MS - The effect of drug structure on fragmentation DBE and the nitrogen rule
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
What is mass spectrometry?
Process of ionization, separation and detection if ions according to their mass-to-charge (m/z) ratio
What are the uses of mass spec?
Molecular weight – how big?
Fragmentation/isomers
Isotope analysis
Drug metabolite analysis
What are the advantages and disadvantages of mass spec?
Advantages - smallest sample size 10-12 g, accurate mwt to 4 dec. pl, exact molecular formulae
Disadvantages - destructive technique, no sample recovery, requires expert operator, and high-cost equipment
What is ionisation?
High energy electrons impact on neutral drug to remove e- from the drug to give molecular ion = molecular weight (M)
What is fragmentation?
Charged molecular ion is a high energy radical cation that can break/fragment to give daughter ions that are separated according to mass-to-charge (m/z) ratio (M+.)
What is detected in mass spec?
Only charged structures detected, not radicals or neutrals
High vacuum required because of the highly reactive structures formed
M- and M+ or M+. and M
What is the base peak in mass spec?
The tallest, most intense peak in the spectrum, representing the most abundant ion.
It is assigned a relative abundance/intensity of 100%
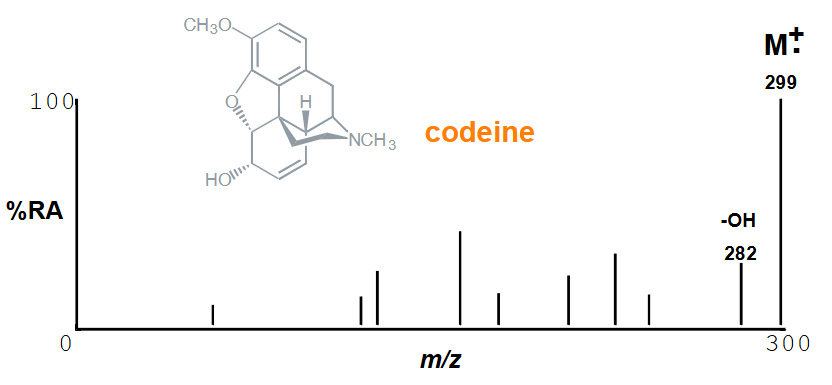
Is the M+. always the base peak and why?
No
Branching gives a weak M+. as it can fragment and break
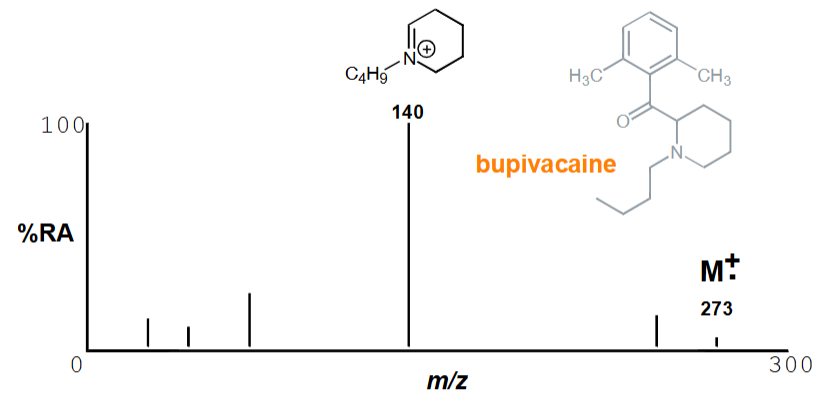
Why does CH₃CH₂Cl have 2 peaks for M+. ?
Due to Cl having 35 and 37 isotopes
In a 3:1 ratio
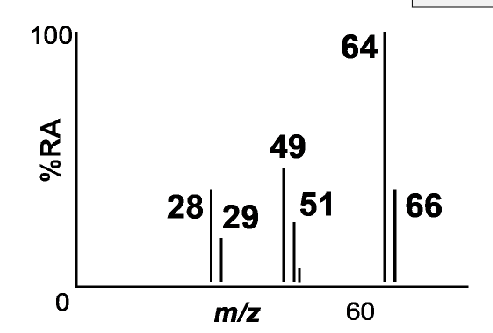
What are the 3 fragments CH₃CH₂Cl can break into?
Homolytic cleavage - radical formation, breaking C-C
CH₃CH₂Cl+. → CH2=Cl+ → m/z 49,51
Heterolytic cleavage - carbocation formation, breaking C-Cl
CH₃CH₂Cl+. → CH3CH2+ → m/z 29
Loss of neutral fragment - lose uncharged, stable molecule, HCl
CH₃CH₂Cl+. → CH2CH2+ → m/z 28
Give examples of neutral molecules that can be lost
H₂ — loss of hydrogen
H₂O — dehydration (common in alcohols)
CO — carbonyl-containing compounds
CO₂ — carboxylic acids, esters
NH₃ — amines, amides
HCl, HBr, HI — haloalkanes (e.g., chloroethane)
CH₄ — alkanes
C₂H₄ (ethene) — common in alkyl chain rearrangements
How many chlorines coresspond to number of molecular ions?
