iPSCs and Cancer Cells
1/72
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
73 Terms
What are ESCs
embryonic stem cells
What are iPSCs
induced pluripotent stem cells
What are the 3 approaches to nuclear reprogramming to pluripotency
somatic nuclear transfer or nuclear transfer, cell fusion, or transcription factor transduction
What is somatic nuclear transfer or nuclear transfer
The nucleus of a somatic cell (which is diploid, 2n) is transplanted into an enucleated oocyte
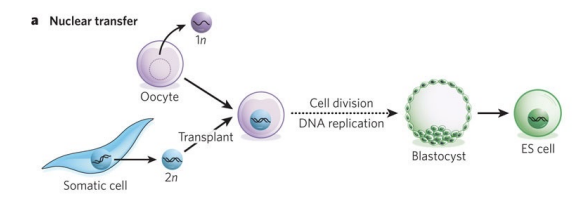
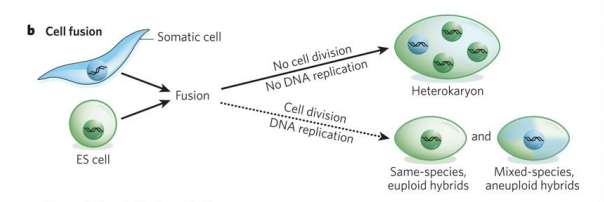
What’s cell fusion
2 distinct cell types are combined to form a single entity
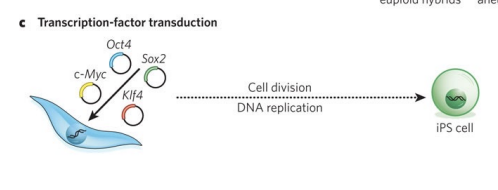
What’s transcription-factor transduction
This approach can be used to form induced pluripotent stem (iPS) cells, which have similar properties to ES cells and can be generated from almost any cell type in the body through the introduction of four genes (Oct4, Sox2, Klf4 and c-Myc) by using retroviruses
Blastula versus blastocyst
Blastocyst is a specific type of blastula found in only mammals. Blastula is a hollow, single-layered sphere of undifferentiated cells
Whats compaction in early mammalian cleavage
tight, adherent junctions form between blastomeres
What occurs during compaction and why is it important
blastomeres of preimplantation embryos begin to flatten and increase cell–cell contacts with their neighboring cells. Thus, the embryo reduces its surface area. This flattened morphology is essential for later morphogenetic events and lineage specification
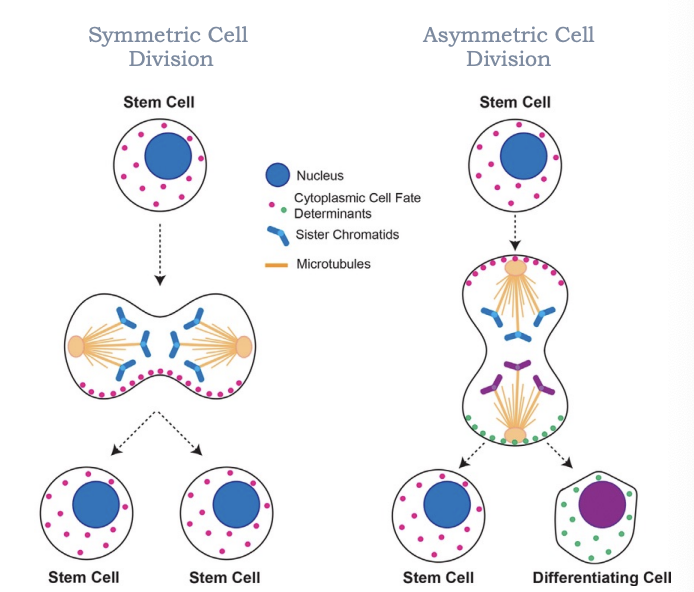
Whats polarization
the asymmetric organization of cellular components leading to cell asymmetry
Are polar cells symmetric
No
are apolar cells symmetric
yes
How does germ cell establishment in C. elegans occur
by inheritance of germ plasma and asymmetric divisions
What do symmetric and asymmetric cell division generate
cell diversity
What is blastocyst established by
essential transcription factors
What is trophectoderm (TE)
forms outer layer of the embryo
How do inner cell mass (ICM) cells interact
they stay closely associated to each other
What is OCT4 used for
(1) expressed in the ICM and (2) required for establishing the ICM in vivo
What does CDX2 do
(1) expressed in the TE and (2) required for implantation
What does NANOG do
promotes rapid cell division and entry into the S phase, ensuring ICM cells stay undifferentiated (pluripotent)
What occurs in the absence of Cdx2
Oct4 is ectopically expressed in the TE

What occurs between Oct4 and Cdx2 and what does it do
mutual antagonism establishes the blastocyst
What is gastrulation
early embryonic development phase where the single-layered blastula reorganizes into a three-layered structure called the gastrula.
