Liver & biliary system (+pancreas) pathology 4
1/67
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
68 Terms
What are the different sources of toxins?
Plants —> phytotoxins
Fungi —> mycotoxins
Inorganic chemicals —> drugs, metals
What are the different forms of a toxin that can cause an effect?
Uptake as toxic substances
Toxic after biotransformation
Toxic after metabolisation by GI microbes
What is the difference between obligate and idosyncratic toxin?
Obligate —> predictable toxic effects, dose dependent, direct or indirect effect on hepatocytes
E.g. paracetamol
Idiosyncratic —> only in some individuals, unpredictable, dose-indenpendent, usually immunologic (hypersens against hepatocytes)
What are the two modes of actions of obligate toxic substances?
Direct effect
oxidation of membrane lipids (cell, organelle, mitochondria)
Denaturing of structural proteins
Inhibition of enzymes
Indirect effect
Blocking of receptor or transport proteins
Modification of proteins
Binding to nuclear proteins, DNA, RNA or ribosomes
What is the most common entry of exogenous poisons to liver?
Via portal blood (intestine)
Because most ingested
What are the roles of hepatocytes in detoxification?
Biotransformation —> hydroxylation & conjugation of lipophilic substances → excretion via bile
Mainly centrolobular hepatocytes
Carrier systems for uptake of water-soluble substances from blood & secretion via bile
Activation of Kupffer cells (macropahges) via receptors (e.g. by endotoxins)
Release of free radicals & inflam mediators → damage to surrounding tissue
What are the phases of biotransformation?
Phase I —> oxidation (CytP450 oxidase enzyme)
Phase II —> conjugation of metabolic product with water
Damage occurs to hepatocytes during biotransformation
What zone dominates when damage occurs to hepatocytes during biotransformation?
Zone 3
Zone 1 much less common but dominates with direct acting toxicants such as metal satls
What gross findings are associated with acute toxic hepatosis?
ascites
oedema of gall bladder wall
petechial haemorrhages in serosa (DIC)
What histological findings are associted with acute toxic hepatosis?
usually centrolobular (periacinar) to massive necrosis, often with fatty / hydropic change of adjacent hepatocytes
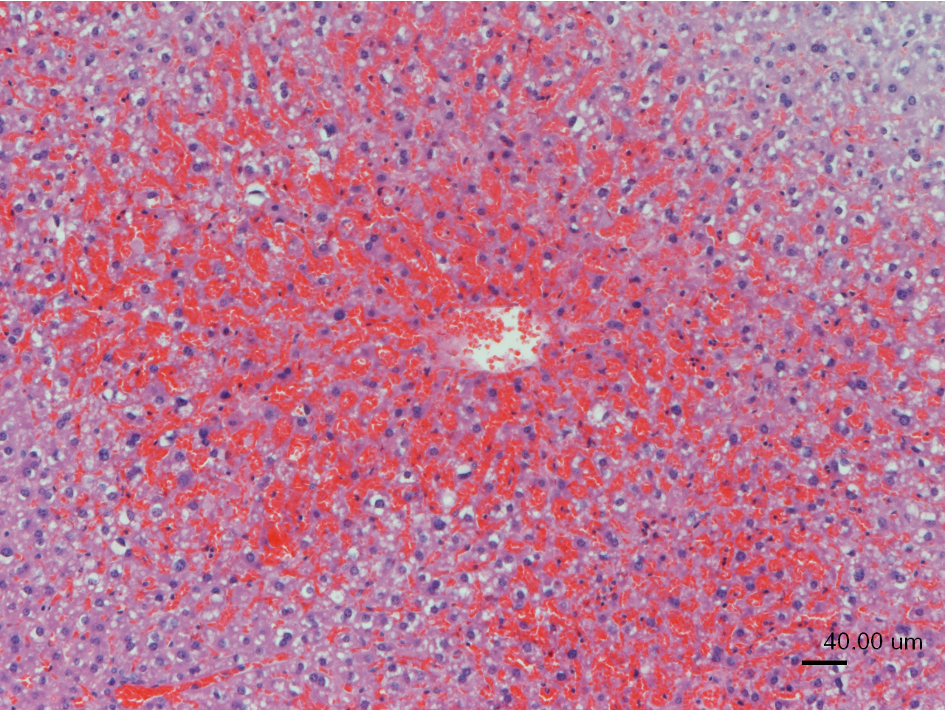
early stage —> degenerate/necrotic hepatocytes still orderly arranged
later stage —> dilated, blood-filled sinusoids due to loss of hepatocytes
What are the overall toxin effects on the liver?
One-time submassive necrosis —> regeneration of liver possible
One-time massive necrosis with destruction of the reticular framework → fibrosis (repair)
Chronic or recurrent toxin application —> chronic active hepatitis → cirrhosis
Some toxins → hepatic neoplasms
What effects do toxic substances have on the hepatocytes?
diffuse or zonal metabolic derangement → hydropic degeneration (swelling + vacuolation = reversible)
lipidosis or necrosis
List some examples of specific hepatotoxins
Copper toxicosis
Pyrrolizidine alkaloidosis (seneciosis, ragwort poisoning)
Aflatoxicosis
Blue-green algae poisoning
What does copper deficiency cause in sheep and cattle?
Sheep = swayback
Cattle = coat & pigment abnormalities
What can cause copper toxicity?
High intake
Reduced biliary excretions
Familial predisposition (bedlingtons, WHWT, North Ronaldsay sheep)
What are the causes of copper toxicosis in sheep?
Low tolerance of dietary coopper
Inadvertent feeding of high Cu diet
Pasture contam (slurry from other species e.g. pigs / poultry)
Nutritional imbalance —> Cu & Molybdenum/Sulfur form complexes and reduce uptake and increase Cu excretion
Consumption of hepatotoxic plants containing puyrrolizine alkaloids
Breed susceptibility —> Merino most resistant; Texels and North Ronaldsay most susceptible
Stress e.g. transport, movement, altered environ
How does copper toxicosis present?
Haemolytic crisis
Release of Cu from necrotic hepatocytes into blood → inapp, jaundice, haemoglobinuria, death
May be precipitated by ingestion of hepatotoxin / stress
What histological changes are associated with copper toxicosis?
liver necrosis (zone 3)
renal tubular Hb casts
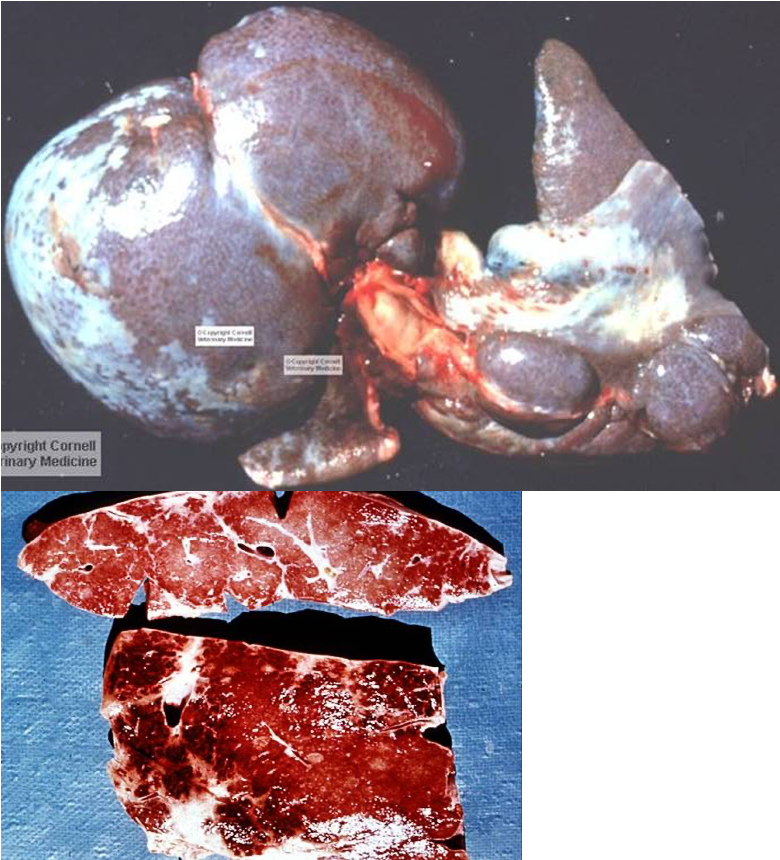
What has caused these lesions?

