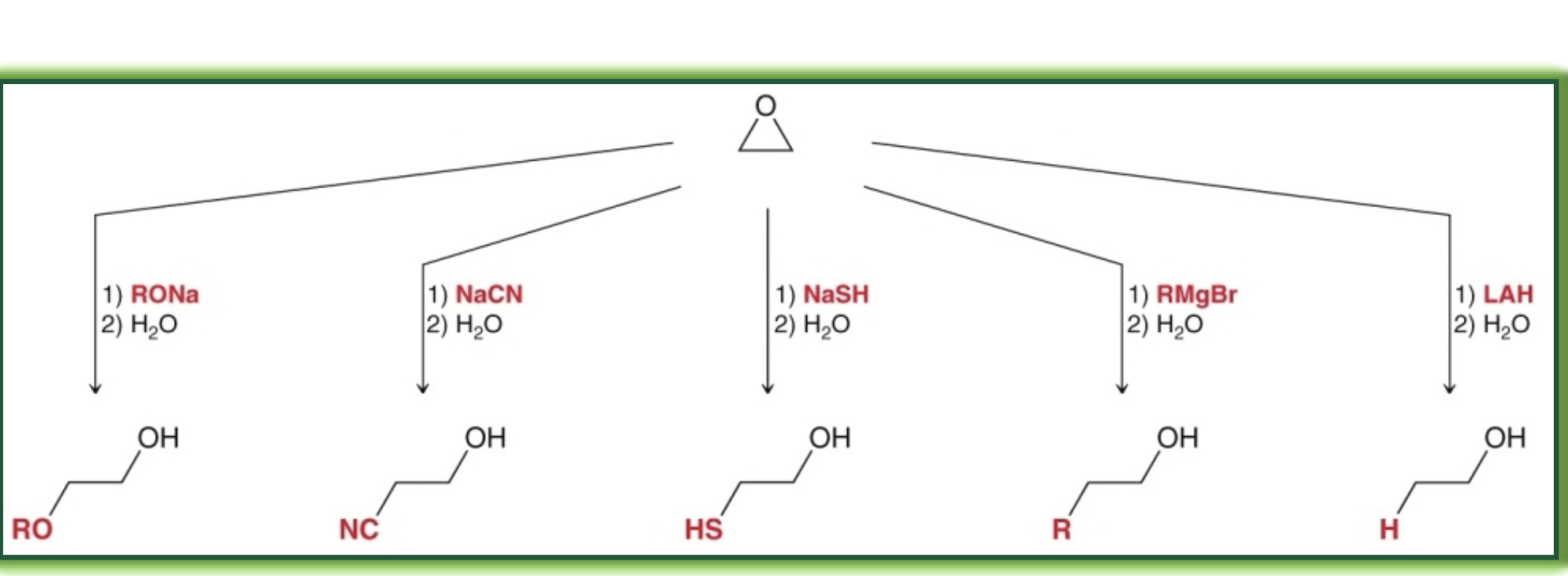
epoxide reactions with strong nucleophiles reagents
1) strong nucleophile
2) H2O
strong nucleophiles that often react with epoxides
RONa
NaCN
NaSH
RMgBr
LAH
stereochemistry of epoxide reactions with strong nucleophiles
basically SN2, nucleophile attacks less substituted carbon, stereochemistry inverts on attacked carbon
epoxide reactions with weak nucleophiles what it looks like
if primary/secondary Cs only
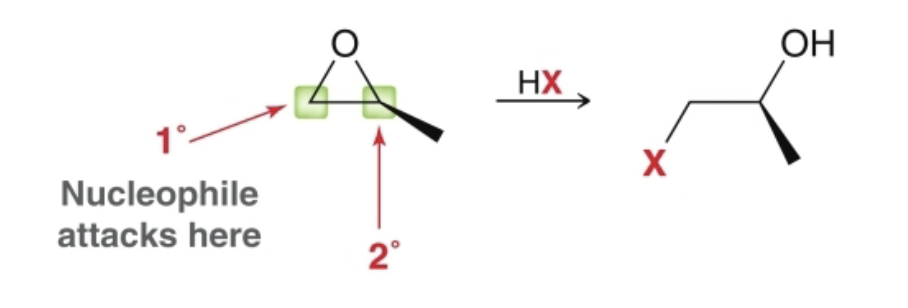
epoxide reactions with weak nucleophiles reagents
weak nucleophile
common weak nucleophiles reacting with epoxides
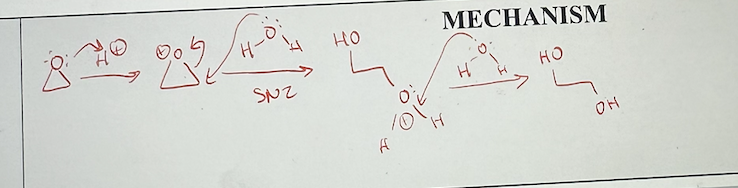
H2O, alcohols, HX
weak nucleophiles reacting with epoxides neutral nucleophiles (H2O, alcohols)
