practical techniques
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
what is distillation used for?
the separation of a pure liquid from its impurities
what is one of the most common distillation practicals?
the oxidation of primary alcohols to aldehydes
what is the process of distillation to produce an aldehyde from a primary alcohol?
reaction mixture containing primary alcohols and acidified potassium dichromate solution placed into a pear shaped flask
anti bumping granules added to promote smooth boiling
distillation apparatus set up
reaction mixture heated until it boils using a heating mantle - control of temperature
water enters at bottom of condenser and removed by drainage pipe at top
distillate formed in condenser drips into receiving vessel as it condenses after vaporisation
aldehyde produce has lower BP than alcohol, so can be distilled off as soon as it forms
draw a diagram for how the distillation apparatus should be set up
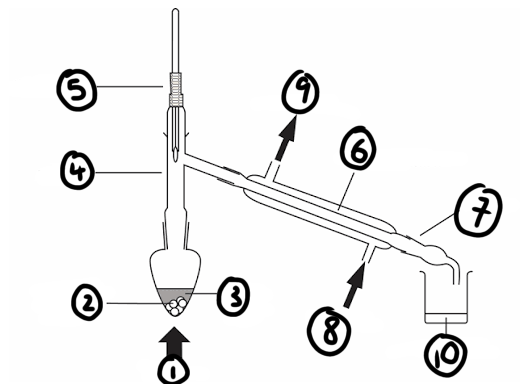
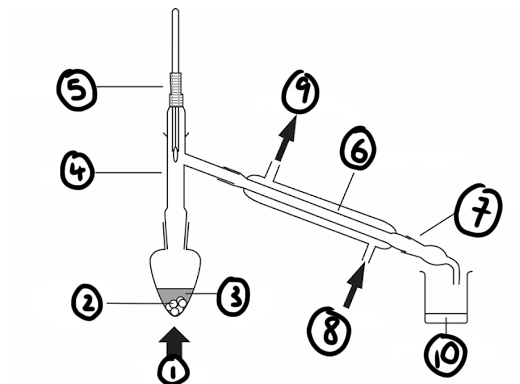
label the diagram of the distillation apparatus
1) heat
2) anti-bumping granules
3) impure liquid
4) still head
5) thermometer
6) Liebig condenser
7) receiver
8) water in
9) water out
10) pure liquid
what is reflux used for?
for a liquid to be continually boiled whilst the reaction occurs, preventing volatile components escaping and flask from boiling dry
what are three common reflux practicals?
the production of a carboxylic acid from a primary alcohol using acidified potassium dichromate
the production of a ketone from a secondary alcohol
the production of an ester from an alcohol and acid in presence of an acid catalyst
what is the process for reflux?
reaction mixture placed into a pear-shaped flask
anti-bumping granules added to promote smooth boiling
reflux apparatus set up
reaction mixture heated until it boils using a heating mantle - control of temperature
water enters at bottom of condenser and removed by drainage pipe at top
vapour from mixture rises up condenser and then condenses to drip back into flask
draw a diagram for how reflux apparatus should be set up
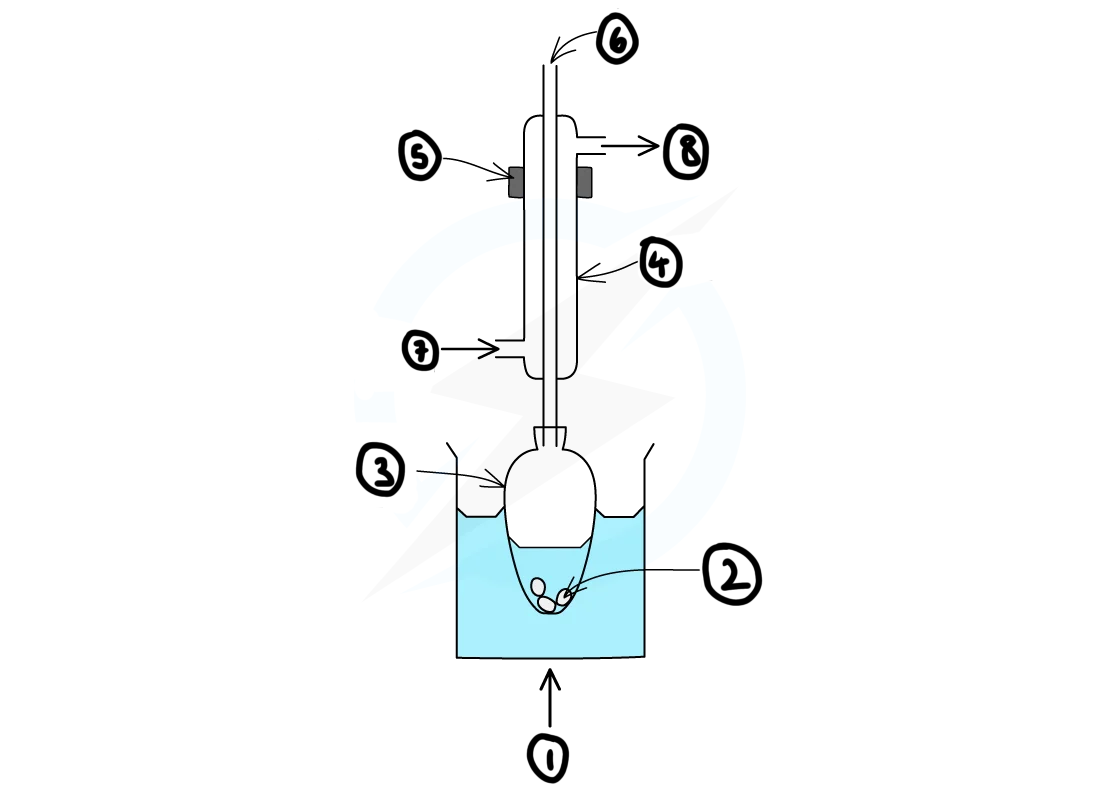
label the diagram of the reflux apparatus?