Copper toxicosis —> haemolytic crisis, acute chromoproteinaemic, nephrosis, liver degen, icterus
How is copper toxicosis diagnosed?
liver copper 500-1000µg/g
(normal = <400)
What dog breed is associated with copper toxicosis?
Bedlington terriers
Autosomal recessive mutation
Can lead to progressive hepatitis & ultimately cirrhosis

What is being shown in these images?
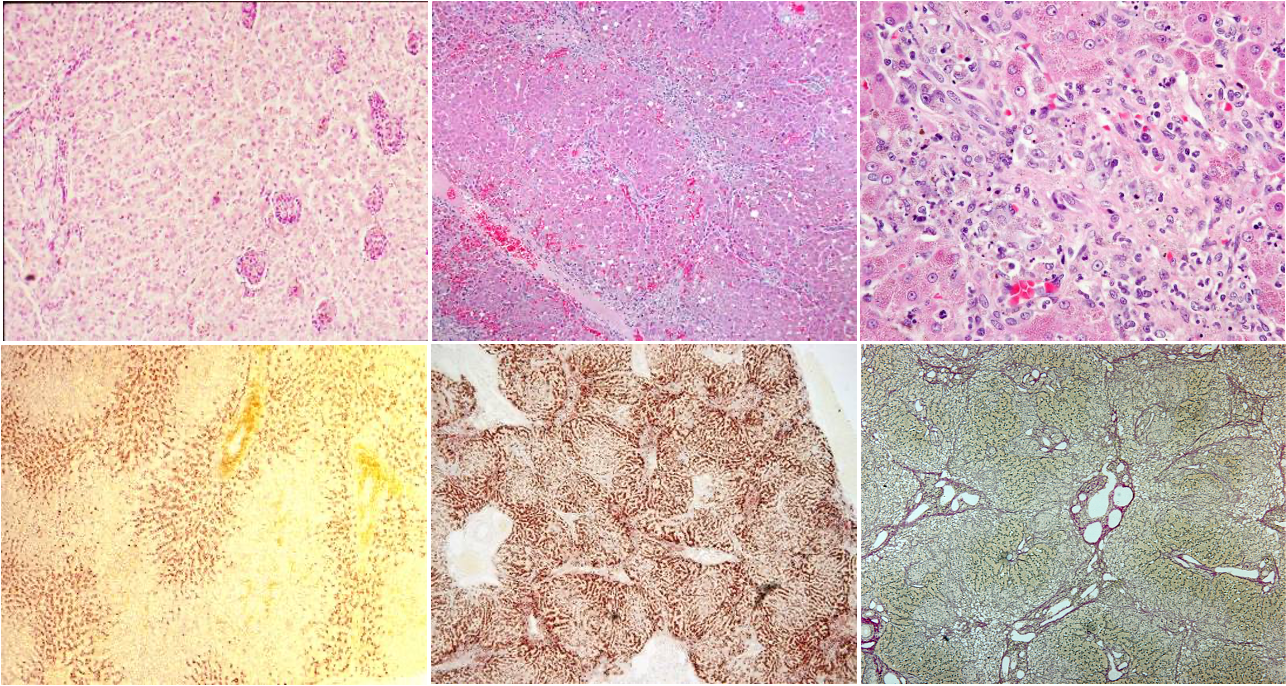
Familial copper toxicosis in dogs (progression)
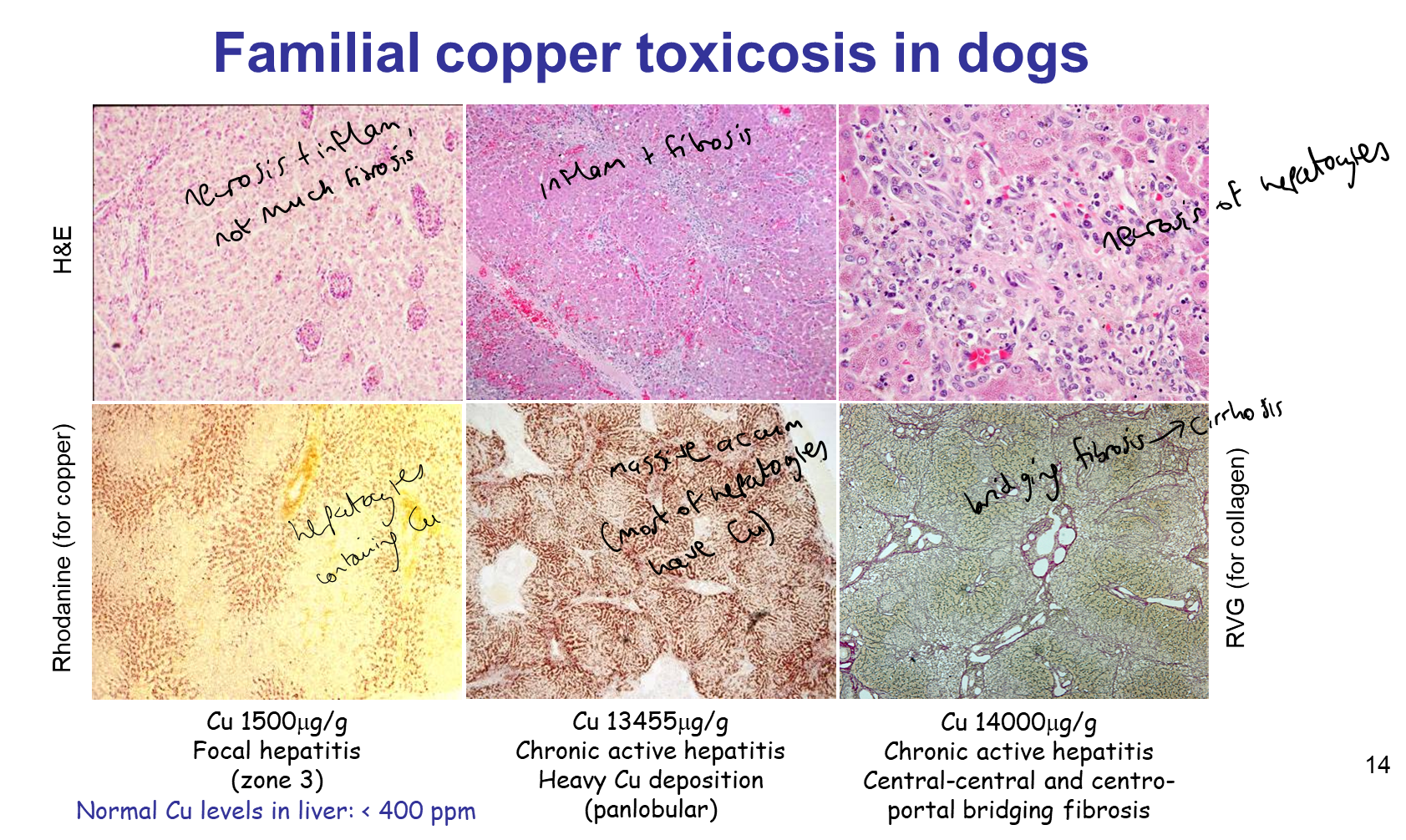
What can copper retention occur secondary to in dogs?
