Part 5a - 2026 Vascular intro, OCT, OCTA, FANG, photocoagulation, MAs, retinal hemes
1/63
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
64 Terms
What is the normal permeability characteristic of retinal arteries?
Retinal arteries form a tight, non‑leaky barrier due to the inner blood-retinal barrier, so they should not leak under normal physiologic conditions.
Where are retinal arteries located relative to retinal layers, and what structural attachment do they have?
Arteries within the nerve fiber layer have strong anchoring connections to the internal limiting membrane (ILM), which is in turn attached to the vitreous cortex.
Why are retinal arteries prone to rupture during posterior vitreous detachment (PVD)?
Because arteries in the nerve fiber layer are tightly adherent to the ILM-vitreous interface, traction during PVD can cause these vessels to break, leading to vitreous hemorrhage.
Why are retinal veins more prone to bleeding than retinal arteries?
Retinal veins have thin walls with elastic tissue and lack the strong barrier characteristics of arteries, making them more susceptible to leakage and bleeding under stress or increased venous pressure.
What happens at retinal arterial‑venous (A/V) crossings, and why is this clinically relevant?
The arterial and venous walls share a common adventitial sheath at A/V crossings. This can lead to venous compression at crossings-important in disorders like branch retinal vein occlusion (BRVO).
Which retinal layers are supplied by the central retinal artery, and through how many capillary networks?
The central retinal artery supplies the inner two‑thirds of the retina via three capillary networks (superficial, intermediate, and deep capillary plexuses).
Why is the RPE crucial in the retina?
Barrier between the sensory retina & choroid (it prevents new, leaky blood vessels in the choroid from leaking into the retina)
Processes metabolites for the retina (RPE is essential for maintaining the photoreceptors)
Absorbs & neutralizes the toxic effects of UV light
What is Bruch’s membrane and where is it located?
Bruch’s membrane is the basement membrane of the RPE, forming a key interface between the RPE and the choriocapillaris.
Important in diseases like AMD where it thickens or accumulates drusen.
What structural feature of the choriocapillaris allows for efficient metabolic exchange with the retina?
It is highly fenestrated, creating a sponge‑like effect that facilitates rapid nutrient exchange and metabolic clearance for the outer retina.
What region of the retina has the greatest hemodynamic activity, and why does this predispose the macula to vascular disease?
The fovea has the highest blood‑flow demand and the densest choriocapillaris.
→ Therefore, the macula is frequently involved in disorders affecting retinal or choroidal blood supply (e.g., AMD, macular ischemia).
What are the two major physiologic functions of the choroid?
Supplies nutrition to the RPE and outer third of the retina (including photoreceptors).
Disperses heat generated by light absorption in the RPE and from high metabolic activity of photoreceptors.
What is Venous Pulsation?
Rhythmic collapse and re‑expansion of the central retinal vein at the optic disc
It is present in most adults, perfectly normal; occurs because the walls of the veins are thin
Venous pressure is about 11 mm (close to IOP)
What is arterial pulsation?
Rhythmic collapse and re-expansion of the central retinal artery at optic disc
It is unusual, abnormal, and often indicates underlying systemic disease
Indicates IOP is > ophthalmic artery pressure (usually about 30-40mm)
IOP is very high
ophthalmic artery pressure is very low
How do microaneurysms and intraretinal hemorrhages appear on OCT?
As hyper-reflective spots within the retina
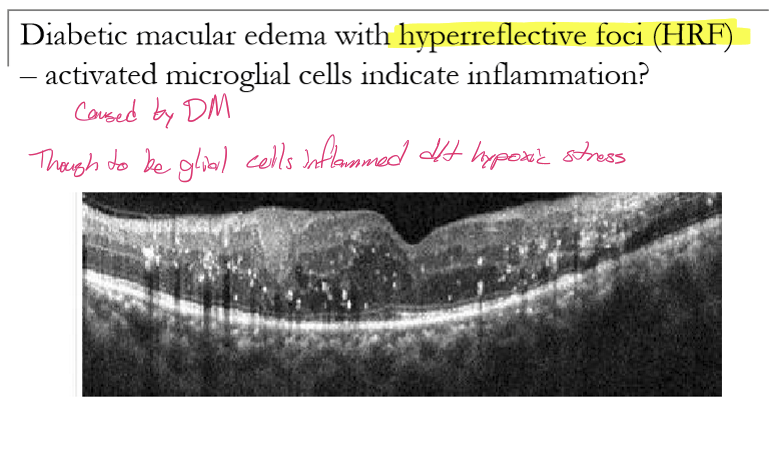
What are hyperreflective foci (HRF) seen on OCT in diabetic macular edema?
Small, punctate, hyperreflective dots on OCT without posterior shadowing. They appear within the retinal layers in DME.
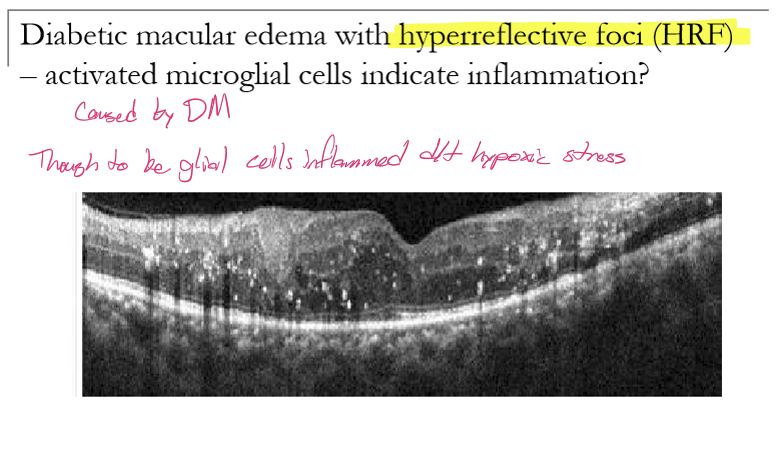
What do HRF represent histologically/biologically in diabetic macular edema?
