CHEMISTRY Exam 2023
5.0(2)
Studied by 0 peopleCard Sorting
1/28
Earn XP
Description and Tags
Last updated 1:37 PM on 5/29/23
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
1
New cards
chromatography
a method of separating mixtures and analysis of a mixture of a soluble chemical substance
2
New cards
what is the stationary phase
paper
3
New cards
what is the mobile phases
solvent (liquid → usually water but not always)
4
New cards
do substances that are more attracted to the mobile move farther up the paper?
YES
5
New cards
chromatography method
1. draw baseline in pencil, 1 cm from the bottom of paper
2. draw samples/dots onto base line
3. add chromatography paper to beaker so solvent paper but below the baseline
4. wait until solvent has traveled up to almost the top of paper, draw a new line in where it reached in pencil (solvent front)
5. calculate Rf of substances
6
New cards
how to calculate the Rf value
distance traveled by substance (colour) DIVIDED BY distance traveled by solvent (liquid or solvent front)
7
New cards
what does do simple distillation?
separates 2 liquids
the mixture is heated enough so one liquid boils, the vapour moves up and into the condenser where it is cooled and turned back into a liquid.
the mixture is heated enough so one liquid boils, the vapour moves up and into the condenser where it is cooled and turned back into a liquid.
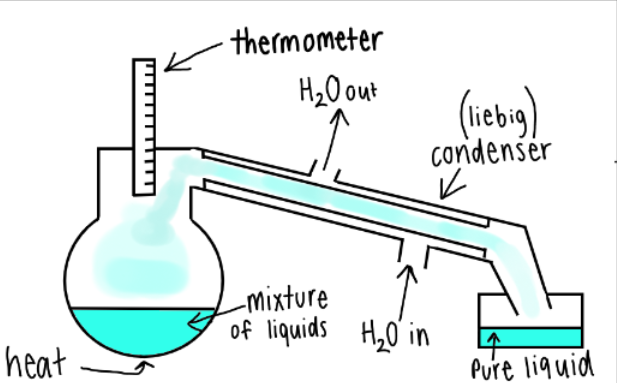
8
New cards
fractional distillation
used to separate 3 or more liquids/gases into individual fractions
9
New cards
filtration
used to remove an insoluble solid from a liquid
10
New cards
crystallisation
used to remove water from an insoluble salt solution (evaporation)
11
New cards
atomic structure
protons and neutrons in the nucleus and electrons in the outer shells
12
New cards
electron relative mass and relative charge
relative mass - 1/2000
relative charge - -1
relative charge - -1
13
New cards
neutron relative mass and relative charge
relative mass - 1
relative charge - 0
relative charge - 0
14
New cards
proton relative mass and relative charge
relative mass - 1
relative charge - +1
relative charge - +1
15
New cards
history of the atom
John Dalton in 1803 thought that all matter was made of particles called **atoms**
Nearly 100 years later, J J Thomson carried out experiments and discovered the **electron**. This led him to suggest the **plum pudding model** of the atom.
In 1909 Ernest Rutherford designed an experiment to test the plum pudding model. In the experiment, positively charged **alpha particles** were fired at thin gold foil. Most alpha particles went straight through the foil. But a few were scattered in different directions.
Niels Bohr adapted Ernest Rutherford's nuclear model to suggest that electrons orbit the nucleus in shells. The shells are at certain distances from the nucleus.
Further experiments led to the idea that the nucleus contained small particles, called **protons**. Each proton has a small amount of positive charge.
In 1932 James Chadwick found evidence for the existence of particles in the nucleus with mass but no charge. These particles are called **neutrons**. This led to another development of the atomic model, which is still used today.
Nearly 100 years later, J J Thomson carried out experiments and discovered the **electron**. This led him to suggest the **plum pudding model** of the atom.
In 1909 Ernest Rutherford designed an experiment to test the plum pudding model. In the experiment, positively charged **alpha particles** were fired at thin gold foil. Most alpha particles went straight through the foil. But a few were scattered in different directions.
Niels Bohr adapted Ernest Rutherford's nuclear model to suggest that electrons orbit the nucleus in shells. The shells are at certain distances from the nucleus.
Further experiments led to the idea that the nucleus contained small particles, called **protons**. Each proton has a small amount of positive charge.
In 1932 James Chadwick found evidence for the existence of particles in the nucleus with mass but no charge. These particles are called **neutrons**. This led to another development of the atomic model, which is still used today.
