Biochem - Mid sem 1
1/93
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
94 Terms
Why are phi and psi so important?
Assuming all peptide bonds are trans, all the conformational freedom in the backbone of the polypeptide is due to these two rotations.
Phi rotations occur between which two atoms along the backbone?
Nitrogen and Alpha carbon
Psi rotations occur between which two atoms along the backbone?
Between the alpha carbon and the carbonyl
Is psi or phi located on the x-axis of a Ramachandran plot?
Phi is
How do we change the peptide conformation from trans to cis?
Flip the peptide psi or phi bond by 180 degrees.
What are the residual patterns that indicate an alpha helix may be formed?
Heptad repeats - 1st and 4th positions in the repeat are typically hydrophobic. The pattern repeats every 7 amino acids.
During protein folding, does the entropy of the surrounding water that the protein is dissolved in increase, stay the same, or decrease?
Increase - it is a condensation reaction
Paralogue definition
Homologous proteins that perform different but related functions within one organism with >25% identity
Orthologue definition
Homologous proteins that perform the same function in different species with >25% identity
Homologues definition
If two sequences show >25% identity, share a common protein domain ancestor and have the same fold
What are the phi and psi angles of beta-pleated sheets?
-130, +130
What is secondary structure?
Refers to the local spatial arrangement of the polypeptide backbone. Two regular arrangements: Alpha helix and beta sheet
What is the difference between tertiary and quaternary structure?
Tertiary structure describes the fold of each domain in a subunit (single chain) while quaternary describes the subunit arrangement (multiple chains).
What is the Bohr effect?
Increased CO2 concentrations or lower pH reduces hemoglobins affinity for oxygen, causing it to release oxygen more readily.
What is the percentage of tissue H+ that HB transports to the lungs and kidneys?
Up to 40%
What is the percentage of tissue CO2 that HB transports to the lungs and kidneys?
Up to 20%
At lower pH when __1__ concentrations are higher, the affinity of Hb for O2 __2___ because __1__ stabilises Hb in the __3__ state
H+, decreased, T
Why is pH lower in tissues?
When muscles move, metabolic processes produce lactic acid and CO2
What kind of amino acid group within the Hb protein would pick up a proton at a pH of around 7?
Histidine
What is BPG?
2,3-biphosphoglycerate
What is BPGs role?
It is a negative allosteric regulator of Hb function and decreases the affinity of Hb for O2
What is the BPG concentration at sea level?
5mM
What is the BPG concentration at high altitudes?
8mM
How does BPG stabilise Hb?
It binds to the + charged residues around the central cavity of Hb that only exists in the T state
What is the formula for Kd?
[P]x[L]/[PL]
When theta equals 0.5, what does Kd equal?
[L]
What is the formula for theta?
theta = [PL]/[P]+[L] AND theta = [L]/[L] + Kd
What doe the ‘L’, ‘P’ and ‘PL’ stand for in regards to protein binding?
Free ligand, Free protein and Protein-ligand complex
What does Kd represent?
Quantifies the binding affinity between a ligand and protein. Represents the concentration of free ligand at which the protein is 50% saturated.
What Kd unit represents a high binding affinity?
Nanomolar (10-9) or smaller
What Kd unit represents a low binding affinity?
Micromolar (10-6) or larger (Millimolar = 10-3)
What is biotin needed for?
Carboxylations
What is biotins role in egg whites?
Very very strongly binds the protein avidin found in raw egg white and avidin binds to bacteria.
The binding of oxygen to myoglobin is associated with a…
Low disassociation constant Kd as myoglobin stores oxygen in tissues.
Does oxygen binding make the haem ring structure more or less planar?
More planar
T state meaning and traits
Tense, More interactions, more stable, lower affinity for O2
R state meaning and traits
Relaxed, fewer interactions, more flexible, higher affinity for O2
How does the conformational change from T —> R come about?
O2 binding results in the breaking of salt bridges between the residues at the alpha 1 and beta 2 interface due to the shape change.
What type of side chains are exposed on the surface of proteins? Give an example.
Polar side chains such as cysteine.
What type of side chains are within the core of proteins? Give an example.
Non-polar side chains such as alanine.
What residues commonly occur in the turns of the backbone? Why?
