Lecture 3: Review of Metabolism
1/134
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
135 Terms
you can classify an organism by their ______ of energy AND by how they ______ energy
source; generate
______ is energy produces by breaking down organic, this is a type of ______
chemoorganotrophy; catabolism
______ is energy produced by oxidizing inorganic compounds like H2, NH4+, or H2S
chemolithotrophy
______ is energy produced by harvesting light, this is a type of ______
phototrophy; anabolism
what is NH4+?
ammonium
respiratory is energy generated through an ______ which participates in a series of ______ reactions
ETC; REDOX
in respiration, an electron ______ feeds high energy electrons to the ETC, and a terminal electron ______ is where the electron is ultimately dumped at the end of the ETC
donor; acceptor
Aerobic respiration uses ______, while Anaerobic respiration uses ___(2)___
oxygen; nitrate, sulfate, etc.
fermentative metabolism is the ______ of ______ compounds ______ coupled to energy production
oxidation; organic; DIRECTLY
redox reactions are how biological system general ______
energy
the electron donor goes from ______ to ______
reduced; oxidized
electron acceptor goes from ______ to ______
oxidized; reduced
true or false: Redox reactions ALWAYS release energy
false; depending on thermodynamics, can release or require require
the ______ the distance between the donor and acceptor, the MORE energy generated (or required) by the Redox reaction
GREATER
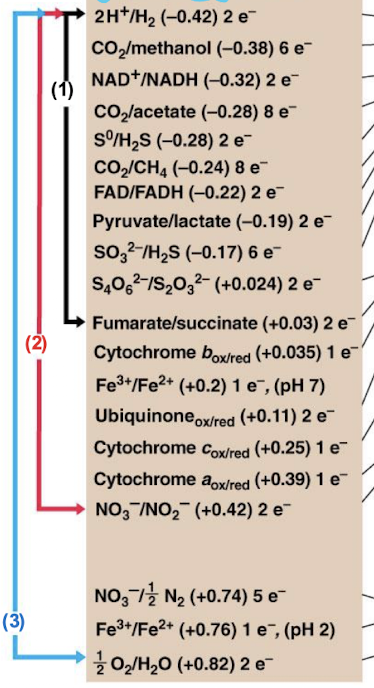
which of these redox partners will release the MOST energy?
blue
E0’ (V) = ______
Reduction potential
the more NEGATIVE the E0’, the greater the electron ______ and the poorer the electron ______
donor; acceptor
the more POSITIVE the E0’, the greater the electron ______ and the poorer the electron ______
acceptor; donor
are the E0’ values consistent? Or do they change?
values gives are at a pH of 7, as conditions change the values will too and certain reactions may become more or less favorable
what are the 2 main important electron accusers in biological systems?
NADH and FAD
NADH carriers two or three times as much energy as ______
ATP
NADH is the ______ form, while NAD+ is the ______ form
reduced; oxidized
overall reduction of NAD+ consumes ______ hydrogen atoms to make NADH
two
because there is a limited amount of NADH in the cell, this requires a redox ______
balance
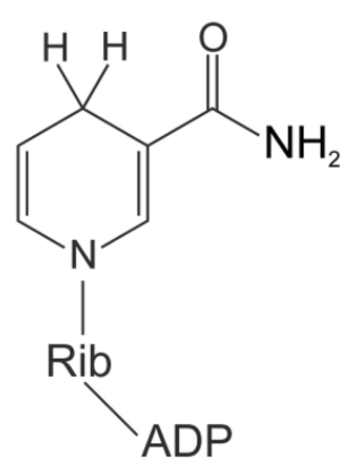
what electron carrier is this?
