(WIP) KMT of Gases - FINAL TERM Chemistry
1/49
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
50 Terms
Gas consists of minute particles; Gas particles constantly move at a rapid and random potion such that they collide with each other and the walls of the container; Gas particles are hard spheres such that their collisions are perfectly elastic; IMF=0; Average kinetic energy is directly proportional to temperature
Enumerate five (5) assumptions of gases:
Photochemical Smog
A common type of air pollution in hot cities with plenty of fumes and which can cause eye irritation and respiratory illnesses.
Photochemical Smog
Results from the reaction of sunlight with nitrogen oxides and volatile organic compounds like CO and formaldehydes.
Vibrates in a fixed position
The movement of particles in a solid _____.
Slide or glide
The movement of particles in a liquid _____.
Rapid or random
The movement of particles in a gas is _____.
Pressure
The force produced when these particles collide with a surface creates _____.
Pressure
The force exerted per area.
Air Pressure
The force exerted on the walls of the container upon collision.
P = F/A
State the formula for Pressure:
Barometer
Measures the atmospheric pressure.
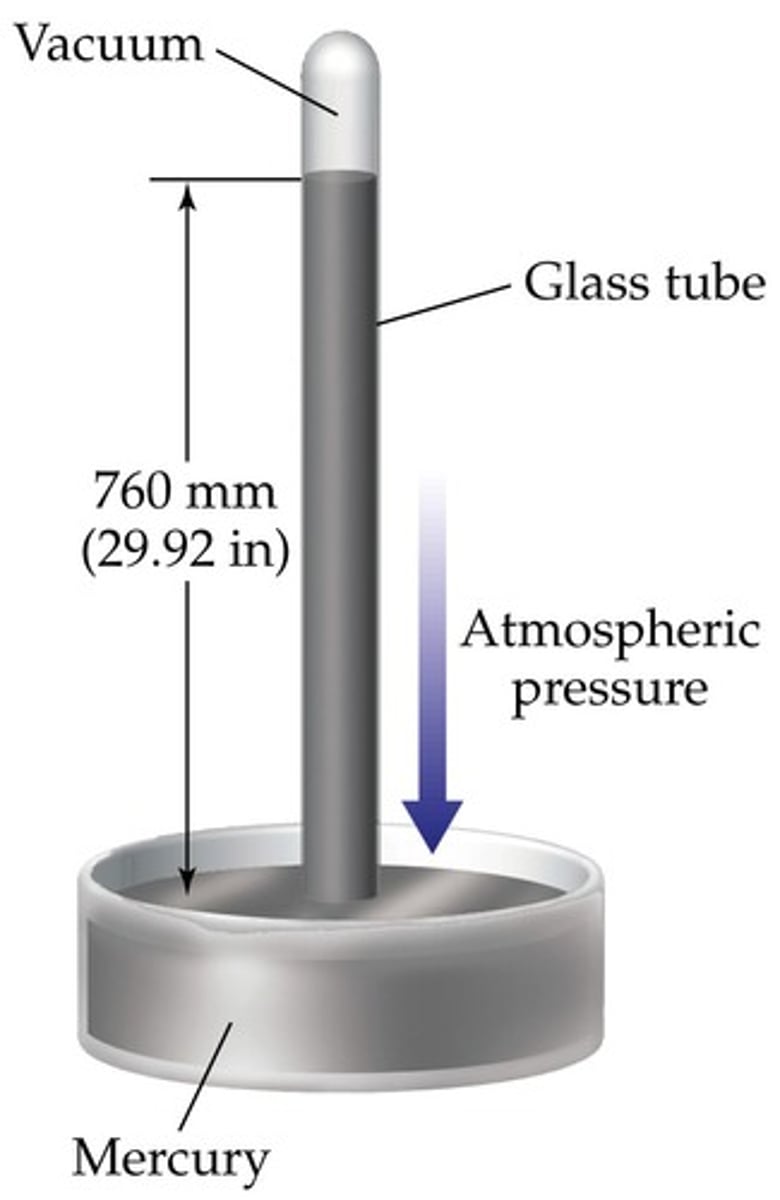
Evangelista Torricelli
Italian physicist who invented the barometer in 1643.
760 mm Hg; 760 torr; 1 atm; 101,325 Pa, 14.7 psi
Enumerate five (5) common units of equivalent pressure at STP:
Manometer
Measures the pressure in a closed system.

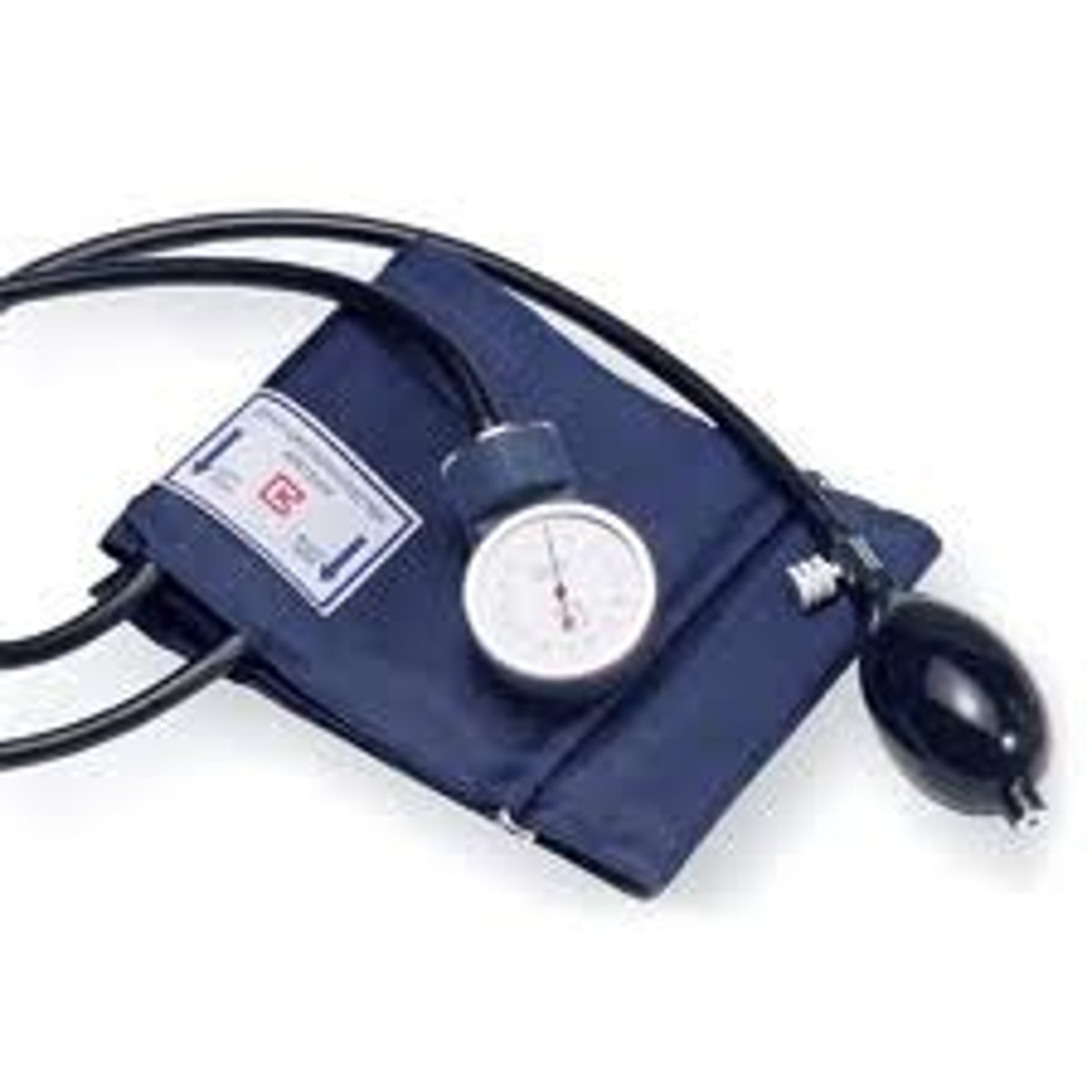
Sphygmomanometer
A medical device used to measure arterial blood pressure non-invasively.