Glycine (Gly) and Proline (Pro) because they are small.
What are four features of folded proteins?
They are compact, water excluded from the interior, non-polar side chains located inside, polar side chains located on the outside.
Why is the folded state more favoured?
Electrostatic forces, van der Waals interactions, hydrogen bonds and hydrophobic interactions stabilise the folded state.
What was Christian Anfinsen’s experiment to show that protein folding is reversible?
Expose a folded protein to a high concentration of urea and mercaptoethanol → protein is denatured → remove urea AND THEN mercaptoethanol→ original conformation re-forms
What does it mean if protein folding is co-operative?
A protein can be either folded or unfolded. If any interactions are disrupted, the rest of the protein structure is also disrupted.
Does disulfide bonding direct folding or does folding direct disulphide bond formation?
Folding directs bond formation
What is the role of chaperones?
They help to prevent protein misfolding. Bind and releases (with thanks to ATP) until the correct fold forms.
Electrostatic interactions: dipoles
Strong, long-range interactions between permanently charged groups. An example is salt bridges. Dipole moments occur with partial double bonds.
London dispersion (van der Waals interactions)
Weak, medium-range attractions between all atoms which contributes significantly to the stability in the interior of the protein.
Favourable hydrogen bonds in proteins
Interaction of N-H and C=O of the peptide bond leads to local regular structures such as alpha helices and beta sheets.
What is the hydrophobic effect?
The release of water molecules from the solvation layer around the molecule as protein folds. Increases the net entropy.
When observing a Ramachandran plot, are all points located in the alpha region involved in alpha helices?
No
Any polar group buried in the protein must form a _____
Hydrogen bond
Number of residues per alpha helix turn
3.6
Average length of alpha helix turn
0.54 nm or 5.4 A
Alpha helix phi and psi angles
-57, -47
One heptad ___ x 100 degrees and __ x 360 degrees
7 and 2
Which residues are strong helix formers (3 letter code)?
Small hydrophobic residues such as Ala and Leu
Which residues act as helix breakers (3 letter code)?
Gly and Pro
What are the components of a beta-pleated sheet
Antiparallel (line up nicer) or parallel beta-strands.
Reverse turns (beta turns): Angle, stabilisation and residues involved
180 degrees over 4 residues, stabilised by a carbonyl to amide hydrogen bond, proline in position 2 or glycine in position 3.
Difference between type 1 and type 2 turns
Type 2 often have Gly in position i+2 due to less steric hindrance. They also differ by the direction of i+1 carbonyl.
Irregular structure names
‘loop’ or ‘random coil’ structure
Super-secondary structure examples
aa-hairpin, bb-hairpin
As chain length increases, solubility and melting point
Solubility decreases and melting point increases
The addition of free fatty acid molecules to an aqueous solution at physiological temperatures favours the formation of:
Micelles
The addition of free phospholipid molecules to an aqueous solution at physiological temperatures favours the formation of:
Bilayers, which go to form liposomes
Do trans fatty acids have a higher or lower melting point than cis forms?
Higher melting point
Do unsaturated cis fatty acids have a lower or higher melting point than saturated cis fatty acids? Due to what?
Lower melting points due to the kink
What type of fatty acid decides the solidity of fats such as oil vs palm oil?
Less C16/C18 saturated fats means less solid.
What is the difference between Omega-3 and Omega-6 fatty acids?
Omega-3 has the last double bond on the 3rd carbon from the end whereas the omega-6 fatty acid has it on the 6th carbon from the end.
20 residues with positive hydropathy values indicate what?
An alpha helix that spans the membrane width
Big drops in hydropathy values indicates what?
The potential to fold into a compact globular protein. The hydrophilic residues will be on the outside and the hydrophobic residues will be in the core.