NADH
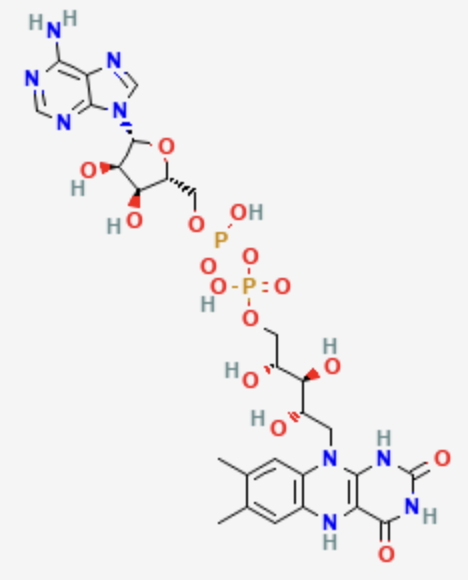
What electron carrier is this?
FADH2
FADH2 is ______ related to NADH that can ______ electrons
coenzyme; transfer
FADH2 is the ______ form, while FAD is the ______ form
reduced; oxidized
unlocked NAD+, FAD is reduced by ______ electrons and ______ protons
2; 2
which electron carrier is weaker? (NADH or FADH2)
FADH2
when FADH2 is combined with a strong electron acceptor such as ______, electrons are transferred and significant ______ is released
oxygen; energy
what is the redox active portion of FADH2?
isoalloxazine ring
what 3 things does respiratory metabolism require
ETC, e- donor, terminal e- acceptor
respiratory metabolism generates a ______ gradient called the ______
proton; proton motive force
in respiratory metabolism, the break down of ______ compounds produces ______; these carriers then donate electrons to the ______. This is an example of ______
NADH/FADH; ETC; chemoorganotrophy
some organisms use high energy ______ compounds that directly feed electrons into the ______; this is an example of ______
inorganic; ETC; chemolithotrophy
an ETC involves a series of ______ reactions that ______ energy
REDOX; energy
the ETC is located in the ______ (prokaryotes) and is used to generate a ______
cell membrane; proton motive force
what is the first step of the ETC?
electron donor donates their e- to the first carrier in the membrane
with each reaction in the ETC, electrons are ______ the membrane
pumped across
at the end of the ETC, the electrons are transferred to a ______
terminal electron acceptor
what are the 4 main REDOX carriers in the ETC?
flavoproteins, cytochromes, Fe/S proteins, quinones
flavoproteins contain a ______ ring
FMN
cytochromes have a ______ ring that is covalently linked to ______ of cytochrome proteins; typically ______ is coordinated at the center
prophyrin; cysteine; iron
quinones are non-______
proteinaceous
what does non-proteinaceous mean?
non-protein (not made from peptides)
quinones are ______ molecules that are integrated into the ______; some can act as ______ carriers too!
hydrophobic; membrane; hydrogen atom
in prokaryotes, the ETC is ______, which allows for the organism to ______ to its changing environment
flexible; adapt
how is the ETC flexible?
cells can change the types of proteins
what is one example of how the ETC can be modified in highly acidic conditions?
complex 1 modified to NDH-2 (a different NADH dehydrogenase) which no doesn't pump protons
what is one example of how the ETC can be modified in low oxygen conditions?
complex 4 converted to Cyt BD which is better at scavenging oxygen
if Cyt BD is better at scavenging low oxygen, why doesn’t the cell use it all the time?
doesn't pump electrons out like complex 4
what is an example of how the ETC can be modified for anaerobic respiration?
uses nitrate reductase complex which can use nitrate instead of O2 as a TEA
what protein converts the PMF to biochemical energy?
F0/F1 ATP synthase
ATP synthase harnesses energy by pumping proteins from ______ to ______ the cell; is this against or along the gradient?
outside; inside; along
As ATP synthase pumps protons, ______ which is used to generate ______
rotates; ATP
Fo subunit is found in the ______, while F1 is found in the ______
membrane; cytoplasm
what is the chemiosmotic theory?
PMF = chemical gradient and the electrical gradient
some organisms can use a ______ gradient to generate ATP (rather than proton)
Na
why would it be useful for ATP synthase to be reversible? (would would you want to use up energy pumping gradients outside?)