Increases
As temperature increases, the kinetic energy (KE) of the particles in a substance _____.
KE = mv²/2
State the formula for Kinetic Energy:
K = °C + 273.15
State the formula for Kelvin:
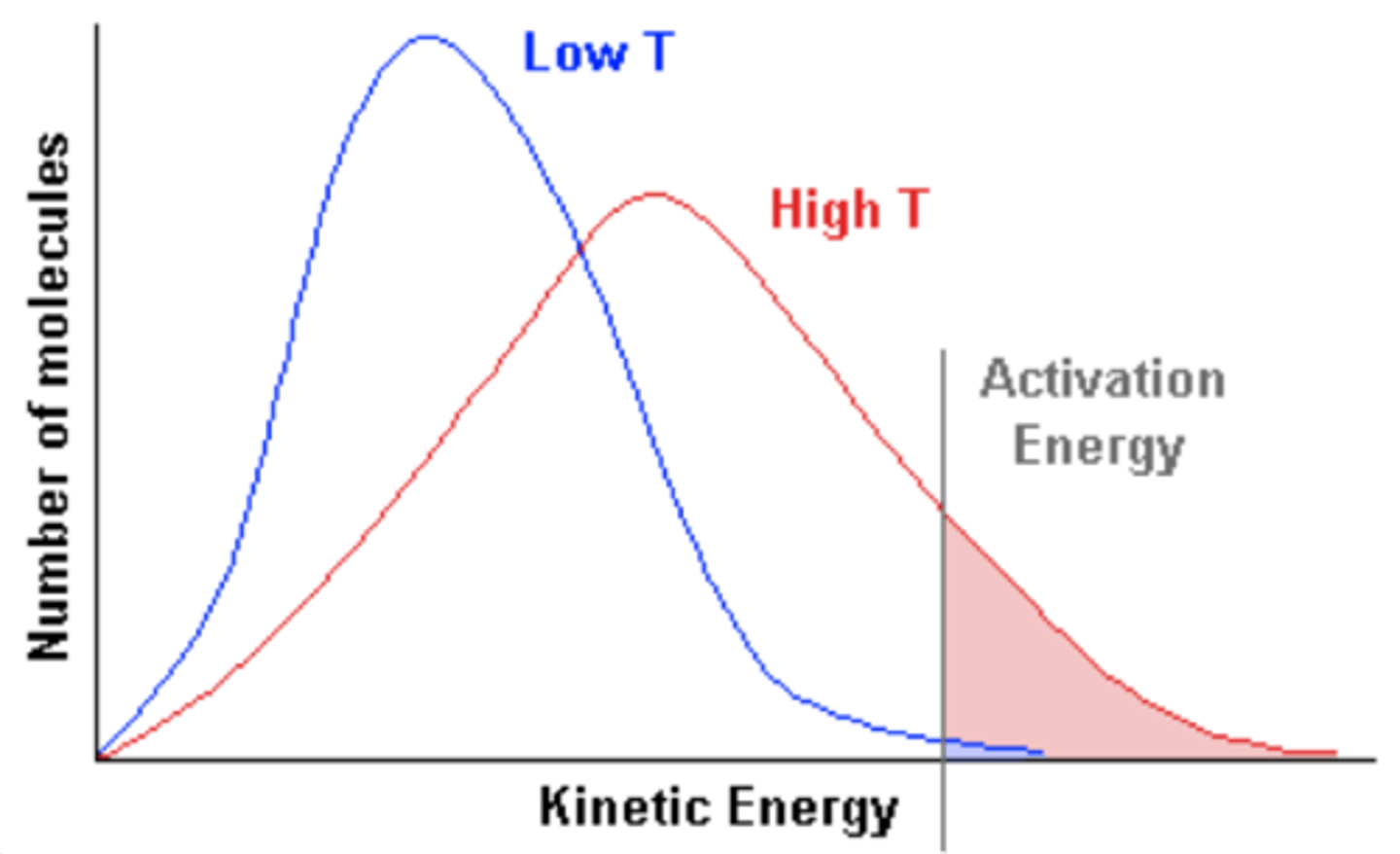
Maxwell-Boltzmann distribution
Describes particle speeds in gases, where temperature determines the distribution shape.