HRF correspond to activated microglial cells, indicating retinal inflammation.
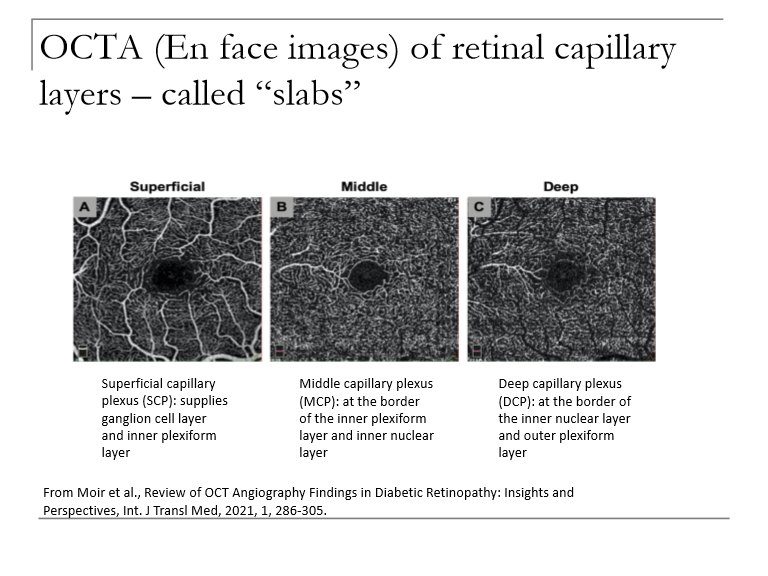
What retinal layers can be visualized on OCT angiography (OCTA)?
OCTA provides en face, high‑resolution maps of the retinal:
Superficial capillary plexus (SCP)
Middle capillary plexus (MCP)
Deep capillary plexus (DCP)
What principle allows OCTA to map retinal and choroidal vasculature?
OCTA acquires multiple sequential OCT images at the same location.
Differences between scans arise from moving red blood cells, and this motion contrast is used to create a vascular map of retina and choroid without dye.
What types of vascular pathology can be identified using OCTA?
OCTA can show:
Changes in vessel shape/architecture
Capillary dropout or non‑perfusion
Abnormal new vessels (neovascularization)
What important vascular findings seen on fluorescein angiography (FA/FANG) are not visible on OCTA?
OCTA does not show:
Actual vascular leakage
Hemorrhages
Hard exudates
Areas of dye pooling or staining
What can the OCTA has some advantages over Fluorescein angiography (FANG)?
Main advantage is no worry about anaphylaxis which can occur in response to the fluorescein injection in FANG
FANG may be “skewed” so that changes in the SCP are seen more easily than changes in the MCP and DCP
Why is OCTA not a substitute for FANG?
Periphery may be difficult to view with OCTA compared to ultra-widefield FANG, however ultra-widefield OCTA is in rapid development
OCTA does not show vascular flow or lesions associated with vascular leakage (ie hemes, hard exudates) the way that FANG does
OCTA has lots of artifacts:
motion artifacts can generate stretched or duplicated vasculature
Segmentation artifacts: inappropriate segmentation of teh capillary layers so that the boundaries of the layer are misinterpreted: may appear there is vasculature in layers where it is not expected
Projection artifacts: superficial vessels are projected into deeper layers so that the same pattern of vessels could appear for 2 different layers
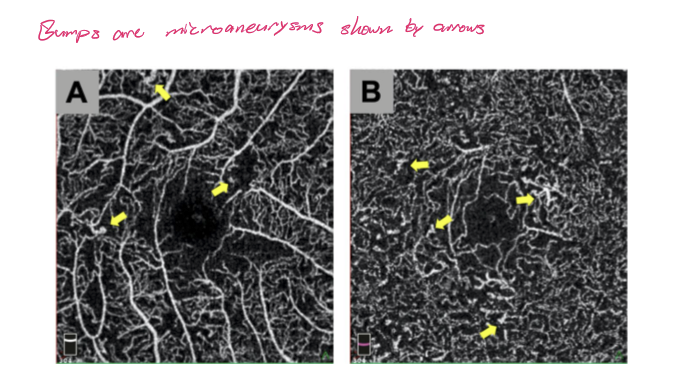
In diabetic vascular changes, what is seen adjacent to areas of capillary nonperfusion/capillary drop-out on OCTA?
Microaneurysms
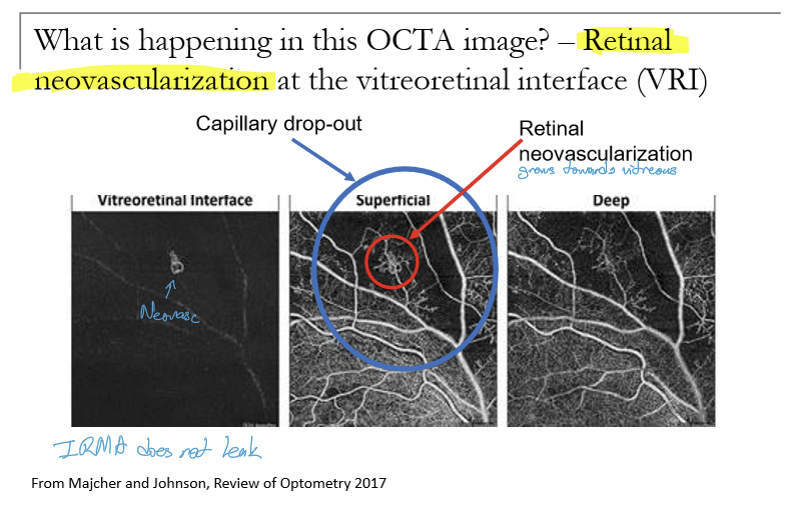
What does retinal neovascularization look like at the VRI on OCT?
