Peripherally Acting Agents to Treat Pain (Hale)
1/52
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
53 Terms
Aspirin
Therapeutic uses:
- Temporary relief of minor aches and pains associated with cold, headache, backache, toothache, menstrual cramps
-Temporary reduction of fever
-Relief of signs and symptoms of rheumatologic diseases
-Prevention and treatment of MI, ischemic stroke, and TIA
MOA: Non-selective, irreversible inhibition of cyclooxygenase (COX) enzymes
Aspirin’s therapeutic effects follow its inhibition of the three (3) known cyclooxygenase enzymes (COX-1,2, and 3)
- COX-1 >> COX-2 >> COX-3
Metabolism: Rapidly de-acetylated to salicylic acid --> salicylic acid is converted by Phase II (non-oxidative) metabolism to Glycine conjugate and Glucuronide conjugates
CI: GI bleeding
Warnings: GI, asthma exacerbation, allergic rxns, pregnancy
DDIs: other NSAIDs, antiplatelets, corticosteroids, diuretics, aldosterone antagonists, warfarin (and anticoagulants), methotrexate, lithium
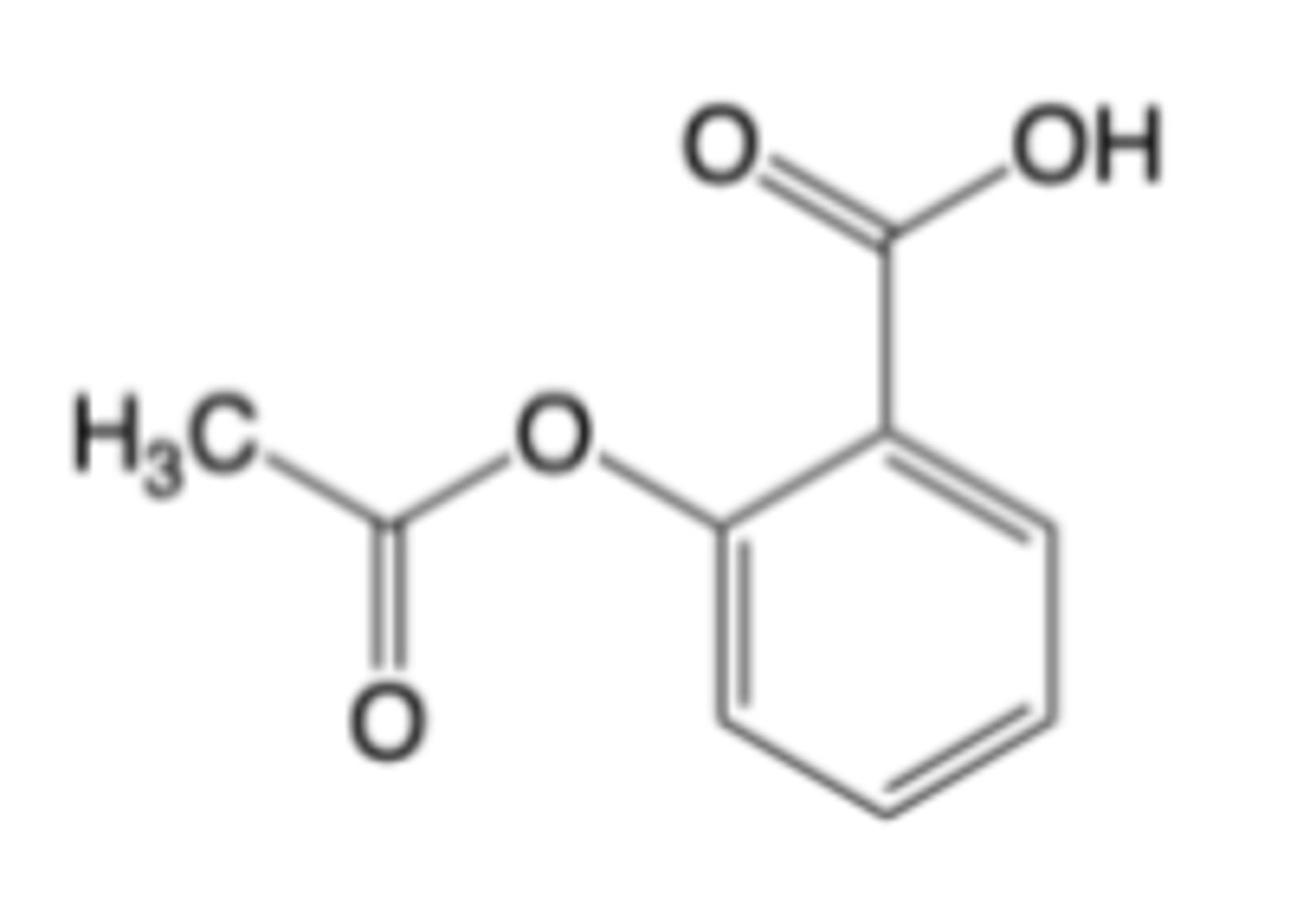
Salsalate
-Salicylic Acid Analog
-non-selective, reversible
-minimal effects on gastric mucosa, minimal symptoms
-No cardiovascular benefit
-no platelet effects
Aspirin (which is available over-the-counter) sees vastly greater clinical use than the prescription salicylates above
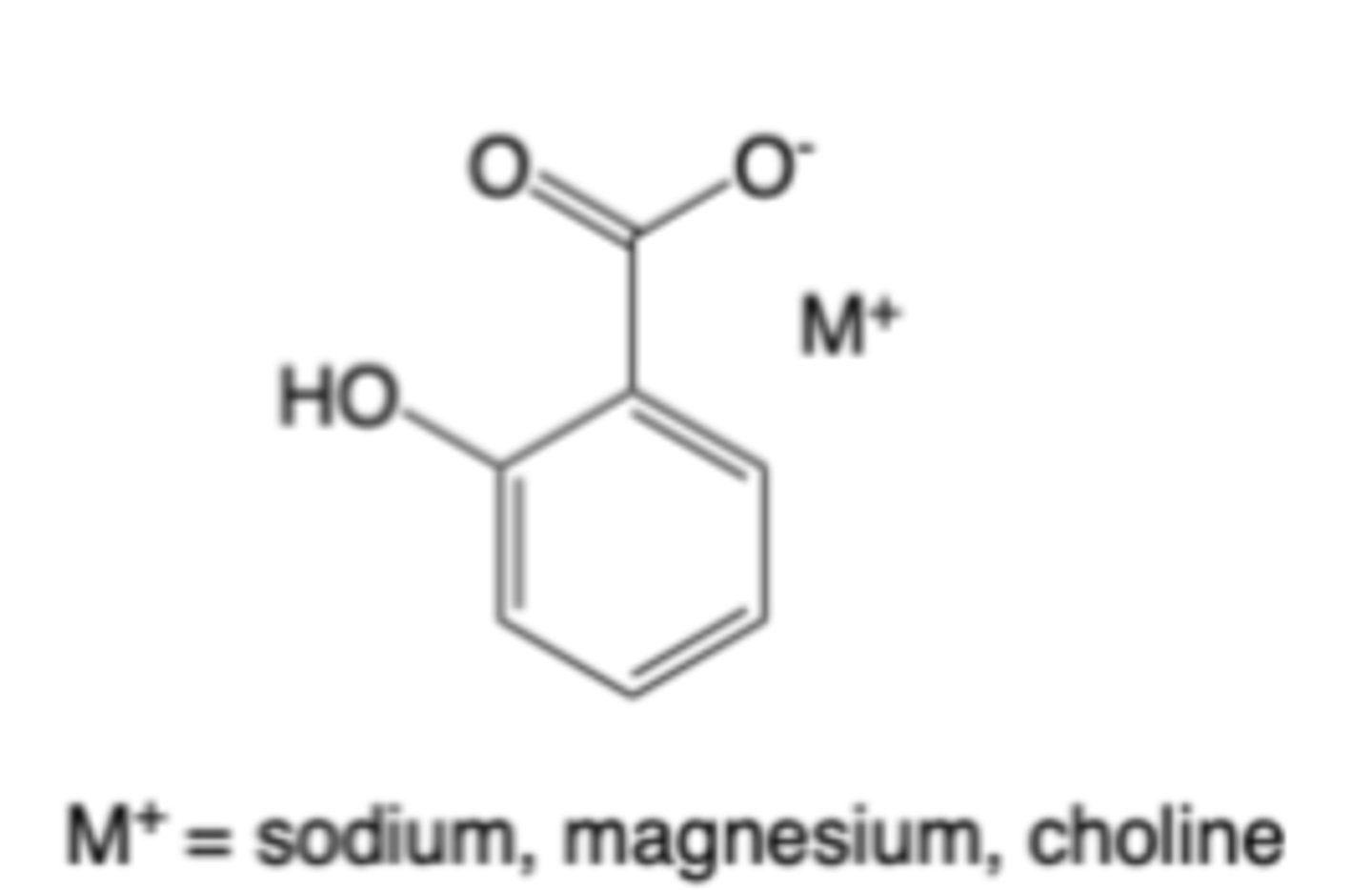
Salicylic acid salts
-non-selective, reversible
-Lower effects on gastric mucosa, milder symptoms
-No cardiovascular benefit, no platelet effects
Aspirin (which is available over-the-counter) sees vastly greater clinical use than the prescription salicylates above
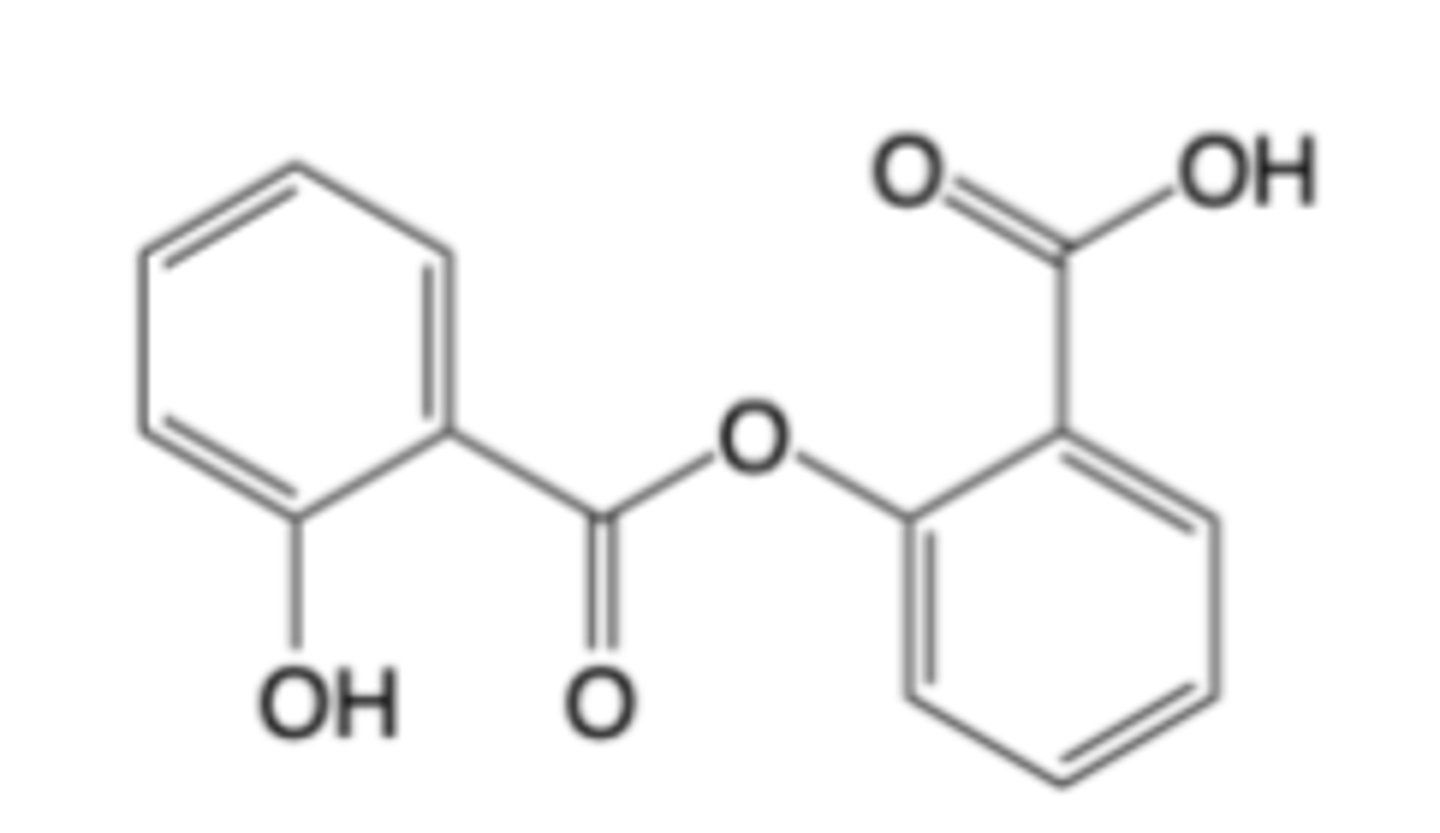
Diflunisal
-Indicated for acute and long-term symptomatic relief of mild-to-moderate pain, osteoarthritis and rheumatoid arthritis
-MOA: Non-selective, reversible inhibition of cyclooxygenase (COX) enzymes -> ~10x more potent than aspirin
-Minimal effects on platelet aggregation
-Overlap with aspirin regarding GI adverse effects, general bleeding renal impairment, mechanism-based DDIs
-Better overall gastrointestinal tolerability (vs. aspirin) and no Reye’s Syndrome risk
Acetaminophen (Tylenol®)
Therapeutic Uses:
-Temporary relief of minor aches and pains associated with cold, headache, backache, toothache, menstrual cramps
-Temporary reduction of fever
NOTE: Analgesic and antipyretic efficacy is similar to that of aspirin
-Acetaminophen does NOT have appreciable anti-platelet effects or anti-inflammatory efficacy
MOA:
-Weak, non-selective inhibition of the peroxidase function of cyclooxygenase (COX) enzymes -> decreases central PGs
-Activation of serotonergic descending inhibitory pathways in the CNS -> suppresses nociception