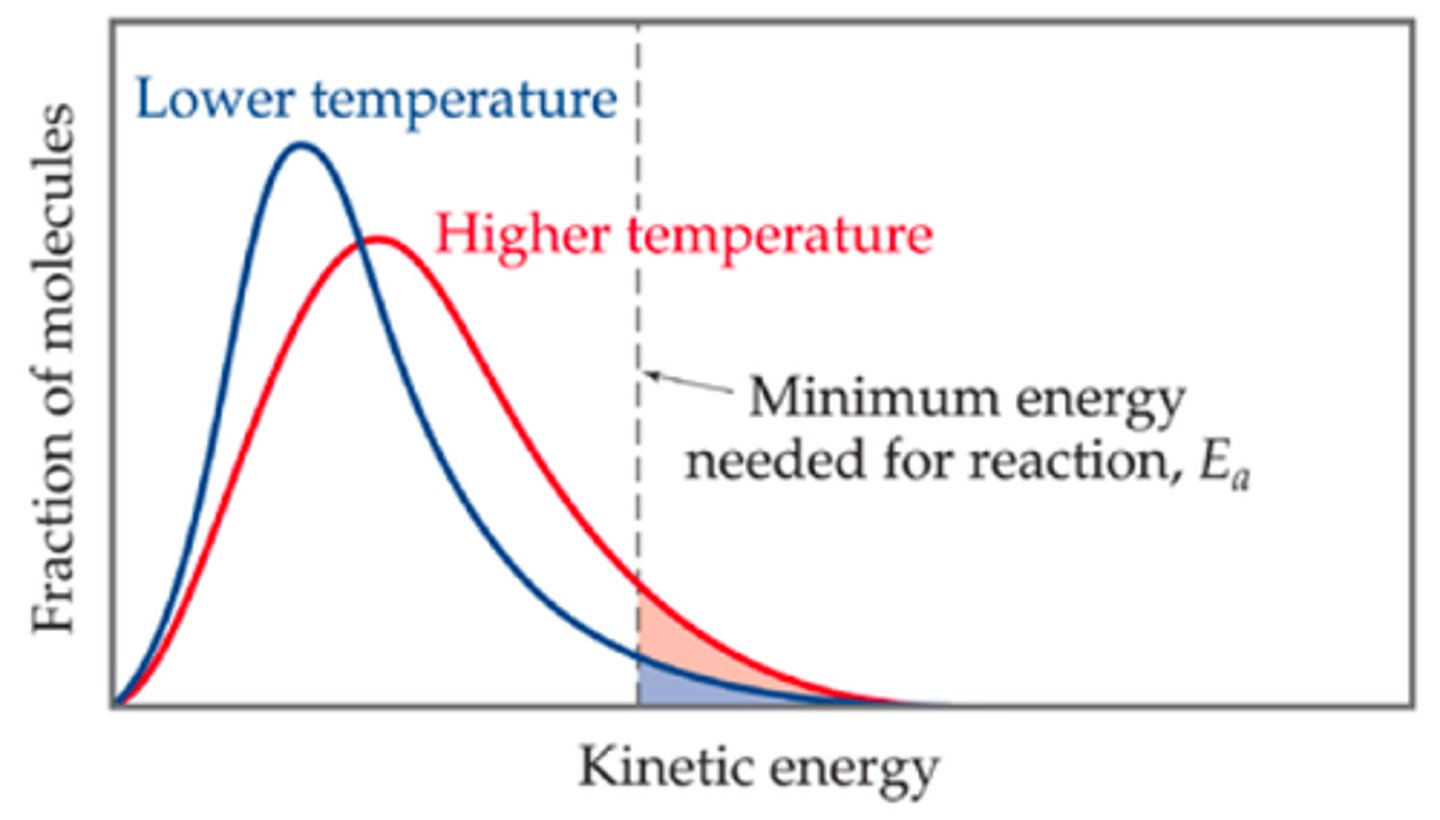
Increased average speed
As temperature increases, particles move faster, shifting the peak to the right.
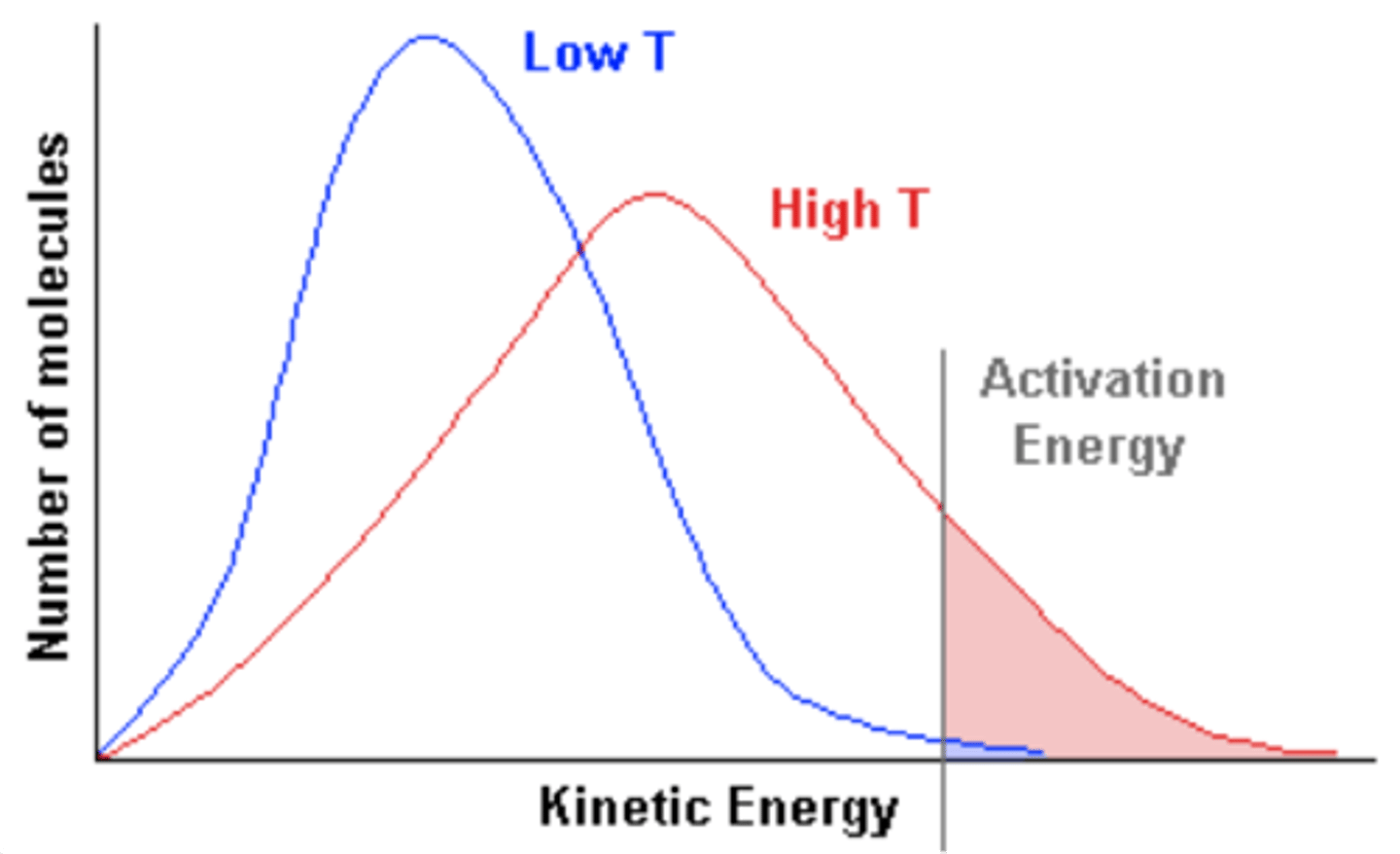
Broader distribution
Higher temperatures result in a flatter, wider curve, meaning a wider range of possible speeds.