A bundle of BV that go nowhere.
What can OCTA quantify?
Foveal avascular zone (FAZ), Vessel density (VD), and fractal dimension (FD)
What happens to the foveal avascular zone (FAZ) on OCTA as diabetic retinopathy or macular ischemia progresses?
The FAZ enlarges with disease progression.
→ Increased FAZ size indicates worsening macular ischemia and capillary rarefaction.

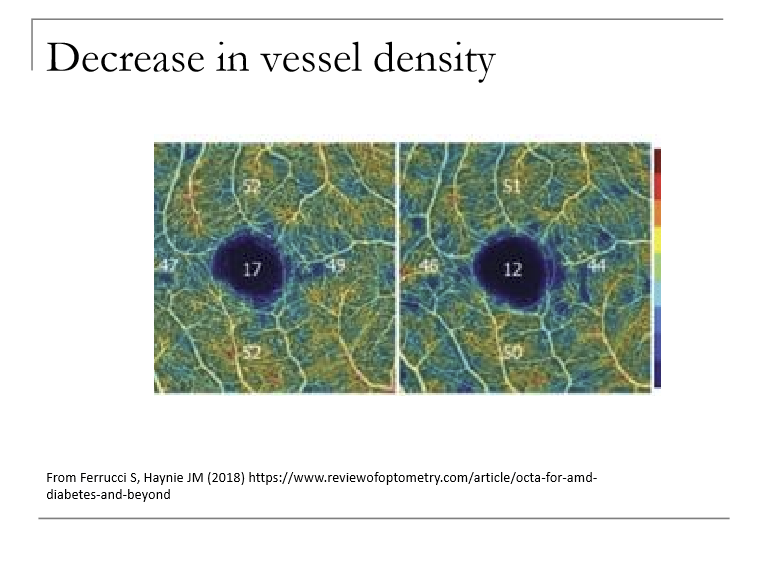
How does vessel density (VD) change on OCTA with progression of retinal vascular disease?
Vessel density decreases with progression.
Reflects capillary dropout, non‑perfusion, and ischemia, especially in the deep capillary plexus.
What is fractal dimension (FD) on OCTA, and how does it change with disease progression?
FD measures how often a similar vascular branching pattern repeats (a marker of vascular complexity).
FD decreases with progression → indicates loss of vascular complexity and increased ischemia.
What is the primary clinical role of fluorescein angiography (FA/FANG/IVFA) in modern retinal practice?
FA is very important for managing retinal vascular diseases, especially in diabetic retinopathy, though it is less essential for diagnosis now due to OCT and OCTA.
It helps evaluate leakage, non‑perfusion, neovascularization, and treatment planning.
What is the basic optical principle behind fluorescein angiography?
Sodium fluorescein, when excited by blue light, emits green/yellow‑green light (fluorescence).
This emitted light is captured to visualize retinal and choroidal circulation.
How does the fundus camera capture fluorescein angiography images?
A white flash is filtered through a blue excitation filter.
Blue light strikes fluorescein in the blood.
Fluorescein emits green light back from the fundus.
This emitted light passes through a barrier filter, allowing only the fluorescence signal to reach the camera.
How is sodium fluorescein delivered during fluorescein angiography?
Injected into an antecubital vein in the arm, allowing rapid entry into the ocular circulation for imaging.
What is the imaging protocol during fluorescein angiography?
Serial fundus photographs are taken:
Every second for the first ~15 seconds (to capture early arterial and arteriovenous phases).
Then at decreasing frequency over the next 2–3 minutes (to capture venous and recirculation phases).
Late frames (~15 minutes) may be taken to evaluate late leakage, staining, or pooling.
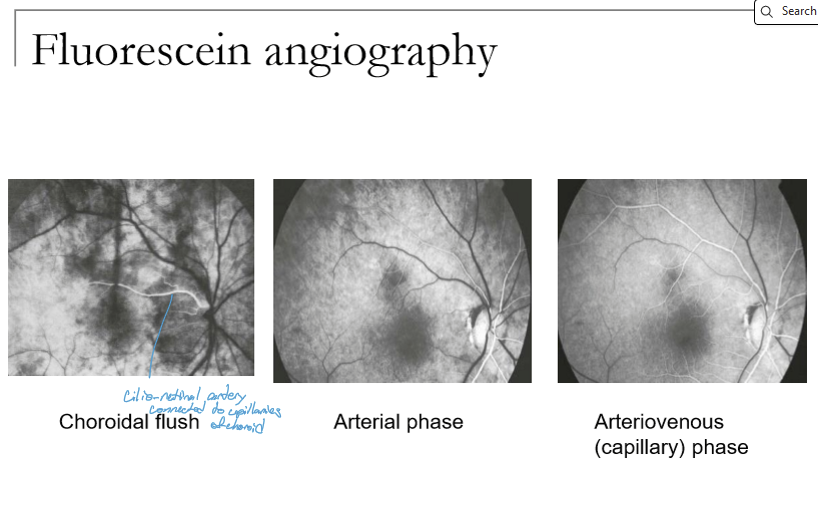
What is the “choroidal flush” on fluorescein angiography, and when does it occur?
The choroidal flush is early filling of the choroidal vasculature with fluorescein, occurring ~10–12 seconds after injection.
The choroid fills first, followed by the fenestrated choriocapillaris, which shows normal early leakage.
What are the main retinal vascular phases of FA after the choroidal flush?