-Activation of endocannabinoids and TRPV1 ion channels in the CNS -> enhances analgesia
Acetaminophen can generally be substituted for aspirin as a pain relief medication and fever reducer in patients with gastrointestinal issues, who are at risk for bleeding and in children with viral infections (no Reyes syndrome risk)
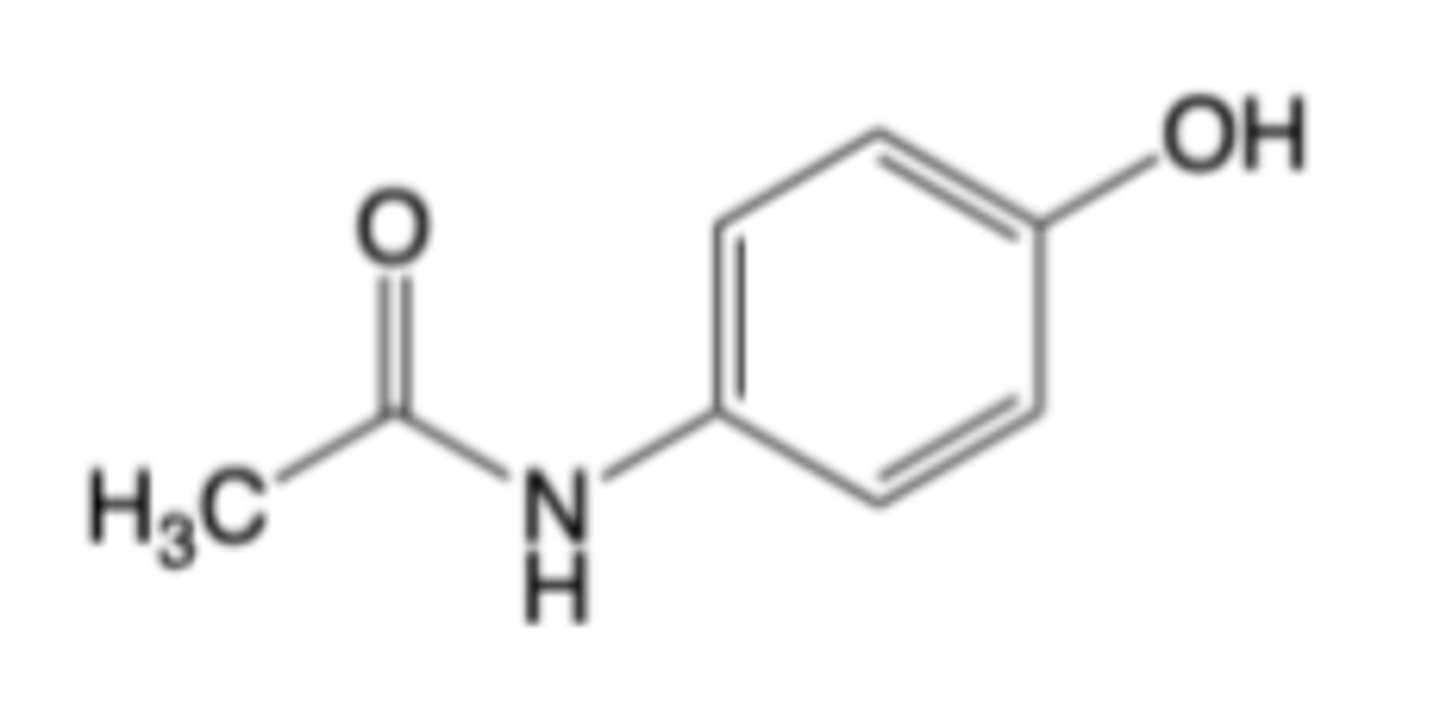
CI: hypersensitivity, hepatic impairment or liver disease
Warnings: administration of higher than recommended may result in hepatic injury
DDI: CYP2E1 inhibitors may alter the metabolism of acetaminophen and increase its hepatotoxic potential
Acetaminophen is primarily converted by Phase II (non-oxidative)
metabolism to:
• Glucuronide conjugate
• Sulfate conjugate
A minor metabolic pathway is oxidation by CYP2E1 and CYP3A4 which ultimately forms chemically-reactive NAPQI (N-acetylimidoquinone)
-NAPQI is normally de-toxified via glutathione conjugation --> Renal or toxicity can occur in instances where glutathione supply is saturated or glutathione --stores stores are depleted
-Ethanol induces CYP2E1 -> greater hepatotoxicity risk of acetaminophen with alcohol consumption
Metabolism in brain: Deacetylation and conjugation with arachidonic acid in the CNS to form AM404 may contribute to central analgesia
Acetaminophen: ADME Properties
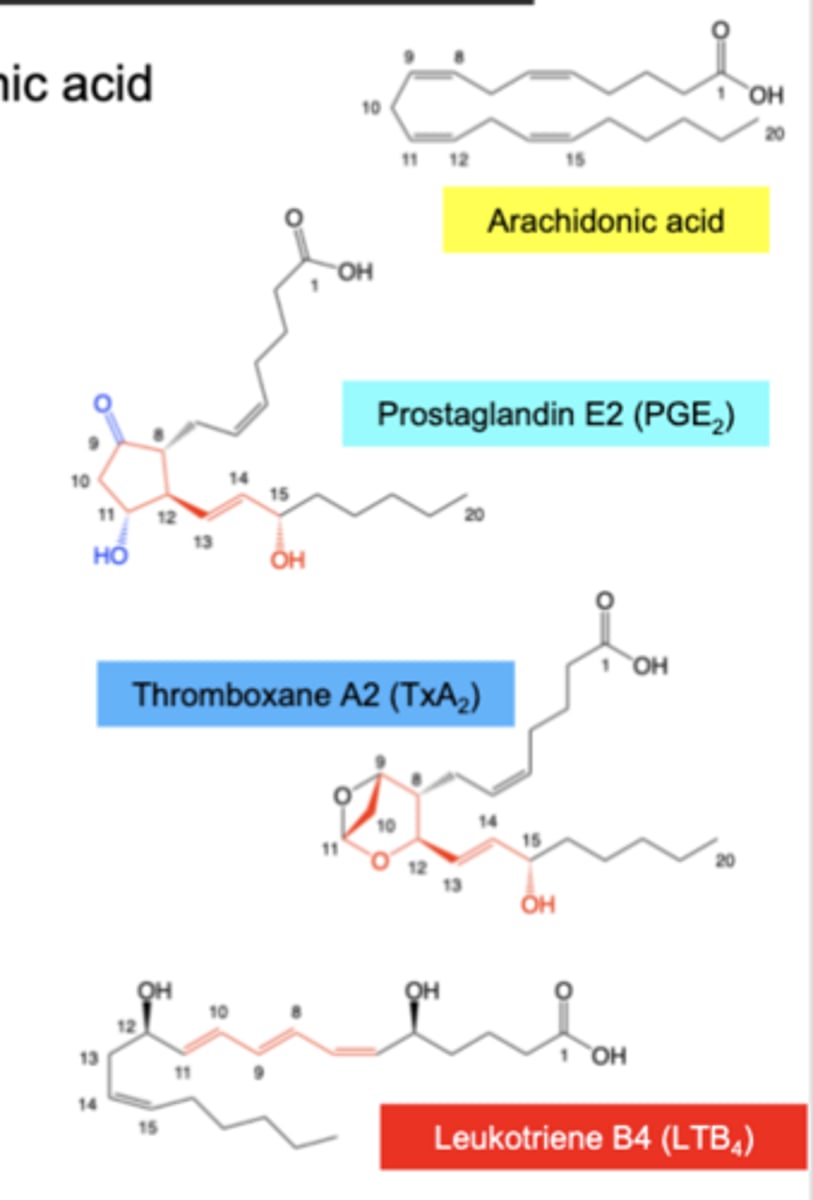
Eicosanoids are locally-acting signaling molecules derived from arachidonic acid
Prostaglandins (PGs)
-Structures contain a 5-membered ring, C13-C14 double bond, C15-alpha-hydroxy (subclass depend on nature and stereochemistry of C9,C11 oxygens)
-Aspirin reduces inflammation and pain by decreasing PG biosynthesis
Thromboxanes (TXs)
-Structures contain a 6-membered ring, C13-C14 double bond, C15-alpha-hydroxy
-Aspirin reduces risk of stroke and heart attack by decreasing TX biosynthesis
Leukotrienes (LTs)
-Structures contain three conjugated double bonds
-Inhibition of PG and TX synthesis by aspirin can increase LT biosynthesis
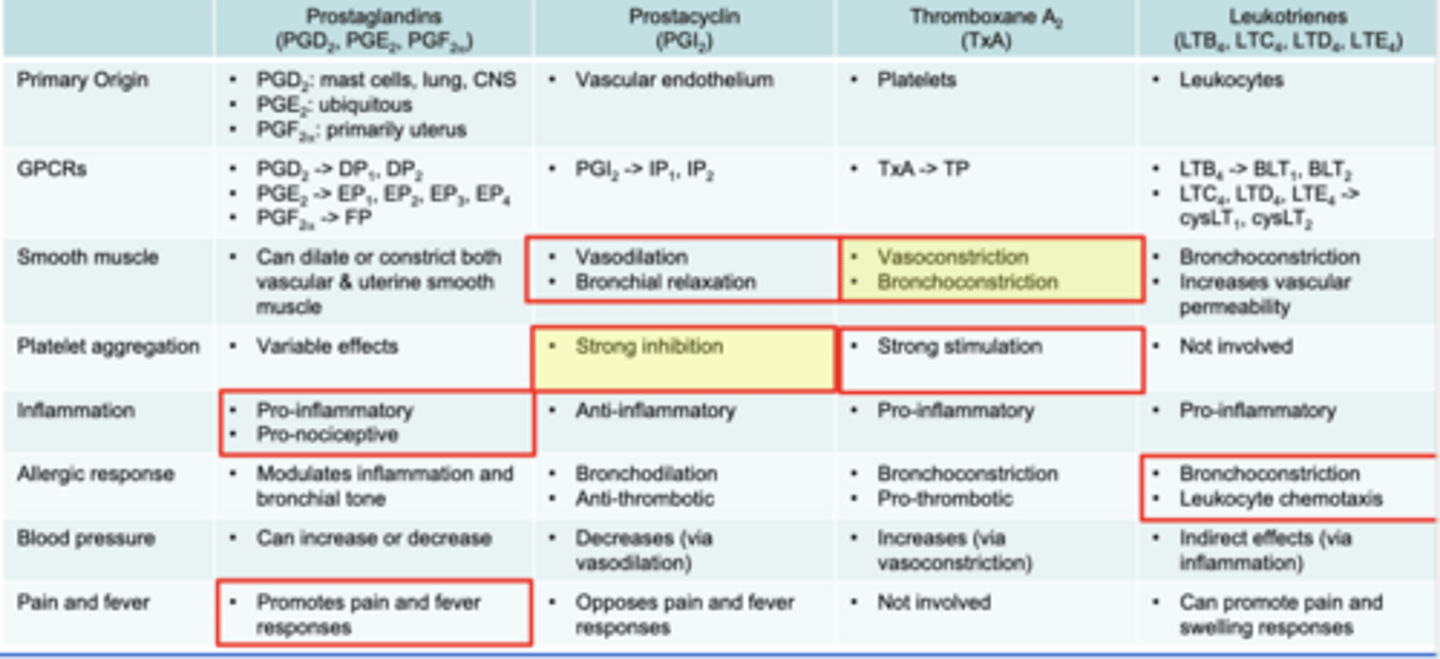
Eiconasoids: Prostaglandins, Thromboxanes and Leukotrienes
Smooth muscle: can dilate or constrict vascular or uterine smooth muscle
Inflammation: pro-inflammatory. pro-nociceptive
Pain and fever: promotes pain and fever responses
PGE1 plays major role in gastrointestinal cytoprotection; PGE1 is production
primarily mediated by COX-1
Prostaglandin Biosynthesis & Functions
Misoprostol
-Prostaglandin Receptor Modulator
-EP1 agonist (PGE1 analog)
-Indicated for gastric ulcers, labor induction
Omindenepag isopropyl
-Prostaglandin Receptor Modulator
-EP2 antagonist
-Indicated for glaucoma, ocular hypertension