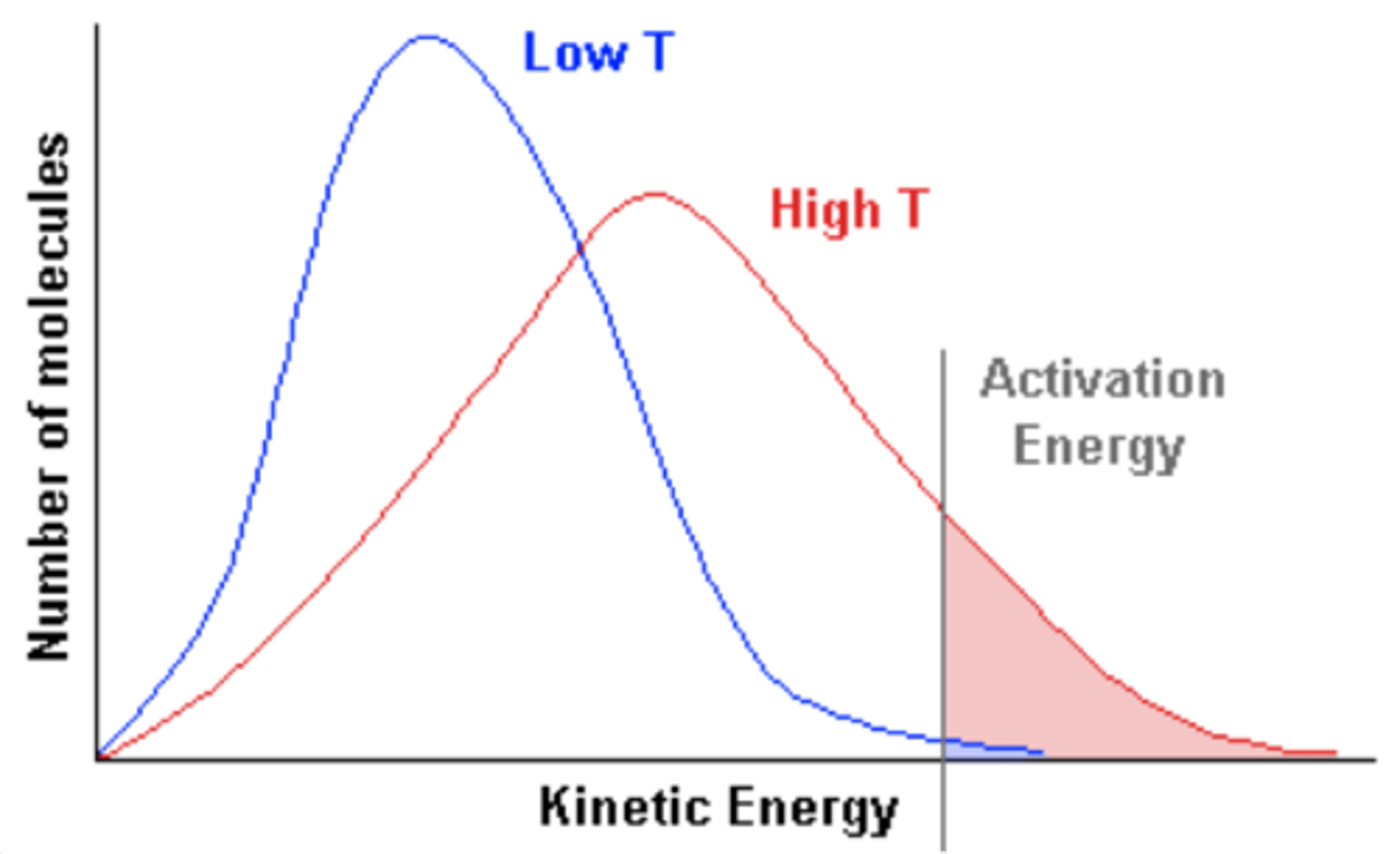
Higher energy fraction
A larger proportion of molecules exceed the activation energy, increasing reaction rates.
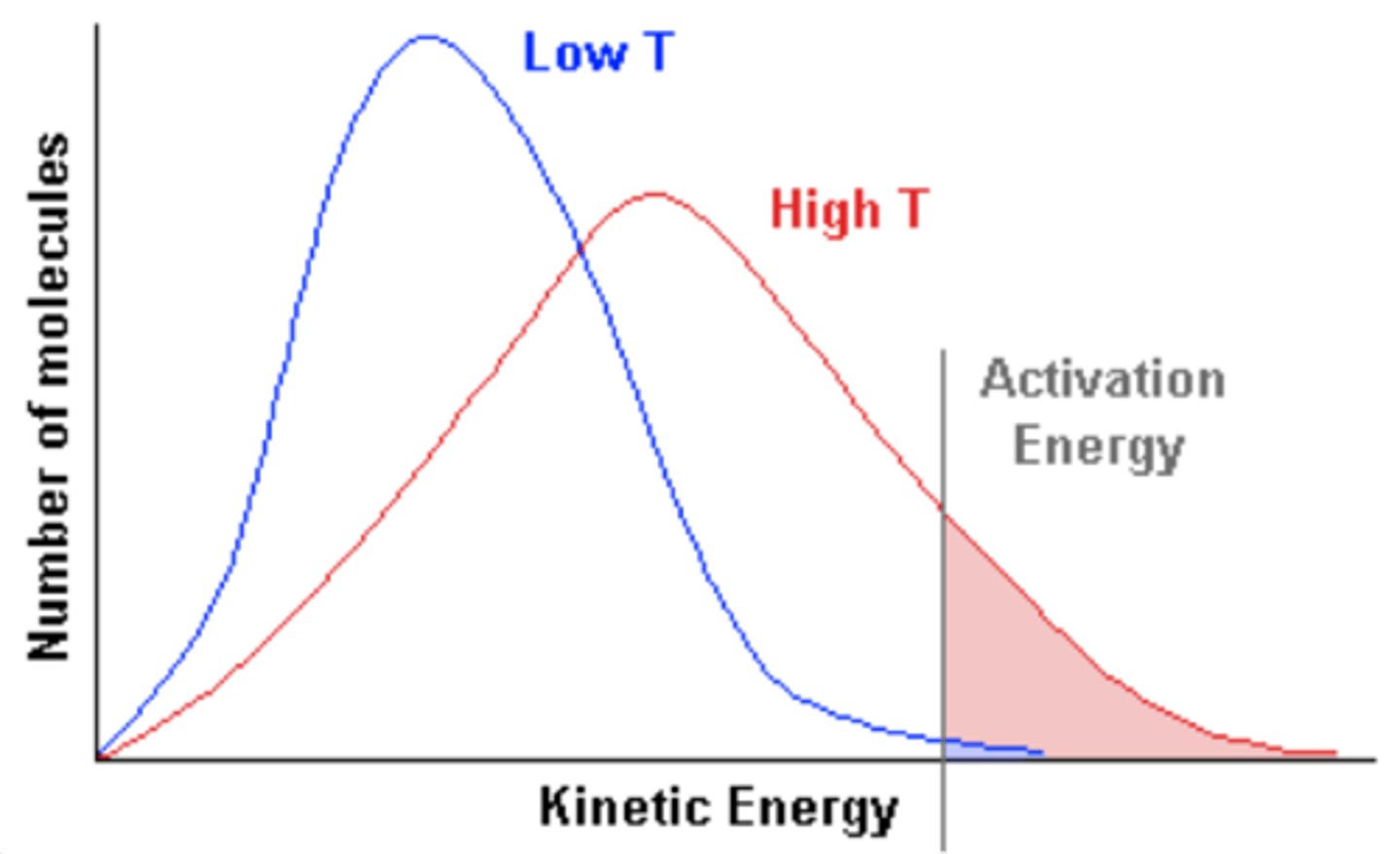
Constant total particles
The total area under the curve remains constant, regardless of temperature changes.
Amount of gas; Pressure; Volume; Temperature
Enumerate four (4) variables that describe the behavior of gases:
n
Variable defined as the amount of particles with the unit: mol.
P
Variable with the units: atm, mm Hg, Torr, Pa
V
Variable with the units: mL, L
T
Variable with the units: °C, °F, K
Robert Boyle
Irish scientist who studied the pressure and volume relationship of a confined gas held at a constant temperature.
Boyle's Law
The pressure of a fixed amount of gas is inversely proportional to its volume at constant temperature.
