low carbon - batteries, supercapacitors, and energy sources Q3
1/16
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
Primary vs. secondary batteries
Primary:
disposable (e.g. zinc-carbon, alkaline)
suffer from self-discharge (losing 8-20% charge yearly at room temp due to side reactions)
Secondary:
rechargeable (e.g. lead-acid, NiCd, Li-ion)
lifespan is limited by internal corrosion, side reactions, and active materials converting into inactive forms over many charge/discharge cycles
Leas-Acid Batteries (standard car batteries): electrochemical reactions
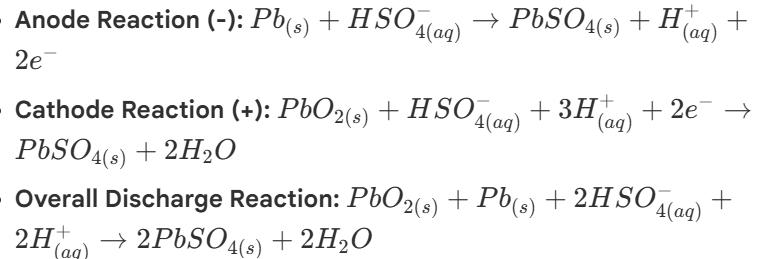
Li-metal vs. Li-ion: Why did Li-Metal fail and the Li-ion solution
Why?
Early primary batteries used pure Lithium metal because very strong reducing agent with high standard potential (+3.04V).
Making them rechargeable failed because pure Li forms "dendrites" (spiky crystals) during recharging, pierce the separator, causing short circuits/explosions.
The Li-Ion Solution:
Instead of pure metal, modern batteries use Chemical Intercalation.
Chemical Intercalation Process
Intercalation: reversible inclusion/extraction of Lithium ions (Li+) into and out of host lattice without destroying host's crystal structure.
anode (graphite): Graphite is composed of layered 2D sheets of carbon. During charging, Li+ ions easily slip between these layers (intercalation) and are stored safely as LixC6, preventing explosive dendrite formation.
cathode: Usually a metal oxide lattice, like Lithium Cobalt Oxide (LiCoO2).
Li-ion Reactions (during charging)
note: during discharge these reactions run in reverse
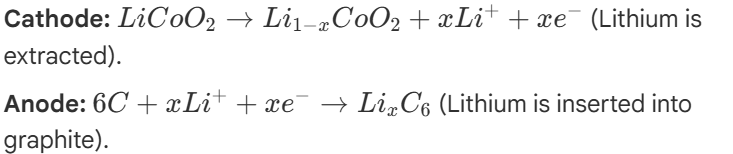
Lithium-Sulfur (Li-S) Batteries
Reactions:
Anode: 2Li → 2Li+ + 2e-
Cathode: S + 2Li+ + 2e- → Li2S
Overall: 2Li + S → Li2S
advantages:
massive theoretical specific energy (2.6 kWh/kg compared to standard Li-ion at ~0.5 kWh/kg)
sulfur is incredibly cheap and abundant.
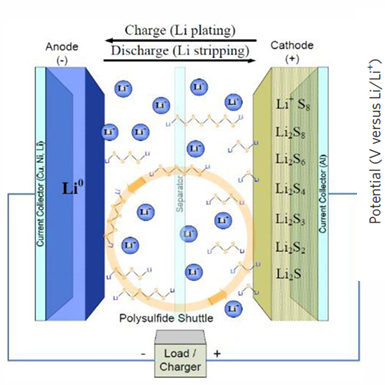
Issues:
polysulfide shuttle effect
Solid sulfur has terrible electrical conductivity (10-30 S/cm)
undergoes massive volume expansion (up to 80%) during discharge, causing severe mechanical stress.
Polysulfide shuttle effect
During discharge at cathode, sulfur does not instantly become Li2S.
S8 → Li2S8 → Li2S6 → Li2S4 → Li2S3→etc. (Discharge @ cathode)
Li2S → Li2S2 → Li2S3 → Li2S4 → Li2S6 → Li2S8 → S8 (Charge @ cathode)
Intermediate polysulfides (like Li2S8 and Li2S6) are highly soluble in organic electrolyte.
They dissolve, "shuttle" across separator to anode, and ruin battery's capacity.
Sodium-Ion (Na-Ion) Batteries
Advantages:
Na is 6th most abundant element on Earth - vastly cheaper than Lithium.
NaO2 discharge products are more stable than Li2O2.