PMF used for other things like motility, want to maintain it
ATP synthase utilization of the PMF is known as ______ phosphorylation
oxidative
fundamentally, there are lots of similarities between the PMF and ______ used in engineering
hydroelectric power systems
what part of the ETC would be analogous to storage reservoir in hydroelectric power plants?
H+/protons
what part of the ETC would be analogous to a dam in hydroelectric power plants?
cell membrane
what part of the ETC would be analogous to a turbine in hydroelectric power plants?
F0/F1 ATP synthase
fermentative metabolism is the generation of ATP by ______ phosphorylation, carried out in the ______ of terminal electron acceptors
substrate-level; absence
what is a facultative fermenter?
organisms can do fermentation AND respiration
EXAM: Substrate level phosphorylation ALWAYS = ______
FERMENTATION
why would you want to be an obligate fermenter if it is less efficient?
they fill a specific niche
true or false: obligate fermentors do not have a PMF
FALSE: they still need to generate a PMF in absence of ETC for motility and active transport!
fermentation is a fairy ______ form of metabolism because you need a LOT of ______
inefficient; substrate
why is respiration more efficient?
you can oxidize more completely in respiratory metabolism
fermentative pathways are named by their ______
WASTE products
you can classify organisms by their ______ of carbon for biosynthesis
source
heterotrophs use carbon derived from ______ of ______ organic compounds
catabolism; external
autotrophs use carbon derived from ______
fixing CO2
many chemolithotrophs are phototrophs are ______
autotrophs
how can an organisms be a chemolithotroph and ALSO an autotroph?
chemolithotroph refers to the source of ENERGY, which autotroph is the source of CARBON
what is the willow tree experiment?