Retinal circulation follows these phases:
Arterial phase
Arteriovenous (capillary) phase
Early venous phase
Mid‑venous phase
Late venous phase
Late fade of both retinal and choroidal fluorescence
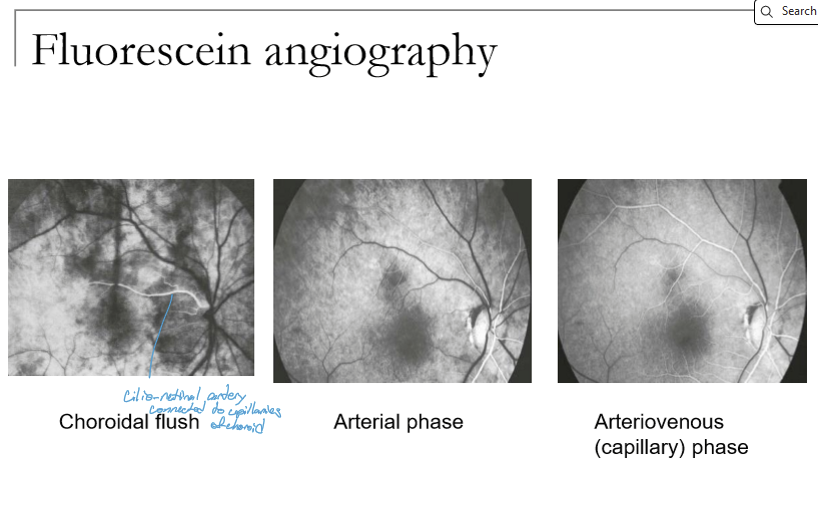
What occurs during the arterial and arteriovenous phases of FA?
Arterial phase: Begins 2–3 sec after choroidal flush as retinal arterioles fill.
Arteriovenous (capillary) phase: Overlaps with arterial; capillaries fill but can be hard to distinguish separately.
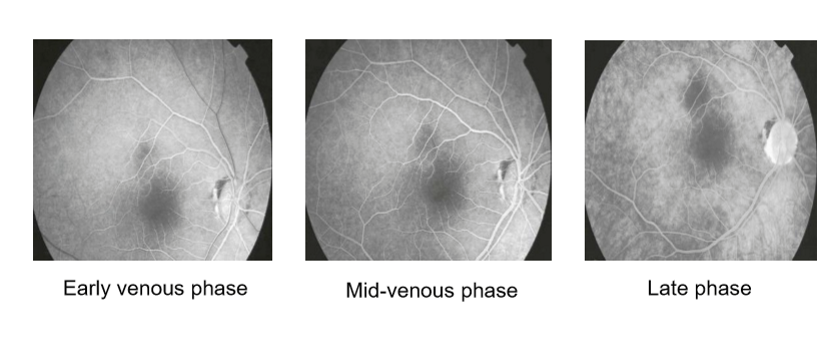
What characterizes the early, mid, and late venous phases in fluorescein angiography?
Early venous phase: Begins 13–15 sec, laminar venous filling; veins fully fill by ~45 sec.
Mid‑venous phase: Arterioles fade; veins continue to fill.
Late venous phase: Arterioles pale; veins fully filled; fluorescence gradually fades from both retina and choroid.
How do healthy retinas look on fluorescein angiography?
Healthy retinal vessels do not leak fluorescein (no fenestrations)
Healthy choriocapillaries vessels are fenestrated and leak freely
RPE filters chorodial glow: if the RPE is absent, more glow (hyperfluorescence) is visible, if there is excess RPE, less glow (hypofluorescence) is visible
What are the two major causes of hyperfluorescence on fluorescein angiography?
1. Leakage: Fluorescein (and fluid) escaping from vessels due to breakdown of the blood–retinal barrier (e.g., DME, CNV, vasculitis).
2. RPE window defect: Decreased RPE pigment allows more choroidal fluorescence to shine through → increased background glow (not actual leakage).
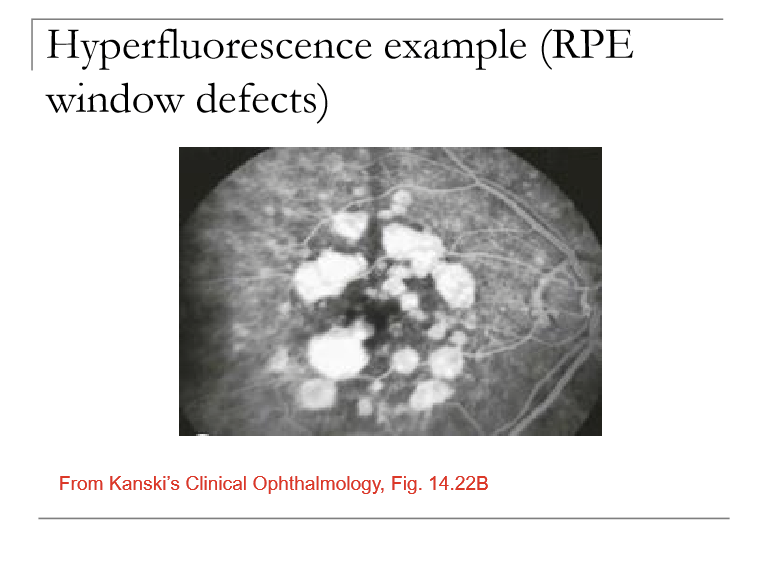
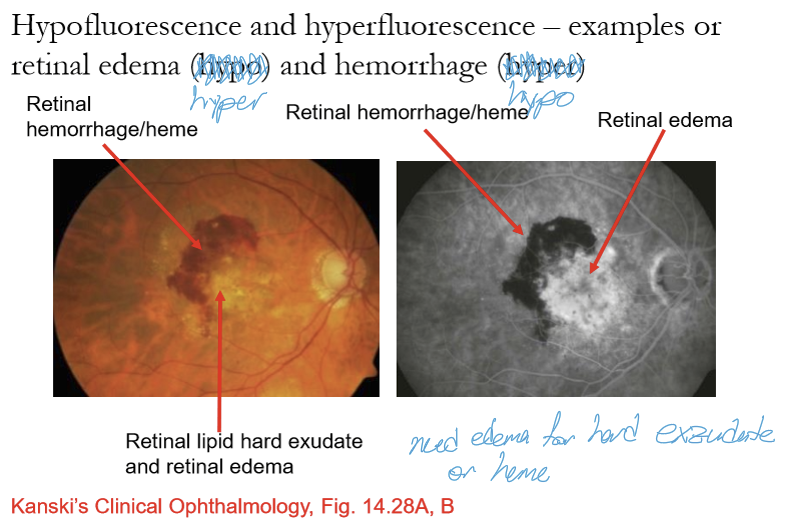
What are the major causes of hypofluorescence on FA?