Chronic liver disease where there is failure / obstruction to bile flow as in:
Chronic active hepatitis in Dobermann Pinschers
Skye terrier hepatitis
(Not as strong association as Bedlington, predisposition?)
In what breed of sheep does familial copper toxicosis occur?
North Ronaldsay sheep
Sheep adapted to Cu deficient environment now have sensitivity to Cu

What is pyrrolizidine alkaloidosis?
Hepatotoxic plant toxins (pyrrolizidine alkaloids) found in different plant genera —> hepatotoxic substances
What plant genera can cause pyrrolizidine alkaloidosis?
Senecio
Ragwort —> normally unpalatable but bigger risk when contaminated other feed stuff e.g. dry in hay / silage
Crotolaria
Heliotropium
What toxic mechanisms causes pyrrolizidine alkaloidosis
Chronic hepatotoxicity
Alkaloids themselves not toxic —> undergoes biotransformation to become toxic:
Cytochrome P450 produce toxic pyrrolic esters —> bind to DNA & RNA —> stop cell & protein replication
What animals are most sensitive to Pyrrolizidine alkaloidosis?
Pig (greatest)
Cattle, horse
Sheep (least sensitive)
What pathological findings are associated with Pyrrolizidine alkaloidosis?
hepatic cirrhosis, with:
single cell necrosis
megalocytes (INDICATIVE OF FINDING) —> regenerative attempt due to inhibition of mitosis but not protein syntehsis
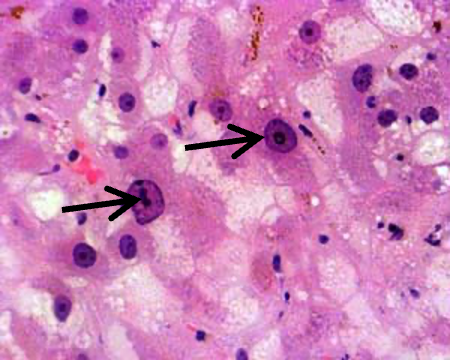
inflammatory infiltration, fibrosis [cattle]
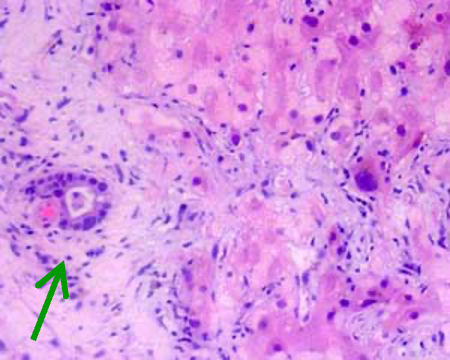
bile duct prolif
hepatoencephalopathy
What is being shown here?
Hepatic cirrhosis due to Pyrrolizidine alkaloidosis
What is aflatoxicosis due to?
Consumption of mouldy feedstuffs
Aspergillus flavus
A. parasiticus
Penicillum puberculum
What are the dangerous features of aflatoxins?
toxic
carcinogenic
teratogenic (effect on unborn fetuses)
mitosis inhibiting
immunosuppressive
What does aflatoxicosis cause?
Chronic hepatotoxicity
Liver changes very similar to seneciosis
Sensitivity —> dog, cat, pig, calf [cattle & horses less sensitive]
In what animals is blue-green algae poisoning seen?
cattle, sheep, horse, pig, dog
What causes blue-green algae poisoning?
Microcystis aeruginosa
How does blue-green algae poisoning present?
Acute hepatotoxicity
hepatotoxin (polypeptide) released when algae disintegrate (in water, in rumen or stomach)
Centrolobular to massive necrosis w/ haemorrhage
What are the two ducts of the pancreas?
Major pancreatic duct —> into duodenum at duodenal papilla
Minor pancreatic duct —> into duodenum at accessory duodenal papilla
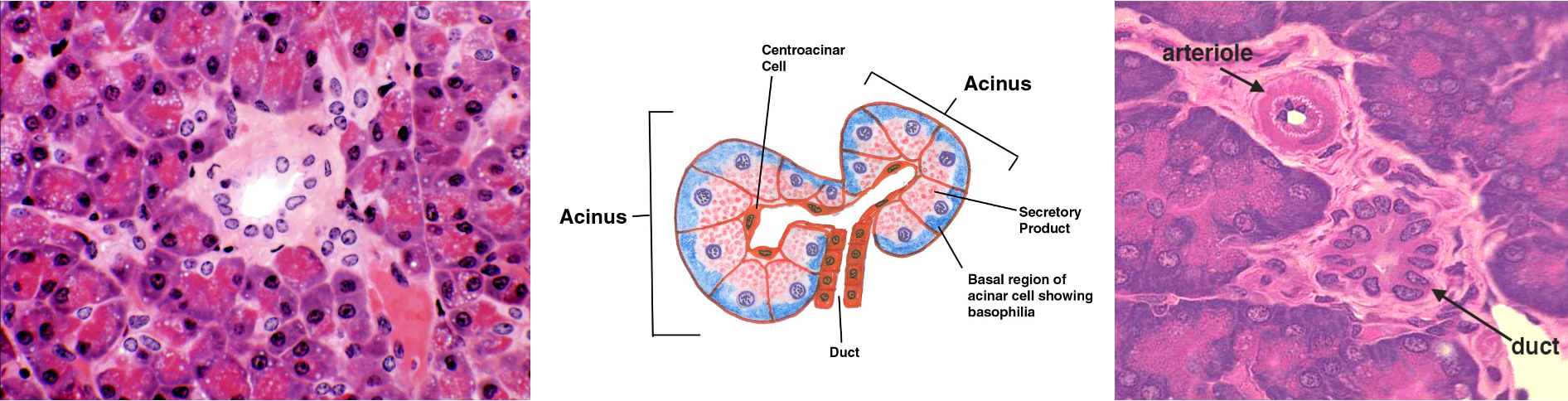
What are the histological features of the exocrine pancreas?
tubulo-acinar gland
secretion of digestive enzymes
regeneration —> by proliferation of differentiated cells
What are the anomalies of development of the pancreas?
Aplasia —> rare, incompatible with life, no exocrine or endocrine tissue
Hypoplasia —> sporadically in calves, clinically = EPI (exocrine pancreatic insuffiency)
Juvenile atrophy
In what age dogs does juvenile atrophy occur?
young dogs esp. german sheperds
How does juvenile atrophy present histologically?
exocrine pancreatic tissue almost absent
islets usually unaffected
What are the clinical signs of juvenile atrophy?
Chronic exocrine pancreatic insufficiency
What is being shown here?
