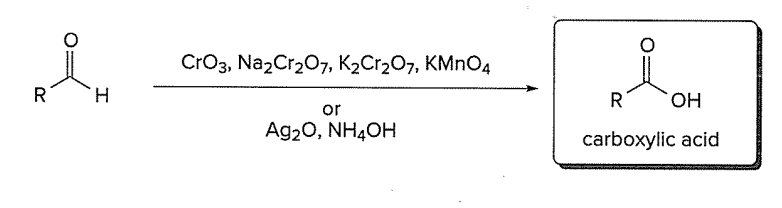Chapter 17 reduction reactions + 1 oxidation reaction
1/7
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
8 Terms
Reagents needed for Reduction of aldehyde and ketone to 1° and 2° alcohols (HINT: THER ARE 3 DIFFERENT SETS, need to know when to use which)
NaBH4, CH3OH
OR
[1] LiAlH4 [2]H2O
OR
H2, Pd-C
![<p>NaBH4, CH3OH</p><p>OR<br>[1] LiAlH4 [2]H2O </p><p>OR</p><p>H2, Pd-C</p>](https://assets.knowt.com/user-attachments/c44d2314-d5d3-4710-847b-ff89e1308470.png)
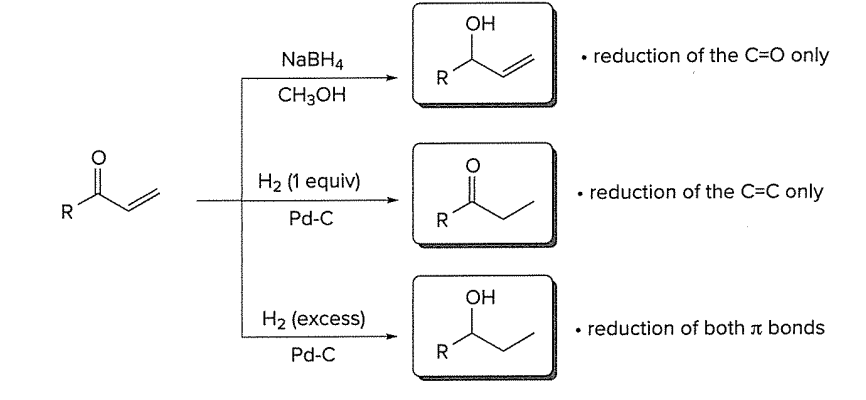
Reagents for Reduction of a,b-unsaturated aldehydes and ketones (diff reagent in diff amount for reducing different parts (C=O only, C=C only, or reduction of both pi bonds)
NaBH4, CH3OH for reduction of C=O only
H2 (1 equiv), Pd-C for reduction of C=C only
H2 (excess), Pd-C for reduction of both pi bonds
Reagents for enantioselective ketone reduction
[1] (S) or (R) - CBS reagent
[2] H2O
**ONLY FORMS A SINGLE ENANTIOMER DEPENDING ON R or S
![<p>[1] (S) or (R) - CBS reagent </p><p>[2] H2O</p><p>**ONLY FORMS A SINGLE ENANTIOMER DEPENDING ON R or S</p>](https://assets.knowt.com/user-attachments/91cf8644-a5ff-4130-ad82-a44507fc5ab7.png)
Reagents for reduction of acid chlorides (2 diff sets, one for making 1° alcohol, one for making aldehyde)
[1] LiAlH4
[2] H2O for 1° alcohol
OR
[1]LiAlH[OC(CH3)3]3
[2] H2O for aldehyde
![<p>[1] LiAlH4 </p><p>[2] H2O for 1° alcohol</p><p>OR</p><p>[1]LiAlH[OC(CH3)3]3</p><p>[2] H2O for aldehyde</p>](https://assets.knowt.com/user-attachments/2bec485a-d200-4551-a512-52ea83673694.png)
Reagents for reduction of esters (one for reducing to 1° alcohol, other for reducing to aldehyde)
[1] LiAlH4
[2] H2O for 1° alcohol
OR
[1] DIBAL-H
[2] H2O for aldehyde
![<p>[1] LiAlH4</p><p>[2] H2O for 1° alcohol</p><p>OR</p><p>[1] DIBAL-H</p><p>[2] H2O for aldehyde</p>](https://assets.knowt.com/user-attachments/f39ff99d-6dc7-4f39-8205-b863c45fc015.png)
Reagents for reduction of carboxylic acids to 1° alcohols
[1] LiAlH4
[2] H2O
![<p>[1] LiAlH4</p><p>[2] H2O</p>](https://assets.knowt.com/user-attachments/036ab7ff-e823-4924-a11a-46243806b42a.png)
reagents for Reduction of amides to amines
[1] LiAlH4
[2] H2O
(for turning amides to …)
![<p>[1] LiAlH4</p><p>[2] H2O </p><p>(for turning amides to …)</p>](https://assets.knowt.com/user-attachments/e1257f34-123e-4675-8d03-9173b800e2b4.png)
Oxidation of aldehydes to carboxylic acids
** all Cr6+ reagents except PCC oxidize RCHO to RCOOH
** Tollens reagent (Ag2O = NH4OH) oxidizes RCHO only, and does NOT react with 1° or 2° alcohols