1 Chlorine Atom: Produces two molecular ion peaks with a 3:1 intensity ratio. M and M+2
2 Chlorine Atoms: Produce three molecular ion peaks with a 9:6:1 intensity ratio. M and M+2 and M+4
3 Chlorine Atoms: Produce four molecular ion peaks with a 27:27:9:1 ratio. M and M+2 and M+4 and M+6
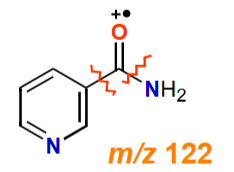
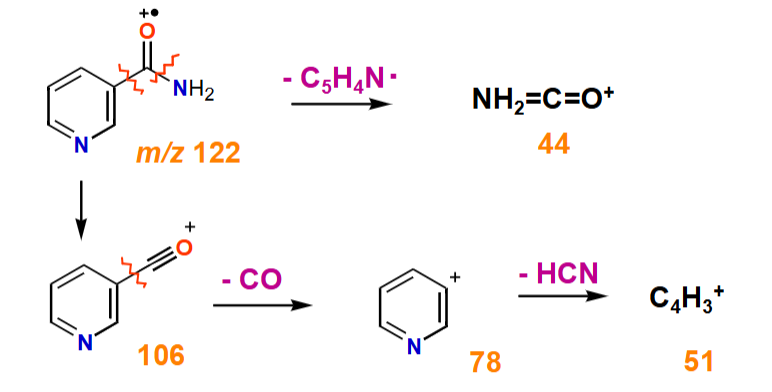
Why does nicotinamide have 2 fragmentation pathways?
Proportion of molecular ions fragment at the C-C bond
Other molecular ions fragment at the amide C-N bond
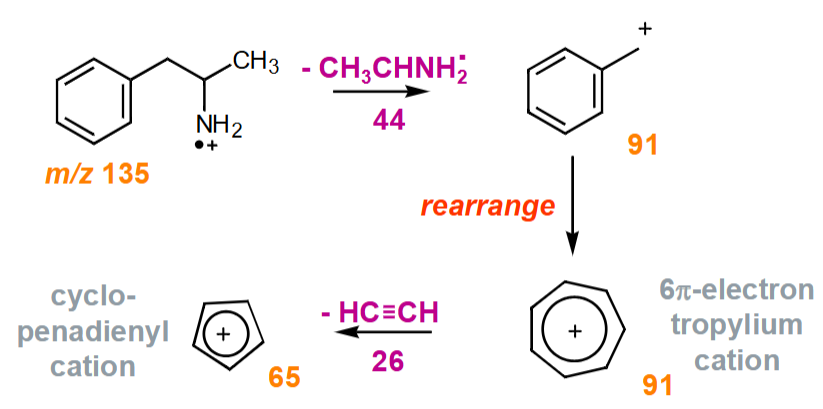
Why does amphetamine give m/z = 91 not 77 in the MS?
The fragmentation process leads to the formation of a highly stable benzyl carbocation (tropylium ion)
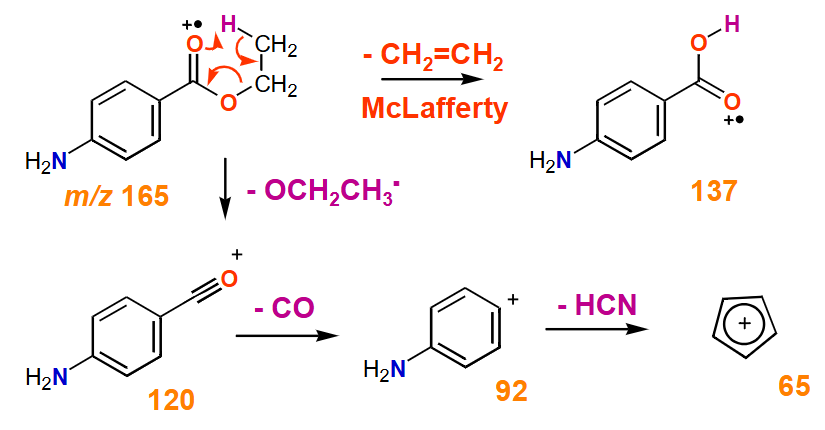
What is Mclafferty rearrangement?