Latanoprost
-Prostaglandin Receptor Modulator
-FP agonist
-indicated for glaucoma, ocular hypertension
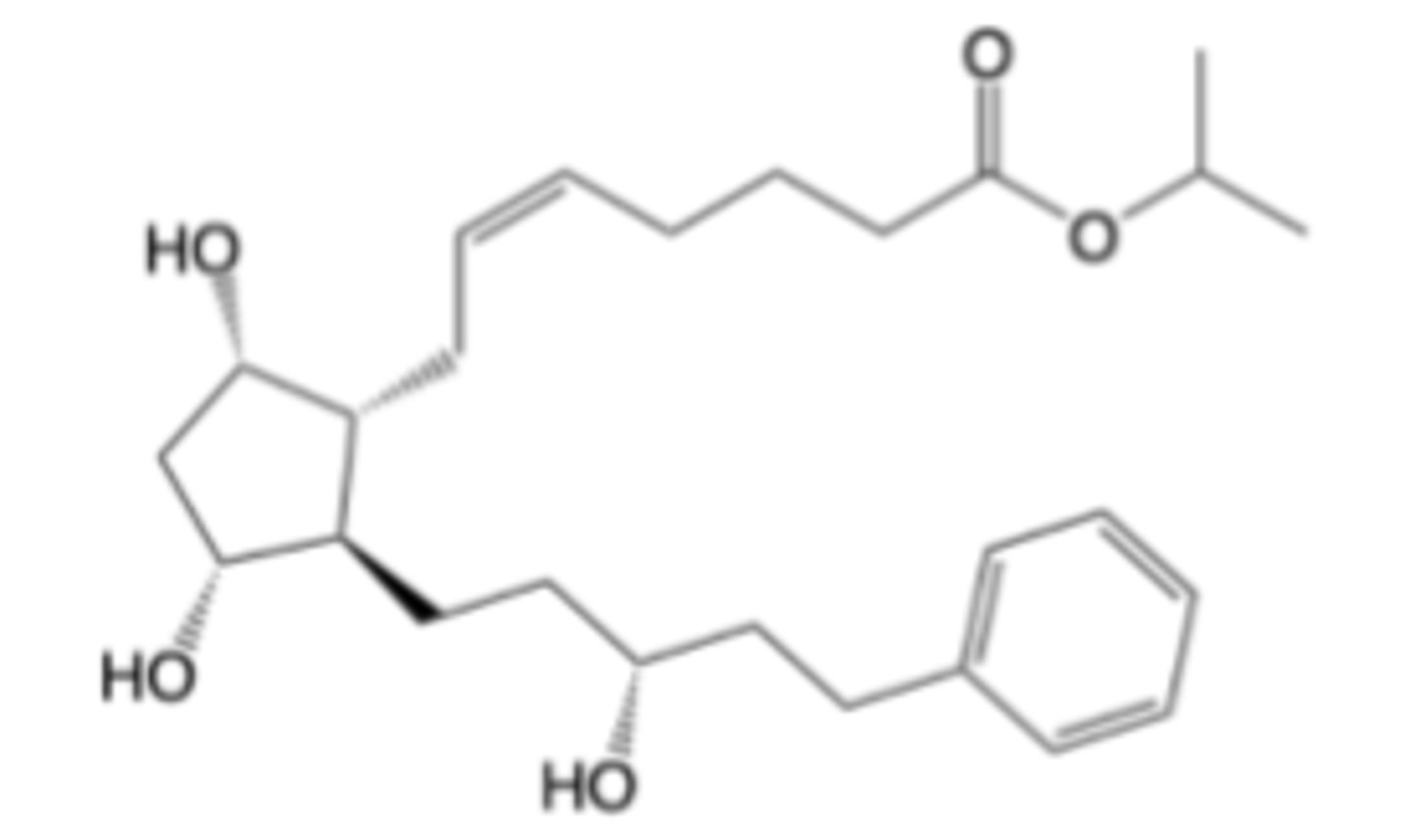
Prostacyclin (PGI2)
-Smooth muscle: vasodilation, bronchial relaxation
-Strong inhibition of platelet aggregation
Thromboxane A2 (TxA)
-Smooth muscle: Vasoconstriction, Bronchoconstriction
-strong stimulation of platelet aggregation
Prostacyclin & Thromboxane Biosynthesis and Function
Selexipag
-Prostacyclin & Thromboxane Receptor Modulator
-IP agonist
-Indicated for pulmonary hypertension
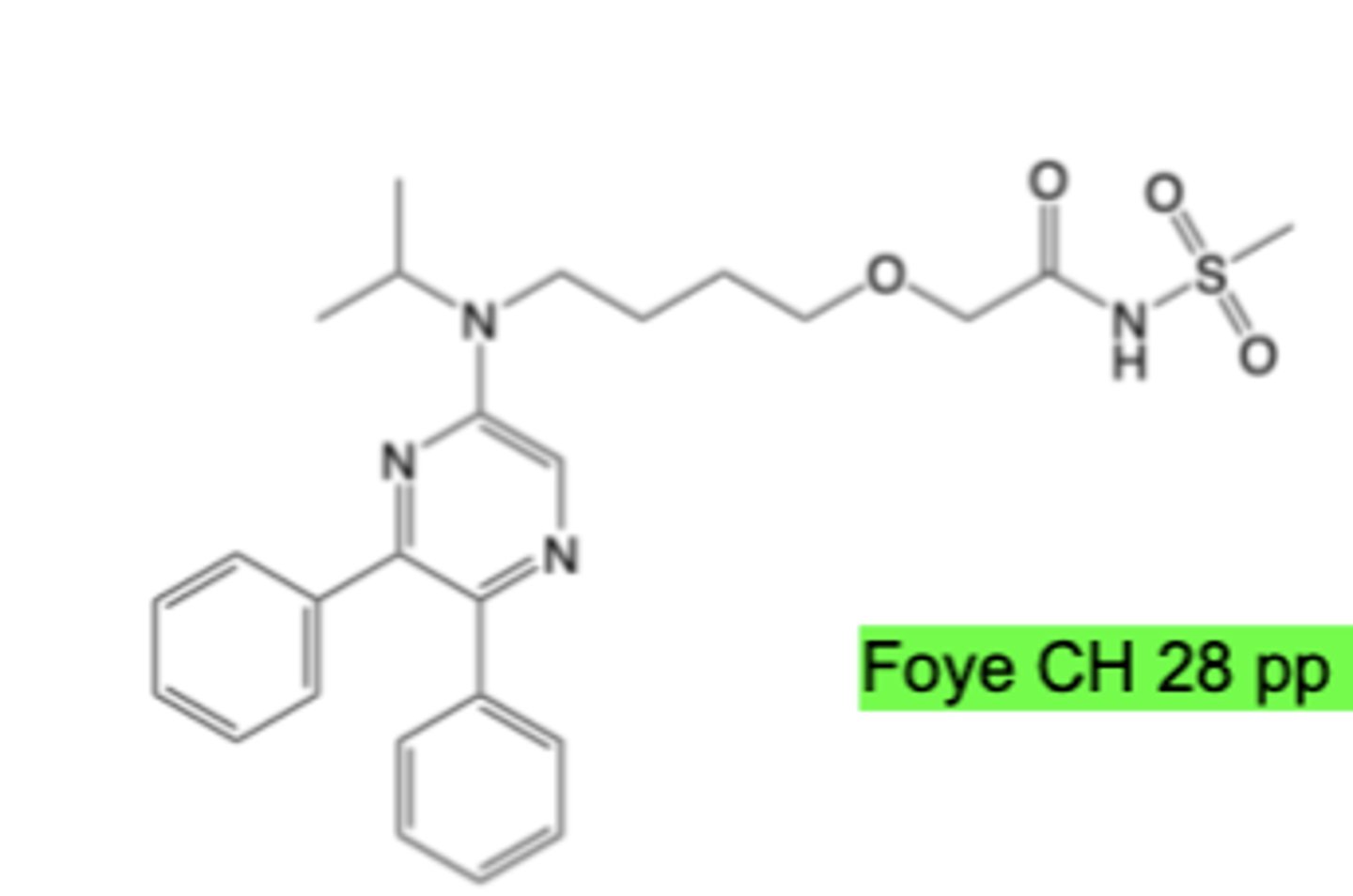
Leukotrienes (LTB4, LTC4, LTD4, LTE4)
-Pro-inflammatory
-Allergic response: Bronchoconstriction, Leukocyte chemotaxis
Leukotriene Biosynthesis and Function
Montelukast
-Leukotriene Receptor Modulator
-cysLT1 antagonist
-Indicated for asthma, exercise-induced bronchoconstriction, allergic rhinitis
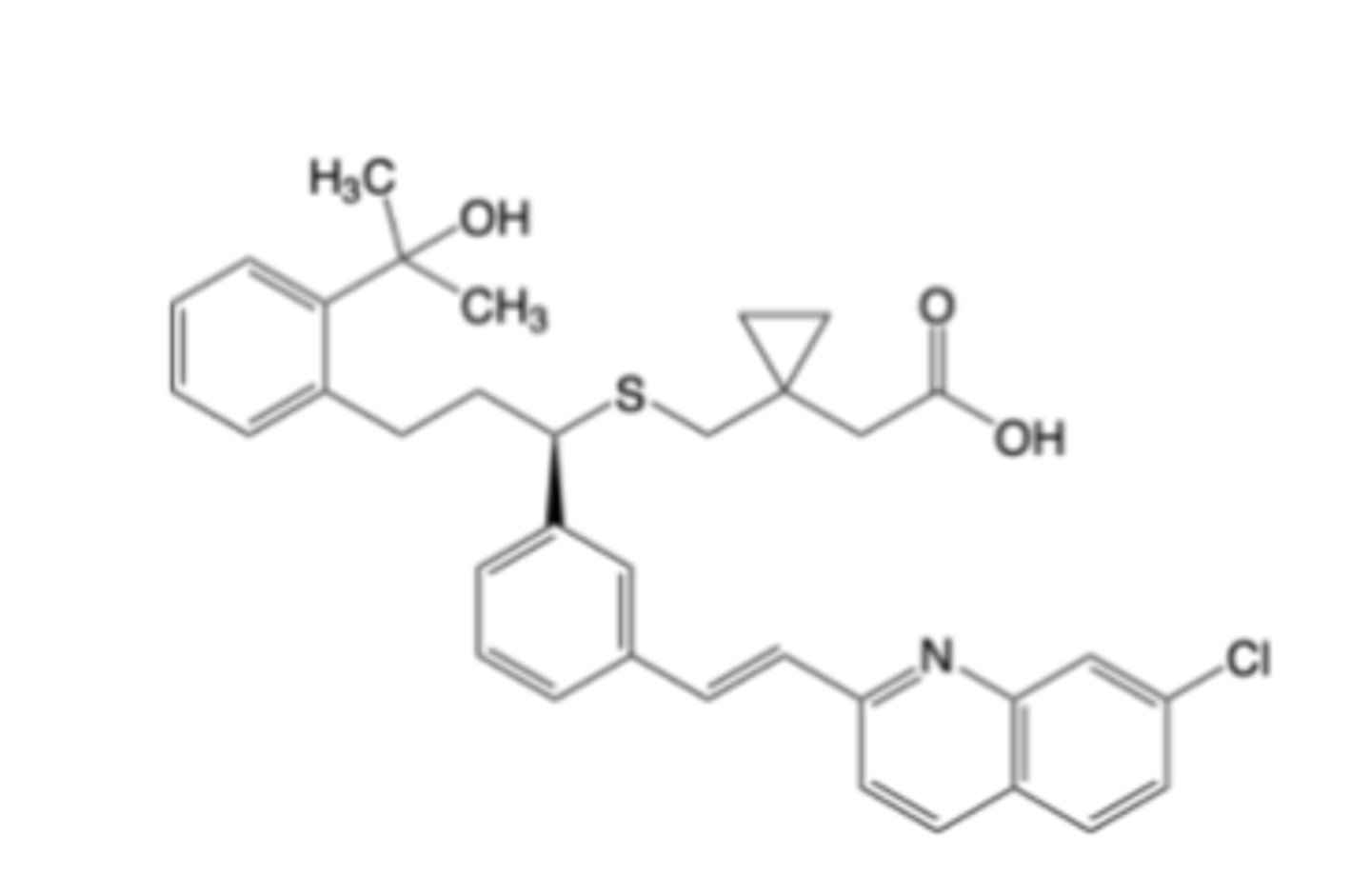
Zafirlukast
-Leukotriene Receptor Modulator
-cysLT1 antagonist
-Indicated for asthma, exercise-induced bronchoconstriction, allergic rhinitis
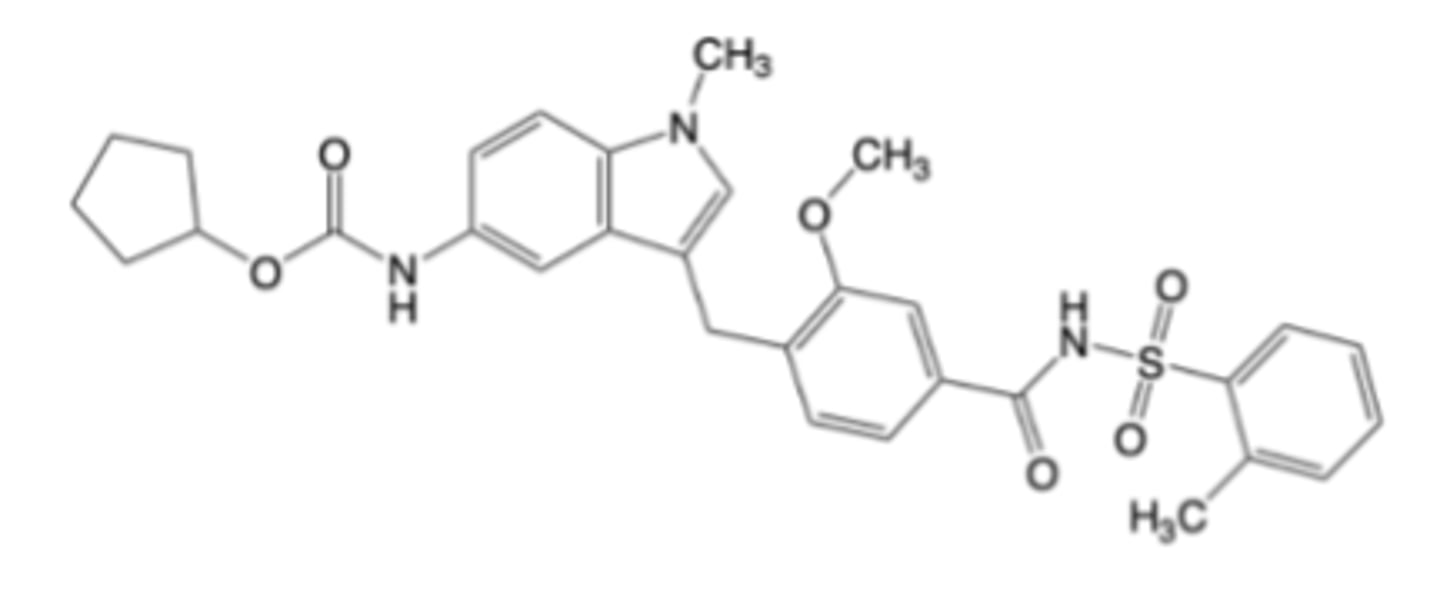
Zileuton
-Leukotriene Receptor Modulator
-5-lipoxygenase inhibitor
-indicated for asthma
Consequences of COX-1 Inhibition
-Beneficial effects: Contributes to anti-platelet effects that reduce the risk of blood clotting, which is clinically exploited by low-dose aspirin for cardiovascular protection
-Detrimental effects: Often causes GI irritation and damage, increased risk of gastric ulcers and bleeding, platelet dysfunction leading to bleeding tendencies, and can cause bronchospasm in some individuals
Consequences of COX-2 Inhibition
-Beneficial effects: Effectively reduces pain and inflammation; May also reduce vascular inflammation & slow the progression of atherosclerosis by stabilizing plaques and improving endothelial function
-Detrimental effects: Disrupts the balance between vasodilatory PGI2 and pro-thrombotic TXA, which can increase the risk of CV events such as MI, stroke, HTN, CHF, and thrombotic events due to unopposed platelet activation and vasoconstriction; Can elevate blood pressure
Non-Steroidal Anti-Inflammatory Drugs (NSAIDs or COX Inhibitors)
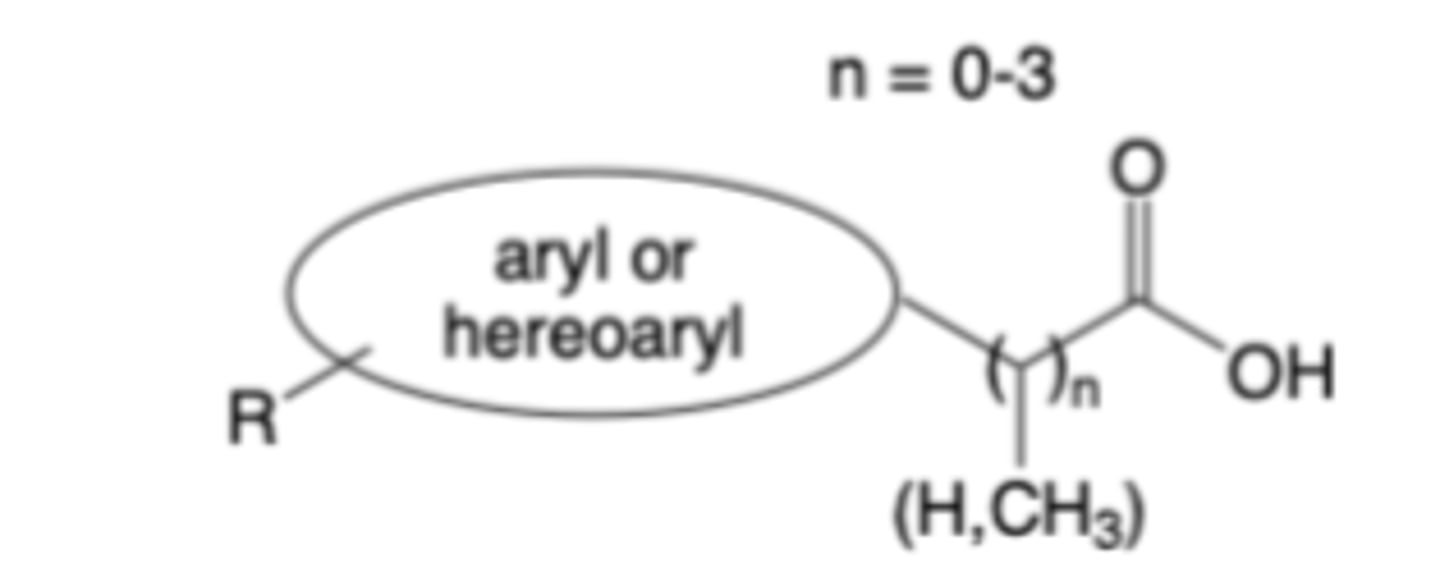
General structure of many COX inhibitors consists of:
– A carboxylic acid (or carboxylate bioisostere)
– A carbon linker (n = 0-3, with n=1 most common)
– A central aryl or heteroaryl ring(s)
– An appended hydrophobic R group (or groups)
These features mimic the interactions of arachidonic acid
with COX enzymes that facilitate binding to the active site
-Carboxylic acid ion-pairs with Arg120