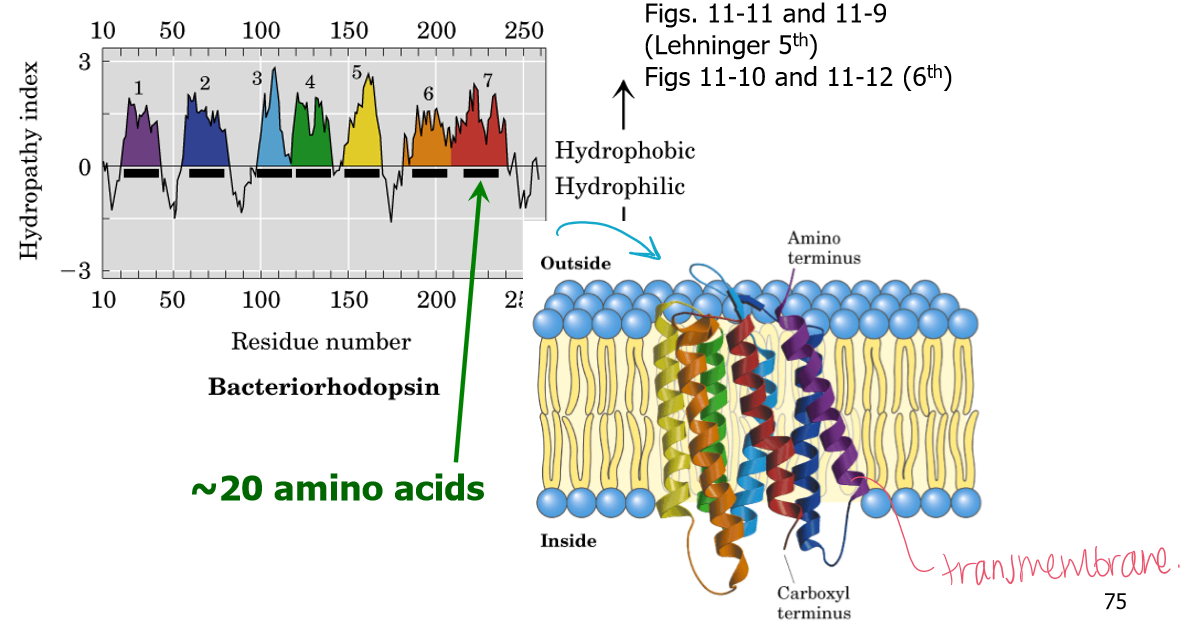
If there is 9 transmembrane alpha-helices in a protein membrane, will the N- and C-termini of this protein be on the same or opposite sides of the membrane bilayer?
Opposite sides
What is lateral diffusion in membranes?
Membrane lipids and proteins exchange places with their immediate neighbours.
How can lateral diffusion be measured?
Through Fluorescence Recovery After Photobleaching (FRAP) experiments.
What is the transbilayer movement?
Flip-flop
Flippase
P-type ATPase flips phospholipid from inside layer of bilayer to the direct opposite outerside.
Floppase
ABC transporter that flops phospholipid from inside to outside membrane layer.
What are p-type ATPases
Active membrane proteins that use ATP hydrolysis to pump ions against concentration gradients
How do carrier proteins work?
Binding of molecule to transporter induces a conformational change and works like a revolving door to transport molecules in and out of the cell.
What is G?
Gibbs free energy change. The energy of the reaction available to do work.
Exergonic (ΔG, fav/unfav, spont./non-spont.)
ΔG is negative - free energy released, favourable, spontaneous
Endergonic (ΔG, fav/unfav, spont./non-spont.)
ΔG is positive- free energy absorbed, unfavourable, non-spontaneous
What is ΔGo?
The standard free energy change - A constant measured under standard conditions.
What is the Free Energy and Equilibrium equation?
ΔG = ΔGo + RTln[C]ic[D]id / [A]ia[B]ib
What are the biochemistry standard conditions?
298K, Gases at partial pressure of 101.3kPa (1atm), [H+] = 10-7 Mg2+ = 1mM
What question does the ΔG = ΔH - TΔS equation answer?
What is the ‘driving force’ of this reaction? (enthalpy, entropy or both)
What two questions does the ΔG’o = -RTlnK’eq equation answer?
What is the position of the reaction at equilibrium? Generally speaking, is this reaction likely to be spontaneous or not?
What question does the ΔG = ΔGo + RTln[C]ic[D]id / [A]ia[B]ib equation answer?
Under a given set of conditions will our reaction be spontaneous?
How can you make unfavourable reaction go?
The concentration of products should be much lower than the concentration of reactants.
Beta strand residue patterns
Alternate between polar and non-polar to get one polar and one non-polar side