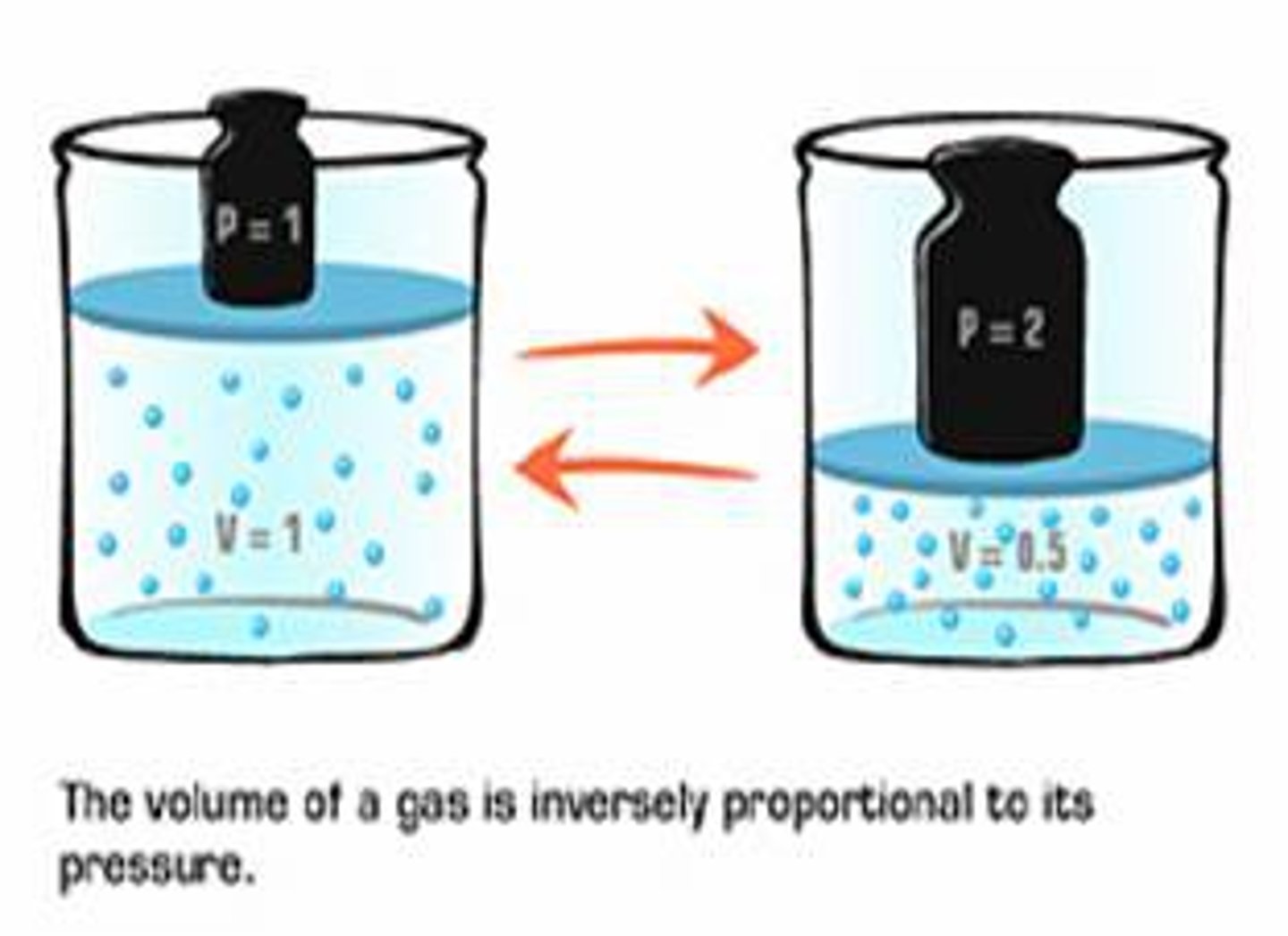
P₁V₁=P₂V₂
State the formula for Boyle's Law:
Joseph-Louis Gay-Lussac
French chemist and physicist who discovered that all gases expand equally over a temperature change.
Gay-Lussac's Law
The pressure of a gas is directly proportional to its absolute temperature when the volume is kept constant.
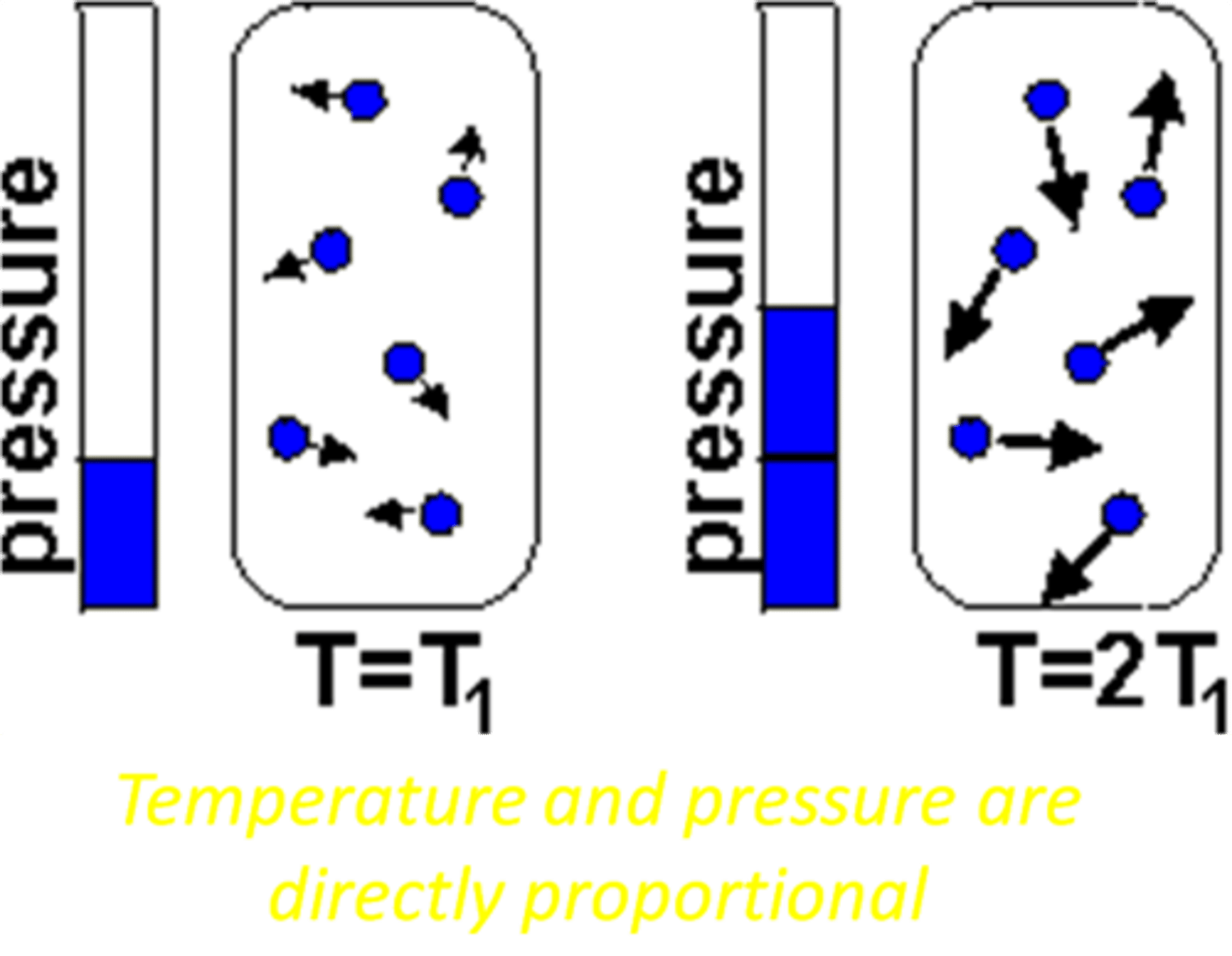
P₁/T₁=P₂/T₂
State the formula for Gay-Lussac's Law:
Jacques Charles
French chemist who discovered volume-temperature relationship of gases at a constant pressure (together with Gay-Lussac).