-Aryl or heteroaryl rings interact (via pi-stacking) with tyrosine (Tyr) and tryptophan (Trp) residues in both COX isoforms
-Substituents (R) on the aryl or heteroaryl rings optimize steric
fit and enhance inhibitory potency
Many COX inhibitors are non-selective or moderately selective
-Selective COX-2 inhibitors (COXIBs) have been pursued to
provide anti-inflammatory efficacy while minimizing GI toxicity
Structure-Activity Relationships (SAR) of COX Inhibitors
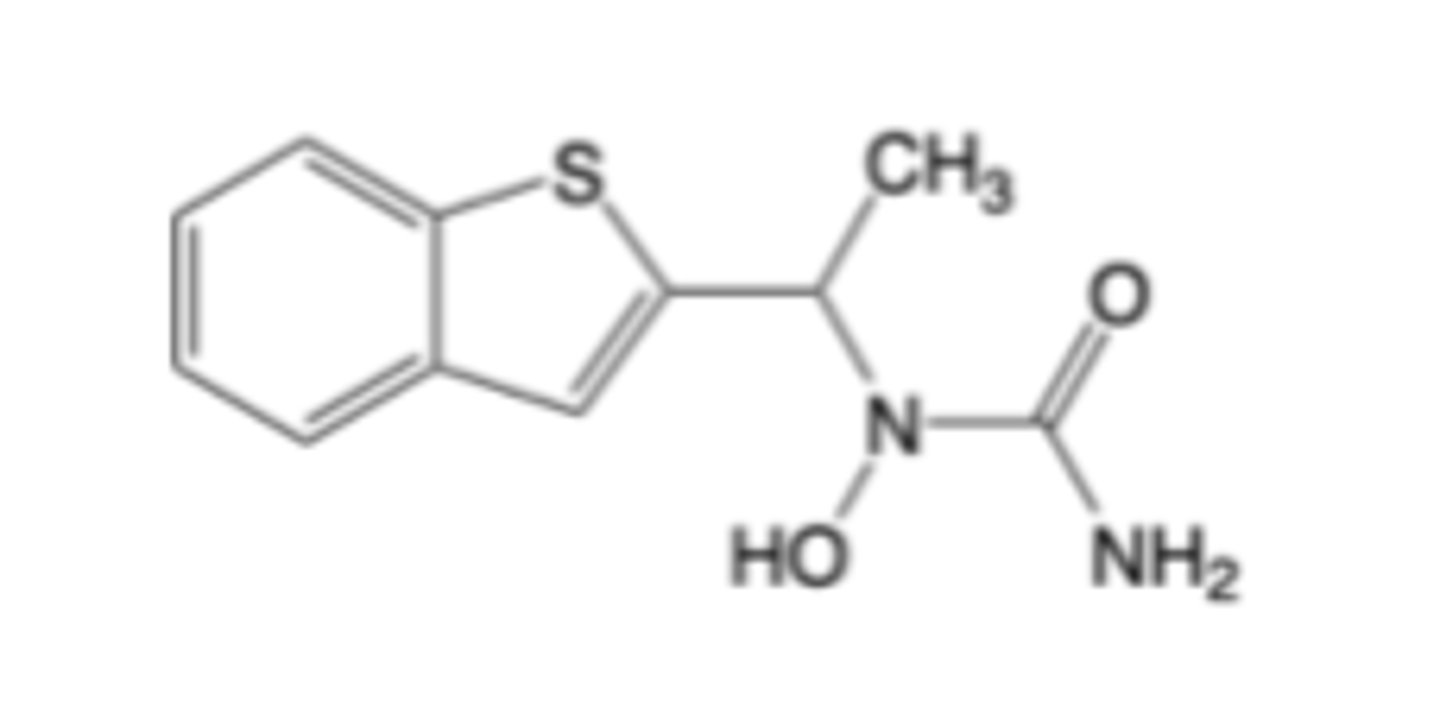
Indomethacin
-Oral formulation for chronic use in moderate-to-severe osteoarthritis, rheumatoid arthritis and ankylosing spondylitis
MOA:
-Non-selective, pseudo-irreversible inhibition of cyclooxygenase (COX) enzymes
-Potent suppression of PG synthesis -> Analgesic & anti-pyretic potency -> 10x vs. aspirin
Warnings:
-overlap with aspirin
-AEs: headache, neutropenia, thrombocytopenia (can limit dose, lead to discontinuation)
-BBW: CI post-coronary bypass, increased risk of CV thrombotic events, significant GI bleeding and ulceration risk (from potent PG inhibition)
ADME:
-Phase I (CYP2C9 oxidation -> O-demethylation), Phase II (glucuronidation)
and amide hydrolysis contribute to indomethacin’s metabolism
-All metabolites are inactive
SAR:
-20methyl enhances COX binding
-amide linker preferred
-C5 substitution of methoxy blocks metabolism
Diclofenac
-Formulated as sodium salt for oral (Voltaren®) and topical (Voltaren Gel®)
administration for chronic use in moderate-to-severe osteoarthritis, rheumatoid arthritis and ankylosing spondylitis
MOA:
-Potent reversible inhibition of cyclooxygenase (COX) enzymes -> ~10x selectivity for COX-2 over COX-1
-Additional analgesic and anti-inflammatory activities -> inhibition of 5-lipoxgenase, inhibition of phospholipase A2
-Analgesic & anti-pyretic potency -> ~100x vs. aspirin, ~10x vs. indomethacin
Warnings:
-BBW: CI post-coronary bypass, increased risk of CV thrombotic events
-hypersensitivity and severe liver injury
-COX-2 selectivity is sufficient to diminish GI adverse effects
ADME:
-Phase I (CYP3A4 oxidation) metabolism predominates
-4’-Hydroxy is major metabolite and forms reactive qunioneimine -> analogous to acetaminophen metabolite
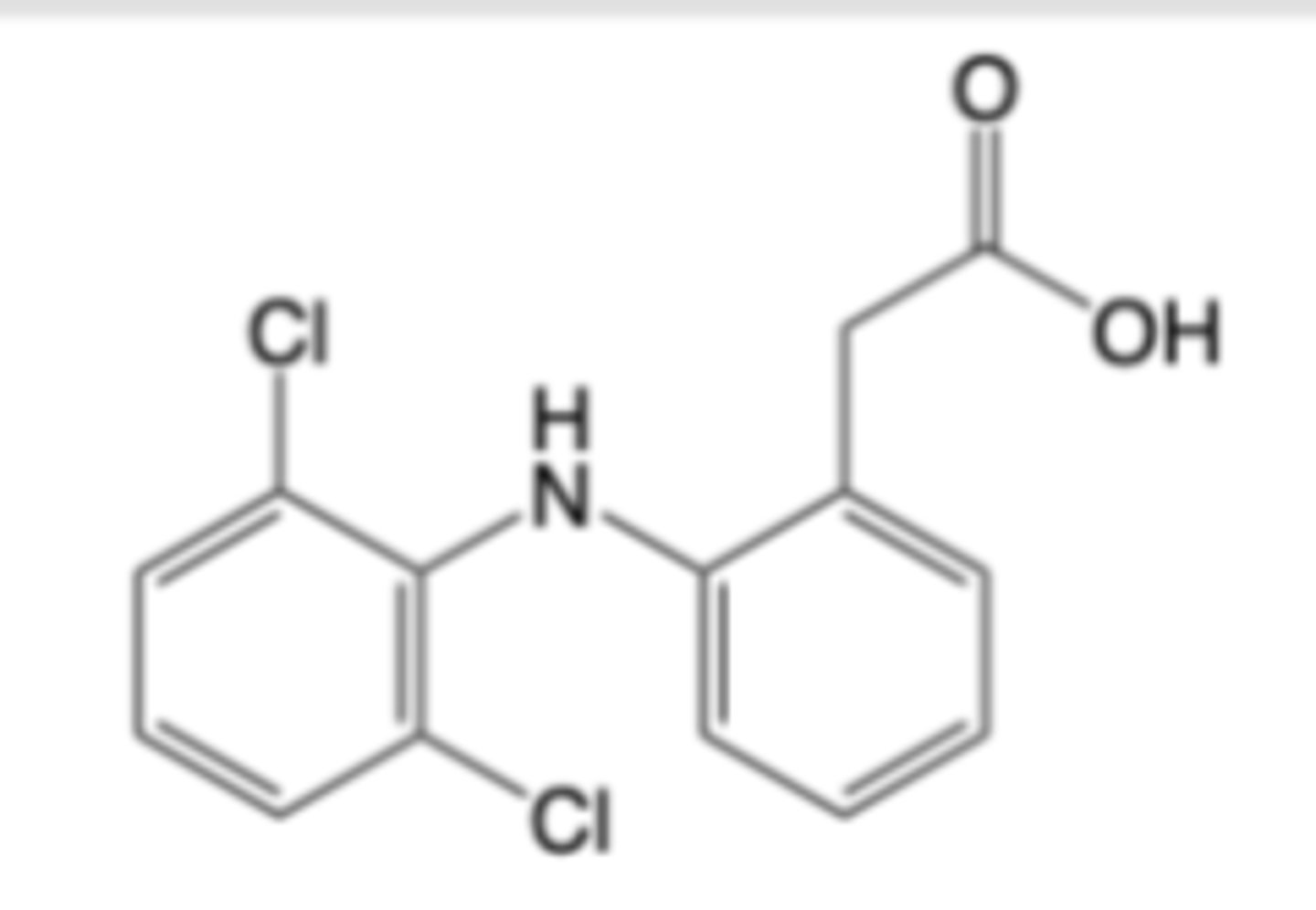
Ibuprofen
-Multiple over-the-counter (OTC) formulations for oral administration (Advil®, Motrin®) for temporary relief from headache, minor arthritis pain, backache, toothache, menstrual cramps, muscle aches, fever and pain from influenza
MOA:
-Non-selective, reversible inhibition of cyclooxygenase (COX) enzymes -> essentially equipotent to aspirin
Warnings:
-Gastrointestinal and bleeding risks present (but OTC doses are better tolerated than aspirin, indomethacin)
