Organic Chemistry: Reagents & Conditions
1/61
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
62 Terms
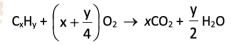
Combustion of Alkanes
R&C: excess oxygen
requires a spark: high activation energy
only burn in the gaseous state
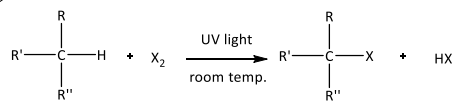
Free radical substitution
R&C: Cl₂ (g) or Br₂ (l), UV light at room temperature
Observations: Greenish-yellow / Reddish-brown decolourised
Initiation: Cl-Cl bond homolytically broken to give chlorine free radicals, with energy coming from light absorbed / heat supplied
Propagation: Highly reactive Cl• collides with methane molecule, abstracts H atom to produce HCl and •CH₃, CH₃ reacts with another molecule of Cl₂ to yield product & regenerate Cl•
Termination: Radicals needed for propagation are consumed, reaction cycle broken & chain reaction teminated
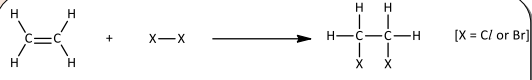
Electrophilic addition with halogens
R&C: Cl₂ / Br₂ dissolved in CCl₄, room temperature
Observation: Greenish-yellow / Reddish-brown decolourised
Non-polar Br₂ molecule approaches π electron cloud, high density of π electron cloud polarises electron cloud of Br₂
Br-Br undergoes heterolytic fission, forming bromide anion + bromide atom bonded to alkene to form (+)-charged carbocation intermediate
Bromide anion acts as nucleophile to attack carbocation intermediate to form product
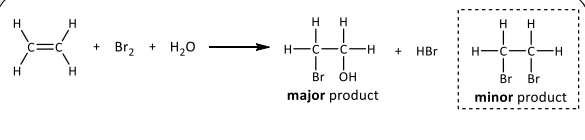
Electrophilic addition with bromine water
R&C: Br₂, room temperature
Observation: Yellow-orange decolourised
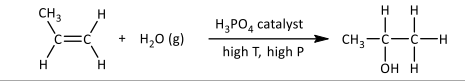
Electrophilic addition of water/steam
R&C:
industrial: steam, H₃PO₄ catalyst, high temperature & pressure
lab: concentrated H₂O, H₂SO₄, cold
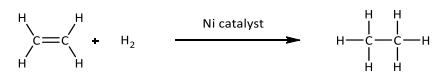
Reduction (Hydrogenation)
R&C: H₂ gas, Ni/Pt/Pd catalyst
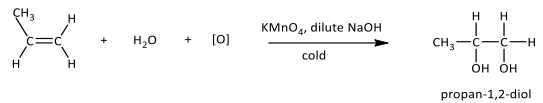
Mild oxidation
R&C: KMnO₄, dilute NaOH, cold
Observation: Purple KMnO₄ decolourised, brown precipitate of MnO₂ formed
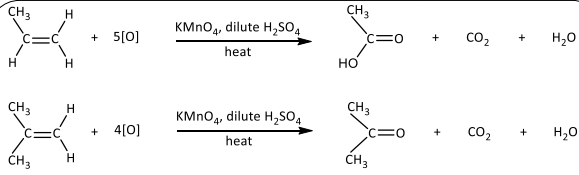
R&C: KMnO₄, dilute H₂SO₄, heat
Observation: Purple KMnO₄ decolourised, possible effervescence of CO₂
Different substrates give rise to different products:
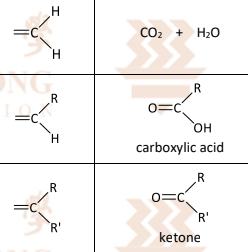
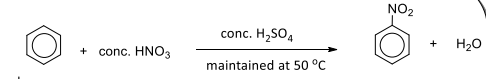
Electrophilic substitution with concentrated nitric acid
R&C: concentrated HNO₃, concentrated H₂SO₄, heat at 55℃
Observation: pale yellow oily liquid formed
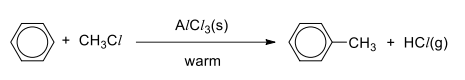
Friedel-Crafts Alkylation
R&C: RCl, anhydrous AlCl₃ catalyst, warm/heat
Observation: white fumes of HCl
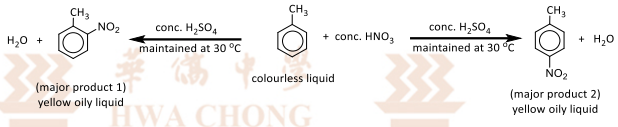
Electrophilic substitution with concentrated nitric cid
R&C: concentrated HNO₃, concentrated H₂SO₄, maintained at 30℃
Observation: yellow oily liquid formed
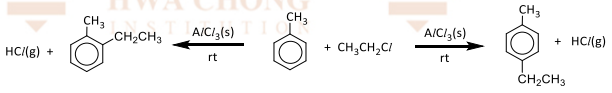
Electrophilic substitution with chlorine/bromine
R&C:Cl₂ / Br₂, AlCl/Br₃ as Lewis acid catalyst, room temperature and absence of UV
Observation: decolourisation of greenish-yellow/reddish-brown, and white fumes of HCl/HBr
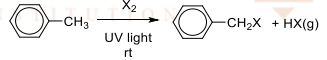
Free radical substitution (side-chain halogenation)
R&C: Cl₂ (g) or Br₂ (l), UV light at room temperature
Observation: greenish-yellow / reddish-brown decolourises slowly
limited Cl₂ / excess alkylbenzene usually used to get monohalogenation
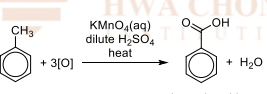