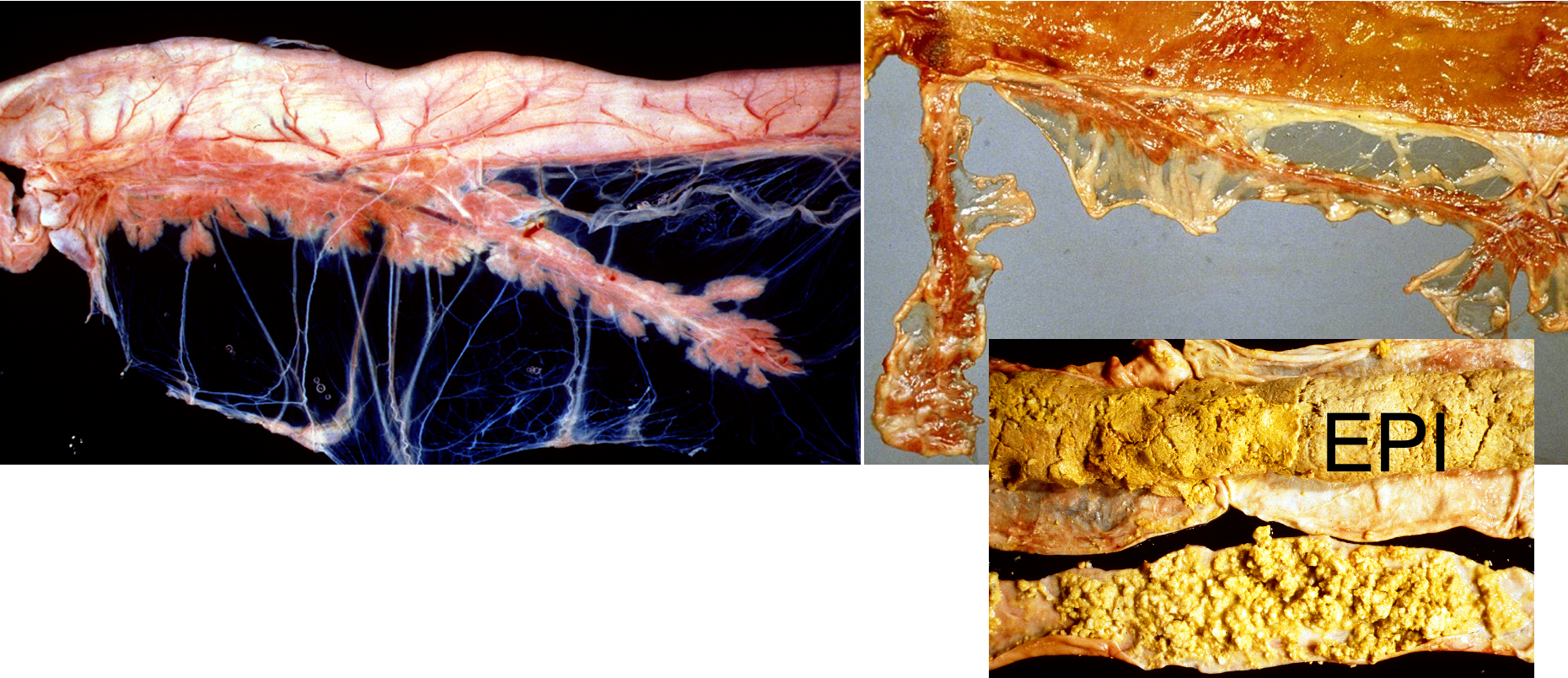
Juvenile atrophy
Severe diffuse atrophy, no fat in mesentery
Steatthorhea
Cannot digest food properly → high fat content in faeces (lipases not synthesised to breakdown & absorb fat ingested)
What ciruclatory disorder can be seen in the pancreas?
haemorrhage
List the reasons you may see pancreatic haemorrahge?
with coagulation disorders
a) infectious diseases [canine infectious hepatitis]
b) intoxications [dicumarol]
c) DIC
What are the differences between acute and chronic pancreatitis?
Acute pancreatitis
Necrotising
Painful
Chronic pancreatitis
Fibrosis
Insufficiency
What are the causes of pancreatitis?
Most idiopathic
Systemic infections (canine infectious hepatitis, FIP, FMD)
Migrating parasite (strongyles in horses)
Zinc poisoning (sheep, calves, dogs)
