Arrangement of electrons
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
Energy level
the fixed energy value that an electron in an atom may have
Ground state
of an atom is one in which the electrons occupy the lowest available energy levels
What is the name given to the visible regein of the hydrogen line spectra?
Balmer series
Which energy level do electrons fall to to produce the Balmer series?
n = 2
What is an emission spectrum?
light is emitted from a sample of a gaseous element which has been excited
If hydrogen has only one electron, why is there more than one line on its emission spectrum?
with a sample of hydrogen there are millions of hydrogen atoms
when energy is supplied not all atoms receive the same amount
some may jump to higher energy levels
those that jump to high energy levels may not drop directly back to n = 1, but could drop to an in between level first emitting energy of 2 different wavelengths
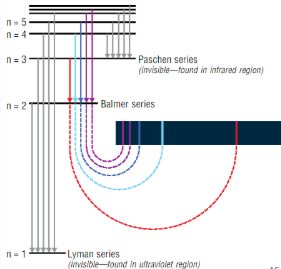
What are the different colours produced in the emission spectrum of hydrogen?
Red n=3 → n=2
Blue/green n=4 → n=2
Indigo n=5 → n=2
Violet n6 → n=2
How is the energy of the emitted light related to its wavelength?
higher energy transitions produce shorter wavelengths → blue and violet light
lower energy transitions produce longer wavelengths → red light
Why is the line spectrum unique for each element?
each element has a unique number of electrons in each of its atoms and thus has a unique electron configuration
in atoms of any element the energy difference between levels is unique to that element
the electronic transitions/moving from one level to another are unique in terms of energy requirements and energy emission
Bohr’s theory states:
electrons occupy fixed energy levels
in the ground state, electrons occupy the lowest available energy level
if an tom receives energy, it becomes excited and jumps to a higher energy level
the energy absorbed by the atom must equal the differences between the ground state and the excited state/the low energy level and the high energy level
the excited state is unstable and the electrons fall back down to lower energy levels
this releases energy in the form of light, of a definite wavelength which correspond to the different colours seen on line spectra
the frequency of light emitted depends on the 2 energy levels and is represented by the equation E2 - E1 = hf - E2 is the higher energy level, E1 is the lower energy level, h is planck’s constant and f is the frequency of light emitted
What is an absorption spectrum?
shows a continuous spectrum of light with certain wavelengths missing that are absorbed by the element
Atomic Absorption spectrometry - principle
atoms in the ground state absorb light of a particular wavelength characteristic of the element, and absorbance is directly proportional to concentration
Atomic Absorption spectrometry - process
sample solution is sprayed into the flame and the sample element is converted into atoms in the element
ground state atoms absorb radiation from a source made from the element
absorbance is measured
Atomic Absorption spectrometry - use
detection and measuring concentration of heavy metals
detection and measuring concentration of water pollutants
How does modern atomic theory describe the behaviour of electrons?
de Broglie suggested that electrons can behave as a particle and as a wave/ demonstrates wave particle duality
Heisenberg’s uncertainty principle
states that it is impossible to measure at the same both the velocity and the position of an electron
Limitations of Bohr’s theory
only worked well for hydrogen + wasn’t accurate for higher elements
did not take into account the fact that the electron had a wave motion
heisenberg’s uncertianty principle was in conflict with Bohr’s theory
did not explain sublevels
did not account for the existence of orbitals
Orbital
a region in space within which there is a high 95% chance of finding an electron
Who calculated the probability of finding an electron within a certain region in space?
Schroedinger
What shape are s orbitals?
sphericalwhat
What shape are p orbitals?
dumbell shaped
Sublevel
a subdivision of a main energy level and consists of one ore more orbitals of the same energy
Aufbau principle
when building up the electron configuration of an atom in its ground state the electrons occupy the lowest available energy levels
Write the electron configuration of Al

Write the electron configuration of Ca

What are the exceptions when writing electron configuration + why?
Chromium + Copper
a full or exactly half filled energy sublevel has extra stability
Hund’s rule of maximum multiplicity
states that when 2 or more orbitals of equal energy are available, the electrons occupy them singly before ever filling them in pairs
Pauli exclusion principle
states that no more than 2 electrons may occupy an orbital and they must have opposite spin