Order of development
Zygote into blastula into gastrula into difference cells
What cells do gastrula give rise to
ectoderm, mesoderm, endoderm, and germ cells
What does ectoderm give rise to
outer surface (epidermal cells), central nervous system (neurons in brain), neural crest (pigment cell)
What does mesoderm give rise to
Dorsal (notochord), paraxial (bone tissue), intermediate (tubule cell of kidney), lateral (red blood cells), head (facial muscle)
What does endoderm give rise to
digestive tube (pancreatic cell), pharynx (thyroid cell), respiratory tube (lung cell)
What do germ cells give rise to
sperm and egg
What iwas leg on the head and what’s the lesson
ectopic expression of the Meis1 gene triggers antennal development. It shows how a single gene expression can change the cell’s “destination”
What was known before yamanaka’s experiments
(1) Several transcription factors that were highly expressed in ESCs had
been studied by knockout (KO) experiments in mice and other model
organisms (Oct4, Sox2, Nanog, etc)
(2) Nuclear reprogramming was possible using the whole somatic nucleus
in frogs and mammals.
(3) Re-expression of a single transcription factor in one cell type leads to
trans-differentiation into another cell type (ex: drosophila)
Whats drosophila
legs growing where antennas were supposed to grow
What did yamanaka’s experiments try to answer
What are the specific factors driving somatic cell reprogramming
What tools were used by Yamanaka
Fbx15 mouse gene is expressed in stem cells and then insertion of neomycin (neo) resistance gene into Fbx15. Neomycin is not expressed in mammalian cells. The neo-fbx15 mutant mouse was viable and used to harvest fibroblasts (somatic cells). Cloning a cDna library of 24 genes expressed by ESCs into retroviral vectors. Then the modified retroviruses were used to transduce the fibroblast cells
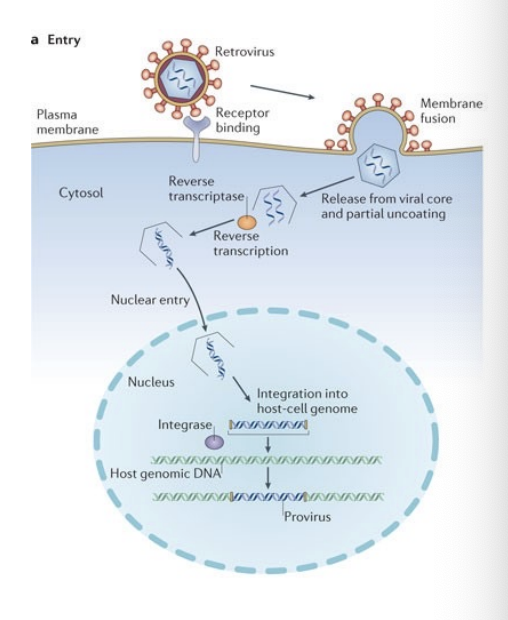
How did Yamanaka test candidate factors
Generation of iPS cells from mouse embryonic fibroblasts (MEF) cultures
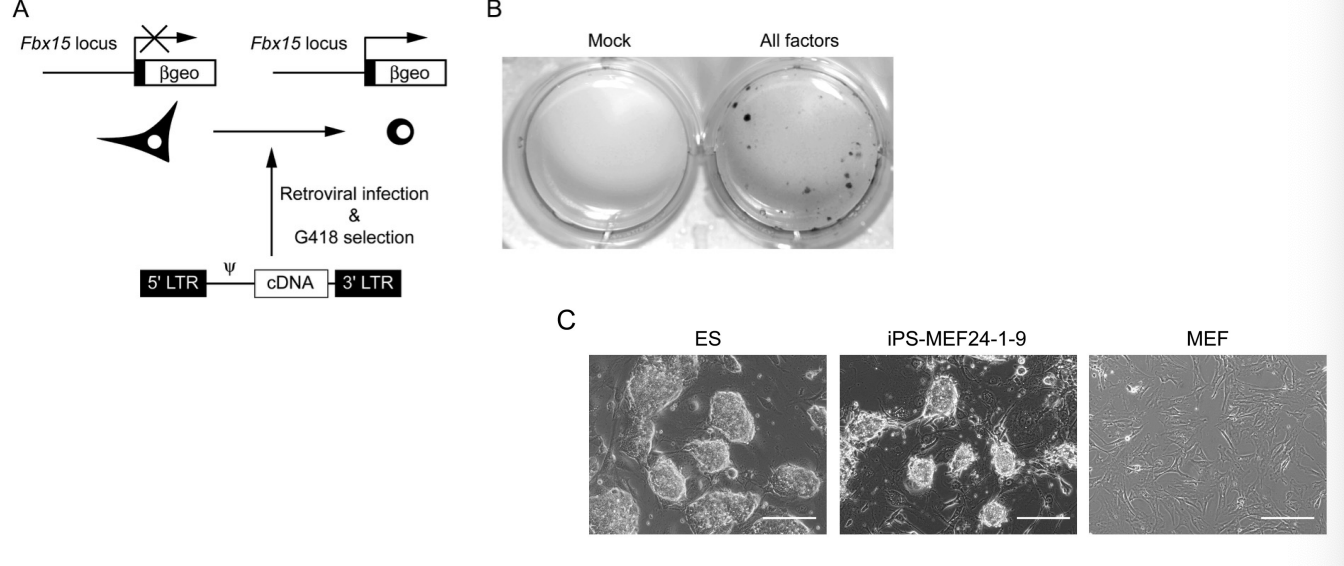
The process of how Takahashi and Yamanaka’s experiment discovered four core nuclear reprogramming factors
Take fibroblasts from Neo-Fbx15 mice (resistant to neomycin) then used antibiotic selection (only cells expressing NeoFbx15 would survive). Only ESCs express Fbx15, while Fibroblasts do not express Fbx15. So, cells expressing Fbx15 should be ESCs. They started with adding 24 viruses encoding DNA from 24 known transcription factors found in ESCs. Fibroblasts differentiated into ESC-like cells that are resistant to neomycin. Started a process of elimination for key factors by taking 1 factor out at a time. Narrow it down to 10 factors, then 4. Found four key factors that drive nuclear reprogramming