Vessel closure / non‑perfusion → no fluorescein entering the capillary bed.
Blockage from pigment or blood (melanin, hemorrhage, CHRPE).
Cotton wool spots: axoplasmic stasis blocks underlying choroidal fluorescence.
When is laser photocoagulation performed?
After anti-VEGF infections fail/not very effective
How does laser photocoagulation work at the tissue level?
Most laser light is absorbed by the RPE and choroid, generating heat that coagulates these layers.
This thermal effect then coagulates the overlying retina, reducing oxygen demand and sealing leaking vessels or breaks.
What are the major types of retinal laser photocoagulation?
Three main types:
Focal laser
Grid laser
Panretinal photocoagulation (PRP)
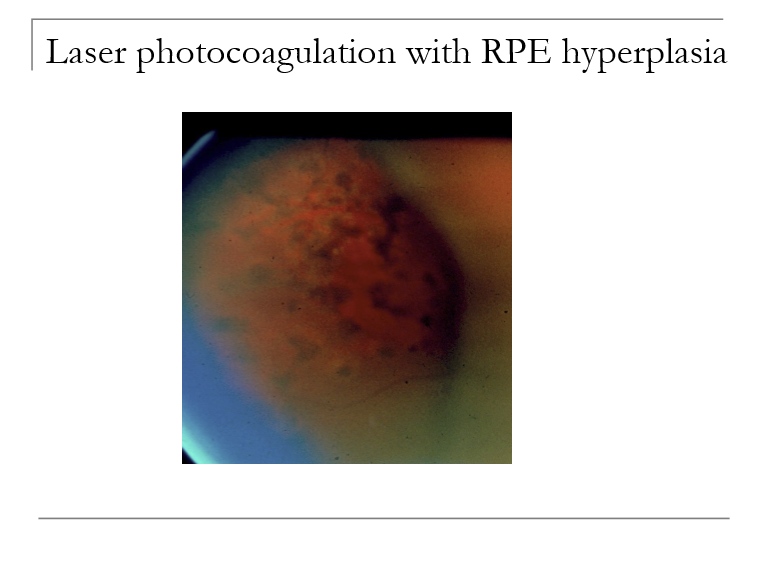
What is the difference between focal and grid laser photocoagulation?
Focal laser: Applied to localized leaking microaneurysms or specific retinal breaks.
Grid laser: Used for diffuse leakage or diffuse macular edema where focal points can't be targeted individually.
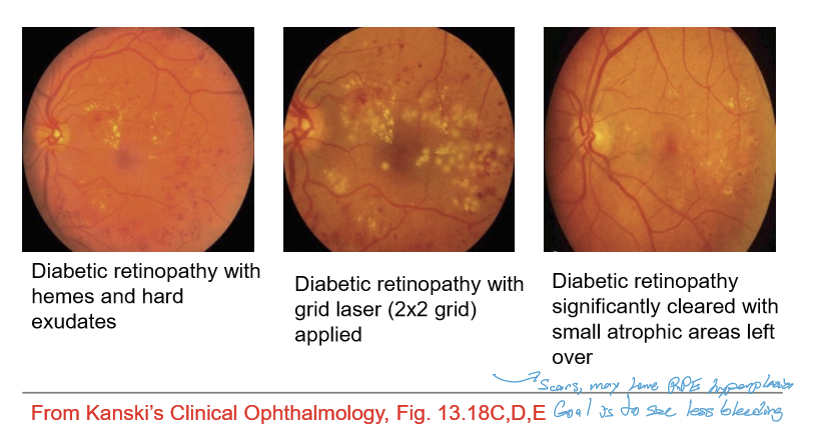
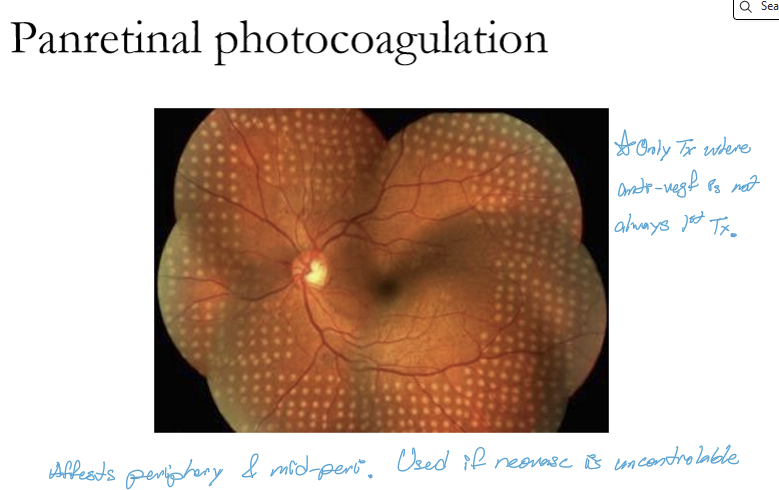
When is panretinal photocoagulation (PRP) indicated?
PRP is used when there is widespread retinal hypoxia, leading to neovascularization, such as in proliferative diabetic retinopathy or retinal vein occlusions with NV.