Side-chain oxidation
R&C: KMnO₄ (aq), dilute H₂SO₄, heat
Observation: purple KMnO₄ decolourises, white ppt. of benozic acid formed
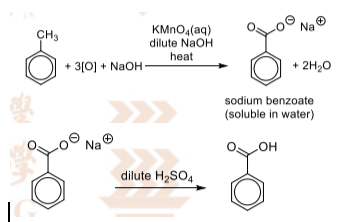
Side-chain oxidation
1 –
R&C: KMnO₄ (aq), dilute NaOH, heat
O: purple KMnO₄ decolourises, brown ppt. of MnO₂ formed
2 –
R&C: dilute H₂SO₄
O: white ppt. of benzoic acid formed
Distinguishing test for arenes and alkenes (Br₂ in CCl₄)
Add bromine in tetrachloromethane dropwise with shaking to 1cm³ of each compound in separate test tubes
Observations —
Benzene: Remains reddish brown
Alkene: Reddish-brown decolourises
Distinguishing test for alkenes and arenes (bromine water)
Add aqueous bromine dropwise with shaking to 1cm³ of each compound in separate test tubes
Observations:
Benzene: remains yellow-orange
Alkene: yellow-orange decolourises
Distinguishing test between alkenes and alkylbenzenes
R&C: KMnO₄, dilute H₂SO₄
Observations —
Alkene: effervescence of CO₂, purple KMnO₄, decolourises, no white precipitate (oxidative cleavage)
Alkylbenzene: purple KMnO₄, decolourises, white ppt. of benzoic acid
Distinguishing test for alkylbenzenes and tert-butylbenzene (3 alkyl groups attached to 1 alkyl group attached to benzene ring)
Add 5 drops of KMnO₄ to 1cm³ of dilute H₂SO₄ in test tube
To 1cm³ of each compound in separate test tubes, add 2 - 3 drops of the mixture with shaking. Warm this test tube in water bath for a few minutes
Observations —
Alkylbenzene: purple KMnO₄, decolourises, white ppt. of benzoic acid
tert-butylbenzene: KMnO₄ remains purple, no white ppt.
lacks a H atom to the benzcylic carbon
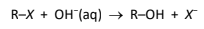
Alkaline hydrolysis
R&C: dilute NaOH, heat
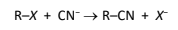
R&C: KCN in ethanol, heat
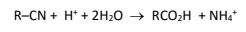
R&C: dilute H₂SO₄, heat
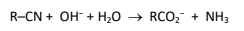
R&C: dilute NaOH, heat
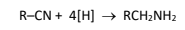
R&C: LiAlH₄ in dry ether / H₂ (g),Ni catalyst
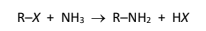
R&C: NH₃ in ethanol, heated under high pressure
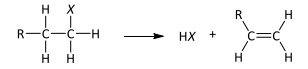
R&C: NaOH in ethanol, heat
OH⁻ behaves like base > nucleophile in ethanol, removing Hᐩ from C atom adjacent to C-X bond
Electron pair from C-H bond moves to form C=C bond
C-X breaks heterolytically to give X⁻, HX formed
Distinguishing test for different types of halogen substitutents
Add NaOH & heat
Add excess dilute HNO₃ (remove unreacted OH⁻ so brown Ag₂O ppt will not be formed in next step)
Add AgNO₃, observe colour of precipitate formed (Agᐩ(aq) + X⁻(aq) → AgX(s))
Decant and wash precipitate with deionised water, add NH₃(aq) to check for solubility
Cl: white ppt.
Br: cream ppt.
I: yellow ppt.
rate of formation: Cl > Br > I (fastest)
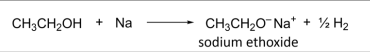
R&C: Na(s), room temperature
Observation: slow effervescence of hydrogen gas
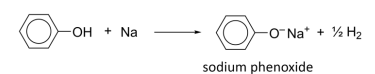
R&C: Na(s), room temperature
Observation: rapid effervescence of hydrogen gas
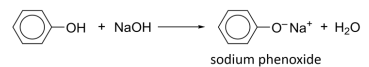
R&C: NaOH(aq), room temperature
Observation: cloudy mixture dissolves to form colourless homogeneous solution
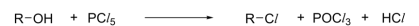
R&C: anhydrous PCl₅
Observation: dense white fumes of HCl produced
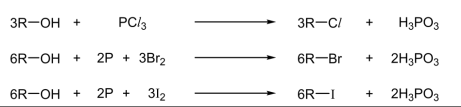
R&C: PCl₃, room temperature / P, Br/I₂, heat