What are the four key factors that drive nuclear reprogramming
OKSM: OCT4, SOX2, Klf4, and C-Myc
What do Oct4 and Sox2 do
essential for generating mature pluripotent stem cells
What do Klf4 and c-Myc do
increase efficiency and accelerate iPS generation
What are key programming events during direct reprogramming
(1) The somatic program needs to be shut down
(2) cell proliferation programs need to be activated
(3) endogenous pluripotency programs need to be activated
(4) exogenous OKSM factors needs to be shut down for maturation
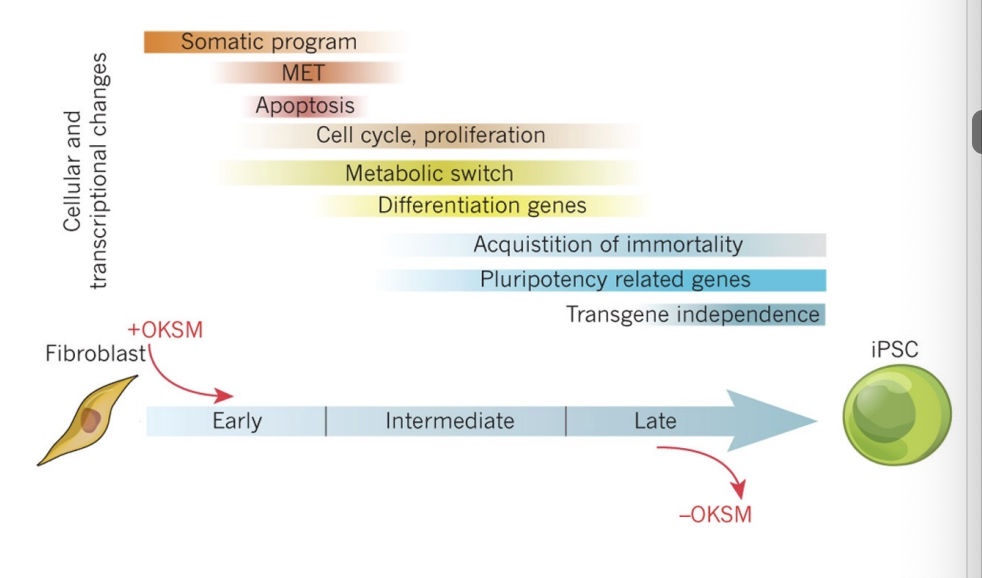
How is activation and repression of different gene programs achieved
in part by changes in epigenetic programs
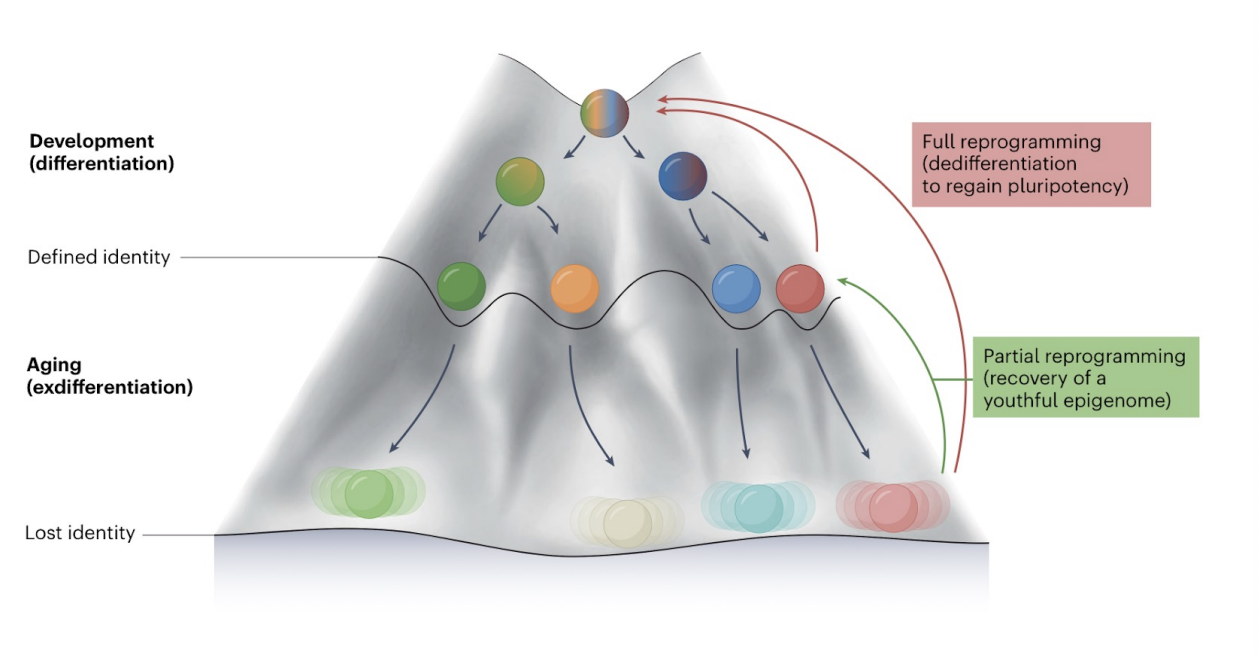
Waddington’s epigenetic landscape for reprogramming
metaphor depicting cell differentiation as a ball rolling down a rugged landscape into valleys (stable states). In reprogramming, this model is reversed, forcing differentiated cells (in the valley) to move uphill back to a pluripotent state
What is cancer
a group of diseases characterized by the uncontrolled growth and division of cells that can invade surrounding tissues or spread to other parts of the body