Trauma, obstruction of pancreatic duct
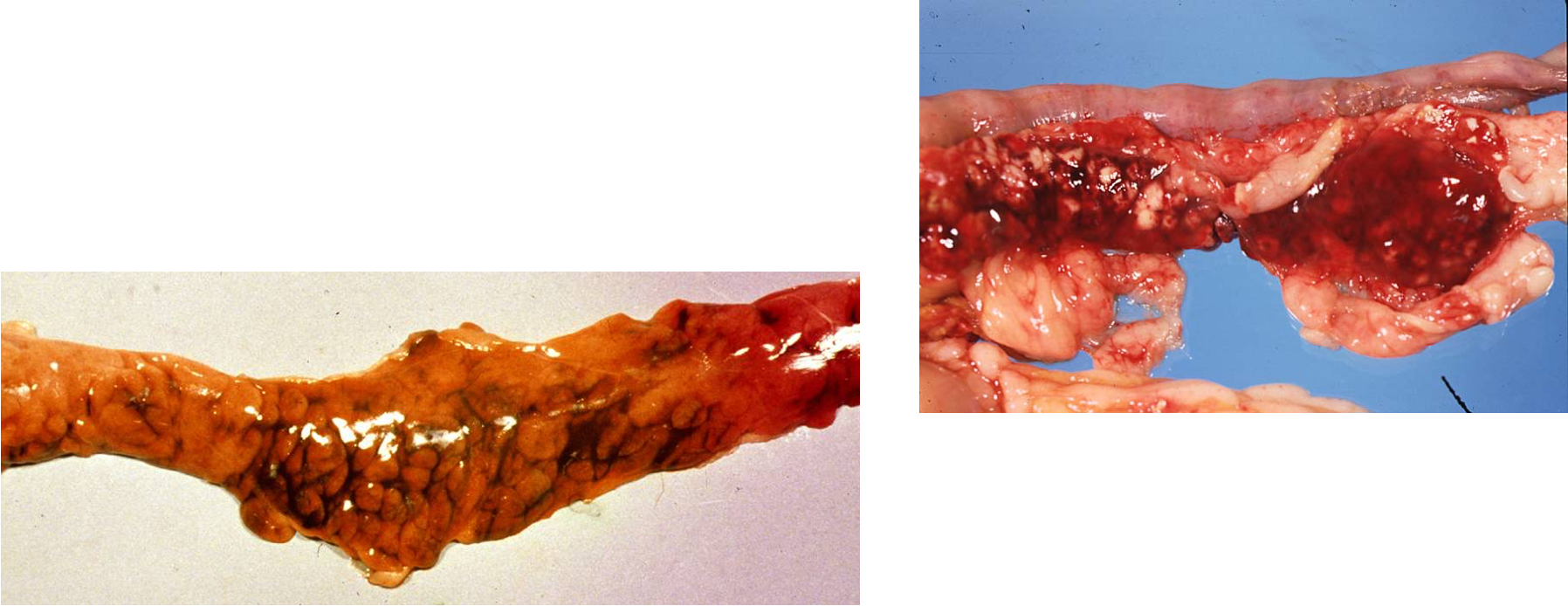
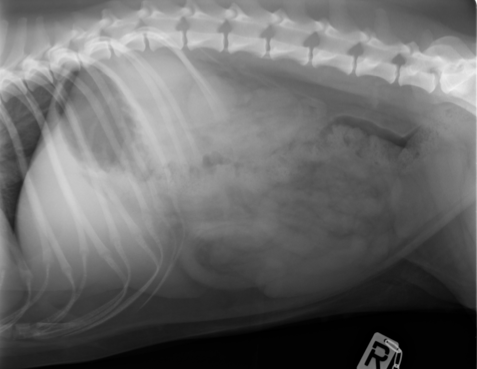
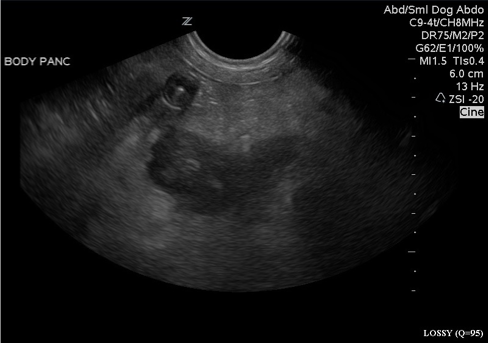
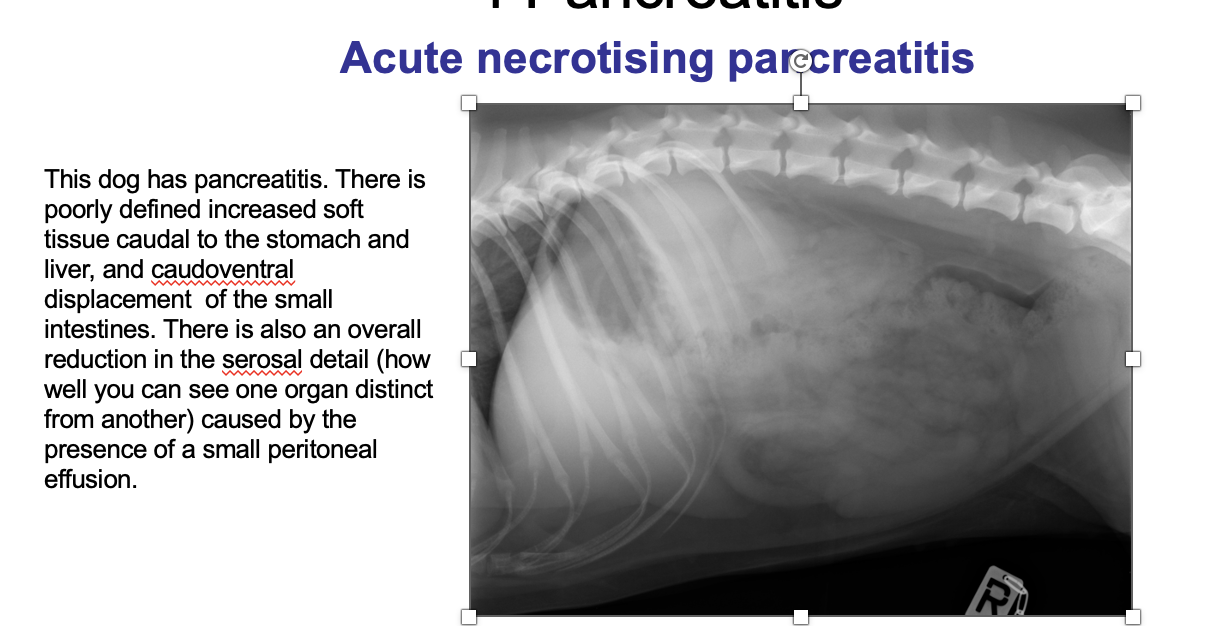
What is being shown here?
Acute necrotising pancreatitis
What is the physioloigcal cause of acute necrotising pancreatitis?
Release & activation of pancreatic enzymes within the pancreas
What lesions are associated with acute necrotising pancreatitis?
focal necrosis, haemorrhage, thrombosis, oedema
followed by inflammatory infiltration & fat necrosis
What are the clinical signs of acute necrotising pancreatitis?
suddenly decreased appetite
dullness, vomiting, diarrhoea, thirst
abdo pain
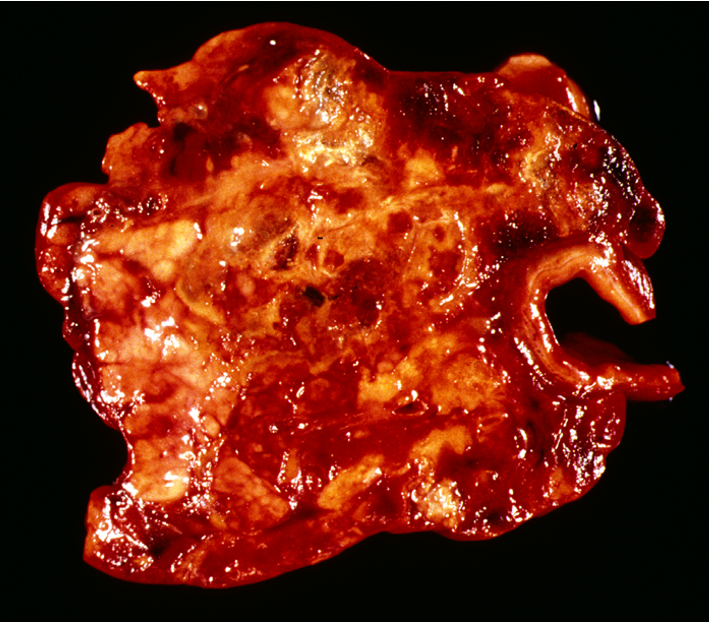
What are these lesions associated with?