Charles's Law
The volume of a gas is directly proportional to its absolute temperature at a constant pressure.
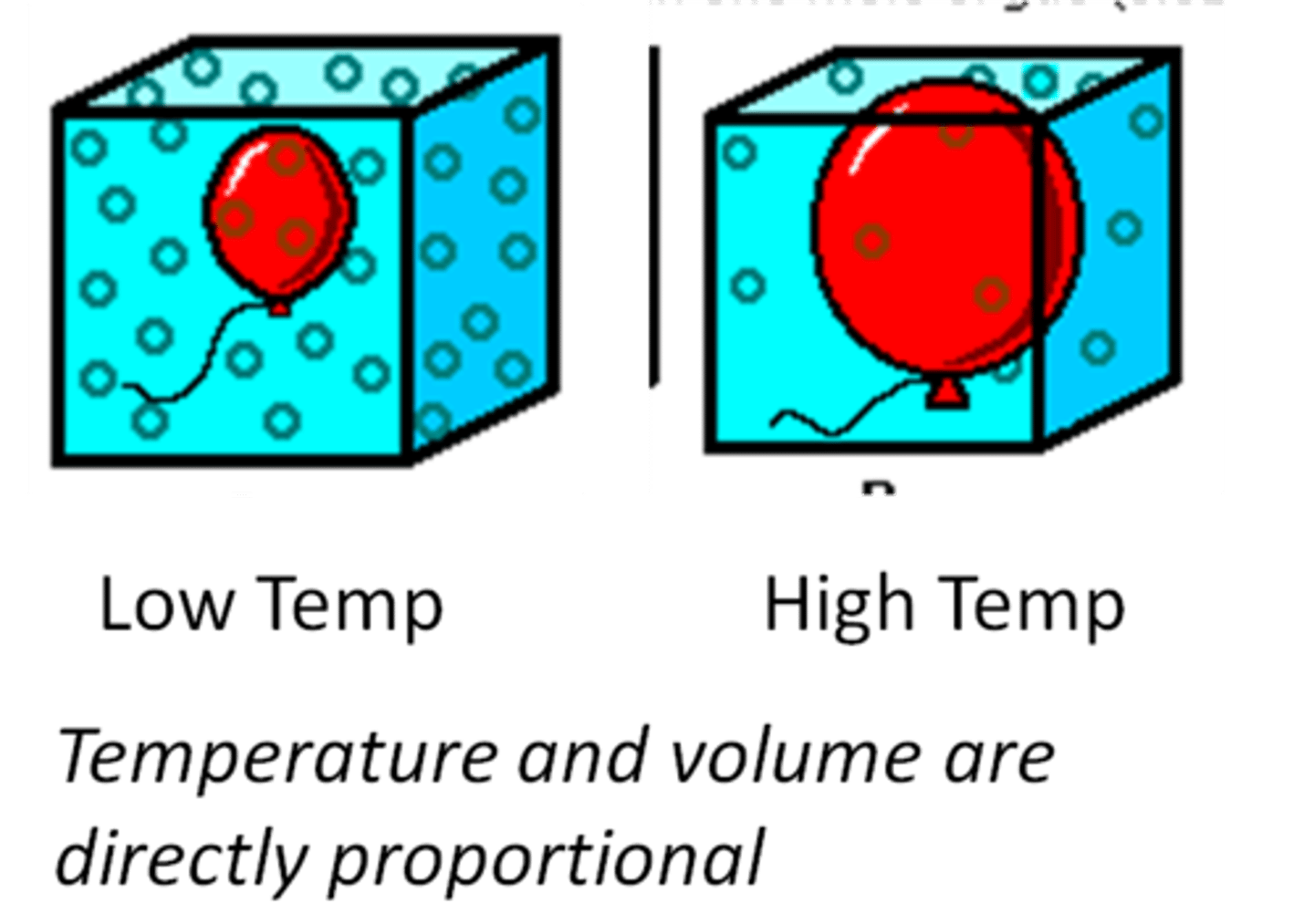
V₁/T₁=V₂/T₂
State the formula for Charles's Law:
Combined Gas Law
The gas laws of Boyle, Charles, and Gay-Lussac combined into a single equation.