Is cancer a single disease
No, it’s many different types of diseases that share the common feature of abnormal cell proliferation
What is the leading risk factor for cancer incidence
Aging
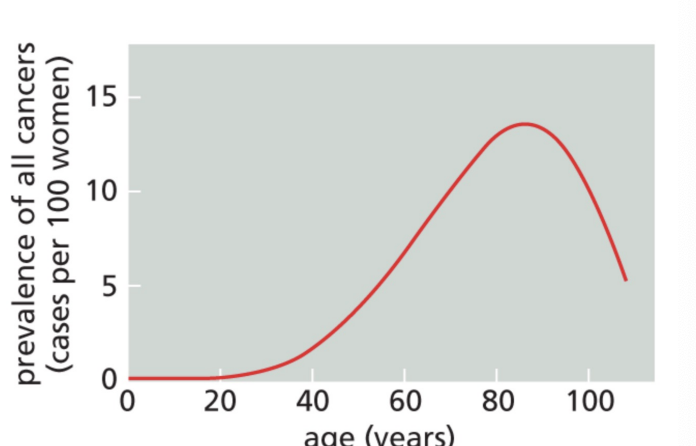
How do cancer cells act
invade surrounding tissues and metastasize to distnace sites
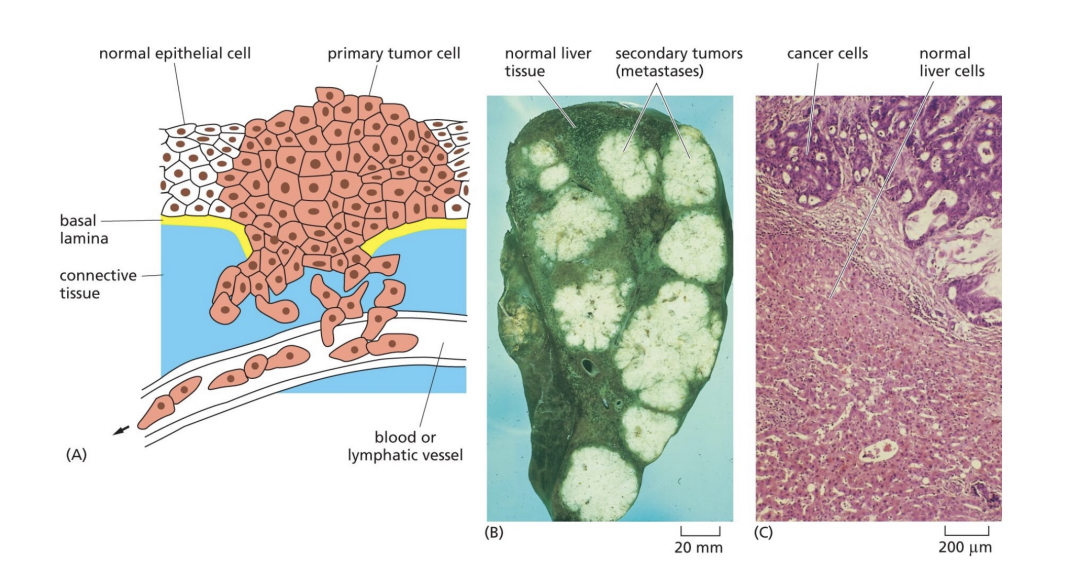
How is genetic instability shown in cancer cells
highly abnormal chromosomes in cancer cells
What are modifiable risk factors for cancer
life style
What are non-modifiable risk factors for cancer
genetic, epigenetic, age, ethnicity, gender
What are two main ways genetic mutations lead to cancer
proto-oncogene (stuck gas pedal, dominant) or tumor suppressor gene (broken brakes, recessive)
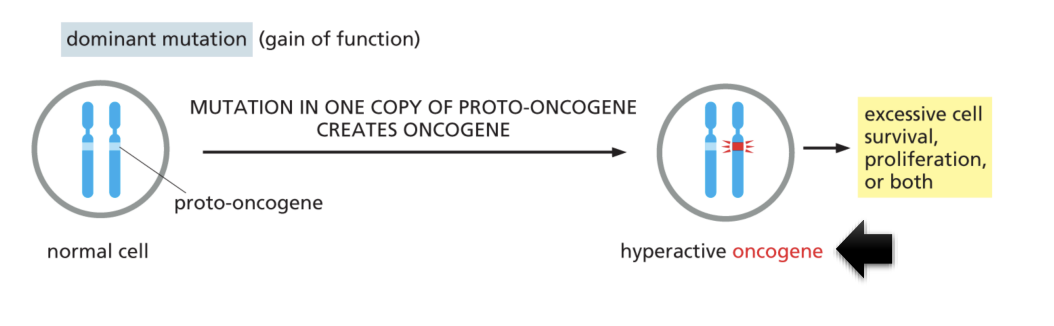
Result of mutation in one copy of proto-oncogene
It creates hyperactive oncogene which leads to excessive cell survival, proliferation, or both
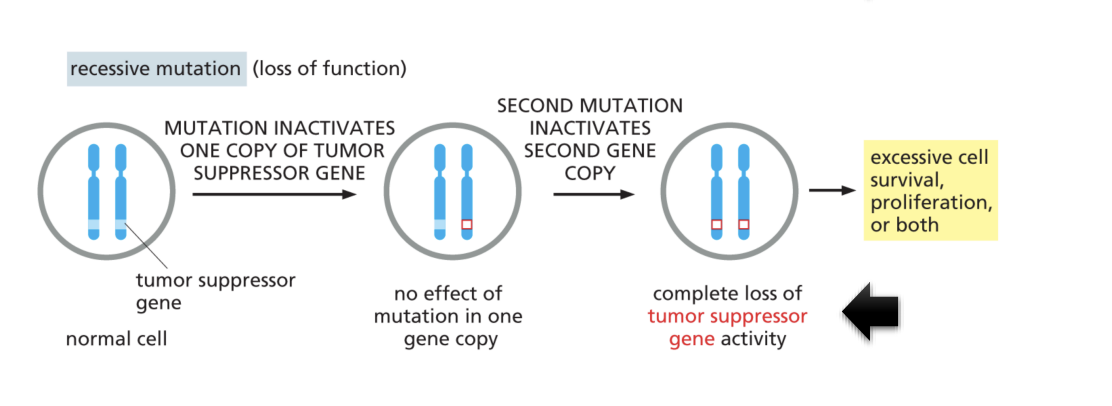
Result in mutation of tumors suppressor gene
It inactives the tumor suppressor but there’s no effect of mutation in one gene copy. So, if a second mutation inactivates the second gene copy, it leads to complete loss of tumor suppressor gene activity which causes excessive cell survival, proliferation, or both