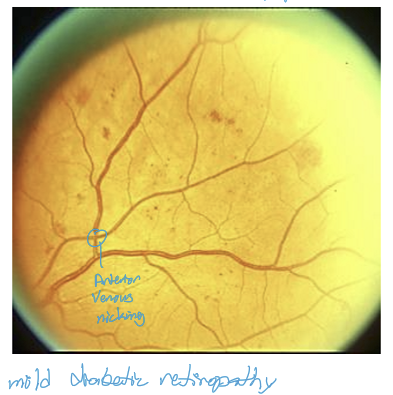
What are retinal microaneurysms and how do they form?
Retinal microaneurysms are saccular outpouchings of capillary walls that occur where the capillary wall is thinned or weakened.
They represent the earliest structural sign of retinal microvascular damage, especially in diabetes.
What is the typical appearance of retinal microaneurysms, and how are they detected?
They are very small, round, red dots adjacent to retinal vessels.
They may be clinically visible, or subclinical, requiring OCT, OCT‑A, or fluorescein angiography for detection.
What types of leakage can occur from retinal microaneurysms?
Blood (causing dot/blot hemorrhages)
Fluid (causing edema)
Maybe hard exudates/lipids
What is the most common systemic cause of retinal microaneurysms?
Diabetes mellitus. Microaneurysms are often the first or one of the earliest clinical signs of diabetic retinopathy.
What do retinal hemorrhages require?
They requires a systemic evaluation most of the time
How do peripheral retinal hemorrhages typically present?
Peripheral retinal hemorrhages can be small or large, often appearing as red, blot-like lesions in the far periphery. Their appearance can vary depending on depth (NFL, INL, subretinal, etc.).
What are common causes of peripheral retinal hemorrhages?
Can result from:
Any retinal vascular disease (diabetes, RVO, hypertension)
Valsalva maneuver (sudden ↑ intrathoracic pressure)
Vitreoretinal traction (PVD, lattice degeneration)
Idiopathic cases (may be part of normal peripheral degeneration)
Under what specific conditions might a small peripheral retinal hemorrhage not require systemic vascular work‑up?
If ALL the following are true:
Fewer than 4 hemorrhages
Peripheral location
Small (≈ diameter of a retinal vessel)
Patient > 50 years old
Then the hemorrhages may reflect benign peripheral retinal vascular degeneration, and some clinicians may choose not to refer.
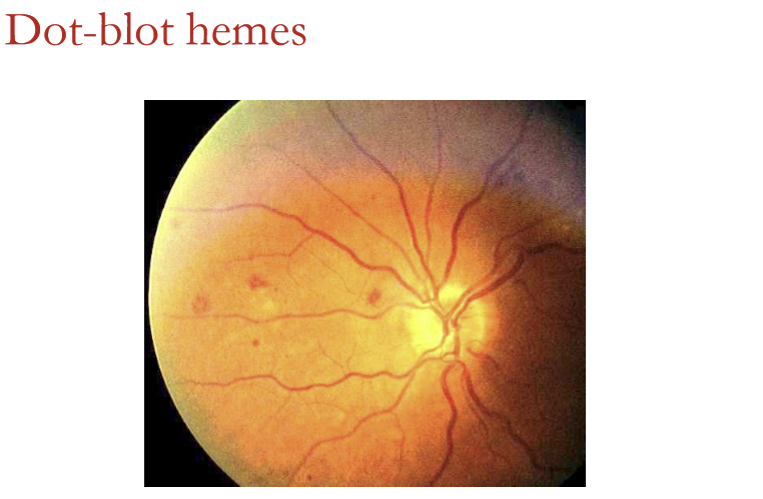
When should a patient with a peripheral retinal hemorrhage be referred for systemic vascular evaluation?
Refer unless all benign criteria are met.
If the patient is younger, has ≥4 hemorrhages, larger lesions, non‑peripheral location, or other findings → systemic vascular work‑up is recommended (e.g., diabetes, hypertension, vascular disease).
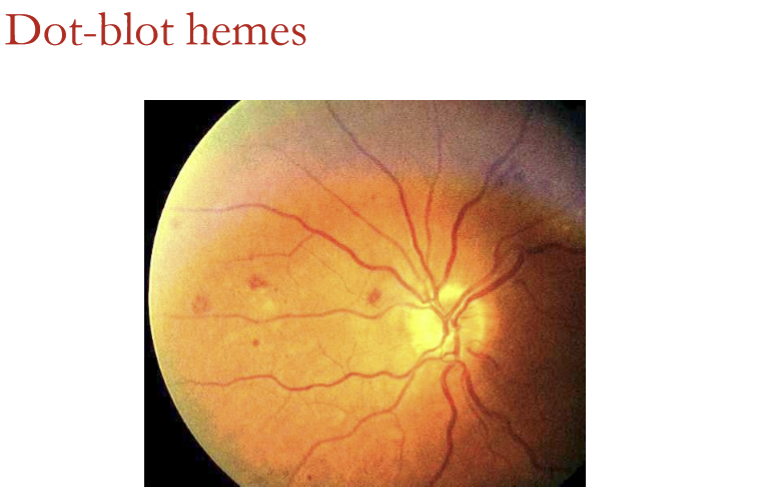
In which retinal layers do dot‑blot hemorrhages form?
Dot‑blot hemorrhages occur in deeper retinal layers:
Inner nuclear layer (INL)
Outer plexiform layer (OPL)
Sometimes the outer nuclear layer (ONL)
They follow the contours of retinal architecture, giving them their round/blot-like shape.
What diseases cause isolated dot‑blot hemes versus sectoral or diffuse involvement?
Isolated dot‑blot hemes: commonly seen in diabetes (non‑proliferative DR).
Sectoral involvement: seen in branch retinal vein occlusion (BRVO) or hemi‑CRVO.
