History of the atom
1/13
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
Who came up with the idea that atoms are spheres and when?
John Dalton in 1803

What did Dalton say about elements?
That each element is made from different spheres

Who discovered radioactivity and when?
Becquerel in 1896

What did Becquerel's discovery of radioactivity show?
That particles could come from inside an atom, therefore the atom was divisible
Who discovered the electron?
JJ Thomson

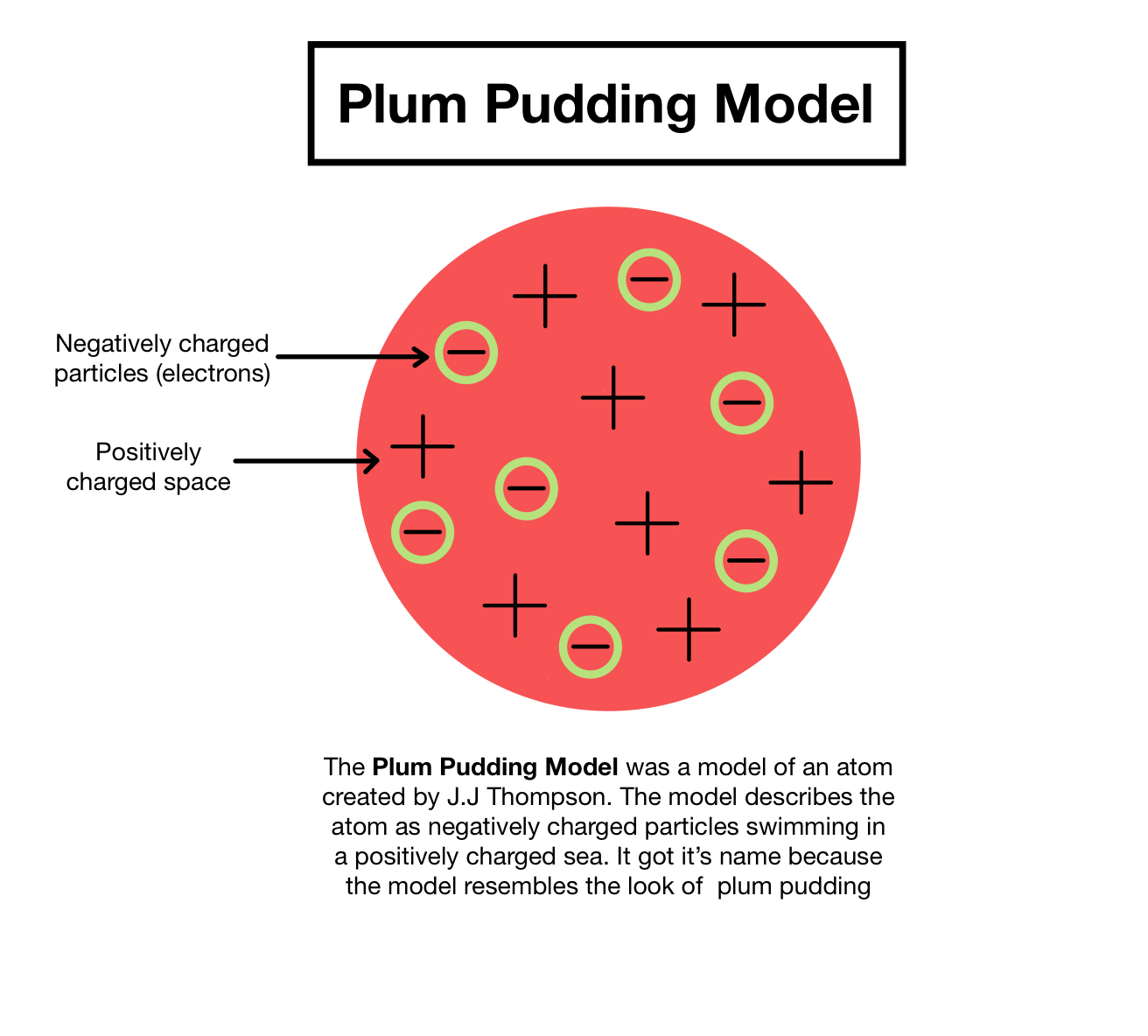
What was JJ Thomson's model and when did he propose it?
Plum pudding model, proposed in 1904
What was the plum pudding model?
An atom is a ball of positive charge with negative electrons embedded in it
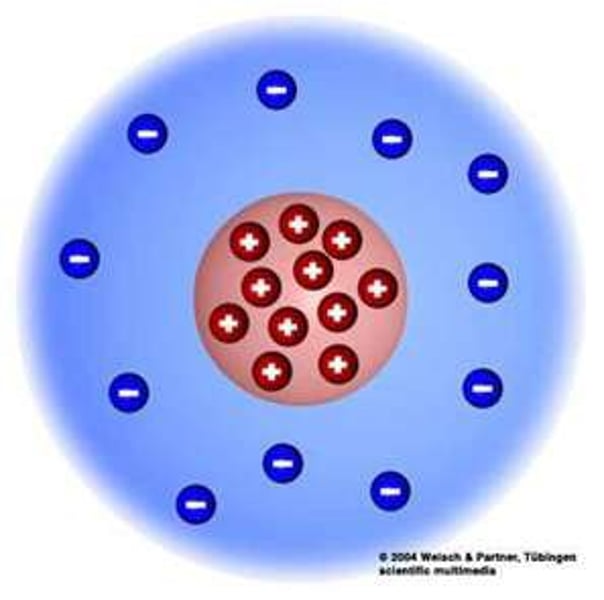
Who discovered the nucleus and when?
Rutherford in 1909