What are proto-oncogenes
normal genes that regulate cell growth and division
What are oncogenes
mutated, overactive version of proto-oncogene that can lead to uncontrolled cell growth and cancer
What converts proto-oncogene to oncogene
gain of function mutations (dominant)
What do 30% of human cancers contain
activating mutations in Ras genes
What does Ras do
In its active state, Ras promotes the activation of a phosphorylation cascade: Ras activates a MAPK signaling module (Mitogen-Activated Protein Kinase) then MAPK phosphorylates various downstream signaling pathways that control gene expression
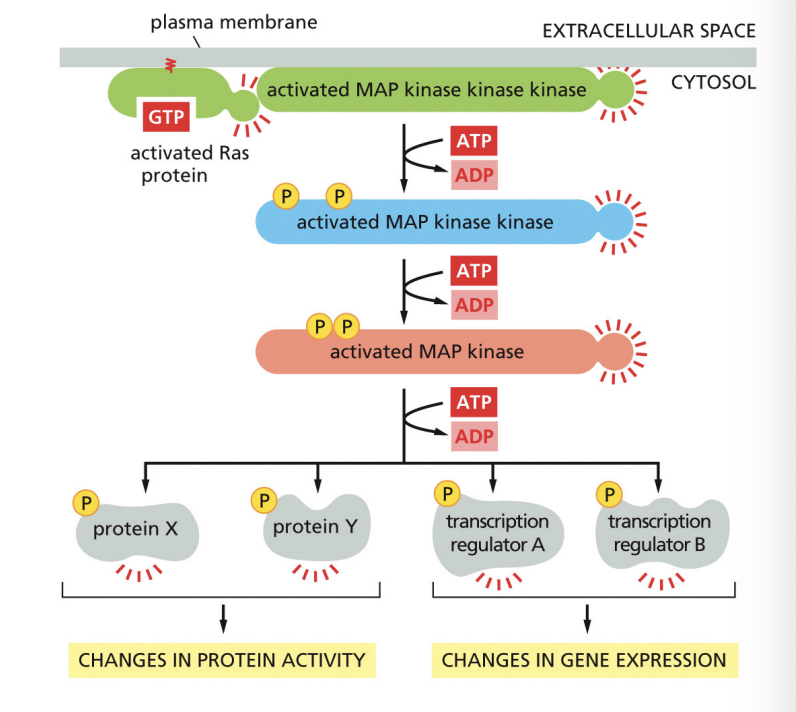
What’s a phosphorylation cascade
a series of serine/threonine protein kinases
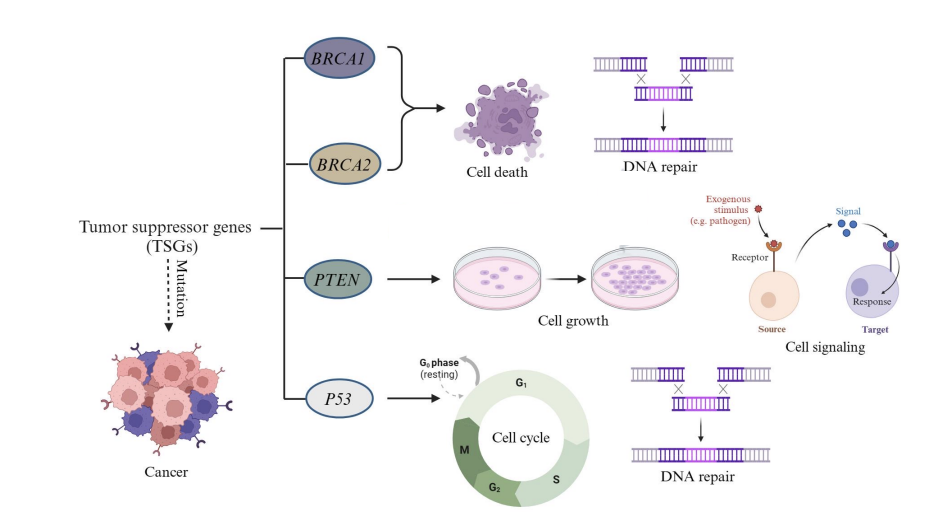
Examples of tumor suppressor genes
BRCA1/2, PTEN, and p53
What are tumor suppressor genes
crucial "brakes" for cell division, slowing or stopping cell growth, repairing DNA, and triggering cell death (apoptosis) to prevent uncontrolled proliferation that leads to cancer
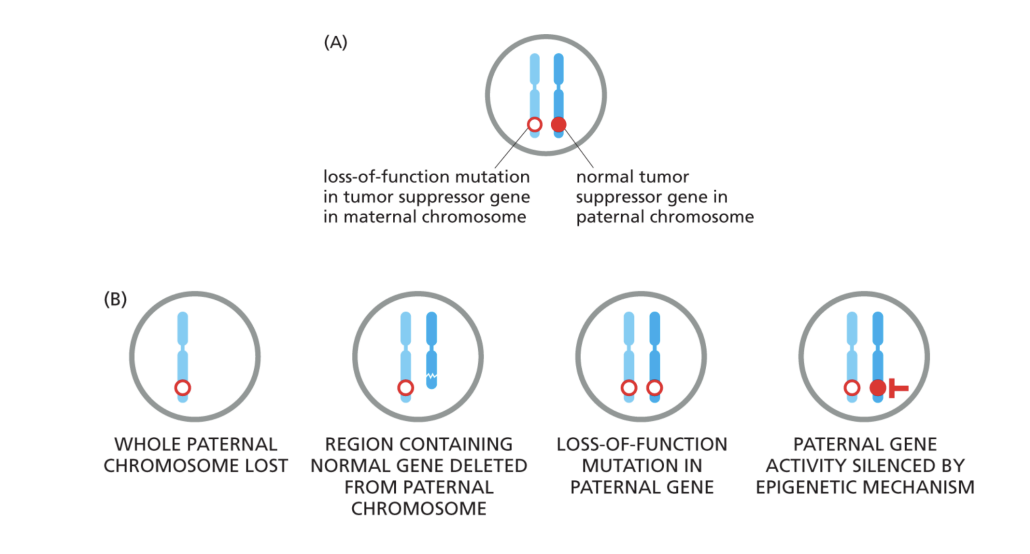
What genetic events can eliminate the activity of a tumor supressor gene