R&C: concentrated HCl
Observation: solution turns cloudy
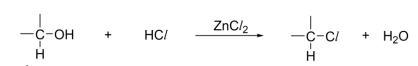
R&C: concentrated HCl, ZnCl₂, heat
Observation: solution turns cloudy

R&C: concentrated HBr / NaBr, concentrated H₂SO₄, heat
For hydrogen iodide, use concentrated HI

R&C: SOCl₂, warm
Observation: SO₂ gas, white fumes of HCl produced
distinguishing test for alcohol if no Carboxylic acid present
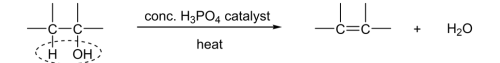
R&C: concentrated H₃PO₄ / H₂SO₄ catalyst, heat
must have at least 1 H atom on the C adjacent to the C bearing hydroxy group
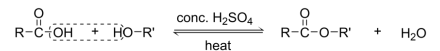
R&C: alcohol, Carboxylic acid, few drops of concentrated H₂SO₄, heat
slow and reversible

R&C: acyl chloride, alcohol, room temperature
Observation: white fumes of HCl observed

R&C: acyl chloride, phenol, room temperature
Observation: white fumes of HCl produced
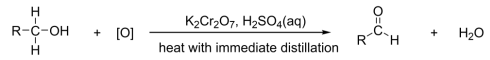
R&C: K₂Cr₂O₇, H₂SO₄, heat with immediate distillation
Observation: orange solution turns green
only K₂Cr₂O₇ selective enough to oxidize primary alcohols to aldehydes (KMnO₄ will oxidize straight to Carboxylic acid)
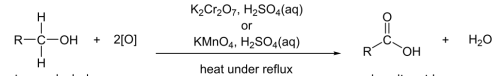
R&C: K₂Cr₂O₇ / KMnO₄, H₂SO₄, heat under reflux
Observation: orange turns green / purple turns colourless
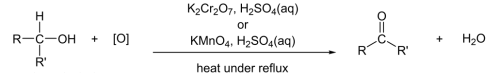
R&C: K₂Cr₂O₇ / KMnO₄, H₂SO₄, heat under reflux
Observation: orange turns green / purple turns colourless
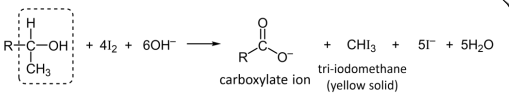
R&C: I₂, NaOH, warm
Observation: yellow ppt. formed
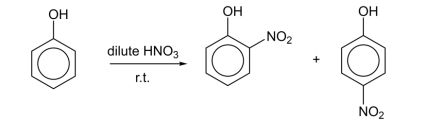
R&C: dilute HNO₃ (2,4,6-trinitrophenol if conc.)
Observation: pale yellow liquid form
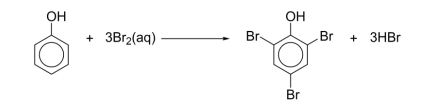
R&C: Br₂ (aq), room temp.
Observation: yellow-orange sol. decolourised, white ppt.
Mono-bromination: Br₂(l) / Br₂ in CCl₄, low temp.
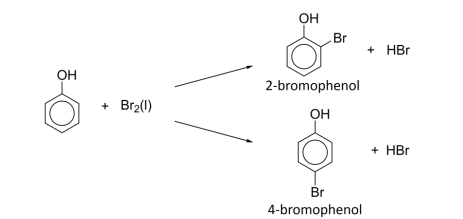
R&C: Br₂ (l) / Br₂ dissolved in CCl₄
Observation: reddish-brown liquid decolourised, white fumes of HBr evolved
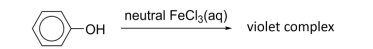