-overlap with aspirin
-increased risk of serious CV thrombotic events
-CI after 20-30 weeks of pregnancy
ADME:
-Oral administration of racemic (+/-)-ibuprofen results in the metabolic formation and excretion of (S)-ibuprofen and (S)-metabolites
-Ibuprofen is a substrate for AMACR, which converts inactive R ibuprofen to active S ibuprofen acyl-CoA after activation by acyl- CoA synthetase
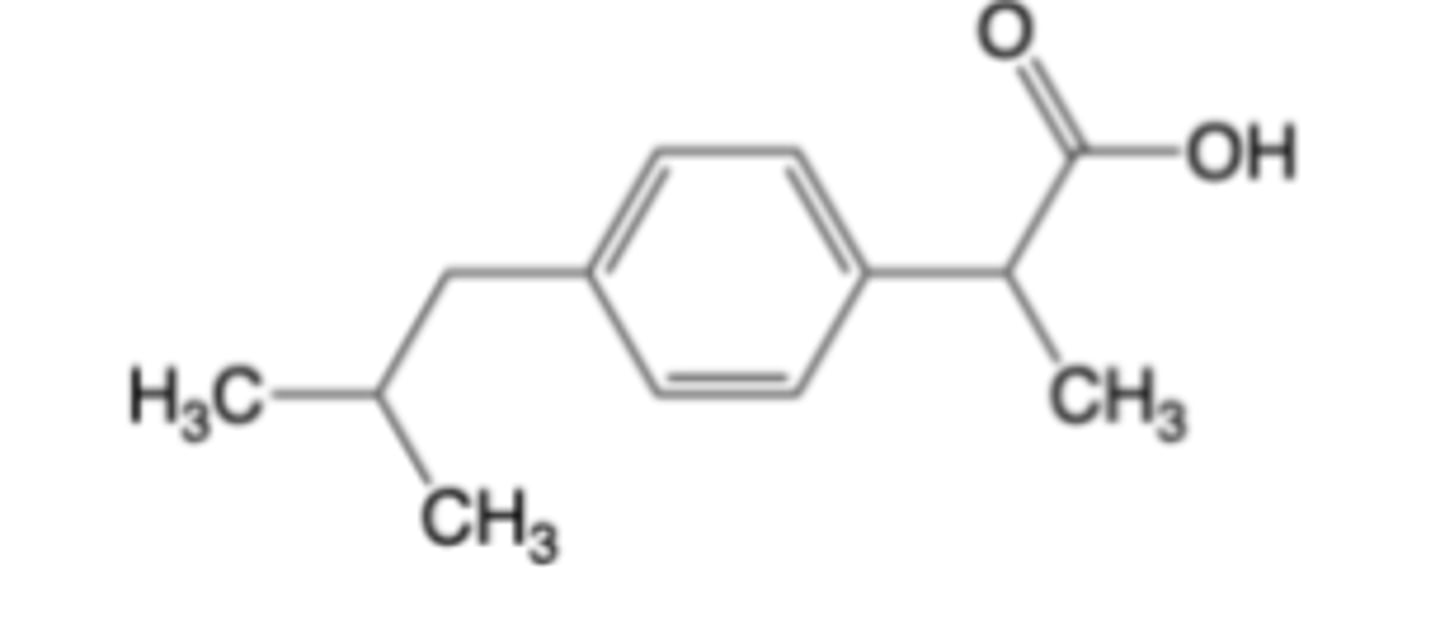
Naproxen
-Multiple formulations of sodium salt for oral administration (OTC = Aleve®, Naprosyn®) for osteoarthritis, rheumatoid arthritis and ankylosing spondylitis, acute painful shoulder (bursitis and/or tendinitis) and gouty arthritis
MOA:
-Non-selective reversible inhibition of cyclooxygenase (COX) enzymes -> essentially equipotent to aspirin, ibuprofen
Naproxen is NOT a substrate for alpha-methylacyl-CoA racemase (AMACR) -> no in vivo de-racemization like ibuprofen
Warnings:
-same as iburpofen, but less CV risk and greater GI risk (with alcohol use)
-CI after 20-30 weeks of pregnancy
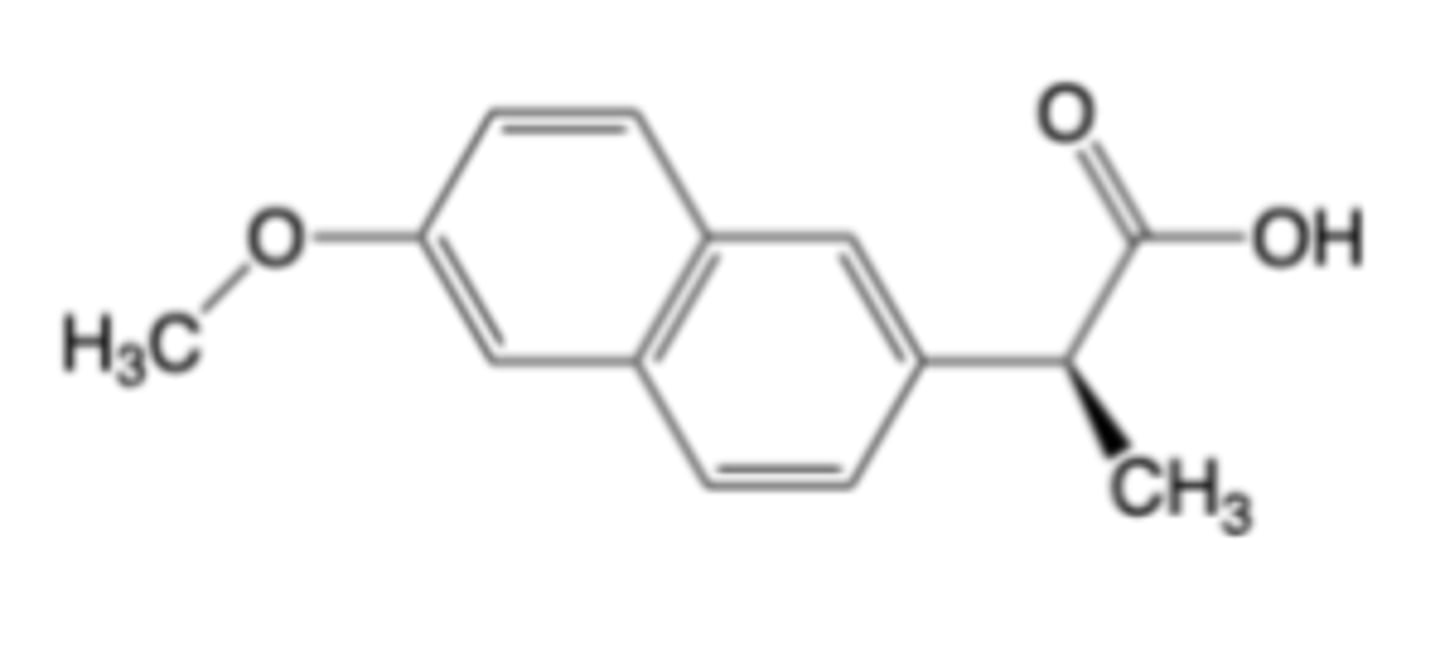
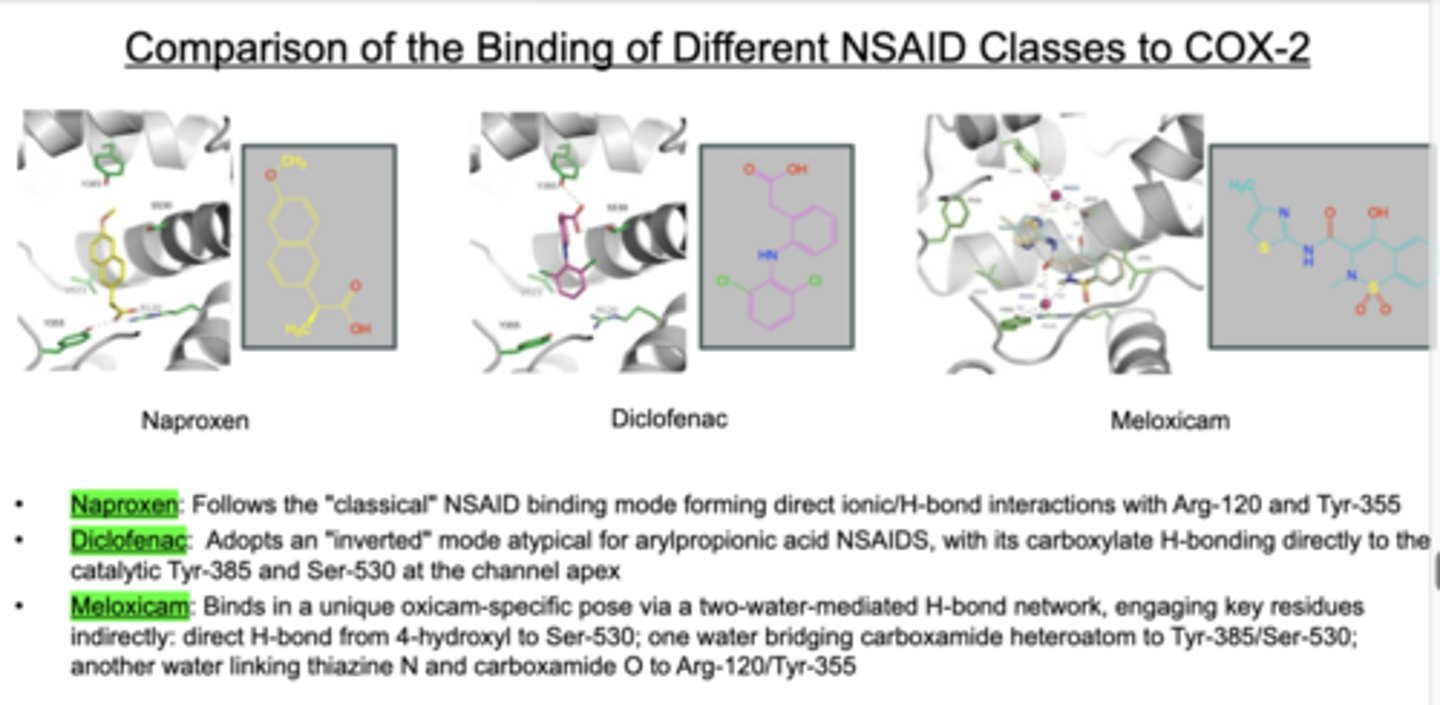
Meloxicam
-Multiple oral formulations for chronic treatment of osteoarthritis and rheumatoid arthritis
-Often preferred for long-term NSAID therapy -> once daily dosing thought to improve compliance
MOA:
-Reversible Inhibition of cyclooxygenase (COX) enzymes -> COX-2 ”preferring”
-Analgesic potency ~100x aspirin, ibuprofen
Warnings:
-BBW: CI post-coronary bypass, increased risk of CV thrombotic events
-analgous to ibuprofen (GI, CV, and renal toxicity monitoring needed)
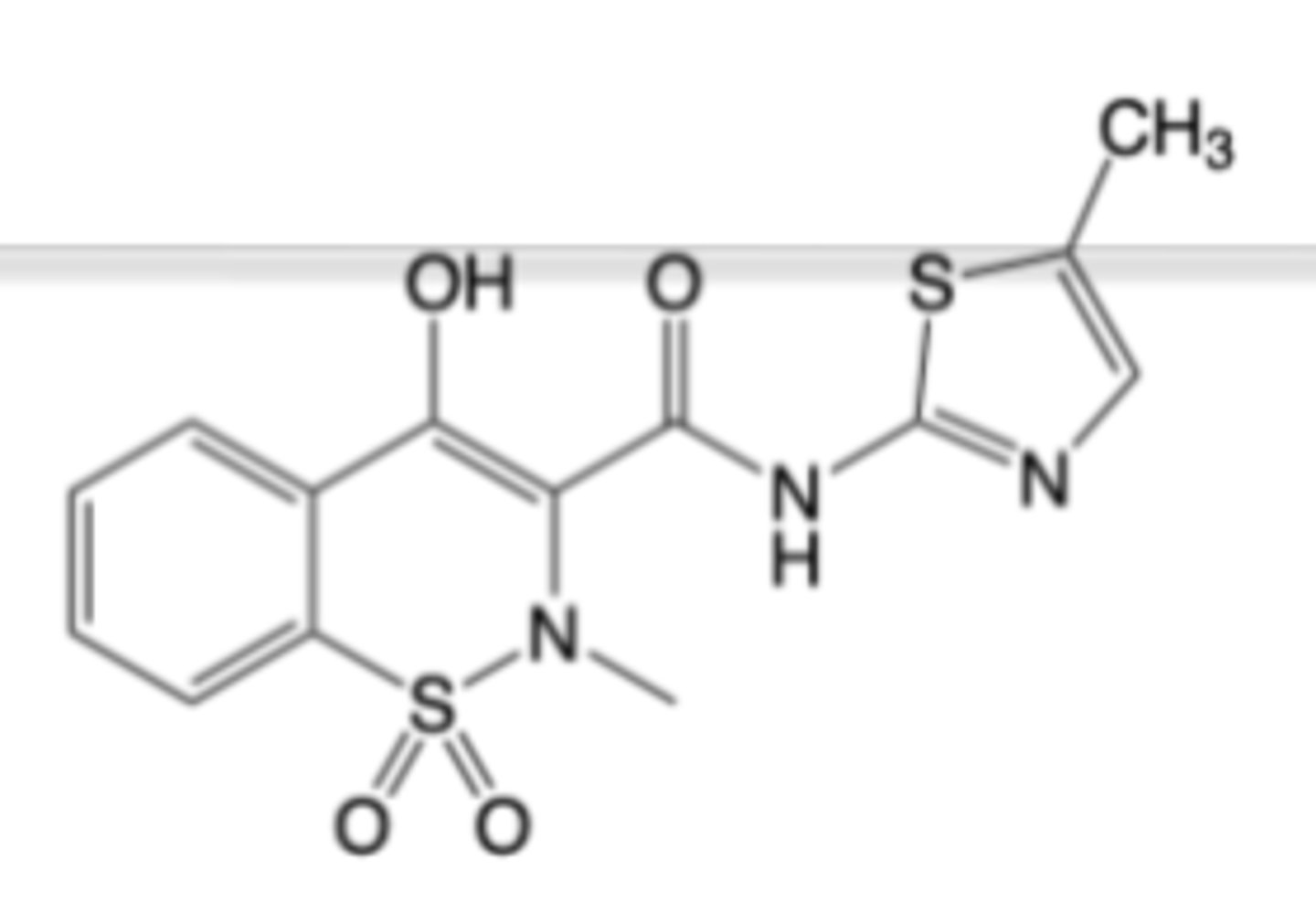
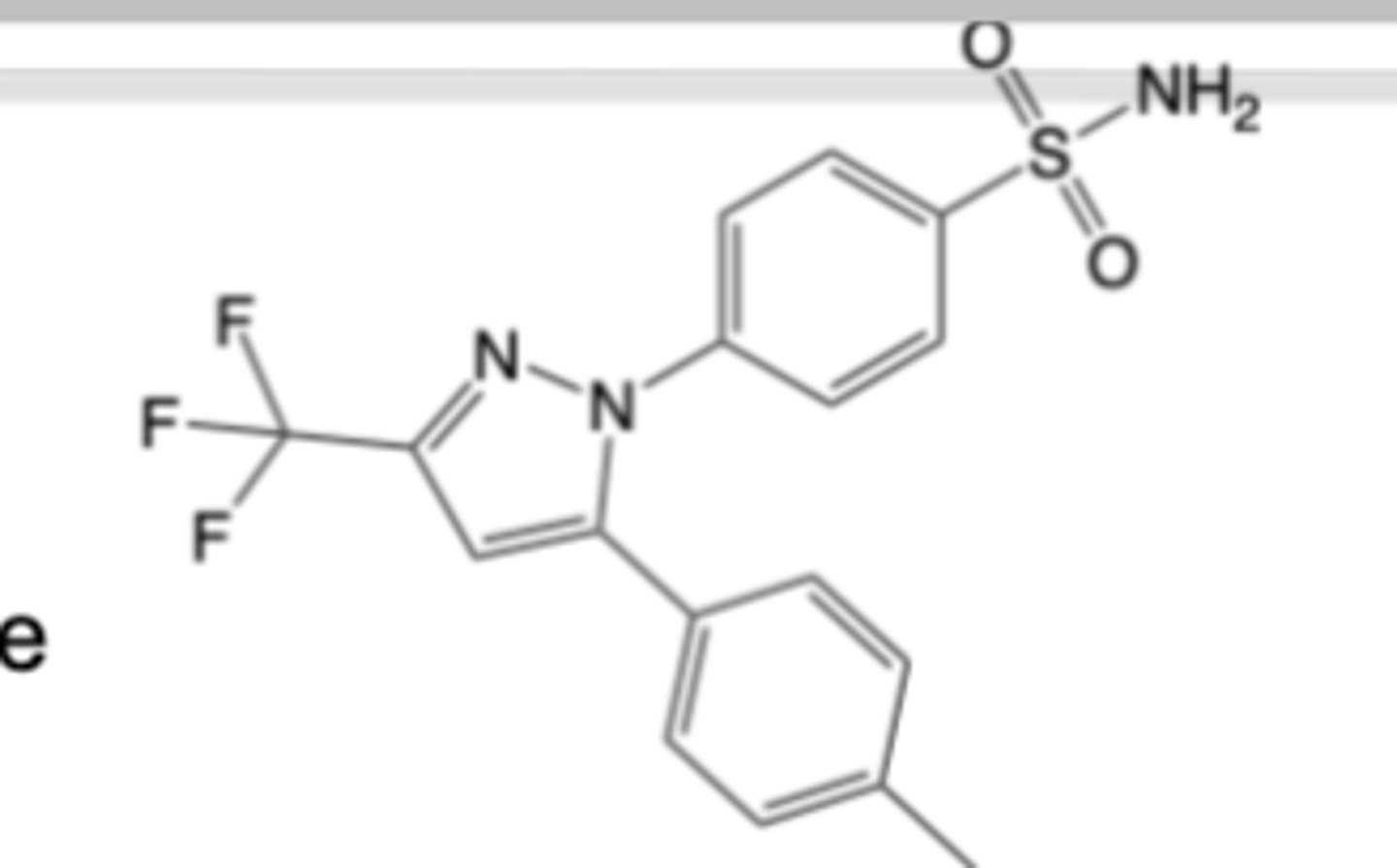
Celecoxib
-Oral formulation for treating acute pain, osteoarthritis, rheumatoid arthritis, ankylosing spondylitis and menstrual pain
MOA:
-Moderately selective reversible inhibition of cyclooxygenase-2 -> ~30x selectivity for COX-2 over COX-1
-Analgesic potency: ~10x aspirin and ibuprofen for arthritis pain relief
Warnings:
-BBW: CI post-coronary bypass, increased risk of CV thrombotic events
-analgous to ibuprofen (GI, CV, and renal toxicity monitoring needed)
DMARDs treat rheumatoid arthritis (RA) by dampening innate & adaptive immune activation, blocking key cytokine signaling, and altering synovial cell behavior reducing joint inflammation and destructive remodeling
Disease-Modifying Anti-Rheumatic Drugs (DMARDs)
- Methotrexate
- Leflunomide
- Sulfasalazine
- Hydroxychloroquine
First‑line therapy before or alongside biologic DMARDs and/or JAK inhibitors
Methotrexate is a first‑line drug for moderate to high activity RA and the most prescribed conventional synthetic DMARD in the U.S.
