BMB 212 exam 1
1/78
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
79 Terms
What type of bonds do you find within a water molecule?
polar covalent
Which of the following compounds are capable of hydrogen bonding with like molecules? Choose all that apply.
CH3CH2CH2CH2CH3
CH3CH2OCH2CH3
CH3NH2
HOCH2CH2OH
CH3NH2, HOCH2CH2OH
Which of the following molecules is hydrophobic?
Sodium chloride
Ethyl alcohol
Hexane
Sucrose
Ammonia
hexane
The slight negative charge at one end of one water molecule is attracted to the slight positive charge of another water molecule. What is this attraction called?
hydrogen bond
When an ionic compound such as sodium chloride (NaCl) is placed in water, the component atoms of the NaCl crystal dissociate into individual sodium ions (Na+) and chloride ions (Cl-). In contrast, the atoms of covalently bonded molecules (e.g., glucose, sucrose, glycerol) do not generally dissociate when placed in aqueous solution. Which of the following solutions would be expected to contain the greatest number of solute particles (molecules or ions)?
1 L of 0.5 M NaCl
1 L of 0.5 M glucose
1 L of 1.0 M NaCl
Correct
1 L of 1.0 M glucose
1 L of 1.0 M NaCl and 1 L of 1.0 M glucose will contain equal numbers of solute particles.
1 L of 1.0 M NaCl
Which of the following ionizes completely in solution and is considered to be a strong base (alkali)?
NaCl
HCl
NH3
H2CO3
NaOH
NaOH
You have two beakers. One contains pure water, the other contains pure methanol (wood alcohol). The covalent bonds of methanol molecules are nonpolar, so there are no hydrogen bonds among methanol molecules. You pour crystals of table salt (NaCl) into each beaker. Predict what will happen.
NaCl crystals will dissolve readily in water but will not dissolve in methanol.
What type of bonds are formed between water molecules?
hydrogen bonds
Which of the following compounds is amphipathic?
H2O
CH3CH2CH2CH2CH3
HOOCCOOH
CH3CH2CH2CH2COOH
Both A and D are correct
CH3CH2CH2CH2COOH
Which of the following statements is true of weak acids dissolved in water?
They are completely ionized in water
They are totally unionized in water
They are partially ionized in water
The dissociation constant is a function of pH
The dissociation constant is a function of solute concentration
They are partially ionized in water
The major chemical elements in living things are:
carbon, hydrogen, nitrogen, oxygen, phosphorus, sulfur
Cell membranes are mostly made of this macromolecule, which keeps them from dissolving in water.
lipids
Carbohydrates contain/are made of units known as
monosaccharides
Notable features of eukaryotes include all of the following except ________.
Rapid cell division
Larger size
A complex internal structure
An endomembrane system
Organelles
Rapid cell division
Enzymes are a type of protein that speed up chemical reactions in living things. They are known as...
biological catalysts
carbohydrates
catalytic converters
Avogadro's number
biological catalysts
The partial negative charge in a molecule of water occurs because
the oxygen atom acquires an additional electron.
the electrons shared between the oxygen and hydrogen atoms spend more time around the oxygen atom than around the hydrogen atom
the oxygen atom has two pairs of electrons in its valence shell that are not neutralized by hydrogen atoms.
the oxygen atom forms hybrid orbitals that distribute electrons unequally around the oxygen nucleus.
one of the hydrogen atoms donates an electron to the oxygen atom.
the electrons shared between the oxygen and hydrogen atoms spend more time around the oxygen atom than around the hydrogen atom
What is the hydrogen ion concentration in a solution at pH 8.3?
5.0 × 10 ^−9 M
5.0 × 10 ^−8 M
0.5 M
5 M
5.0 × 10 ^−9 M
What would be the pH of 1L of water if 1 ml of 1M HCl is added?
3
0
2
13
3
What is the hydrogen ion [H+] concentration of a solution of pH 8?
10^-8 M
What is the pH of a 1 millimolar NaOH solution?
pH 3
pH 8
pH 9
pH 10
pH 11
11
What is the pH of a solution with a hydroxyl ion [OH-] concentration of 10-12 M?
pH 2
pH 4
pH 10
pH 12
pH 14
pH 2
If the pH of a solution is decreased from 9 to 8, it means that the
concentration of H+ has decreased to one-tenth (1/10) what it was at pH 9.
concentration of H+ has increased 10-fold (10X) compared to what it was at pH 9.
concentration of OH- has increased 10-fold (10X) compared to what it was at pH 9.
concentration of OH- has decreased to one-tenth (1/10) what it was at pH 9.
Both B and D are correct.
Both B and D are correct
If the pH of a solution is increased from pH 5 to pH 7, it means that the
concentration of H+ is twice (2X) what it was at pH 5.
concentration of H+ is half (1/2) what it was at pH 5.
concentration of OH- is 100 times greater than what it was at pH 5
concentration of OH- is one-hundredth (0.01X) what it was at pH 5.
concentration of H+ is 100 times greater and the concentration of OH- is one-hundredth what they were at pH 5.
concentration of OH- is 100 times greater than what it was at pH 5
One liter of a solution of pH 2 has how many more hydrogen ions (H+) than 1 L of a solution of pH 6?