R&C: neutral FeCl₃, room temp.
Observation: violet colouration
Distinguishign test for phenols
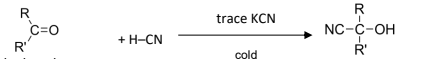
R&C: HCN with trace KCN catalyst, cold
KCN is used as a catalyst, CN- is regenerated at the end of the reaction
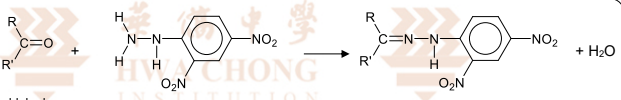
R&C 2,4-dinitrophenylhydrazine (2,4-DNPH), room temperature
Observation: orange ppt. formed
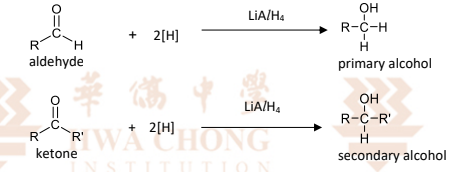
R&C: LiAlH₄ in dry ether, room temperature / H₂, Ni catalyst
alkoxide ion is first formed, then water is added subsequently to protonate the alkoxide to form the alcohol
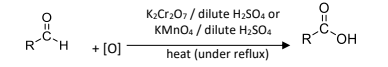
R&C: K₂Cr₂O₇ / KMnO₄, dilute H₂SO₄, heat under reflux
Observation: orange turns green / purple turns colourless
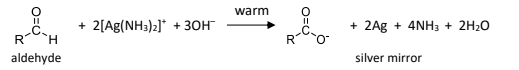
R&C: Tollen’s reagent, warm
Observation: silver mirror formed
[Ag(NH₃)₂]+ is unstable, must be freshly prepared by:
one drop of NaOH is added to 3cm³ of AgNO₃ to produce dark brown precipitate to Ag₂O
Dilute NH₃ is then added dropwise until brown precipitate first formed just dissolves

R&C: Fehling’s reagent, warm
Observation: reddish-brown precipitate formed
ketone, benzaldehyde do not reduce Fehling’s reagent
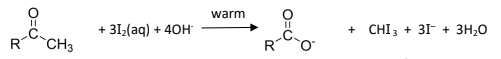
R&C: I₂, with NaOH, warm
Observations: yellow crystals of CHI₃ formed
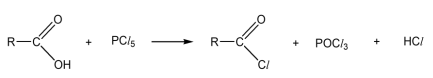
R & C: PCl₅
Observation: white fumes of HCl produced

R & C: LiAlH₄ in dry ether
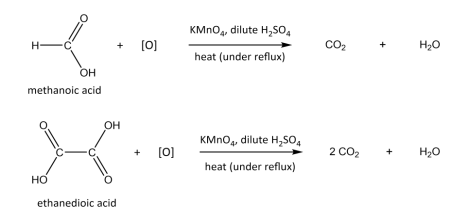
R&C: KMnO₄, dilute H₂SO₄, heat under reflux
Observation: purple solution decolourises, effervescence of CO₂ observed

R&C: water
Observation: rapid reaction forming white fumes of HCl

R&C: primary amines (in excess) + acyl chlorides
acyl chloride react readily with primary amines to form amides via condensation reaction
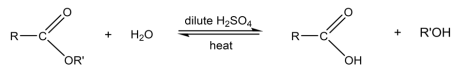
R & C: dilute NaOH, heat
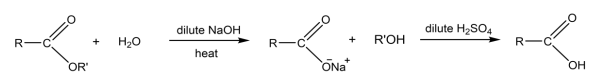
R&C: dilute NaOH, heat, followed by acidification with dilute H₂SO₄