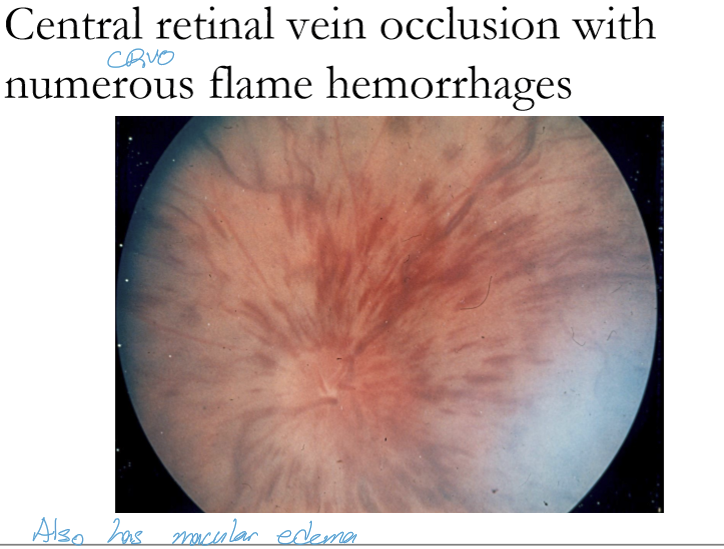
Diffuse/widespread: almost the entire retina can be involved in central retinal vein occlusion (CRVO).
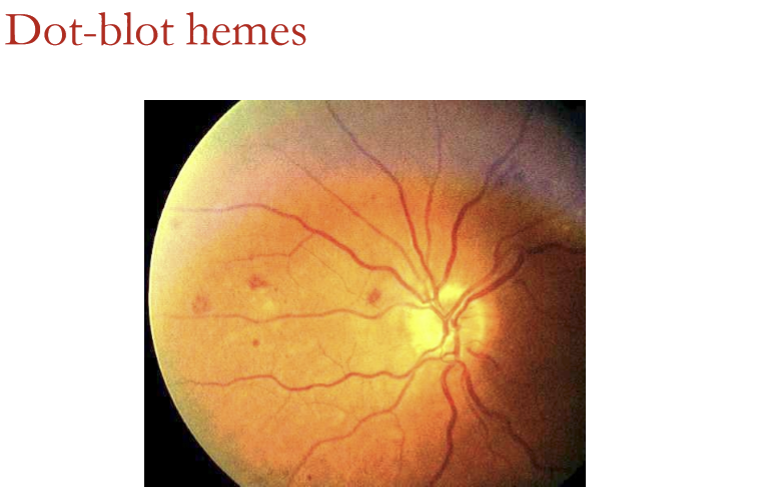
What does dot-blot hemorrhages indicate? Why are they dangerous?
Indicates deep retinal edema
Edema is dangerous to retina because:
It can distort retinal structure
It drowns/kills the retinal cells
Can operculate down to RPE and kill it
What causes superficial flame‑shaped hemorrhages, and in which retinal layers do they occur?
They result from capillary leakage in the ganglion cell layer, with blood accumulating in the nerve fiber layer (NFL). Their appearance reflects the linear contour of the NFL.
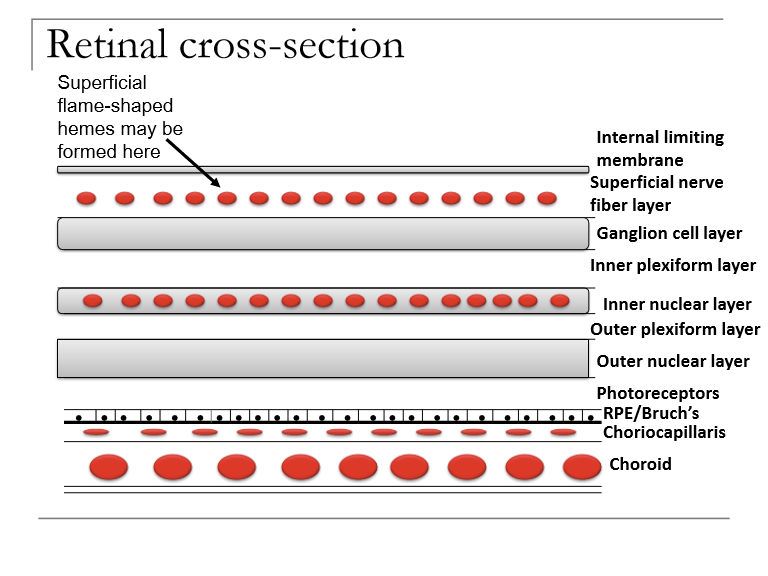
Why do superficial retinal hemorrhages appear flame‑shaped?
Because blood spreads along the radial, feather‑like orientation of nerve fiber bundles in the NFL, producing the characteristic flame‑like shape.
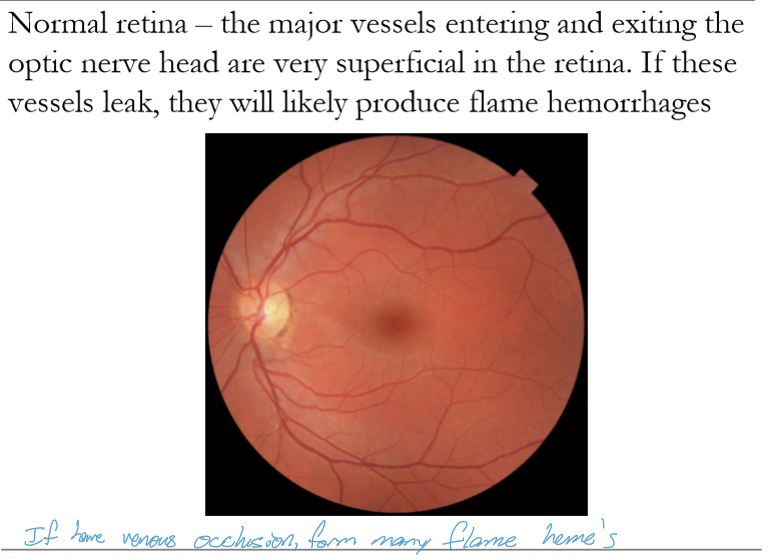
What systemic conditions commonly cause isolated flame‑shaped retinal hemorrhages?