MTX is considered an "anchor drug" for RA and is commonly combined with other DMARDs
Conventional Synthetic DMARDs
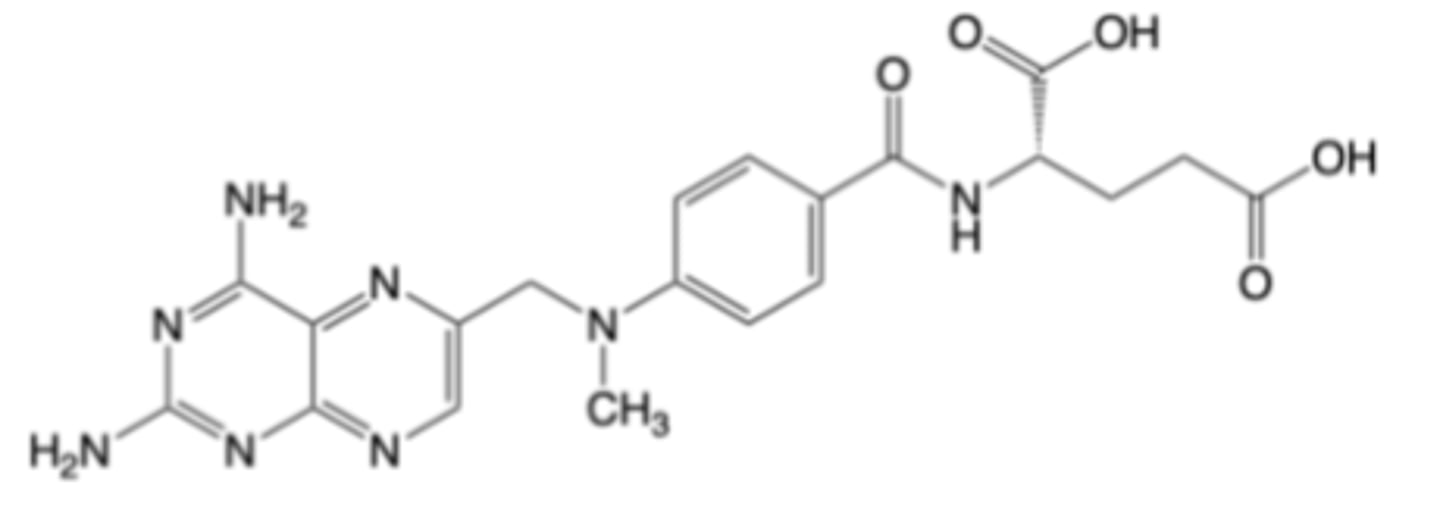
Methotrexate
-Oral formulation for the treatment of RA and other autoimmune disorders (e.g., severe psoriasis)
-Oral formulation for the treatment of acute lymphoblastic leukemia (ALL) and certain B cell malignancies
-once weekly
MOA:
-Inhibition of dihydrofolate reductase (DHFR) -> limits DNA synthesis and proliferation of activated T and B cells -> attenuates of adaptive immune responses in synovium
-Increases extracellular adenosine -> inhibits neutrophil adhesion & LTB4 synthesis, local IL‑1 production, and decreases levels of IL‑6/IL‑8 -> modulates expression of multiple synovial proteinase enzymes
BBW: embyo-fetal toxicity
BBW: serious adverse reactions, including death
Immunizations with live vaccines is not recommended while taking methotrexate
DDIs:
-NSAIDs can reduce MTX renal clearance and displace it from plasma proteins
-antimicorbials exhibit additive effects
-other DMARDs increase toxicity
MTX enters cells via the reduced folate carrier (RFC) and the folate receptor (FR)
-FR is upregulated with increased metabolic activity
MTX is a substrate for the ATP Binding Cassette (ABC) efflux transporter
– Conversion of MTX to MTX-PG by folyl-polyglutamyl synthetase
(FPGS) prevents efflux
MTX-PG inhibits several intracellular enzymes
-Inhibition of TYMS
-Inhibition of dihydrofolate reductase (DHFR)
-Inhibition of ATIC (increases adenosine)
Methotrexate Mechanisms of Action
Leflunomide
-Oral formulation for the treatment of RA
MOA:
-Inhibition of dihydroorotate dehydrogenase (DHODH) -> blocks pyrimidine synthesis and restricts proliferation of activated lymphocytes that rely on the DHODH pathway, reducing auto-antibody and cytokine production
Warnings:
-BBW: Embryo-fetal toxicity, risk of severe hepatotoxicity
-Multiple other warnings (adverse hematologic & infection risks, pulmonary & dermatologic toxicity, can cause peripheral neuropathy and hypertension)
–DDIs with agents that can be hepatotoxicity (e.g., statins) or provide additive immunosuppression
Sulfasalazine
-Oral formulation for the treatment of mild to moderate ulcerative colitis
-Not a first-line agent for RA, but is combined with MTX to treat RA
MOA:
-Metabolized in the small intestine to Sulfapyridine (SP) and 5-aminosalicylic acid (5-ASA)
-Sulfapyridine increases adenosine, suppresses NF-κB activation, reduces pro-inflammatory cytokines (e.g., TNF)
-5-ASA inhibits prostaglandin synthesis
Warnings:
-No pregnancy impairment
-Warnings for hepatic, renal, and hematologic toxicity, including risks of death from hypersensitivity reactions
-DDIs analogous to salicylates and with drugs that provide additive immunosuppression
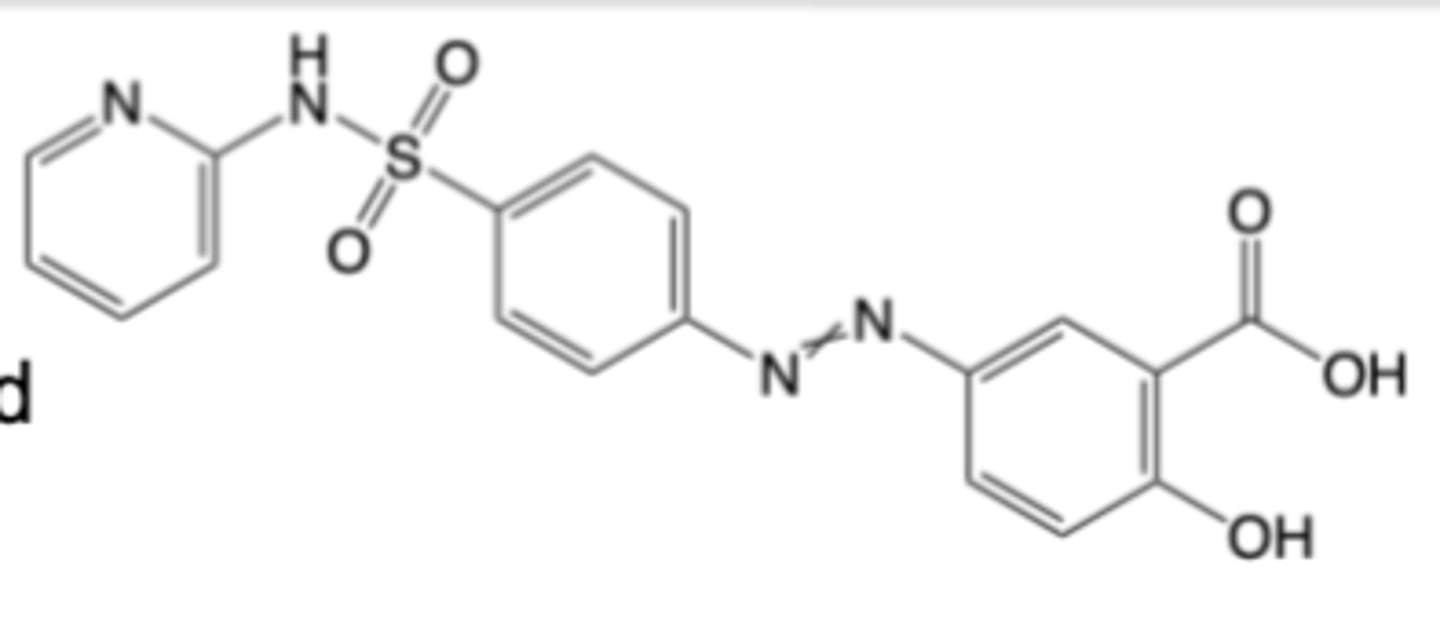
Hydroxychloroquine
-Oral formulation of hydrochloride for us as an anti-malarial and anti-rheumatic (for RA, lupus)
-Not a first-line agent for RA, but is combined with MTX to treat RA
-2x daily, 40-50 day t1/2
MOA: Increases pH in endosomes/lysosomes (blunts T Cell activation), inhibits Toll-like receptors (decreases cytokines)
Warnings:
-Warnings for potentially inducing life-threatening cardiomyopathy and ventricular arrythmias, retinal damage, renal toxicity, hematologic toxicity, serious skin reactions and worsening of psoriasis and porphyria
-DDIs with anti-arryhtmia drugs
-can increase blood levels of narrow TI drugs
-DDIs with CYP inhibitors
Tofacitinib, Upadacitinib, Baricitinib
JAK inhibition has been demonstrated to:
-Suppress T-cell differentiation and proliferation reducing autoantibody production and osteoclast activation
-Block B-cell signaling impairing plasma cell survival and autoantibody persistence
-Inhibit fibroblast-like synoviocytes invasiveness, MMP/IL-6 release, and
chemotaxis toward macrophages, curbing pannus invasion
-Decrease TNF and IL-1 output by limits macrophage polarization and NLRP3
inflammasome activity
For RA, all three (3) JAK inhibitors have higher or non-inferior efficacy vs. methotrexate or TNF blockers (patient population dependent) albeit with trade offs regarding long-term safety
not first line RA agents ->used if methotrexate and/or TNF blockers are inadequate)
Targeted Synthetic DMARDs: JAK Inhibitors
Tofacitinib
-Uses: RA,Psoriatic Arthritis, Ulcerative Colitis
-MOA: JAK1/JAK3 inhibition
-DDIs with CYP inducers or inhibitors
Warnings (all JAK inhibitors) compared to TNF blocekrs :
-Increased risk of serious bacterial, fungal, viral and opportunistic infections
-Higher rate of all-cause mortality