4 times more
400 times more
4,000 times more
10,000 times more
100,000 times more
10,000 times more
One liter of a solution pH 9 has how many more hydroxyl ions (OH-) than 1 L of a solution of pH 4?
5 times more
100 times more
1,000 times more
10,000 times more
100,000 times more
100,000 times more
What is the pH of a solution with a hydrogen ion [H+] concentration of 10-8 M?
pH 2
pH 4
pH 6
pH 8
pH 10
pH 8
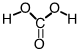
this is the conjugate acid of what
bicarbonate ion
Which of the following would not form a suitable buffer?
Acetic acid/ acetate
Carbonic acid/ bicarbonate
Bicarbonate/ carbonate
Hydrochloric acid/ chloride
Phosphoric acid/ dihydrogen phosphate
The most important buffer in blood is __________.
Carbonate/bicarbonate buffer
Protein buffer
Phosphate buffer
Lactate buffer
Tartrate buffer
Carbonate/bicarbonate buffer
When an individual breathes very rapidly, large amounts of carbon dioxide are exhaled. What effect does this have on blood pH?
Acidosis
Alkalosis
No change
First acidosis then rebounding alkalosis
First alkalosis then rebounding acidosos
Alkalosis
Which of the following molecules are weak acids?
HCl
CO3−2
HNO3
HCO3−
Both B and D
Which of the following species would form a buffer with HPO4−2?
H3PO4
H2PO4−1
CO3−2
All would form buffers with HPO4−2
Both A and B
H2PO4−1
Which of the following species can form a buffer system?
Hydrochloric acid / Acetic acid
Acetic acid / Sodium chloride
Acetic acid / Ammonium chloride
Acetic acid/ Sodium acetate
Phosphoric acid / Sodium phosphate
Acetic acid/ Sodium acetate
Is it possible to prepare a buffer consisting of only carbonic acid and sodium carbonate?
no
Buffers are substances that help resist shifts in pH by
releasing H+ in acidic solutions.
donating H+ to a solution when they have been depleted.
releasing OH- in basic solutions.
accepting H+ when they are in excess.
donating H+ to a solution when they have been depleted, accepting H+ when they are in excess
One of the buffers that contribute to pH stability in human blood is carbonic acid (H2CO3). Carbonic acid is a weak acid that dissociates into a bicarbonate ion (HCO3-) and a hydrogen ion (H+). Thus,
H2CO3 <--> HCO3- + H+
If the pH of the blood drops, one would expect that the system would compensate for it by causing the:
a decrease in the concentration of H2CO3 and an increase in the concentration of HCO3-.
the concentration of hydroxide ion (OH-) to increase.
the concentration of bicarbonate ion (HCO3-) to increase.
the HCO3- to act as a base and remove excess H+ with the formation of H2CO3.
the HCO3- to act as an acid and remove excess H+ with the formation of H2CO3.
the HCO3- to act as a base and remove excess H+ with the formation of H2CO3.
One of the buffers that contribute to pH stability in human blood is carbonic acid H2CO3. Carbonic acid is a weak acid that when placed in an aqueous solution dissociates into a bicarbonate ion (HCO3-) and a hydrogen ion (H+). Thus,
H2CO3 --> HCO3- + H+
If the pH of the blood increases, one would expect the system to compensate by
(Hint: Think in terms of protons generated due to increase in pH. How would they shift the equilibrium)
a decrease in the concentration of H2CO3 and an increase in the concentration of H2O.
an increase in the concentration of H2CO3 and a decrease in the concentration of H2O.
a decrease in the concentration of HCO3- and an increase in the concentration of H2O.
an increase in the concentration of HCO3- and a decrease in the concentration of H2O.
a decrease in the concentration of HCO3- and an increase in the concentration of both H2CO3 and H2O.
a decrease in the concentration of H2CO3 and an increase in the concentration of H2O
The dissociation constant for an acid is 1 × 10−6. What is its pKa?
0.6
-6
-0.6
6
6
A buffer solution at pH 10 has a ratio of [HA]/[A−] of 10. What is the pKa of the acid?
8
9
10
11
12
11
Calculate the ratio of dihydrogen phosphate to hydrogen phosphate in blood at a pH 7.4. The Ka is 6.3 × 10−8.
1:1.6
1.4:1
1.6:1
1:1
1.6:1
The two sets of macromolecules most responsible for energy use in living things.
Proteins
Carbohydrates
Nucleic acids
Lipids
carbs, lipids
Sugar is a macromolecule that falls under which group?
Proteins
Lipids
Nucleic acids
Carbohydrates
Carbohydrates
Cell membranes are mostly made of this macromolecule, which keeps them from dissolving in water.