Acute necrotising pancreatitis
Necrotising pancreatitis w/ fat necrosis
Reddened, haemorrhage
Necrosis of adjacent tissue (adjacent to stomach) = R image
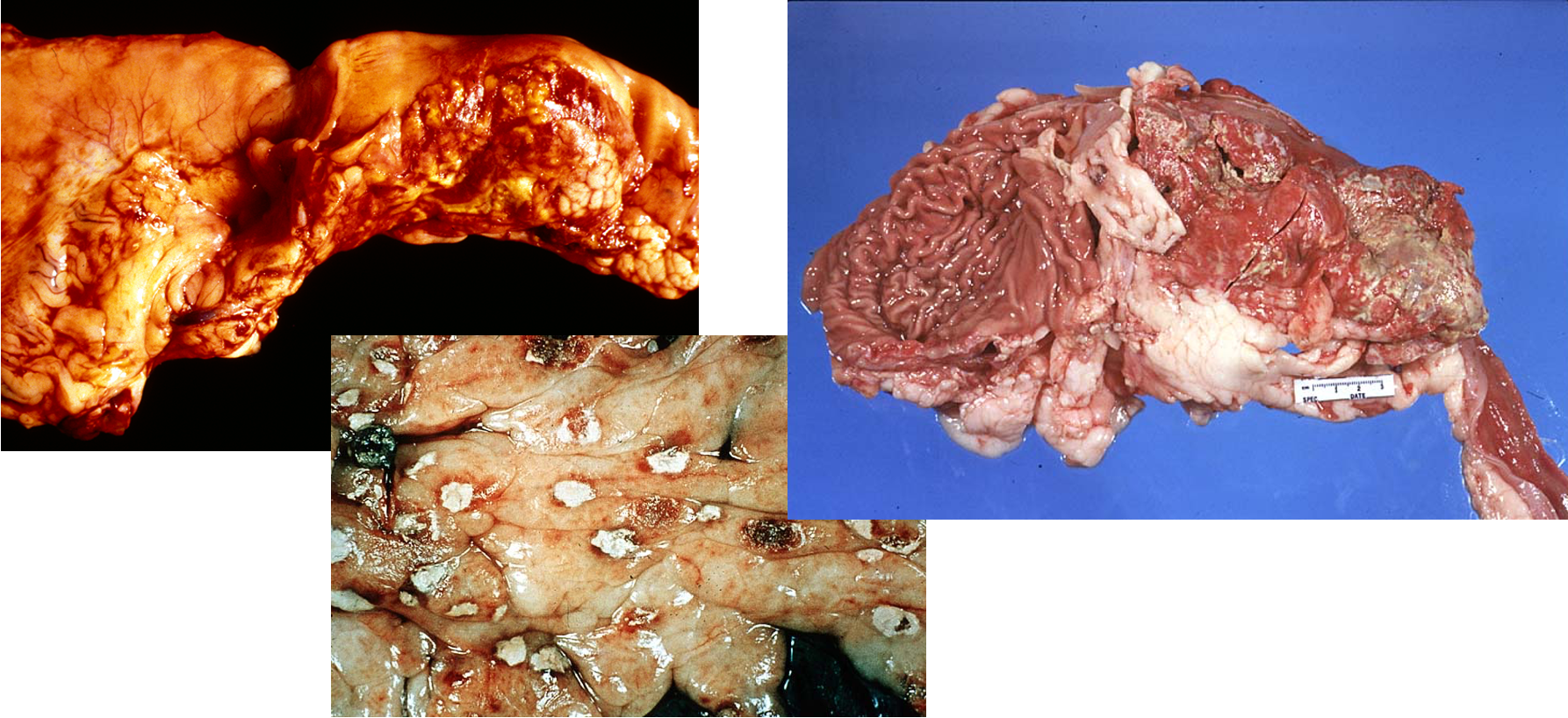
What is being shown here?
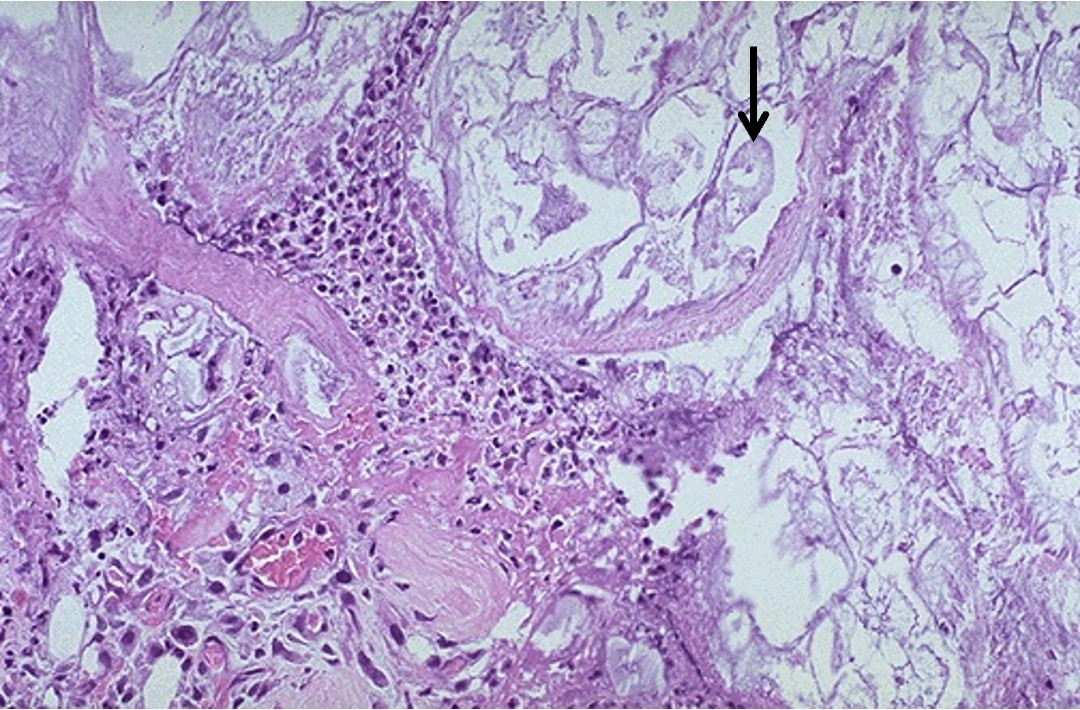
Acute necrotising pancreatitis with fat necrosis
fibrin accum + inflam
What is being shown?
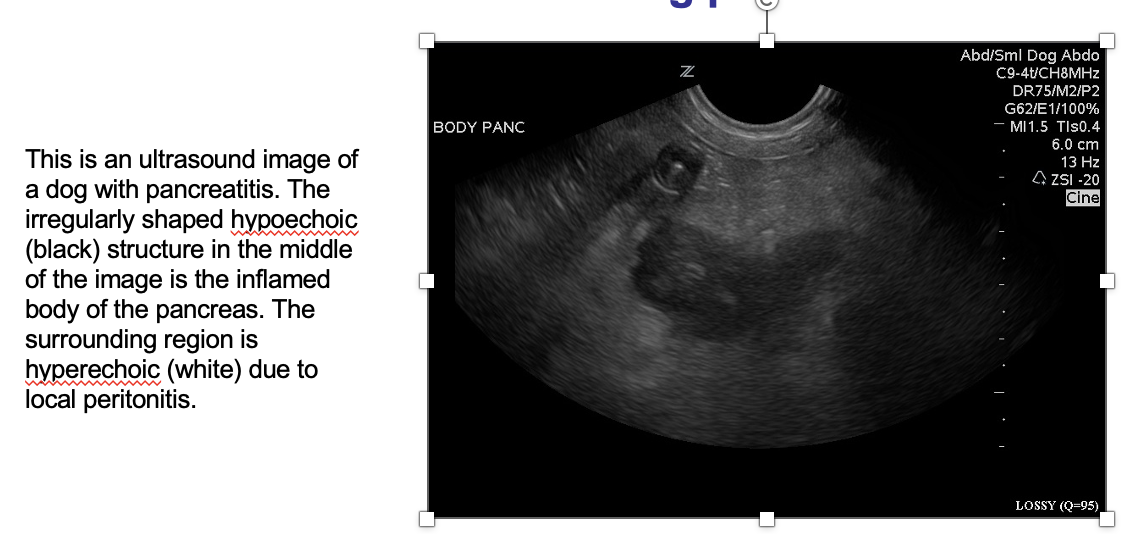
What are the outcomes of acute necrotising pancreaitis?
Death within a few days
Consumption of plasma protease inhibitors →
Activation of kinin, coagulation, fibrinolysis, complement cascades →
DIC, shock →
Animals survive & develop repeated acute episodes
chronic fibrosing pancreatitis
exocrine pancreatic insufficiency, diabetes mellitus
What is chronic fibrosing pancreatitis?
sequel of acute necrotising pancreatitis or w/o signs of acute pancreatitis [cats]
pancreatic tissue replaced by fibrous tissue
How does chronic fibrosing pancreatitis present in cats?
chronic interstitial pancreatitis w/ chronic cholangitis / cholangiohepatitis (& IBD) —> “triaditis”
What are the clinical signs of chronic firbosing pancreatitis?
EPI +/- endocrine deficiency
What is being shown here?