has an extra proton transfer
regioselectivity of weak nucleophiles reacting with epoxides
if primary or secondary carbon are only ones present, the nucleophile attacks the less substituted carbon - steric effect
I tertiary carbon is present, the nucleophile attacks there - electronic effect
less substituted carbon’s stereochemistry will invert, regardless of which carbon is attacked
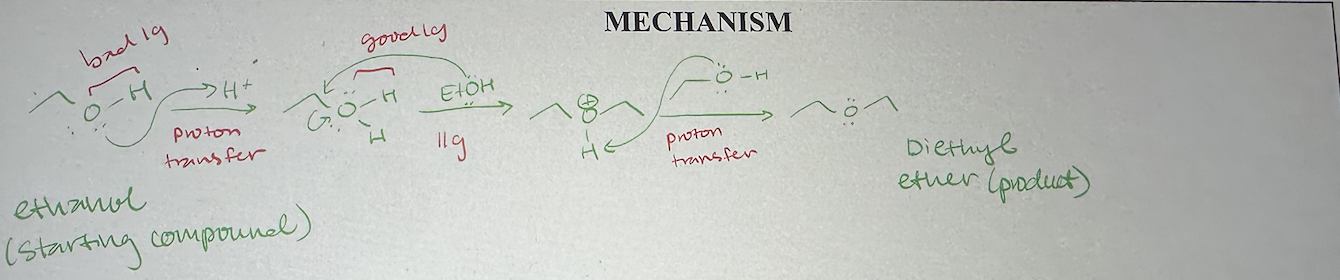
acid-catalyzed dehydration of ethanol mechanism
Williamson ether synthesis mechanism

Alkoxymercuration-demercuration mechanism
Acid cleavage of ethers mechanism
preparing epoxide with peroxy acid mechanism
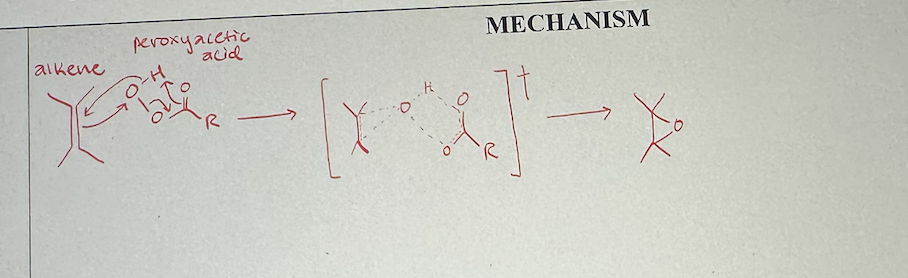
preparing epoxide from halohydrin mechanism
ether reactions with strong nucleophiles mechanism