Proteins
Lipids
Carbohydrates
Nucleic acids
Lipids
Enzymes are a type of protein that speed up chemical reactions in living things. They are known as...
carbohydrates
Avogadro's number
biological catalysts
catalytic converters
biological catalysts
We have long-term energy even if we don't eat for a period of time because of
chemical bonds
enzymes
nucleic acids
proteins
lipids
lipids
Which of the following would be classified as a lipid?
starch—a polysaccharide
catalase—an enzyme
cholesterol—a steroid
alanine—an amino acid
cholesterol
The following molecule is an example of which single class of compounds?
CH3CH2CH(NH2)COOH
Amino acid
Amine
Acid
Alcohol
Ester
Amino acid
The following molecule is an example of which class of compounds?
CH3CH2CH2COOCH2CH3
Aldehyde
Ester
Acid
Hydrocarbon
Ether
Ester
The intermolecular bonds that link the complementary base pairs and help hold the in the two strands of the DNA double helix together are ________.
hydrogen bonds
Adenine is a purine.
True
False
true
A nucleotide consists of only a base and a sugar.
True
False
false
Which of the following is not an example of a macromolecule? (Hint: Macromolecule is the polymeric form.)
Polysaccharide
Protein
Nucleic acid
Amino acid
Enzyme
Amino acid
In the Five Kingdom classification system, human beings would be considered
animals
Which of these eukaryotic kingdoms consists primarily of unicellular organisms?
a. Animals
b. Fungi
c. Plants
d. Protista
e. Both fungi and protista.
Both fungi and protista.
Which of these kingdoms includes only prokaryotic organisms?
a. Animals
b. Fungi
c. Monera
d. Plants
e. Protista
8. If atoms with greatly differing electronegativities form a bond, that bond will be
polar
All eukaryotic organisms have
a nucleus
Which of the following functional groups are not commonly seen in biomolecules?
a. Alkyl halides
b. Amides
c. Carboxylic acids
d. Ethers
e. Phosphate esters
Alkyl halides
The water molecule is polar because:
a. Electrons are not distributed symmetrically in the molecule.
b. The hydrogen atoms are found on one "side" of the molecule.
c. Hydrogen is less electronegative than oxygen.
d. The hydrogen atoms are found on one "side" of the molecule and hydrogen is less electronegative than oxygen.
e. All of these are correct.
All of these are correct
Ionic compounds and polar covalent compounds tend to dissolve in water because of
ion dipole and dipole dipole interactions
Many of the properties of water can be accounted for by the fact that
a. it is polar
b. it forms hydrogen bonds
c. it is a bent molecule
d. all of these are true
all of these are true
How does the strength of hydrogen bonds compare with covalent bonds? a. Hydrogen bonds are much stronger than covalent bonds.
b. Hydrogen bonds are much weaker than covalent bonds.
c. Hydrogen bonds and covalent bonds have similar strengths.
d. The question cannot be answered without knowing which covalent bonds are being referred to
Hydrogen bonds are much weaker than covalent bonds.
Which will dissociate most in water, a weak acid or a strong acid?
A strong acid
Bases are
a. proton donors.
b. proton acceptors.
c. hydrogen bond donors.
d. hydrogen bond acceptors.
proton acceptors
Which has the greater Ka, a weak acid or a strong acid?
a. A weak acid.
b. A strong acid
c. They should dissociate about the same.
d. It's impossible to predict.
A strong acid
Which has the greater pKa, a weak acid or a strong acid?
a. A weak acid.
b. A strong acid
c. They should dissociate about the same.
d. It's impossible to predict
A weak acid
How many hydrogen bonds can water form?
4
Which of the following compounds are capable of hydrogen bonding with like molecules?
A) CH3CH2CH2CH2CH3
B) CH3CH2OCH2CH3
C) CH3NH2
D) HOCH2CH2OH
E) Both C and D are correct
Both C and D are correct
.Calculate the ratio of dihydrogen phosphate to hydrogen phosphate in blood at a pH 7.4. The Ka is 6.3 x10-8 .
1: 1.6
One liter of a solution pH 9 has how many more hydroxyl ions (OH-) than 1 L of a solution of pH 4?
100,000
Which of the following would not form a suitable buffer?
Carbonic acid/ bicarbonate
Phosphoric acid/ dihydrogen phosphate
Hydrochloric acid/ chloride
Bicarbonate/ carbonate
Acetic acid/ acetate
Hydrochloric acid/ chloride
A suitable buffer solution must consist of a blank acid and blank base
weak, conjugate
Which of the following species would form a buffer with HPO4−2?
H2PO4−1
Both A and B
H3PO4
All would form buffers with HPO4−2
CO3−2
H2PO4−1
At pH 6.8 the H+ concentration is 4 times greater than at pH 7.4. (T/F)
true
Which has the greater Ka, a weak acid or a strong acid?
strong
Polar covalent compounds tend to dissolve in water because of
dipole dipole interactions
What is the henderson hasselbach eq (what is ha what is a)
ph = pka log (a/ha) where ha is weak acid and a is conjugate vase
The following molecule is an example of which class of compounds?
CH3CH2CH2COCH3
ketone
The majority of Earth’s species belong to which of the following classifications?
Prokaryotes