grew a willow tree and weighed it, discovered that the biomass cam from something other than soil → CO2
from one molecule of glucose, you receive ______ molecules of ATP (aerobic respiration)
38
how many ATPs are produced in substrate level phosphorylation?
6
how many NADH molecules are used per molecules of glucose?
10
how many FADH molecules are used per molecule of glucose?
2
how many ATPs are made per 10 molecules of NADH?
30
how many ATPs are made per 2 molecules of FADH?
4
how many ATP are needed for predatory reaction? (are are subtracted from net ATP produced)
2
______% of glucose is captures as ATP
38
what part of central metabolism generates the most e- carriers?
TCA cycle
different ______ pathways eventually funnel into central metabolic pathways
catabolic
what are some other metabolic pathways that can funnel into the central metabolism?
polysaccharides, lipids, proteins
chemoorganotrophs can use all kinds of ______ compounds as energy sources
organic
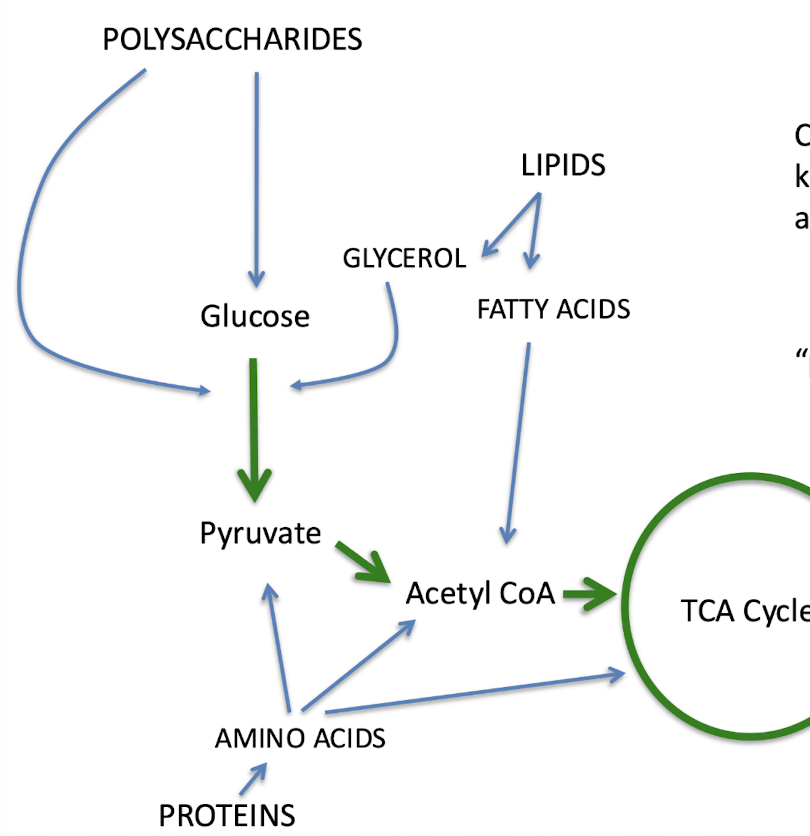
in this image, the blue lines represent ______
peripheral pathways
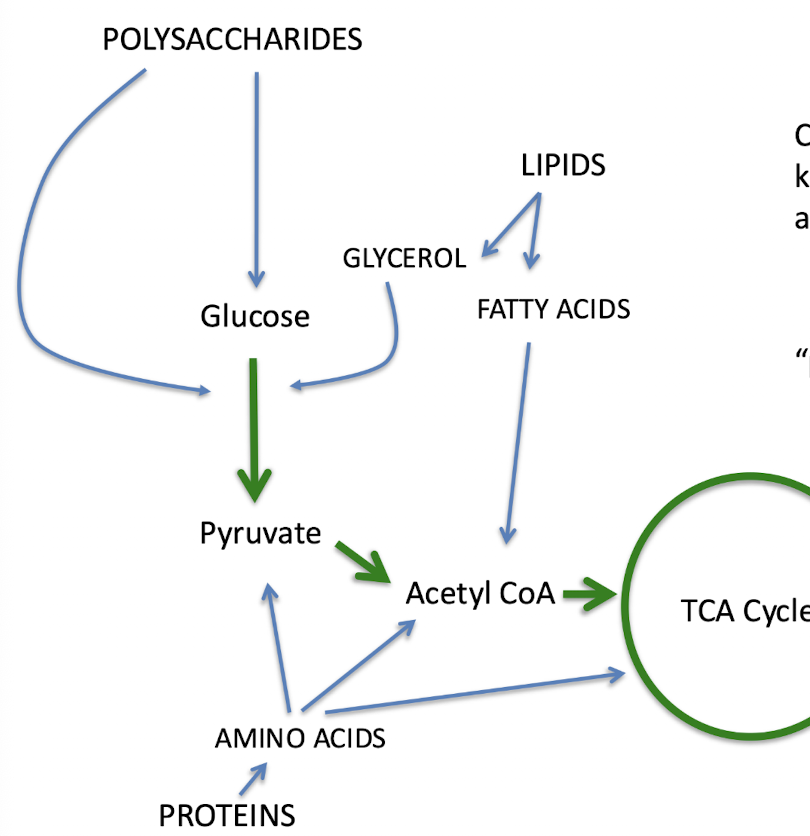
in this image, the green lines represent ______
central metabolism
central metabolic pathways are the interface between _____ and _____
catabolism; anabolism
what is catabolism?
break down
what is anabolism?
build up (synthesis)
through _____ of organic compounds (by _____), the cell ultimately funnels breakdown products into the _____ pathways
catabolism; chemoorganotrophs; central metabolic
central metabolic pathways produce the key _____ precursor molecules required for _____ reactions
12; biosynthetic
why do chemolithotrophs and phototrophs still the same central metabolic pathways?
these pathways are needed to make the 12 key precursors
what are the 3 main pathways involved in central metabolism?
EMP (glycolysis), TCA, Pantone phosphate cycle (PPC)