Differences/disadvantages:
Sodium cation (Na+) is significantly larger than Lithium cation (1.0 Å vs 0.7 Å).
because of larger molecular radius, ions move much slower through electrolyte and lattice, resulting in slower charge/discharge rates.
They have lower specific capacity and lower average voltage (-0.3V compared to Li/Li+).
Mechanism of supercapacitors
store energy in electrostatic fields
when charged, accumulation of oppositely charged ionic species gathers at interface between conductive electrode and liquid electrolyte, creating a ‘double layer’
pros: extremely fast charge/discharge times, 95% efficiency, can endure hundreds of thousands of cycles without degrading
cons: have low energy density (3-5 Wh/kg) compared to batteries
Why are carbon nanomaterials utilised in supercapacitors?
capacitance is directly proportional to surface area of electrodes (C=ϵrϵ0A/d).
They provide incredibly massive specific surface area for ions to accumulate on.
example to cite:
Graphene: Achieves highest energy densities (85.6 Wh/kg) but layers tend to restack.
Aligned Carbon Nanotubes (SWCNTs): High power density.
Carbon Aerogels: Highly porous, offering up to 1500 m²/g of surface area.
why are supercapacitors used in conjunction with batteries?
supercapacitors have huge power density but low energy density, they are paired with battery strings (like in hybrid buses/wind turbines).
supercapacitor handles short, intense power surges (like acceleration or emergency doors), while battery handles steady, long-term energy output.
this drastically reduces cycling strain on battery and extends its life.
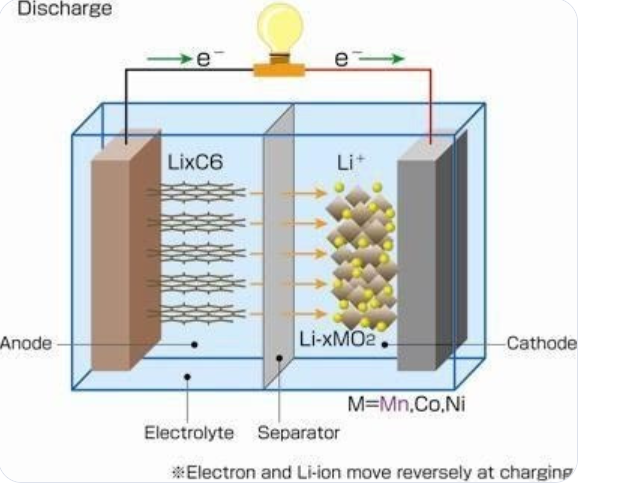
Li-ion chemical intercalation diagram
polysulfide shuttle effect diagram
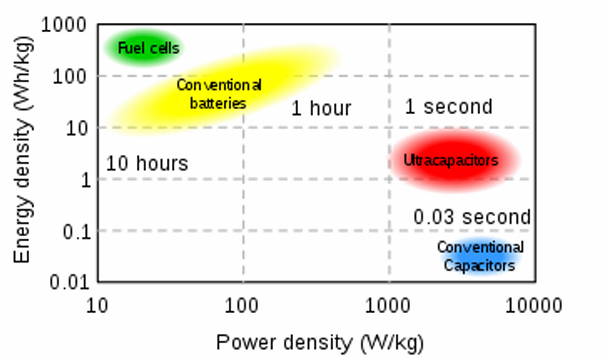
Ragone chart
Metal-air batteries
use a pure metal anode and extract oxygen directly from the surrounding air for cathode reaction, giving them massive theoretical energy densities
metal-air batteries: Lithium-air batteries
Reactions (Aprotic Electrolyte):
Anode: Li(s)→Li++e−
Cathode: Li++e−+O2→LiO2∗ (which then becomes Li2O2)
Advantages:
theoretically offer 5-15x more specific energy than standard Li-ion batteries (approaching the energy density of gasoline at ~12 kWh/kg) - massively increase driving range of electric vehicles.
Issues:
Humidity severely degrades cathode and discharge products.
pure Lithium anode is extremely reactive and forms dendrites that can pierce the separator
Metal-air batteries: Sodium-Air (Na-Air / Na-Oxygen) Batteries
Reactions:
Na+O2→NaO2 (E0 = 2.27 V)
2Na+O2→Na2O2 (E0 = 2.33 V)
2Na+21O2→Na2O (E0 = 1.95 V)
Comparison to Li-ion / Opportunities:
Na is 6th most abundant element on Earth - incredibly cheap and highly scalable for large grid storage compared to Lithium.
NaO2 discharge product is chemically more stable than Li2O2 product found in Li-air batteries