ether reactions with weak nucleophiles mechanism
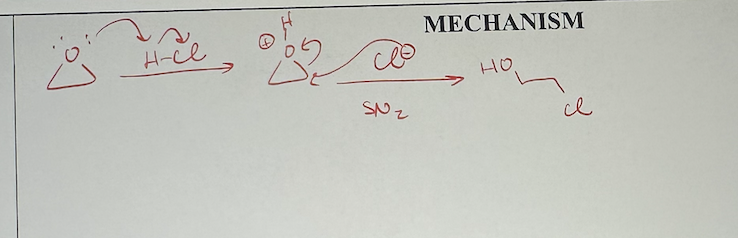
ether reactions with neutral nucleophiles (water, alcohol) mechanism
thiol
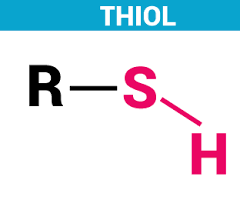
naming thiols
if -SH group, end name in -thiol
if -SH and -OH groups, end with -ol; -SH is mercaptan group, so name substituent mercapto-
reaction to form thiols
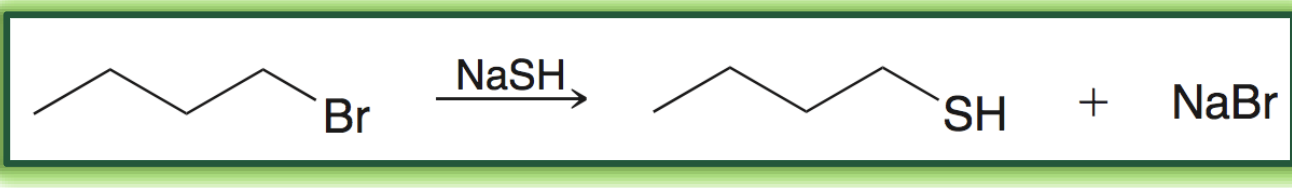
formation of thiols stereochemistry
SN2, inversion of stereochemistry, NaSH will form primary or secondary thiol
oxidation of thiol mechanism
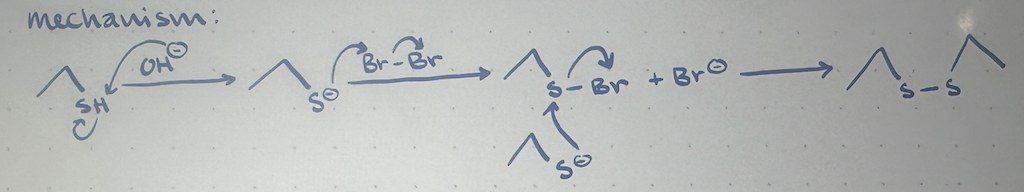
reagents in oxidation of thiols
H2O, Br2
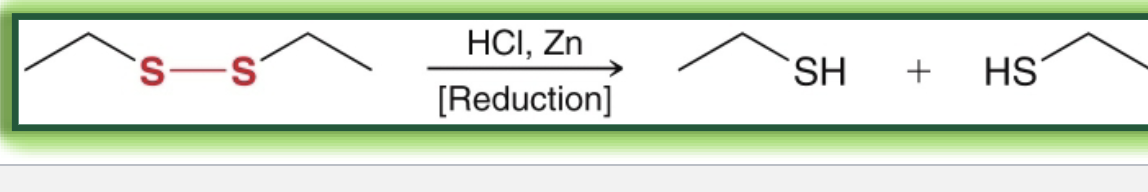
reagents in reduction of disulfide (back to thiol)