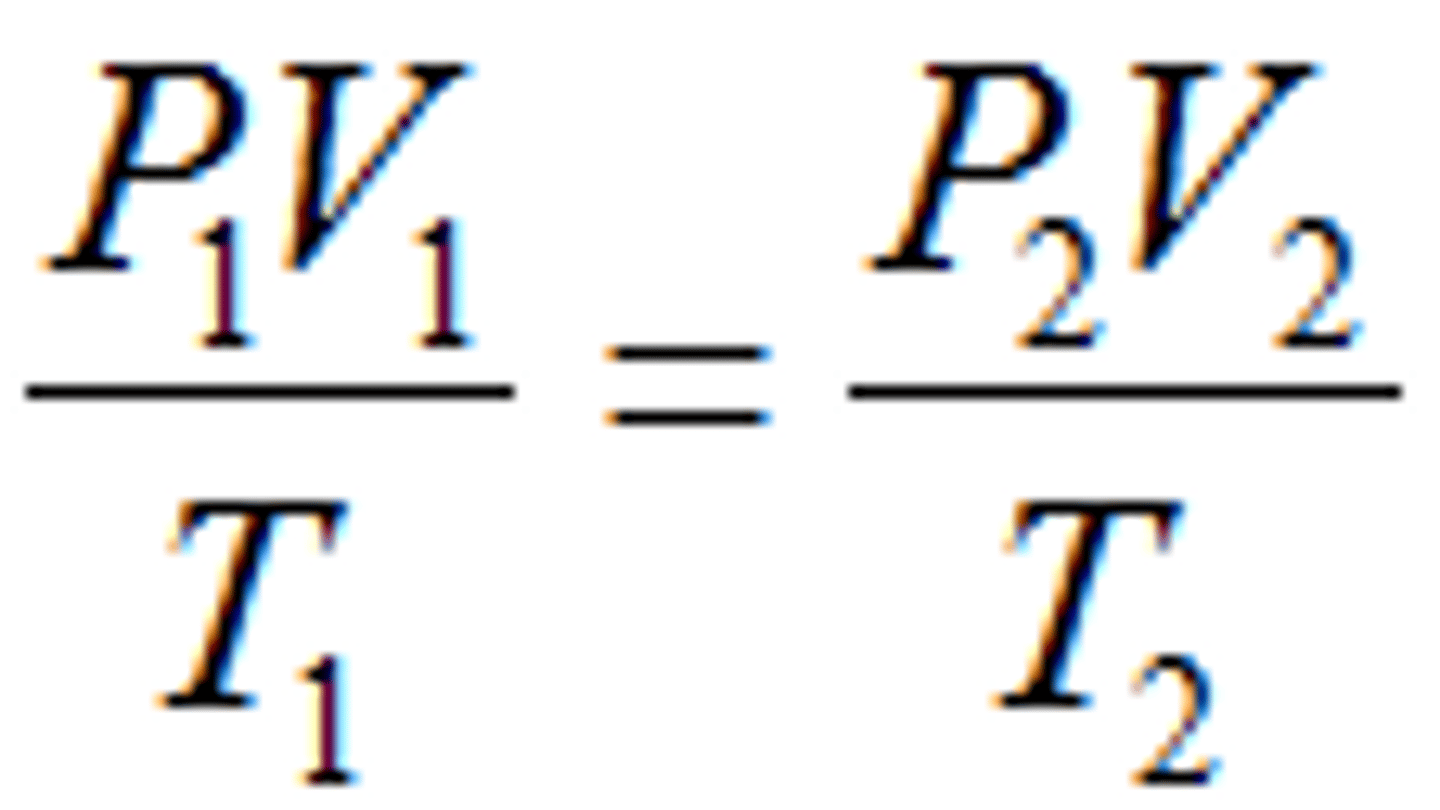
P₁V₁/T₁=P₂V₂/T₂
State the formula for Combined Gas Law:
Ideal Gas
A theoretical gas, which satisfies the assumptions of the kinetics molecular theory.
Ideal Gas Equation
Links all three variables (volume, temperature, and pressure) to the amount of gas expressed in mole..
PV = nRT
State the formula for the Ideal Gas Equation:
Gas constant
What is R in the Ideal Gas Equation?
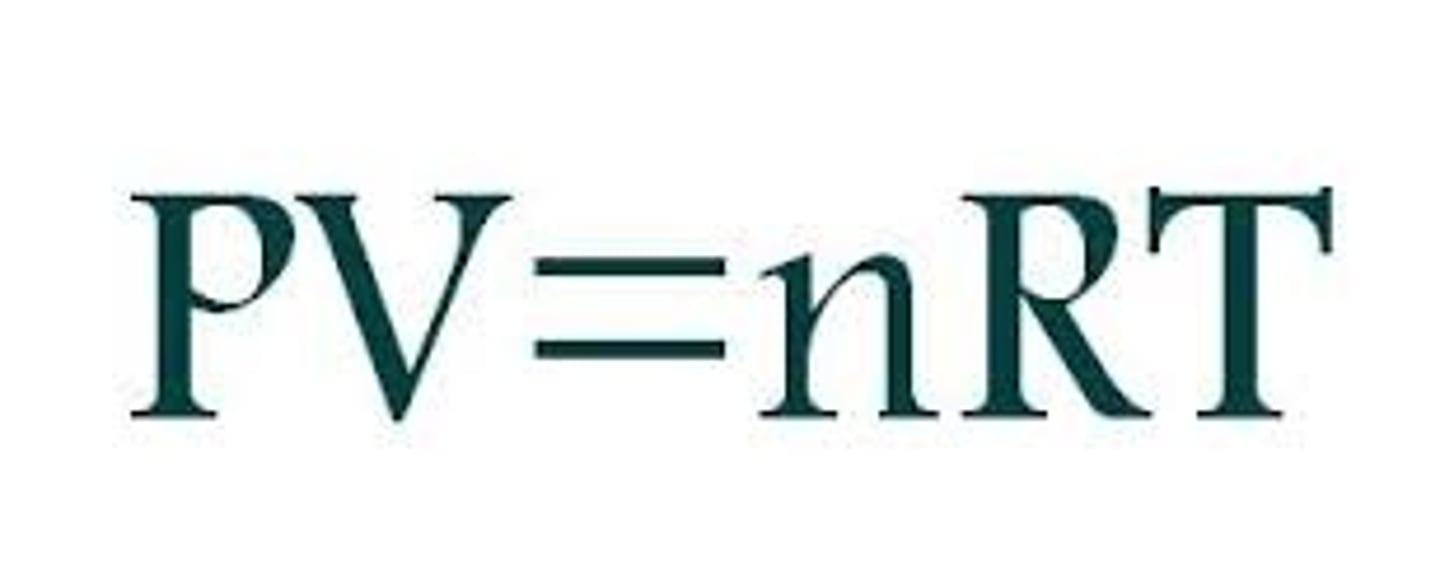
Avogadro's Law
Equal volumes of a gas contain the same number of molecules at the same temperature and pressure.
V₁n₁=V₂/n₂
State the formula for the Avogadro's Law:
Diffusion
The process by which a gas spreads or mixes with another gas.
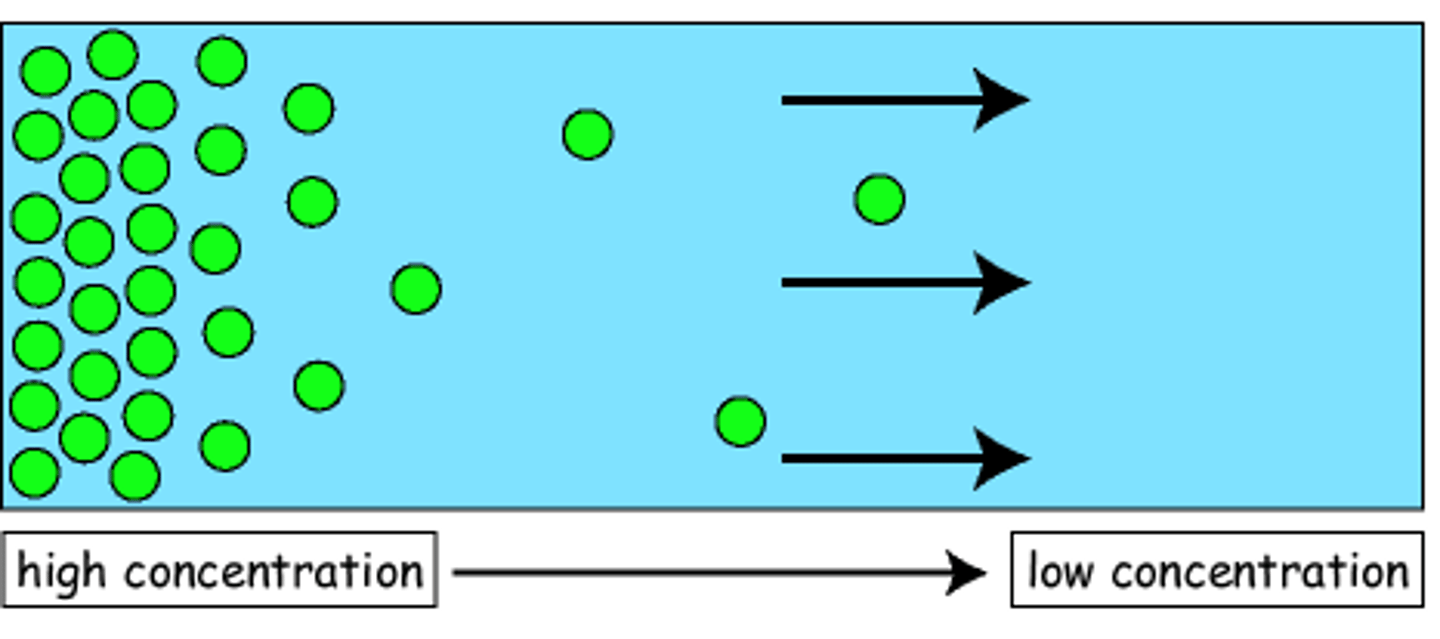
Diffusion
A result from concentration gradient.