Hypertension and diabetes mellitus. Both cause microvascular damage leading to capillary leakage.
Which retinal venous occlusions can cause sectoral or widespread flame hemorrhages?
Branch retinal vein occlusion (BRVO) → sectoral flame hemes
Hemicentral RVO → about half the retina
Central retinal vein occlusion (CRVO) → widespread, nearly entire retina
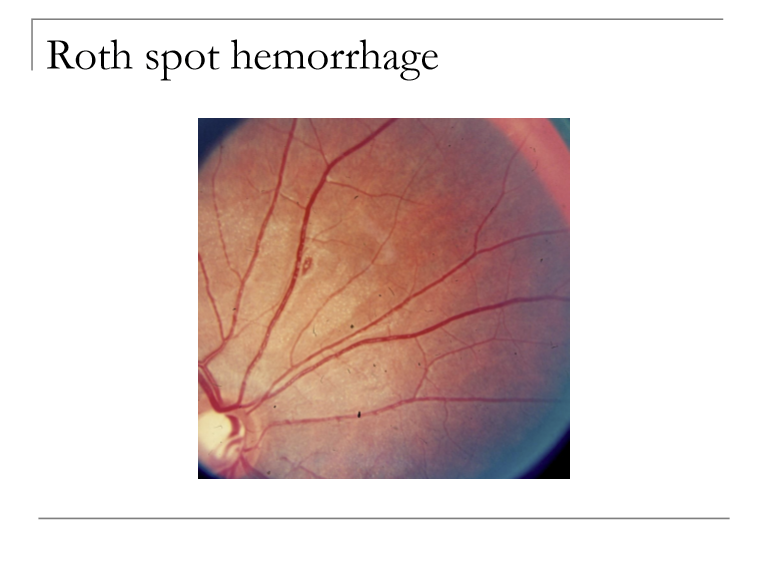
What distinguishes superficial flame‑shaped hemorrhages from Roth spot hemorrhages?
Flame‑shaped hemes: superficial, in the nerve fiber layer, follow NFL contour.
Roth spots: hemorrhages with a white center, associated with systemic disease (e.g., endocarditis, leukemia).
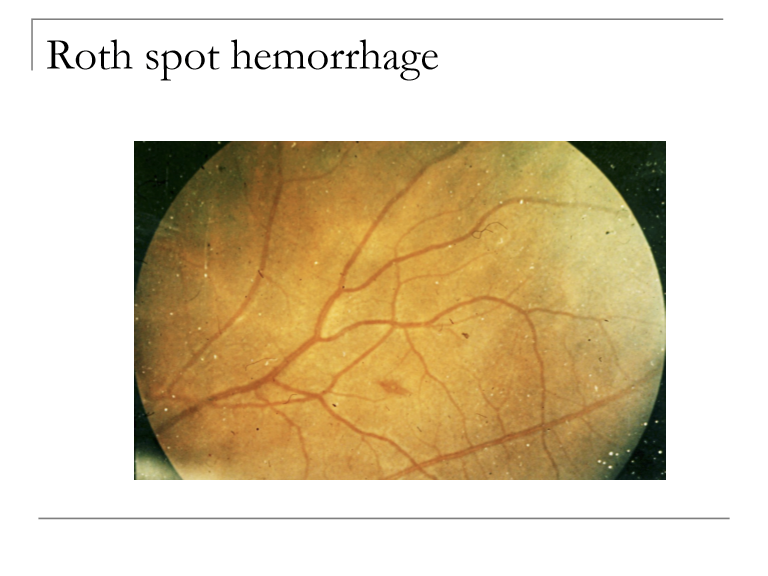
What is a Roth spot, and what is the significance of its white center?
A Roth spot is a retinal hemorrhage with a white central spot, representing focal ischemia or inflammatory material. The white center indicates underlying vascular or systemic pathology.
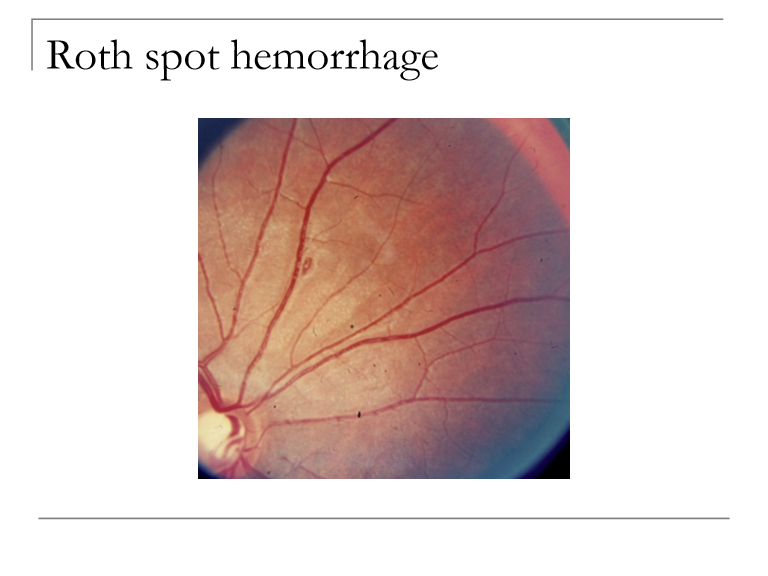
What are the major causes of the white center in Roth spot hemorrhages?
Focal accumulations of WBCs or fibrin from retinal vascular blockage
Systemic inflammatory conditions, especially bacterial endocarditis
Cotton wool spots (localized nerve fiber infarcts)
Leukemic cell foci