(1) loss of function mutation in tumor suppressor gene in maternal chromosomes but normal tumor suppressor gene in paternal chromosomes (2) whole paternal chromosomes lost and region containing normal gene is deleted from paternal chromosome leading to loss of function mutation in paternal gene which leads its activity to be silenced by epigenetic mechanism
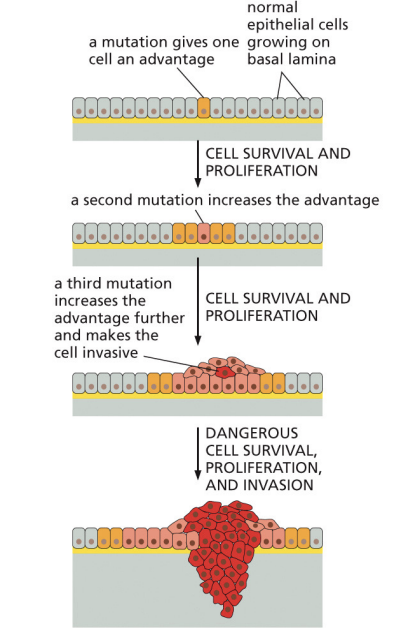
What do malignant tumors contain
cells with multiples mutations in both proto-oncogenes and tumor supressor genes
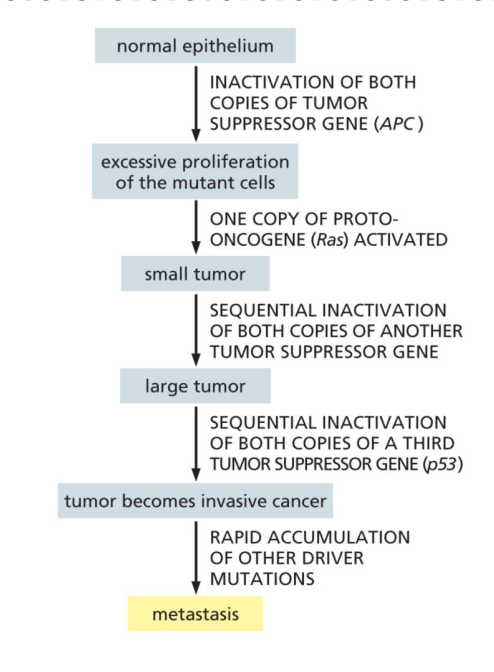
What’s APC
adenomatous polyposis coli, a tumor suppressor gene
In the colon, what happens in mutations inactivate APC
initiate tumors by causing excessive activation of Wnt signaling pathway
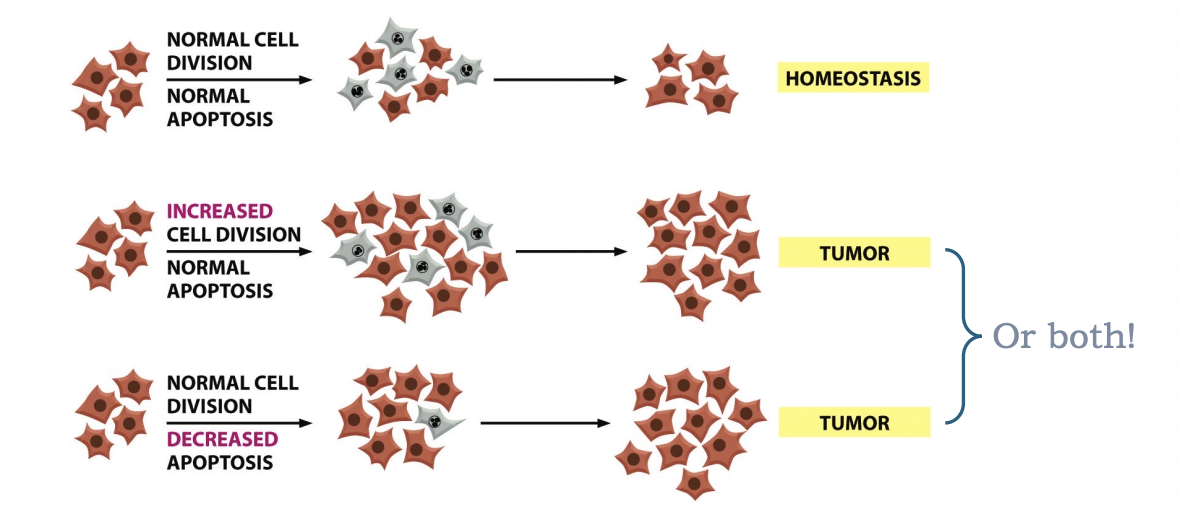
What happens to homeostasis in cancer
normal balance between cell division and apoptosis is disrupted
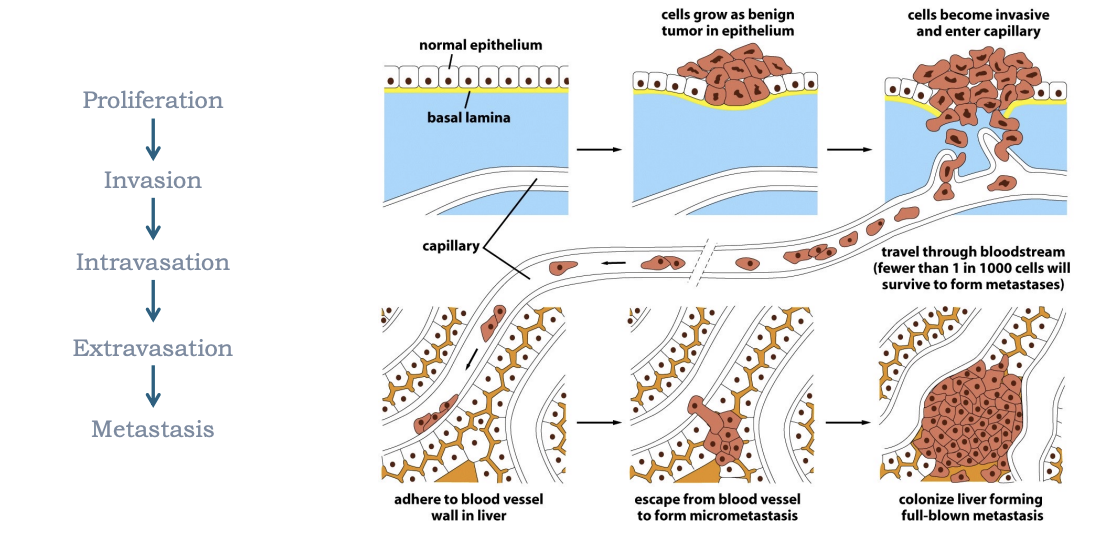
How to cancer cells invade other tissue
Cancer cells attach to the extracellular matrix to invade. Then they use enzymes (metalloproteinases) to break down the ECM. Then they must migrate through the degraded ECM (which may require changes in cell shape and polarization)
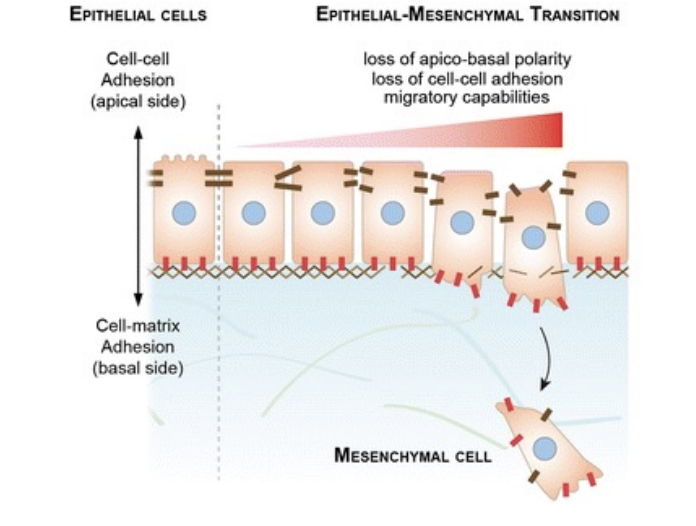
What is epithelial-mesenchymal transition (EMT)
complex, non-linear multi-step process that converts epithelial cells into mesenchymal cells
What does metastasis involve
epithelial-mesenchymal transition