1) heat
2) anti-bumping granules
3) pear-shaped flask
4) liebig condenser
5) clamp
6) open top
7) water in
8) water out
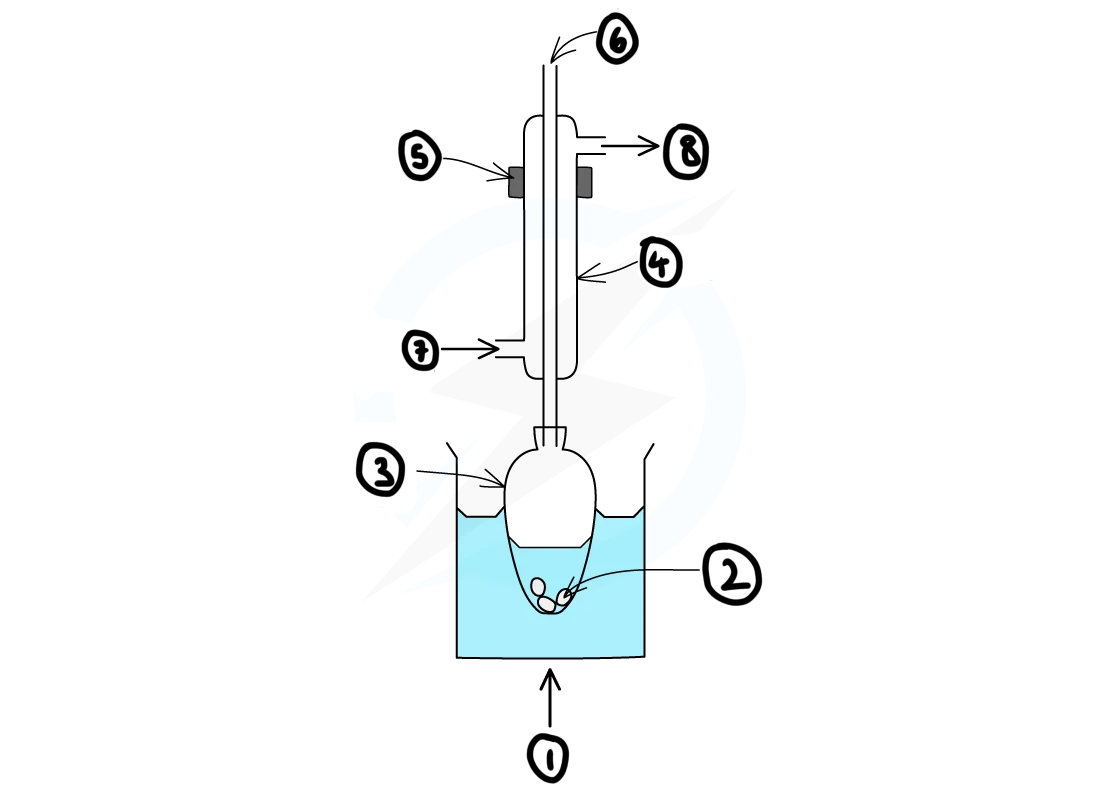
why is an open top used during reflux?
to prevent the apparatus exploding as a result pressure building up as hot air expands in the closed system
what are three methods of purifying samples of organic liquid?
use of a separating funnel, use of drying agents, redistillation
how is a separating funnel used to purify samples of organic liquid?
pour mixture of liquids into separating funnel and a stopper added
separating funnel is inverted and stopcock opened to release pressure
two layers separate
if bottom layer is aqueous, open stopcock so the aqueous layer drains away and organic layer drained into another conical flask
what should be added to the mixture in the separating funnel if acid was used during the preparation process?
aqueous sodium carbonate should be added and shaken with the mixture to remove acid impurities from the product
how are drying agents used to purify samples of organic liquid?
add the organic liquid to a conical flask
a spatula of drying agent is added to the liquid and swirled
if drying agents clump together, there is still water present
more drying agent added until some solid is dispersed in solution as a fine powder
decant liquid, should be clear if dry
how is redistillation used to purify samples of organic liquid?
a second distillation is carried out if the sample contains some organic impurities, due to close boiling points
aims to obtain just the desired organic liquid by having a narrower boiling point range