-Higher rate of MACE (e.g., heart attack, stroke)
-Higher rate of malignancies (e.g., lymphoma, lung cancer)
-Higher rate of thrombosis (e.g., pulmonary embolism, arterial or venous thrombosis)
not first line RA agents ->used if methotrexate and/or TNF blockers are inadequate)
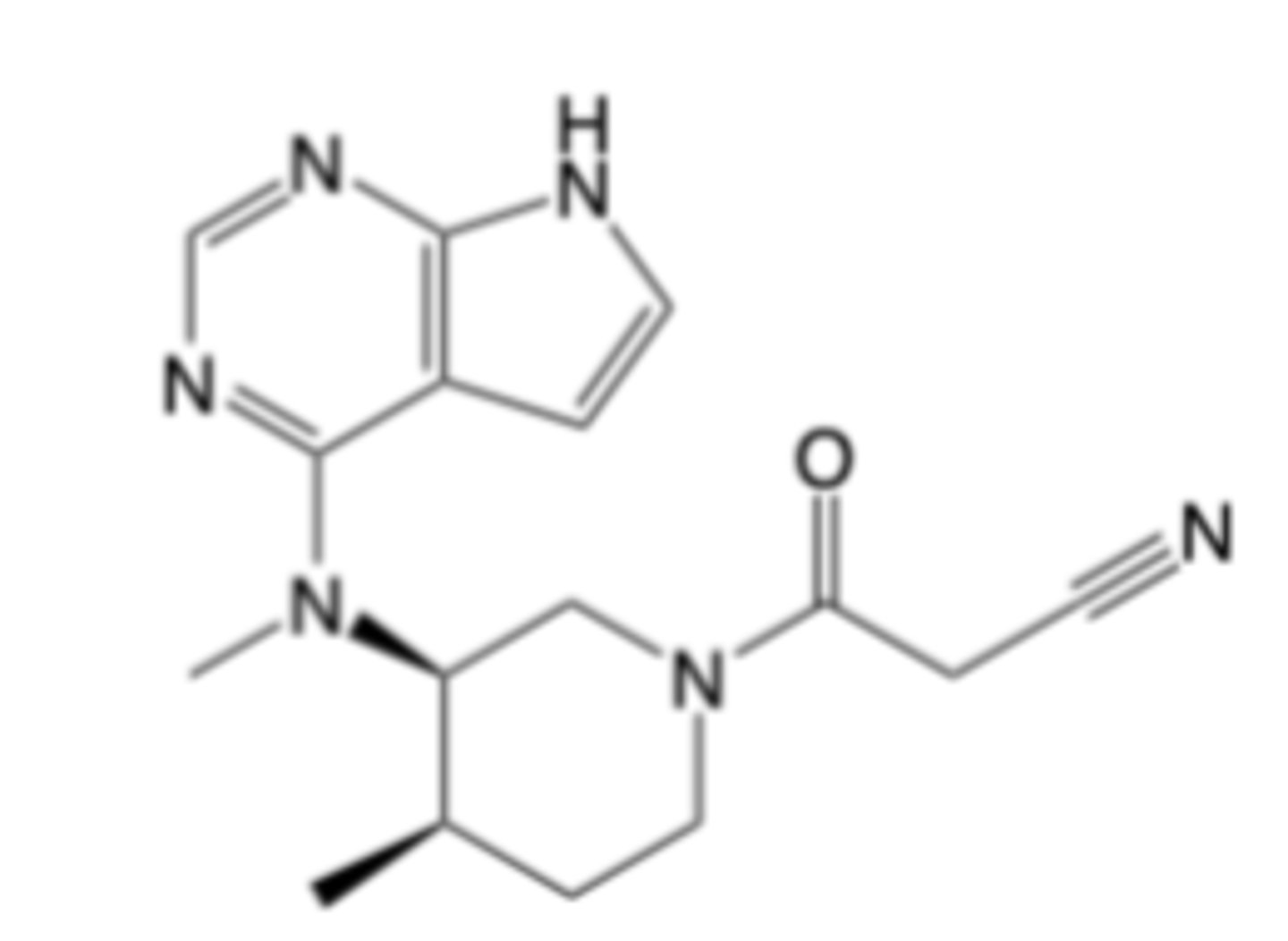
Upadacitinib
-Rheumatoid Arthritis
-Psoriatic Arthritis, Ulcerative Colitis, Atopic Dermatitis, Crohn’s Disease
-MOA: JAK1 inhibition
-DDIs with CYP inducers or inhibitors
Warnings (all JAK inhibitors) compared to TNF blocekrs :
-Increased risk of serious bacterial, fungal, viral and opportunistic infections
-Higher rate of all-cause mortality
-Higher rate of MACE (e.g., heart attack, stroke)
-Higher rate of malignancies (e.g., lymphoma, lung cancer)
-Higher rate of thrombosis (e.g., pulmonary embolism, arterial or venous thrombosis)
not first line RA agents ->used if methotrexate and/or TNF blockers are inadequate)
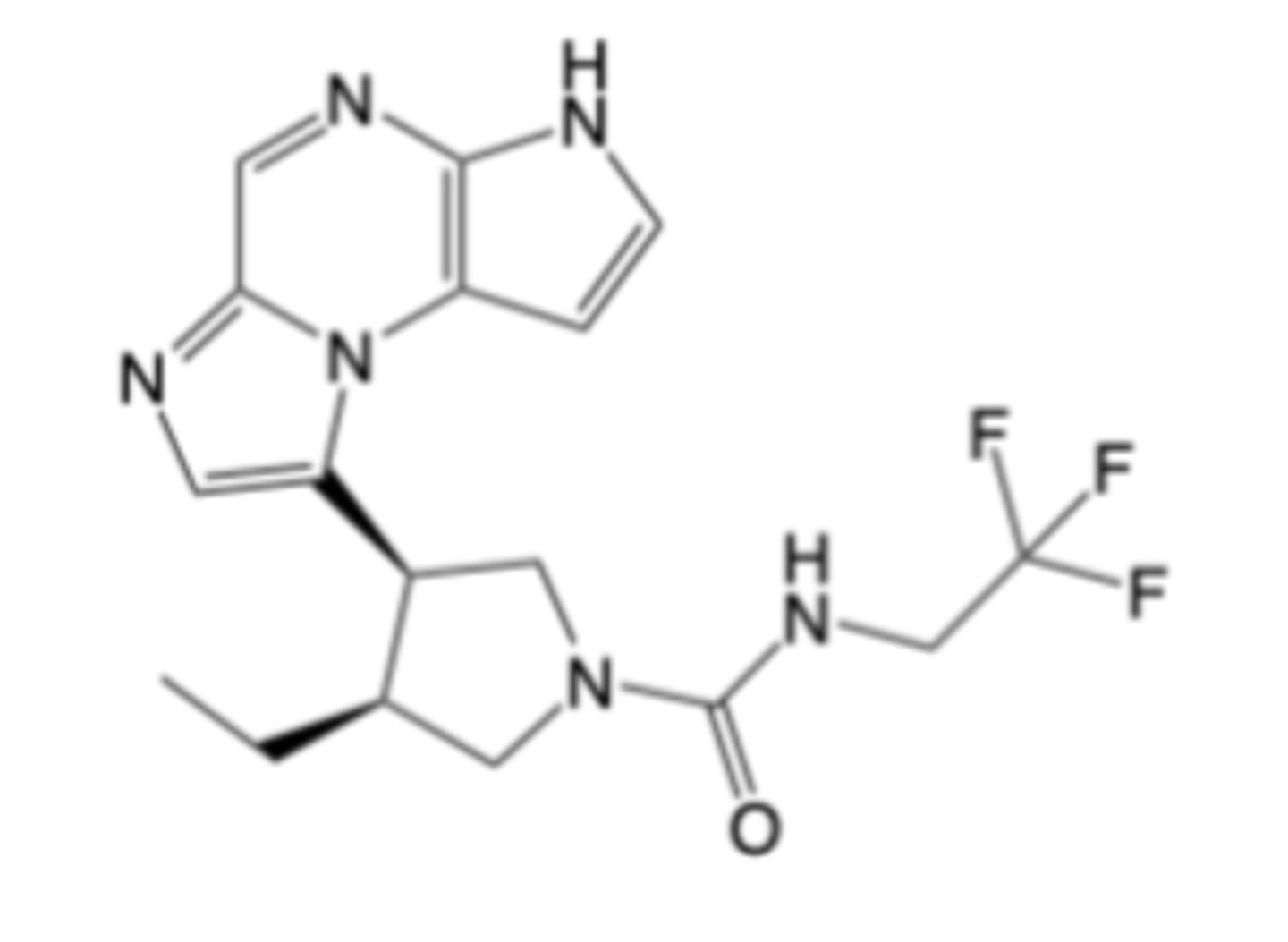
Baricitinib
-Use: RA
-MOA: JAK1/JAK2 inhibition
-Minimal DDIs
Warnings (all JAK inhibitors) compared to TNF blocekrs :
-Increased risk of serious bacterial, fungal, viral and opportunistic infections
-Higher rate of all-cause mortality
-Higher rate of MACE (e.g., heart attack, stroke)
-Higher rate of malignancies (e.g., lymphoma, lung cancer)
-Higher rate of thrombosis (e.g., pulmonary embolism, arterial or venous thrombosis)
not first line RA agents ->used if methotrexate and/or TNF blockers are inadequate)
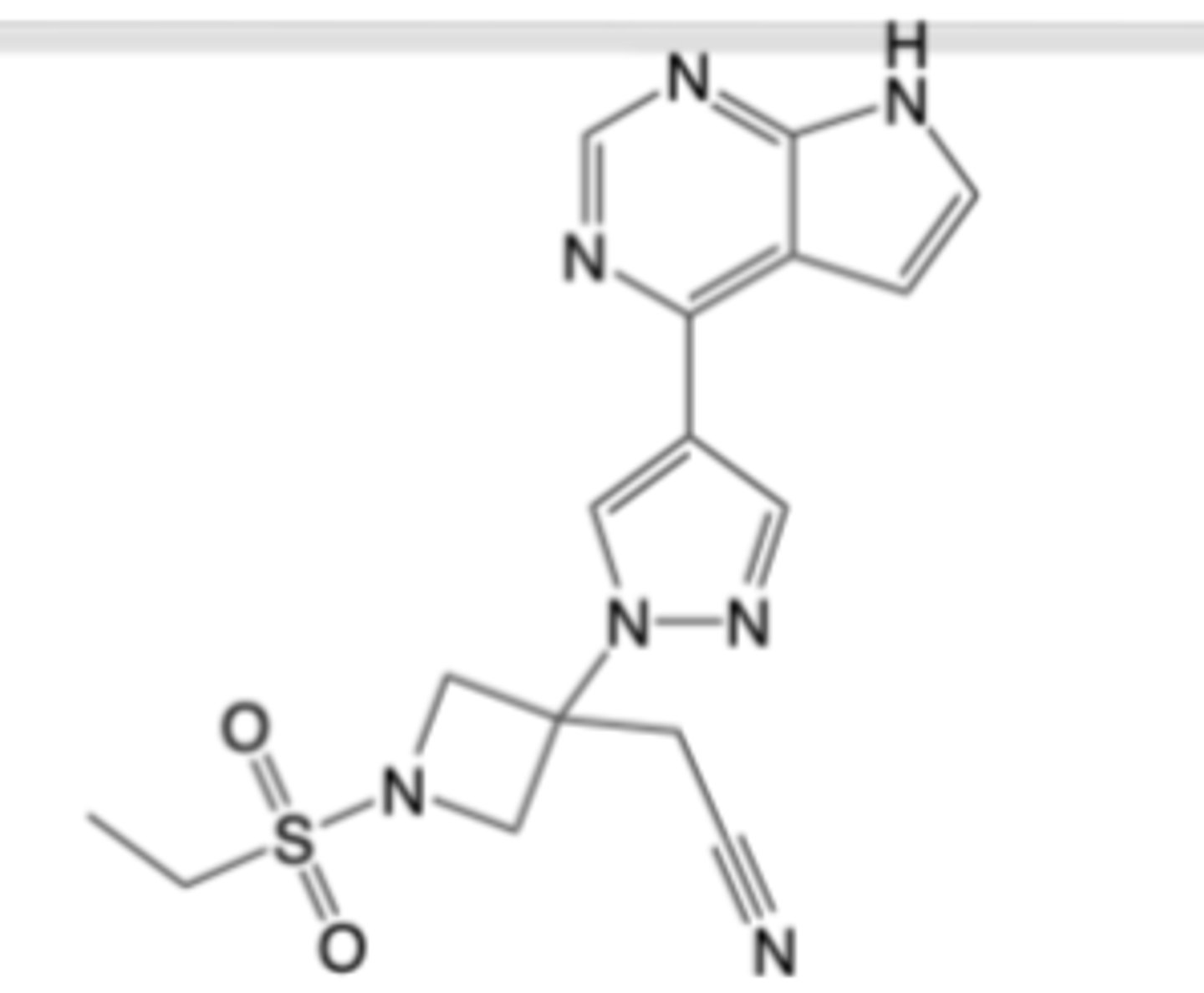
Infliximab
Adalimumab
Golimumab
Etanercept
Certolizumab pegol
neutralize the actions of both mTNF and sTNF
TNF inhibition:
- Improves the symptoms & structural outcomes in RA -> synergistic with methotrexate
- Is associated with increased risk for serious & opportunistic infections (e.g., TB)
- May enhance adverse cardiovascular risk, does enhance malignancy risk
there does not appear to be meaningful differences in clinical efficacy & safety across the 5 drugs
Biologic DMARDs: Tumor-necrosis factor (TNF) inhibitors
-TNF inhibitor (Drugs in this class are generally approved the treatment of rheumatoid arthritis (RA), psoriatic arthritis, ankylosing spondylitis)
-Additional indications Crohn’s disease, Ulcerative colitis
-IV infusion every 8 weeks (all others subQ)
Methotrexate co-administraiton: Required (in drug label)
Class warnings:
-BBWs: Risk of serious infections (including tuberculosis reactivation), risk of malignancy (lymphomas)
-No live vaccine admin, potential HBV reactivation, can induce auto-immune hepatitis or Lupus-like syndrome can worsen heart failure
Warnings:
-Infusion reactions
-Anti-body mediated loss of response
-Acute CV AEs
Mouse/human chimeric monoclonal anti-TNF IgG1 antibody
Infliximab
-TNF inhibitor (Drugs in this class are generally approved the treatment of rheumatoid arthritis (RA), psoriatic arthritis, ankylosing spondylitis)
-Additional indications: Chron's, UC, IBD, multiple otehrs
Not required co-administration with methotrexate for RA (but recommended
Class warnings:
-BBWs: Risk of serious infections (including tuberculosis reactivation), risk of malignancy (lymphomas)
-No live vaccine admin, potential HBV reactivation, can induce auto-immune hepatitis or Lupus-like syndrome can worsen heart failure
Fully human monoclonal anti-TNF IgG1 antibody
Adalimumab
-TNF inhibitor (Drugs in this class are generally approved the treatment of rheumatoid arthritis (RA), psoriatic arthritis, ankylosing spondylitis)
-Additional indications: UC
Methotrexate co-administraiton: Required (in drug label)
Class warnings:
-BBWs: Risk of serious infections (including tuberculosis reactivation), risk of malignancy (lymphomas)
-No live vaccine admin, potential HBV reactivation, can induce auto-immune hepatitis or Lupus-like syndrome can worsen heart failure
Golimumab
-TNF inhibitor (Drugs in this class are generally approved the treatment of rheumatoid arthritis (RA), psoriatic arthritis, ankylosing spondylitis)
Not required co-administration with methotrexate for RA (but recommended
Class warnings:
-BBWs: Risk of serious infections (including tuberculosis reactivation), risk of malignancy (lymphomas)
-No live vaccine admin, potential HBV reactivation, can induce auto-immune hepatitis or Lupus-like syndrome can worsen heart failure
May have reduced malignancy risk
Human TNFR2/human IgG1 Fc region fusion protein
Additionally binds to and neutralizes: lymphotoxin-alpha3 (LT- alpha3)
Etanercept
-TNF inhibitor (Drugs in this class are generally approved the treatment of rheumatoid arthritis (RA), psoriatic arthritis, ankylosing spondylitis)