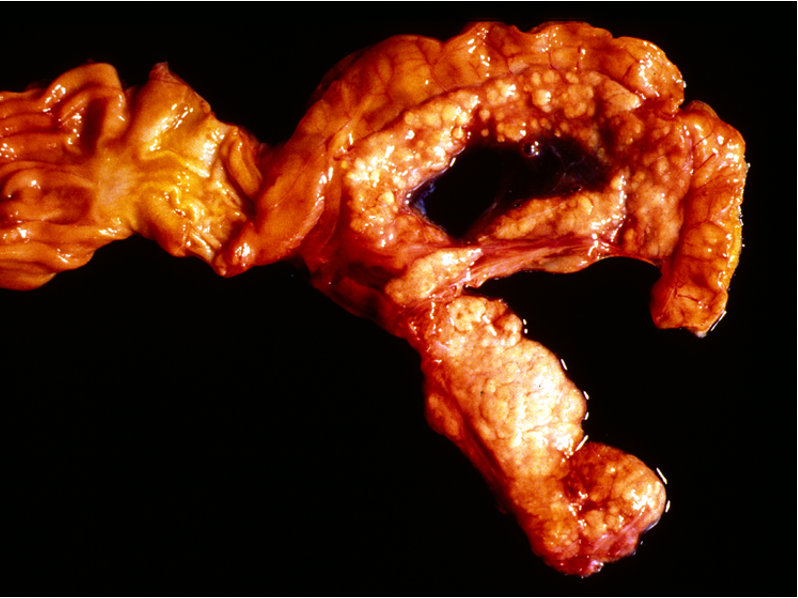
Chronic fibrosing pancreatitis
What is being shown here
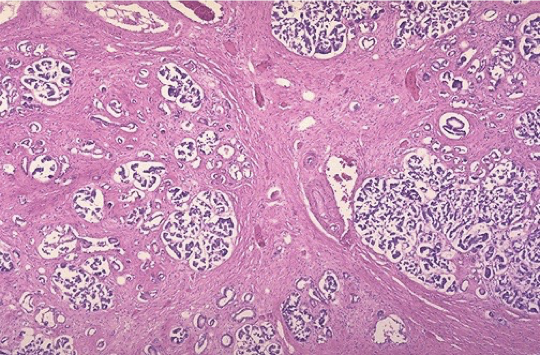
Chronic fibrosing pancreatitis with severe interstitial fibrosis
some islands of exocrine tissue left + fibrosis
What are the causes of exocrine pancreatic insufficiency?
Juvenile atrophy (dogs)
Chronic pancreatitis (cats)
Exocrine pancreatic neoplasia
Hypoplasia (calves)
What are the signs associated with exocrine pancreatic insuffiency?
Diarrhoea and chronic weight loss
pale, soft, voluminous, malodorous faeces (pancreatogenic maldigestion)
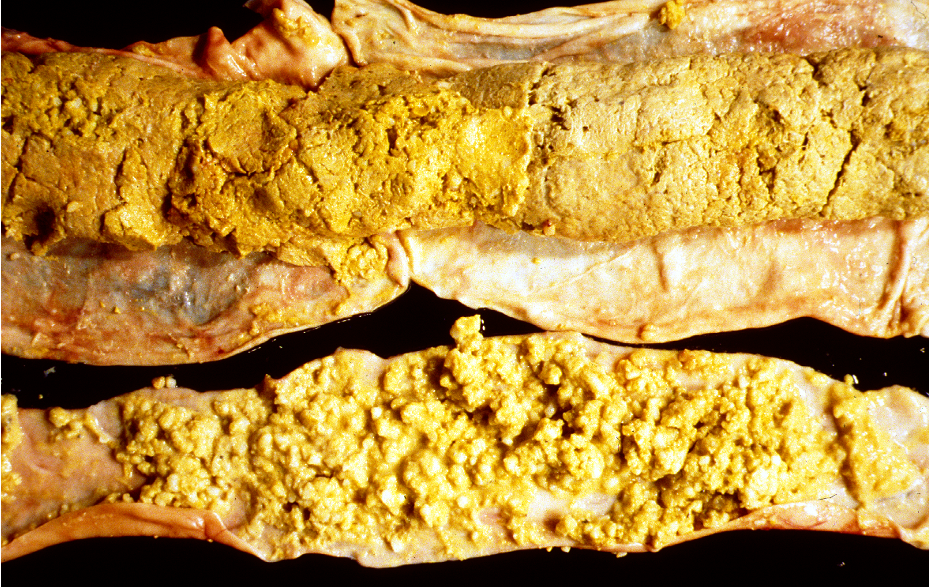
+/- steatorrhoea
bacterial overgrowth (SIBO)
malabsorption of vitamins → hypovitaminosis
+/- diabetes mellitus (depending on underlying aeitiology)
How do you differentiate a nodular hyperplasia from a neoplasia?
nodular hyperplasia = not neoplasm
seen in old dogs, cats and cattle
multiple
no encapsulation
no compression of adjacent tissue
List the neoplasms of the exocrine pancreas
Adenoma
Very rare
Usually solitary
Adenocarcinoma
What are the features of adenocarcinomas
Often arising within centre of pancreas
Cellular origin = acini / ducts
Gross = greyish, scirrhous tissue
V. agressive (early metastases)
implantation metastases (peritoneum, diaphragm → thorax)
Haematogenous (portal vein → liver)
Lymphogenic spread (local LNs)
Local invasion (into duodenal wall)
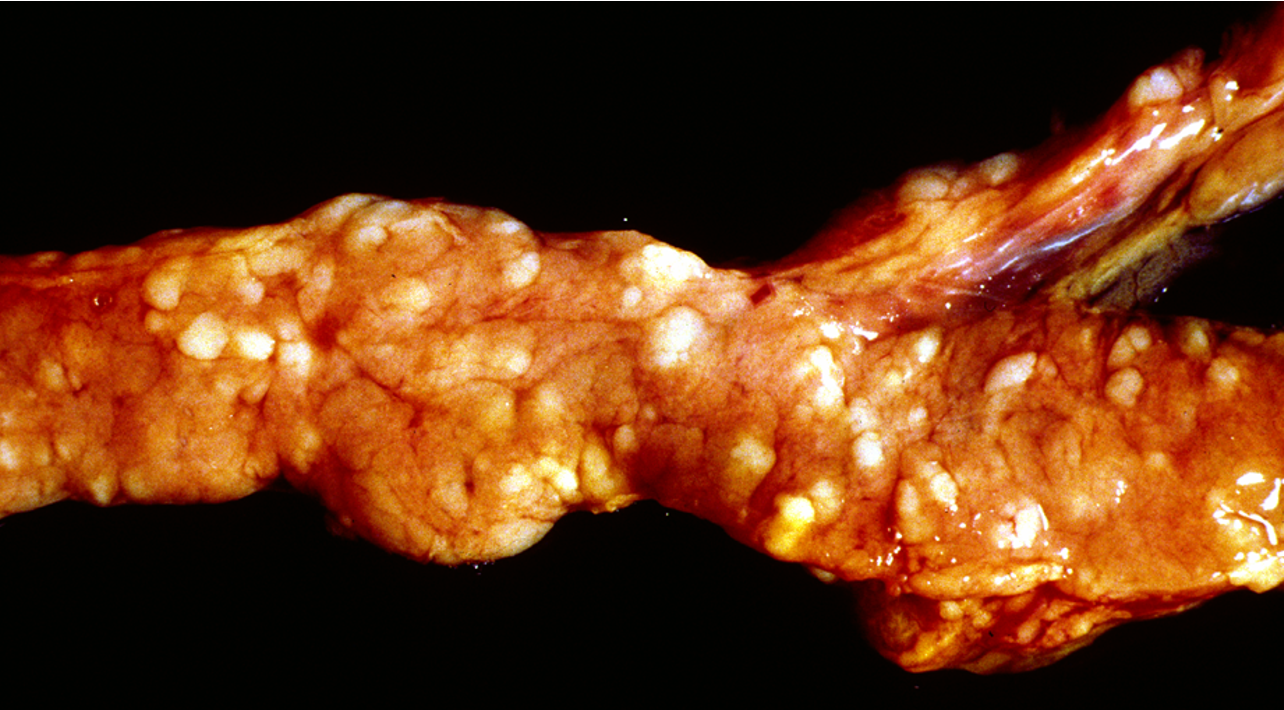
What is being shown here?
