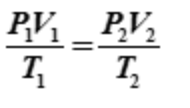chem!
1/72
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
73 Terms
K
C + 273.15
1mL
= 1cm³
1Hz
= 1/s or 1s^-1
1J
= 1kg m²/s²
avogrado’s number
6.022 × 10²³/mol
speed of light in vacuum
3.000 × 10^8m/s
density
d = m/v
kinetic energy
KE = 1/2mv², photon energy - binding energy
frequency and wavelength
v=frequency, c=speed of light, ^=wave length
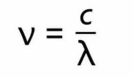
energy of a photon

de broglie relation

M
10^6
k
10³
d
10^-1
c
10^-2
m
10^-3
u
10^-6
n
10^-9
p
10^-12
precision
closeness of all values to each other
accuracy
closeness to actual value or target
multiplying or dividing sig figs
result is same number of sig figs as number with least amount of sig figs
adding or subtracting sig figs
results has same number of decimal places as number with least amount of decimal places
intensive properties
dont change with the amount of a substance
density, temp, color
extensive properties
changes with the amount of a substance, number measures the substance
mass, volume, energy
energy
capacity to do work
work
action of a force applied across a distance
w = F(d)
endothermic reaction
heat enters system, energy of system increases, temp in surrounding decreases
exothermic reaction
heat exits system, energy of system decreases, temp in surrounding increases
pure substances
elements or compunds
element
on periodic table, cannot be separated by chemical means
compund
combination of elements with fixed proportions, can be separated by chemical means
homogenous mixture
uniform, solute fully dissolve, can be separted by physical means
heterogenous mixture
not uniform, solute not fully dissolved, can be separated by physical means
law vs theory
law: summarizes past observations and predicts future ones
theory: model that explains why nature is the way it is
atomic theory
created by john dalton
each element composed of atoms
all atoms of given element have same mass and characteristics (we now know about isotopes)
atoms combine into simple, whole number ratios
atoms in one element cannot change into atoms of another element (we now know about fission and fusion)
electric field
a region where an electrically charged particle experiences a force
magnetic field
a region where a magnetized particle experiences a force
amplitude
amount of photons, measure of light intensity
wavelength
determines color
longest: red, low energy, low frequency
shortest: violet, high energy, high frequency
photon energy (E)
proportional to frequency, can only be positive
E = hv, E = hc/lambda
constructive interference
waves line up in phase when they meet and create a larger wave, makes light brighter
destructive interference
waves are out of phase when they meet and cancel each other out, makes it dark
diffraction
behavior that only happens with waves, not particles
threshold frequency
minimum frequency needed before electrons are emitted
binding energy
energy required to remove electron from metal
atomic spectra
unique patterns of light emitted or absorbed by atoms as their electrons move between energy levels
bohr model
electrons orbit nucleus in quantized energy levels, electrons can move between these levels by emitting or absorbing photons
n
number of orbitals
5d n=5
3s n=3
L
shape of orbital
0=s, 1=p, 2=d, 3=f
always less than n
m
orientation of orbital
-L to L
ms
electron spin
spin up = +1/2
spin down =-1/2
node
where probability density drops to 0
nodes = L
the more nodes the higher the energy
solution dilution
m1v1=m2v2
heat capacity
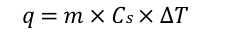
bomb calorimeter
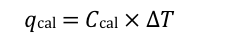
enthalpy from bond energies
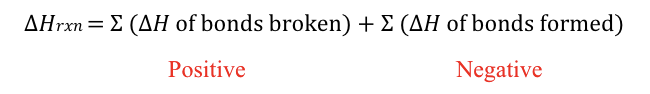
standard enthalpy
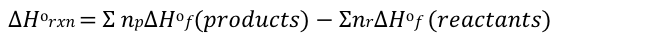
ideal gas law
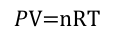
dalton’s law
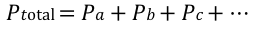
mole fraction
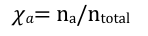
pressure
collisions with the walls
temperature
how fast gas particles are moving
dalton’s law
total pressure equals sum of each gas
mole fraction
what percent of the mixture is this gas
gas collected over water
gas pressure= total-water vapor
graham’s law
lighter gas is faster
ideal gasses
no volume and no attractions
high pressure, low temp= less ideal
hess’s law
if reactions add, their heat change values also add
if you flip reactions, you have to flip sign
lattice energy
stronger attraction when charges are bigger and when ions are smaller
stronger attraction=more energy
diffusion
mixing of gasses from areas of high to low concentration
effusion
escape of gas particle through a tiny hole into a vacuum or lower concentration area
combined gas law