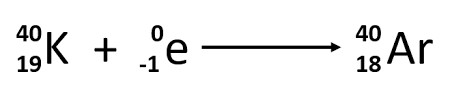Soil Layers and Geochemical Reactions
1/24
Earn XP
Description and Tags
Vocabulary flashcards covering key concepts from the geology and chemistry lecture notes.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
O Horizon
The organic layer of soil, the one closest to the top.
A Horizon
The mineral layer of soil.
E Horizon
Layer of soil made from clay, a zone of leaching.
C Horizon
Parent material in the soil.
B Horizon
Accumulation of clay transported from above.
Al2Si2O5 (Clay)
A common mineral in soil consisting of aluminum, silicon, and oxygen.
Cl2O
Water disinfectant used to purify water.
CaCl2
A simple salt that contains water and is similar to chlorine, sulfate, and carbonate.
Evaporite Reaction
A chemical process that involves the reaction of minerals to yield salts.
Flux
Substance added to reduce melting point and separate other metals.
Primary Mineral
Naturally occurring minerals with a fixed chemical formula, such as KAl2Si2O8.
Secondary Mineral
A mineral that has oxygen, water, and is a clay (silicon/aluminum oxide/hydroxide).
Volcanic Sublimate
A mineral without oxygen that contains elemental sulfur.
Dissolution Reaction
Minerals dissolve directly into water, leaving behind ions.
Hydrolysis Reaction
Water reacts with minerals, particularly silicate minerals, to form new minerals like clay.
Hydration Reaction
Minerals absorb water, causing expansion and reduced resistance to weathering.
Crust Composition
Primarily made of silicon, aluminum, and oxygen.
Acid Hydrolysis
A reaction using a strong acid to catalyze the breaking of molecular bonds by adding water.
Leaching
A hydrometallurgical process that uses chemical solutions to dissolve and extract valuable metals from ore.
Concentration Factor Equation
Percentage mass of metal in ore divided by the average crustal mass percentage.
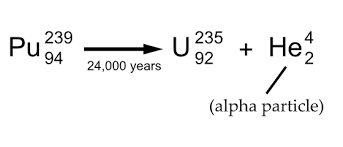
Alpha decay
A helium nucleus, subtracts 4 from the mass number (top) and 2 from the atomic number (bottom)
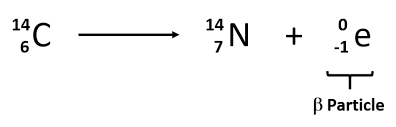
Beta decay
mass number stays the same, atomic number increases by one
Positron decay
mass number stays the same, atomic number decreases by one
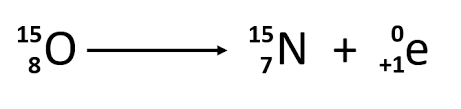
Electron capture
Mass number stays the same, atomic number decreases by one.