16
New cards
relative atomic mass calculations
* To calculate the relative atomic mass:
* Multiply the % amount of each isotope by its mass
* Add these numbers together
* Divide by the total abundance (which when using % abundance, will be 100%)
* Here it is as an equation:
* Multiply the % amount of each isotope by its mass
* Add these numbers together
* Divide by the total abundance (which when using % abundance, will be 100%)
* Here it is as an equation:
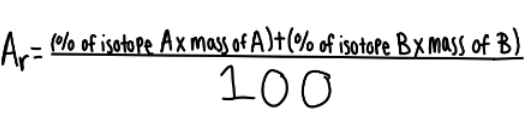
17
New cards
group 1
as you go farther down group 1 the metals get more reactive as the electron needed to be removed is farther away from the nucleus so lower attraction.
nicknamed Alkali Metals
nicknamed Alkali Metals
18
New cards
group 7
Nicknamed the halogens
the closer the outer shells to the nucleus the greater ability to react
the closer the outer shells to the nucleus the greater ability to react
19
New cards
group 0
nicknamed the noble gases
they have full outer shells so do not need to react
they have full outer shells so do not need to react
20
New cards
Earth's atmosphere
made up of multiple layers
21
New cards
Early atmosphere
* lots of water vapour (over 100 degrees)
* high levels of CO2
* high levels of sulfur dioxide
* very hot
* lots of volcanoes and eruptions
* some ammonium
* no free oxygen
* high levels of CO2
* high levels of sulfur dioxide
* very hot
* lots of volcanoes and eruptions
* some ammonium
* no free oxygen
22
New cards
Changes in the earth’s atmosphere - H2O
the earth cooled to below 100.C and water vapour condensed and fell onto the surface
23
New cards
Changes in the earth’s atmosphere - CO2
levels were decreased - absorbed into oceans - used by sea creatures to make shells - locked up in fossil fuels & limestone
24
New cards
Changes in the earth’s atmosphere - O2
organisms evolved to photosynthesis - plants, algae and bacteria
25
New cards
Exothermic
gives out energy to the surroundings
26
New cards
endothermic
takes in energy from surroundings
27
New cards
exothermic profile diagrams
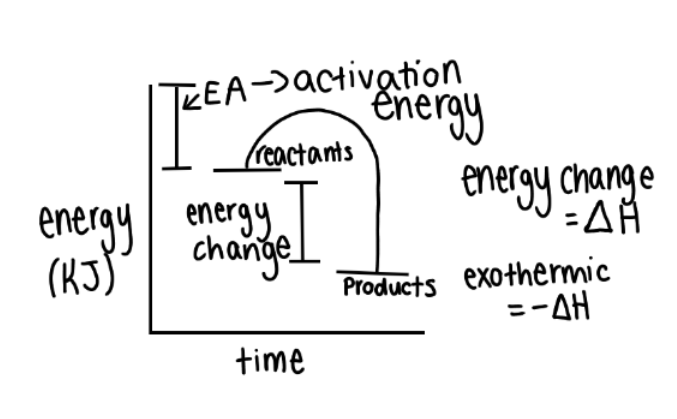
28
New cards
endothermic profile diagrams
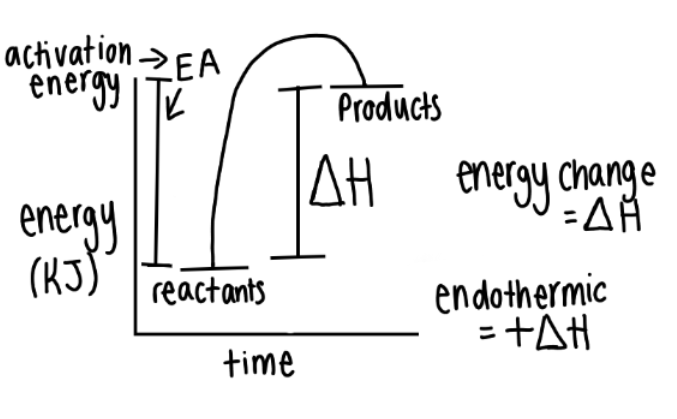
29
New cards
bond energy calculations
1. calculate the energy for each reaction
2. the LEFT HAND SIDE - RIGHT HAND SIDE = ANSWER
3. if answer is positive it is endothermic and if answer is negative it is exothermic