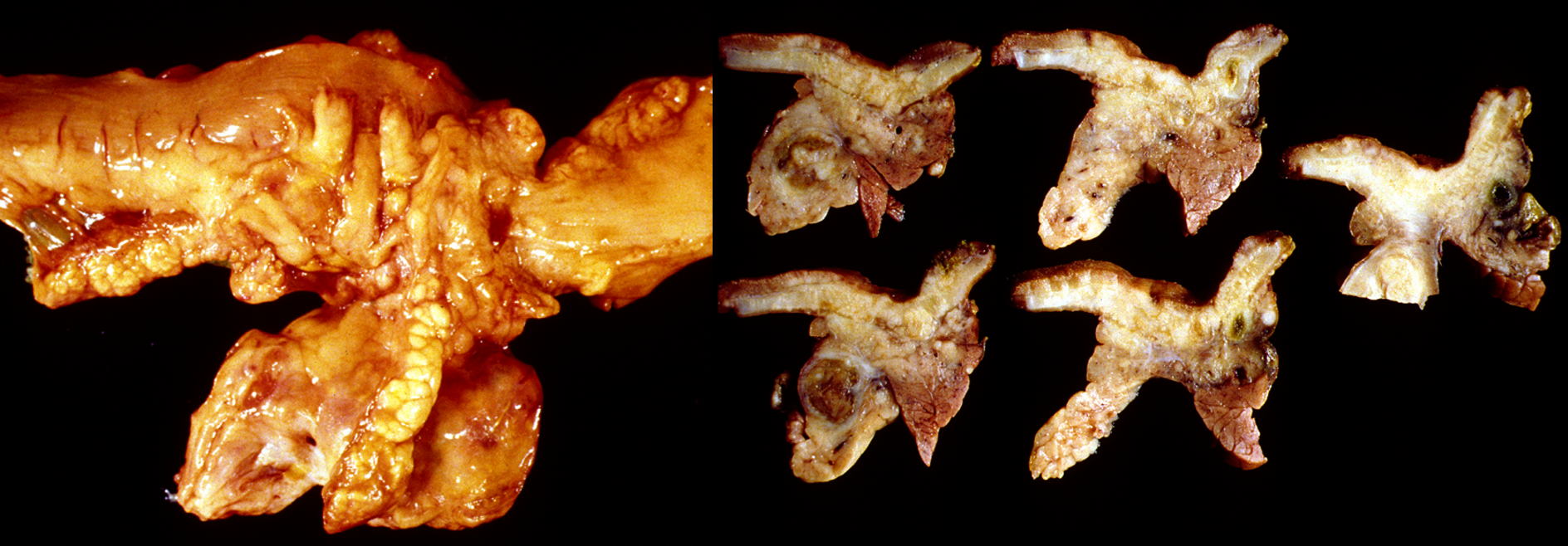
Pancreatic carcinoma (Ddx: duodenal carcinoma)
mass highly infiltrative
What is being shown here?
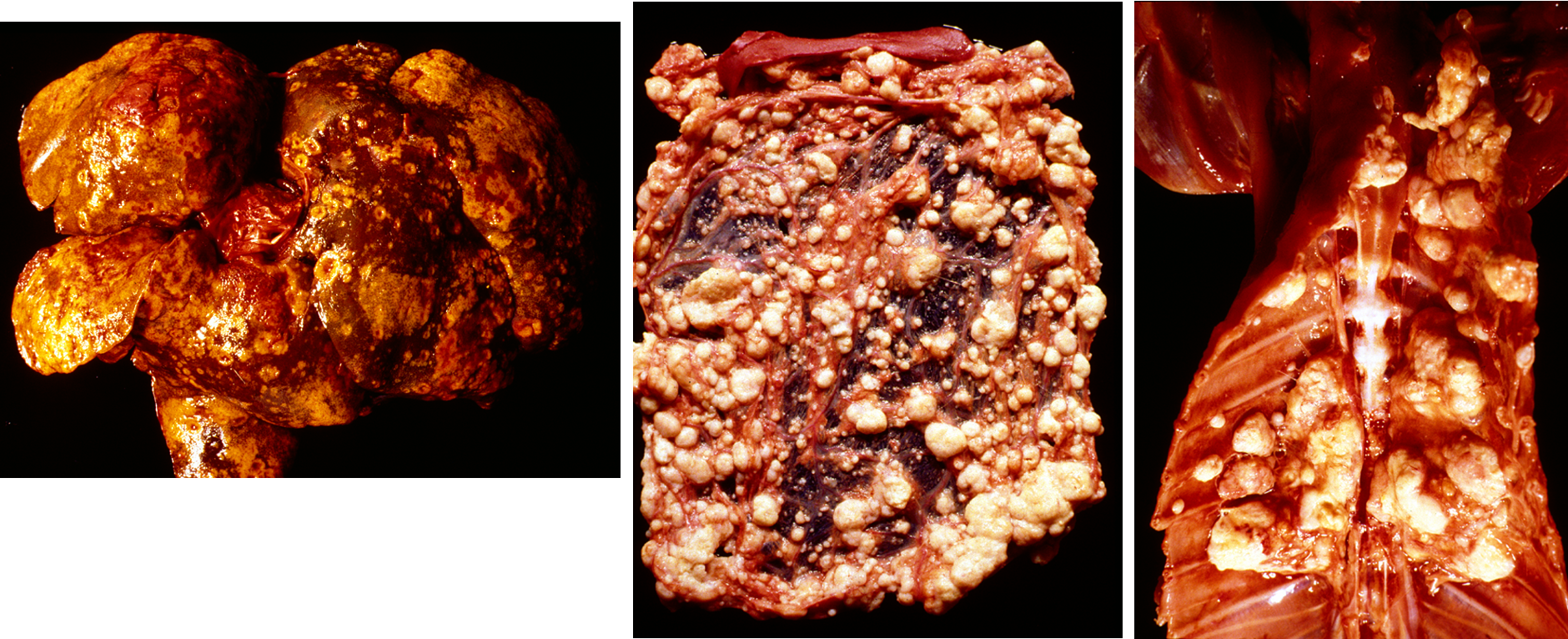
metastases of pancreatic carcinoma:
liver, omentum (spread across abdo cavity), parietal pleura
How can adenocarcinoma of the exocrine pancreas metastasise?
Implantation metastases [peritoneum, diaphragm to thorax]
Haematogenous spread [portal vein to liver]
Lymphogenic spread [local lymph nodes]
Local invasion into duodenal wall