What did Rutherford discover about the nucleus?
The nucleus was very small and was positively charged
What was Rutherford's experiment?
Gold foil experiment
What happened in the gold foil experiment and what did it show?
Positive alpha particles were fired at a thin sheet of gold. Most of the particles went straight through, which showed atoms consist mainly of empty space. However, some particles deflected back, meaning they would have hit a positive nucleus
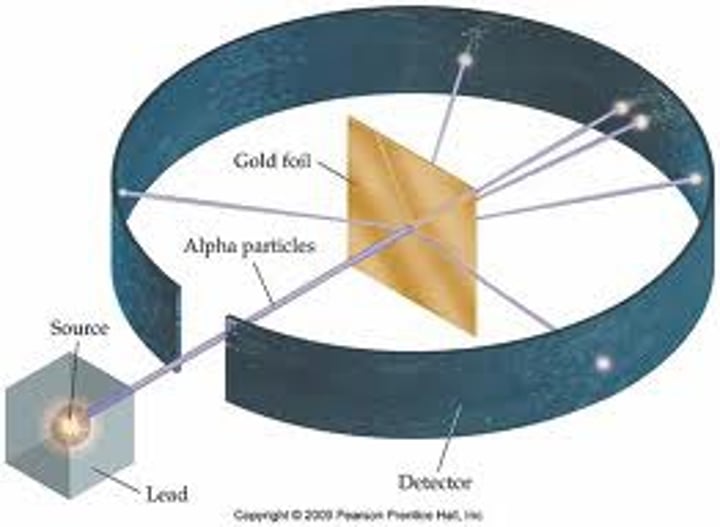
What did Rutherford conclude about the atom?
The atom was mainly empty space and had a positive nucleus
Who discovered a problem with Rutherford's model? What was the problem?
Niels Bohr. He said the cloud of electrons would collapse into the positive nucleus

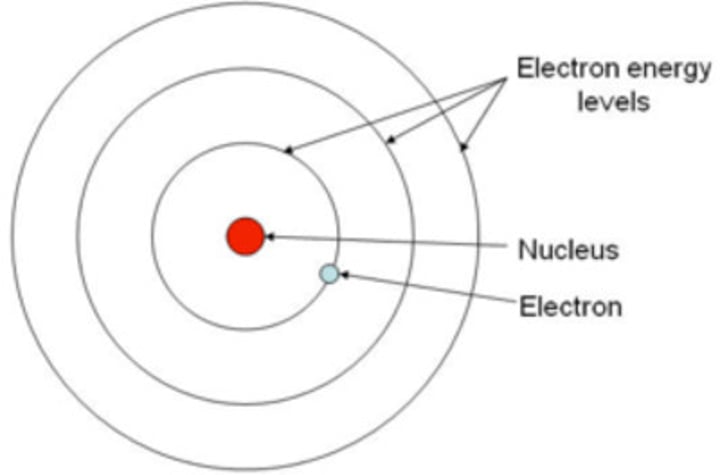
How did Bohr fix this problem? (what did he propose?)
He developed the first quantum theory of the atom, with electrons assigned to fixed orbits of defined energy