-additional indication: Chron's
Not required co-administration with methotrexate for RA (but recommended
Class warnings:
-BBWs: Risk of serious infections (including tuberculosis reactivation), risk of malignancy (lymphomas)
-No live vaccine admin, potential HBV reactivation, can induce auto-immune hepatitis or Lupus-like syndrome can worsen heart failure
may have reduced risk for use during pregnancy
No mTNF-cell cytotoxicity or apoptosis (requires human Fc)
PEGylated Fab fragment of human anti-TNF IgG1 antibody
Certolizumab
These biologic agents (that do not target TNF) are used second- or third-line drugs to treat RA
-These drugs interrupt cytokine action, cellular activation and adaptive immune pathways that drive synovitis, pannus formation, and joint destruction
- Like TNF inhibitors (TNFi), these drugs generally synergize with methotrexate
Anakinra: IL1-inhibitors (Inferior to TNFi)
Tocilizumab and Sarilumab: IL-6 Inhibitors (Comparable to TNFi)
Abatacept: T-Cell co-stimulation modulators (Comparable to TNFi)
Rituximab: B-cell depleterrs (Inferior to TNFi)
Biologic DMARDs: non-TNF Targeting Agents
Gout is a chronic disease caused by monosodium urate (MSU) crystal deposition -> usually in the joints of the lower limbs
Gout is traditionally considered to be a disorder of purine metabolism
Elevated serum uric acid is a major risk factor for MSU crystal
formation and the development of acute and chronic symptoms
Anti-inflammatory drugs (steroids, NSAIDs, colchicine) are widely
used to treat the symptoms of acute flare
– Colchicine is used specifically for gout
– Aspirin is contraindicated to treating acute gout -> competes with and
reduces uric acid secretion in the kidney
Drugs Used To Treat Gout
Colchicine
-Oral formulation for the treatment and prevention of gout flares
-NOT an analgesic medication
-NOT used to treat pain from other causes
MOA:
-Reduction of inflammation due to monosodium urate (MSU) deposition –> does NOT alter serum uric acid levels
-Disrupts microtubule‑dependent neutrophil functions -> reduces cellular response to MSU crystals
-Limits neutrophil influx into gouty joint(s) -> decreases local swelling & inflammation
-Dampens inflammasome‑driven cytokine (capsase-1, IL-1beta release -> reduces inflammation
Wanings:
-Narrow therapeutic index -> fatal overdoses can easily occur
-Toxicity arises from its binding to tubulin, which disrupts microtubule polymerization and impairs multiple cellular processes in rapidly dividing and/or motile cells
CI: renal or hepatic impairment
DDIs: cyp or pgp inhbitors
Usually mild AEs reversible upon lowering dose or pausing treatment (N/V, diarrhea)
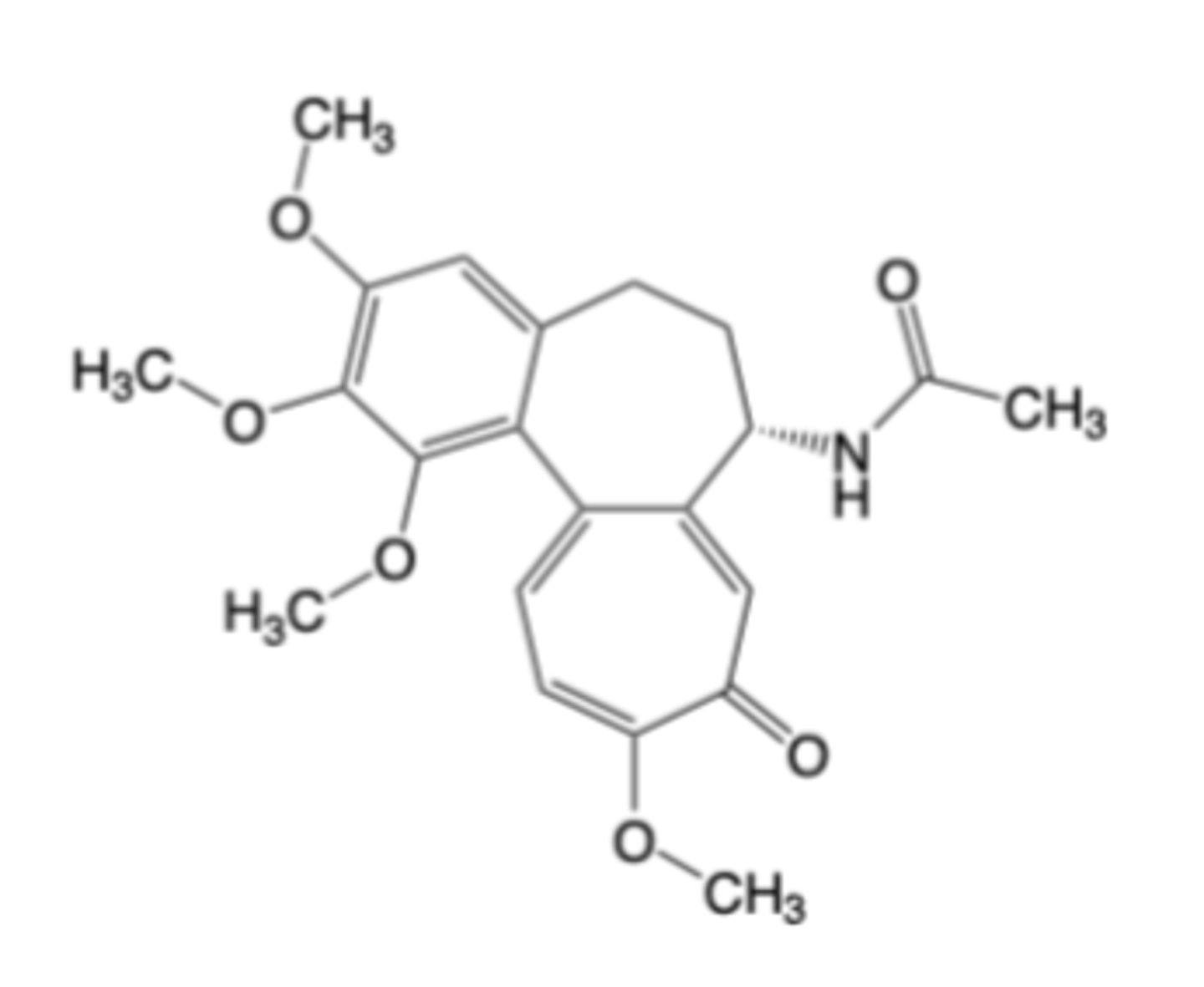
Production (Xanthine Oxidase) Inhibitors
• Allopurinol
• Februxostat
Excretion Enhancers
• Probenecid, Lesinurad
Metabolism Enhancer
• Pegliotase
Uric Acid Lowering Therapies for Gout
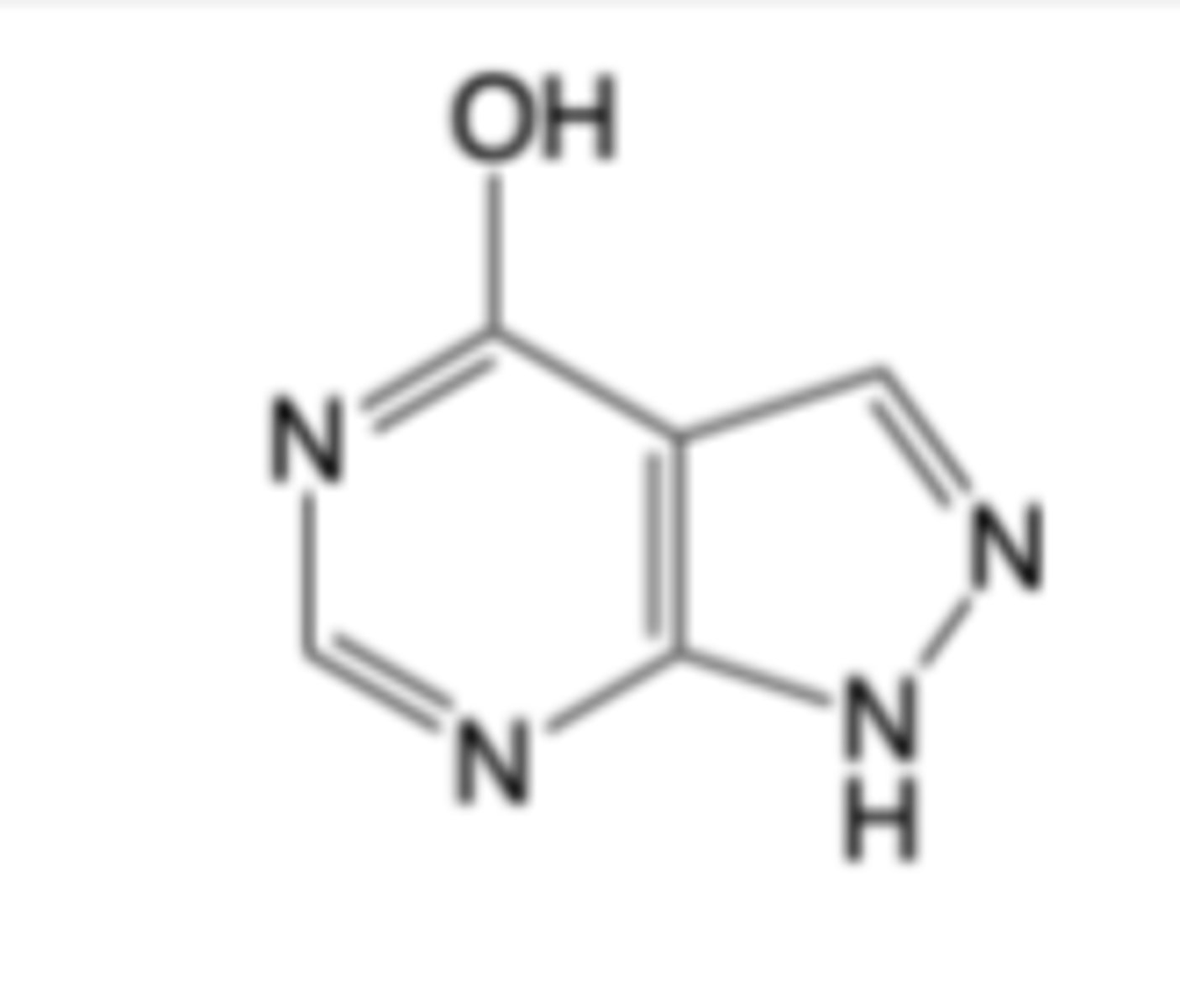
Allopurinol
-purine analog
-Oral formulation for the treatment of acute and chronic gout -> first line treatment
-Oral and intravenous formulations for reducing uric acid elevation associated with cancer therapy
MOA:
-Substrate for and competitive “suicide” inhibitor of xanthine oxidase
-Enzymatic conversion of allopurinol to oxypurinol de-activates xanthine oxidase
Warnings:
-Potentially fatal allopurinol hypersensitivity syndrome (AHS)
-induce acute gout flare
Febuxostat
-Oral formulation for the treatment of chronic gout in patients with inadequate response to or who are intolerant to allopurinol -> 2nd line treatment (urate lowering efficacy is comparable to that of allopurinol)
MOA: Reversible, non-competitive inhibitor of xanthine oxidase
Warnings:
-BBW: higher rates of CV death with disease
-fewer AEs vs allopurinol
-induce acute gout flare
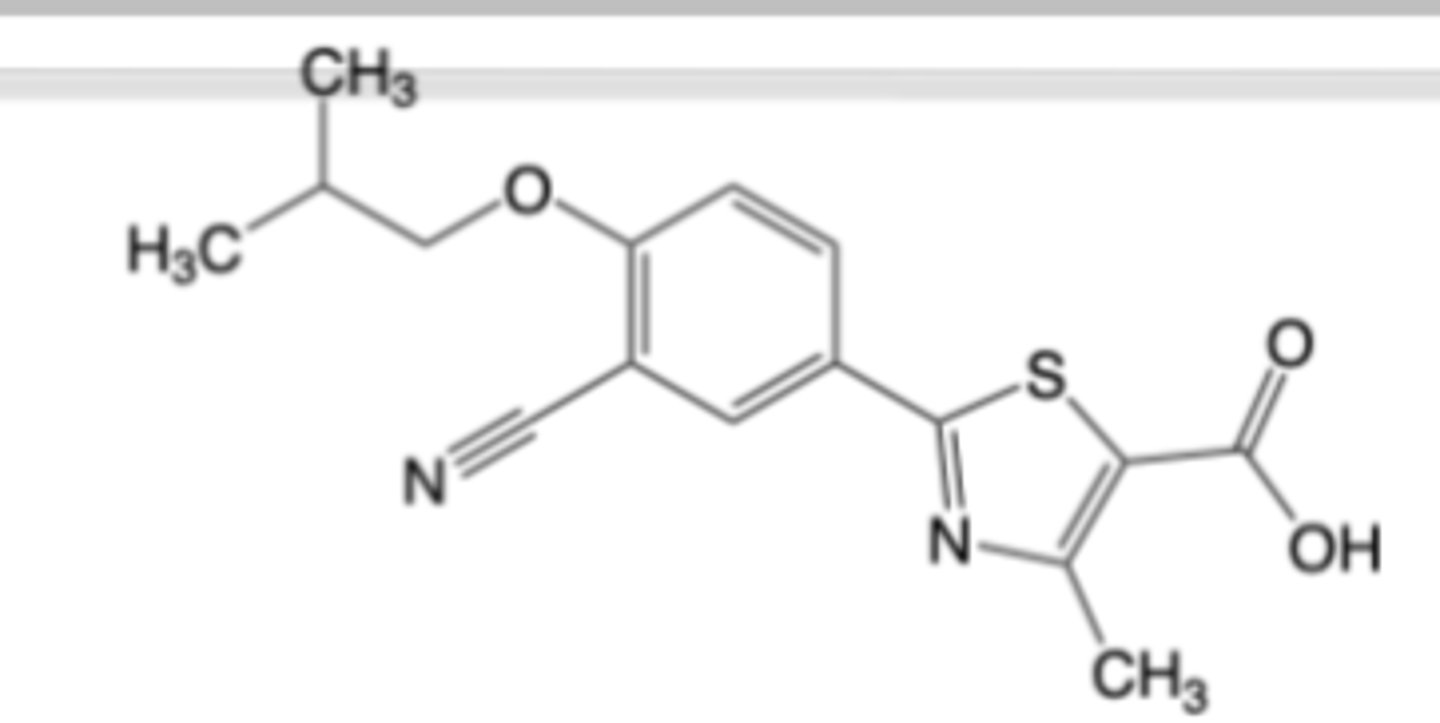
Probenecid
-Used for Chronic gout -> 2nd line drug (in combination with xanthine oxidase
inhibitor)
MOA: Uric acid transporter (URAT1) inhibitor -> less selective vs. lesinurad
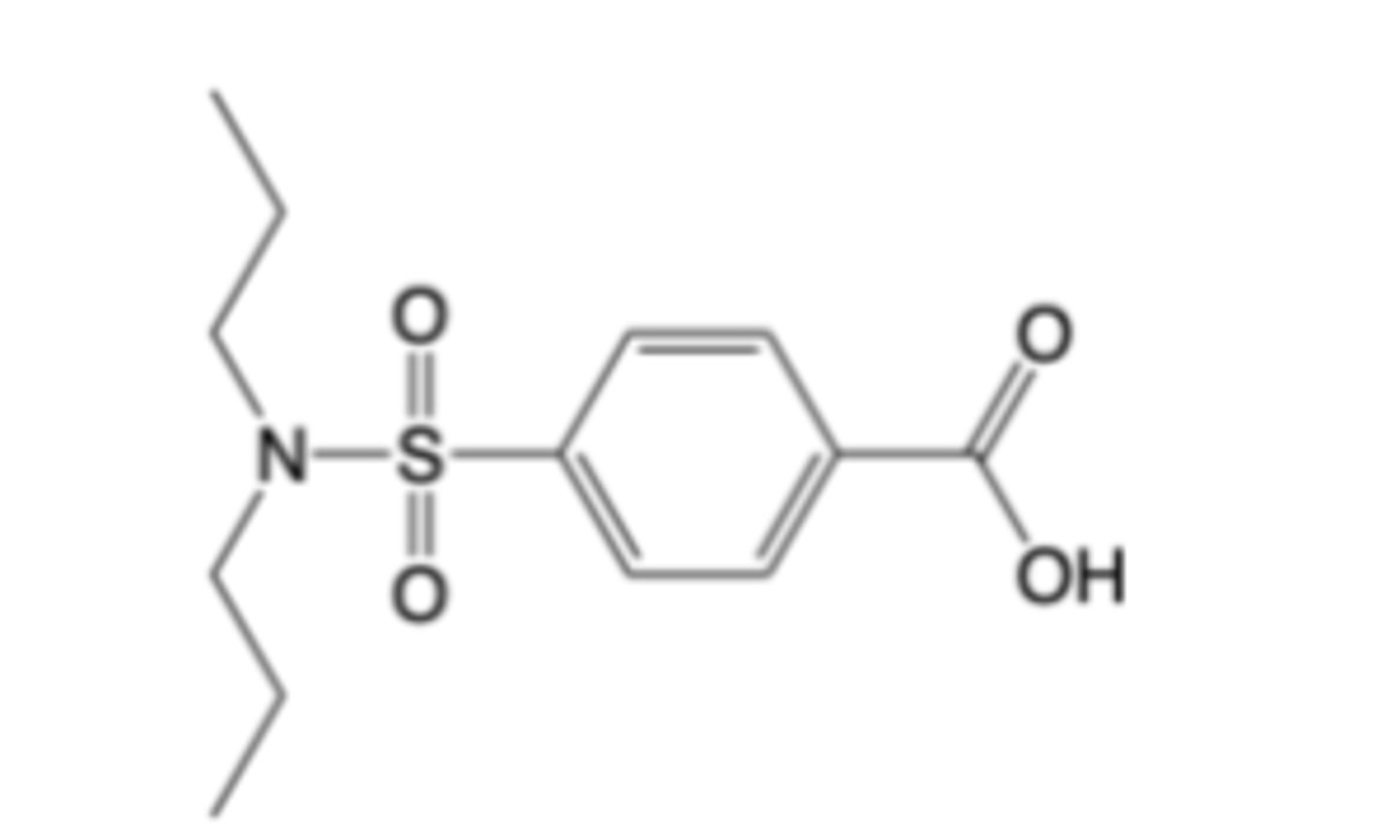
Lesinurad
--Used for Chronic gout -> 2nd line drug (in combination with xanthine oxidase
inhibitor)
-withdrawn
MOA: Uric acid transporter (URAT1) inhibitor -> more selective vs. probenecid
BBW for elevated risks of acute renal failure and major cardiac events
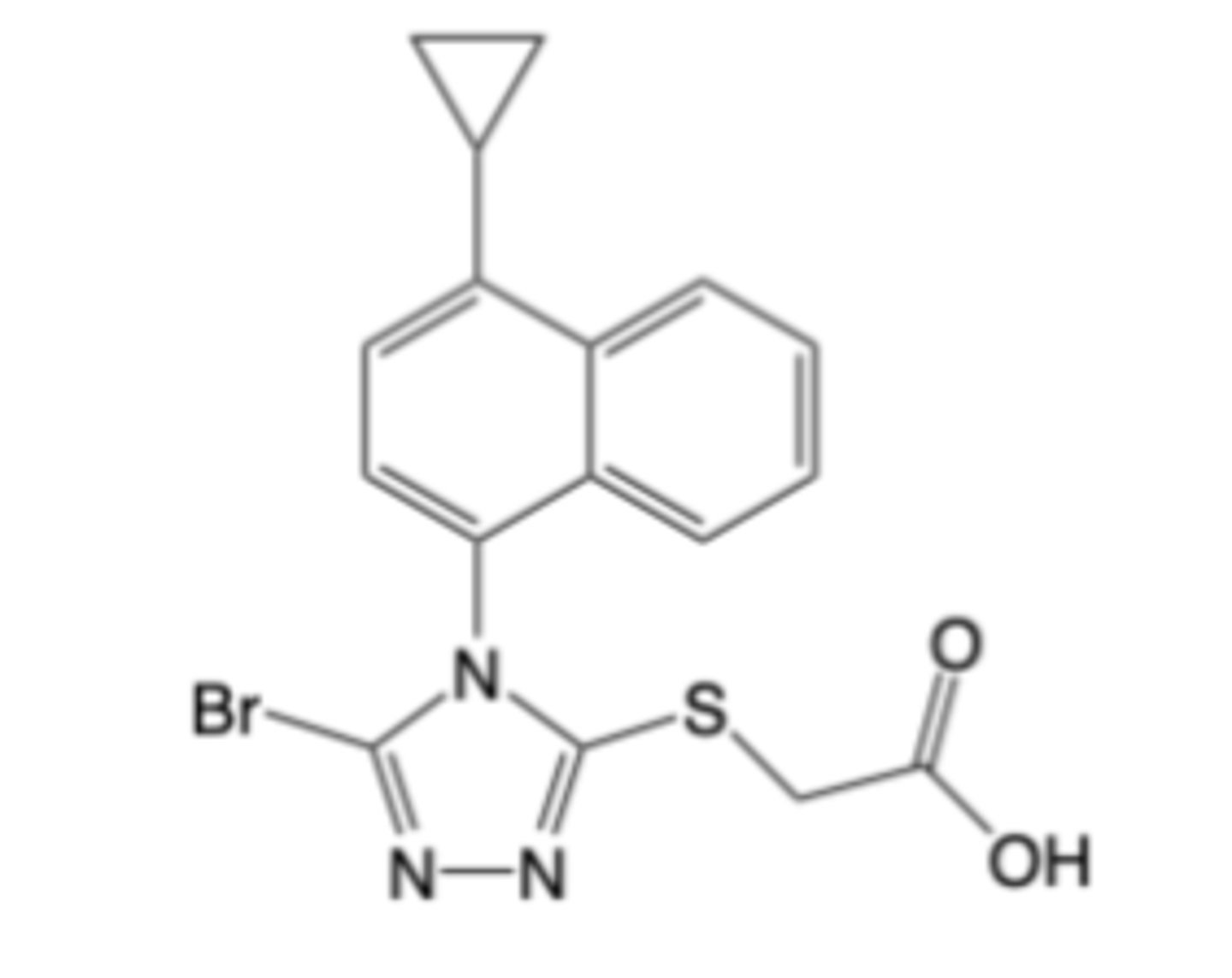
-Chronic gout -> 3rd line drug (in combination with methotrexate, when other therapies fail)
MOA: Uric acid metabolism enhancer (PEG uricase enzyme increases uric acid -> allantoin)
IV
BBW: anaphylaxis risk, infusion reaction risk, G6PH-deficiency associated hemolysis
Pegloticase