When the molecular ion forms, the carbonyl oxygen abstracts the γ‑hydrogen. This occurs through a six‑membered cyclic transition state, leading to:
Transfer of the γ‑H to the carbonyl oxygen
Cleavage of the α–β bond
Formation of:
A charged enol radical cation (detected in MS)
A neutral alkene (not detected)
Happens in ketones, aldehydes, esters, and carboxylic acids that have a γ‑hydrogen.
What is combustion analysis?
Gives drug composition % C,H,N …also F,Cl,P…..
O measured by difference few mg sample needed
What is the empirical and molecular formula?
Empirical formula - gives ratio of each element using % composition from combustion analysis
Molecular formula - calculate exact number of each atom present using empirical formula and molecular weight from MS
Check if % add up to 100 to see if O is present
How do you know a drug is pure using mass spec?
For any new drug, % for all elements measured must be
within +/- 0.3 of the calculated value
What is the double bond equivalent and equation?
Total number of double bonds + rings.
Large DBE value but small number of C suggest rings present in the drug
Divalent sulphur - oxidation state = 2 → makes 2 bonds
Tetravalent sulphur - makes 4 bonds
Hexavalent sulphur - 6 covalent bonds → add 4x number of sulphurs
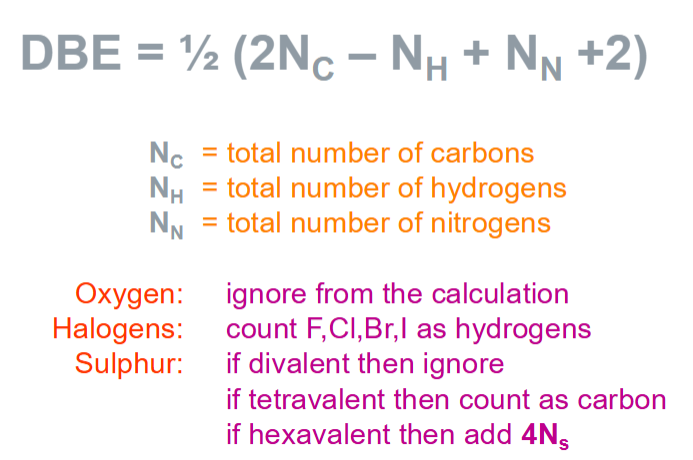

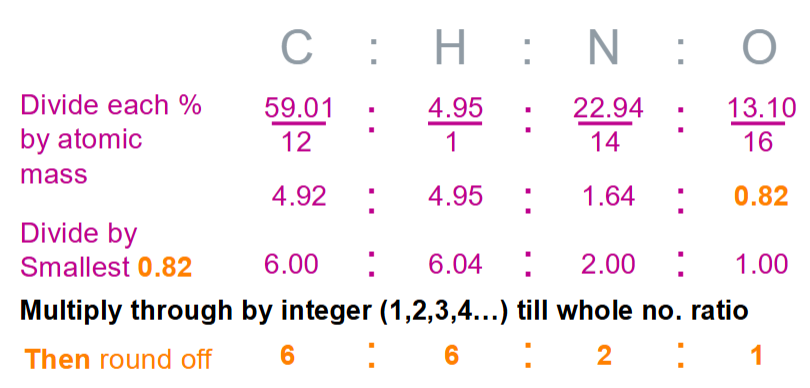
Calculate the empirical formula, molecular formula and DBE of nicotinamide
Molecular formula, M+ = 122
Empirical formula = C6H6N2O = 122
Empirical formula x 1 = Molecular formula
DBE
½ (2NC - NH + NN + 2) = ½ ((2 x 6) – 6 + 2 + 2) = ½ (10) = 5
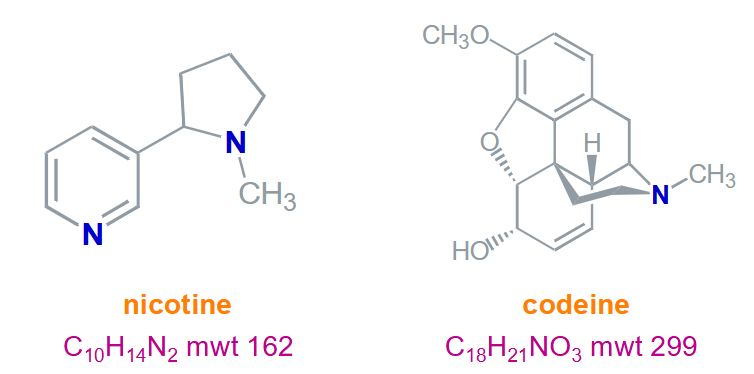
What is the nitrogen rule?
Even number of nitrogens, even molecular weight
Odd number of nitrogens, odd molecular weight (0 is even)

E