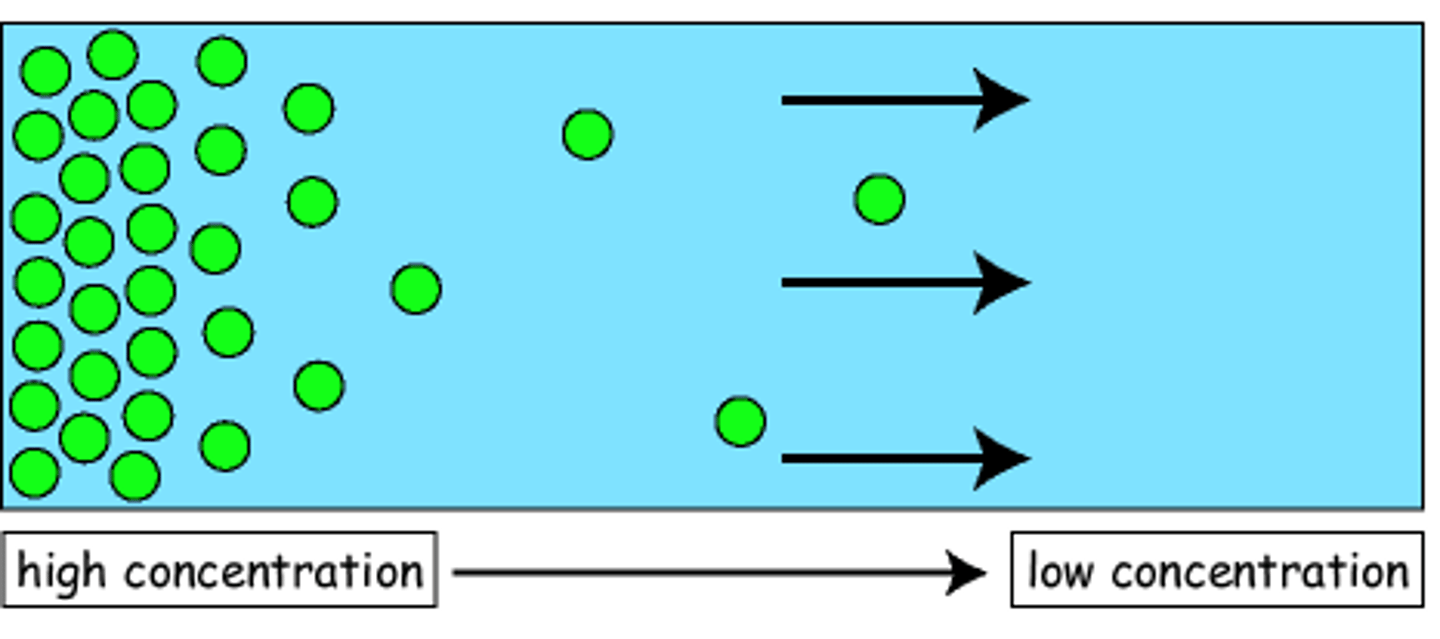
Diffusion
A gas at allocation of high concentration naturally tend to move to a lower concentration.
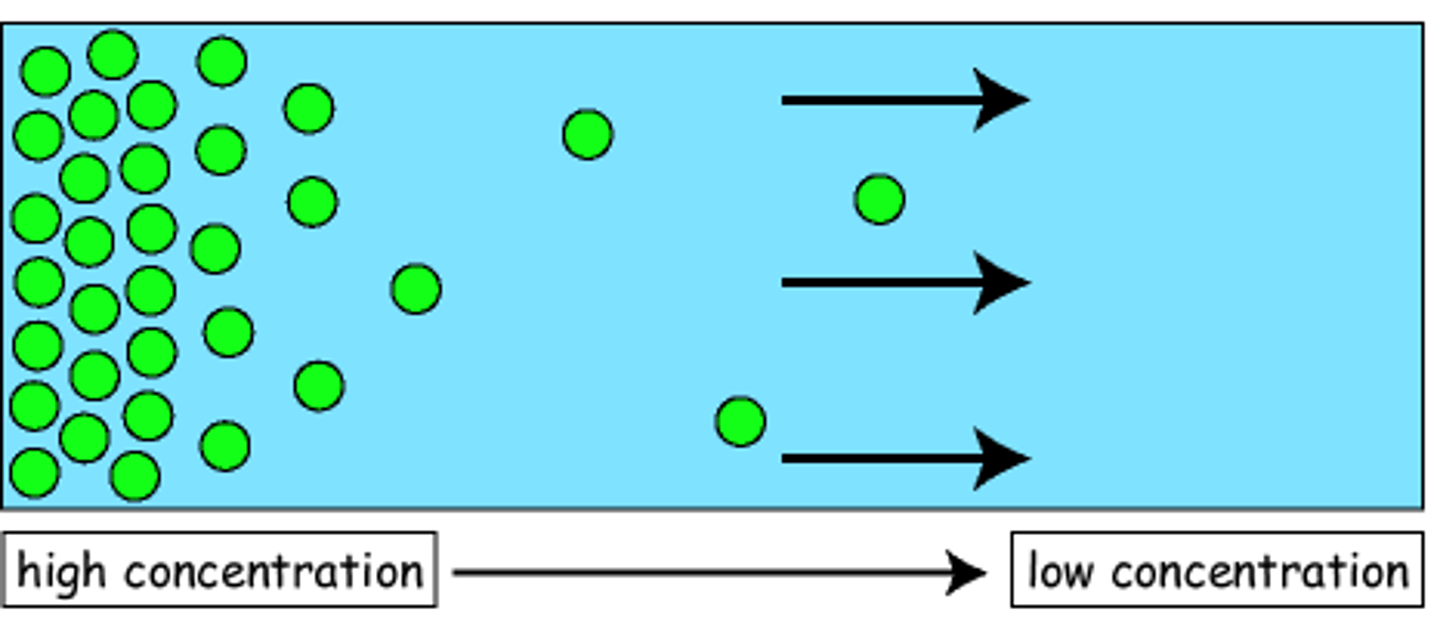
Effusion
The movement of gas particles through a pore or tiny hole.
Graham's Law of Effusion