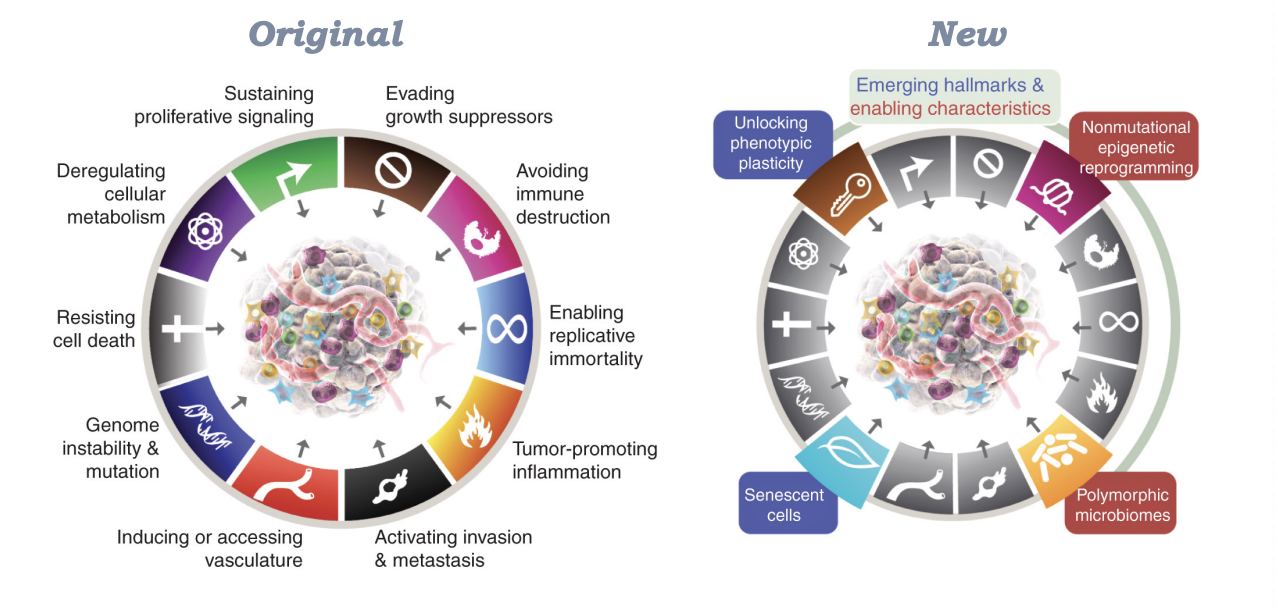
What are hallmarks of cancer
-sustaining proliferative signaling
-deregulating cellular metabolism
-resisting cell death
-genome instability and mutation
-inducing or accessing vasculature
-activating invasion and metastasis
-tumor-promoting inflammation
-enabling replicative immortality
-avoiding immune destruction
-evading growth suppressors
-unlocking phenotypic plasticity
-non mutational epigenetic reprogramming
-senescent cells
-polymorphic microbiomes
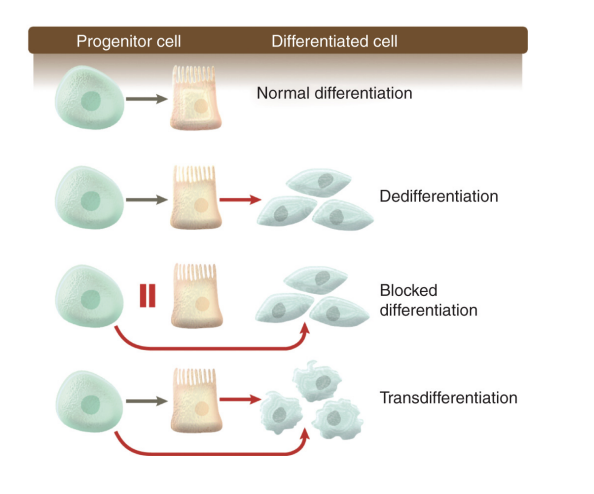
What is a critical component of cancer pathogenesis
unlocking the normal restricted capacity for phenotypic plasticity to evade or escape terminal differentiation
What causes epigenomic changes and what does it do
Nonmutational epigenetic reprogramming caused by aberrant physical properties of tumor microenvironment, leading to clonal outgrowth of cancer cells with enhanced fitness for proliferative expansion
What has profound impact on cancer phenotypes
polymorphic variability in the microbiomes between individuals in a population
What’s the principal mechanism by which senescent cells promote tumor phenotypes
senescence-associated secretory phenotype, which secretes signal molecules that act in a paracrine fashion and contribute to tumor phenotypes