HCl, Zn
what reduction of disulfide looks like
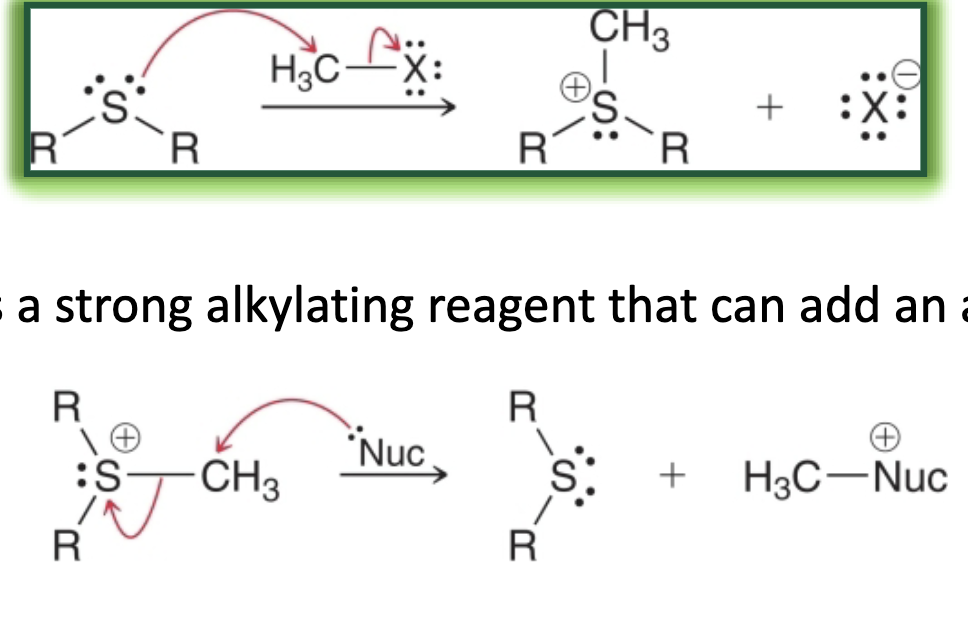
sulfides
sulfur analogs of ethers
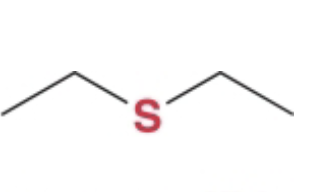
naming sulfides
same as for ethers, can be named as a substituent as a -thio group
preparation of sulfides what it looks like
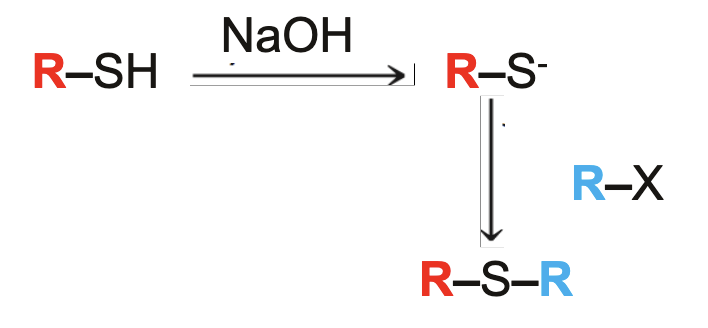
preparation of sulfides reagents
NaOH, R2-X
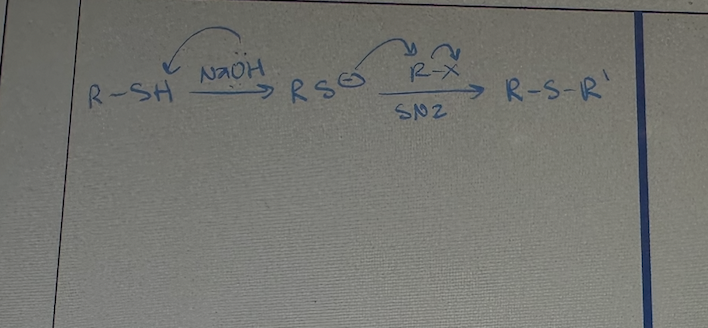
preparation of sulfides mechanism
sulfoxide
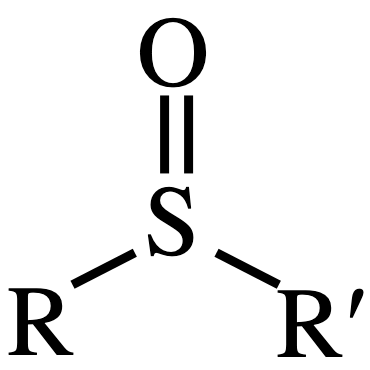
sulfone
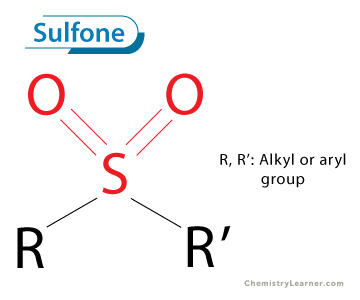
reagents for sulfide - sulfoxide
NaIO4
reagents for sulfide - sulfone
2 H2O2
Role of sulfides as reducing agents
Used in ozonolysis (DMS or DMSO alongside O3)
SN2 Substitution with Alkyl Halide Mechanism