EXAM III Chem 112
1/49
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
50 Terms
kinetics
study of rates of chemical reactions
important things to remember about rates
reaction rates are always positive
rate is expressed as either the appearance of a product (+) or disappearance of the reactant (-)
rates can be expressed in several ways:
M/s or M*s⁻¹
mol/(L*s) or mol*L⁻¹s⁻¹
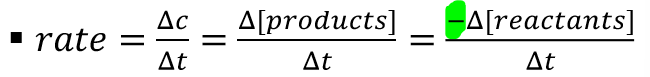
rate can be represented mathematically represented as…
-∆[R]/∆t
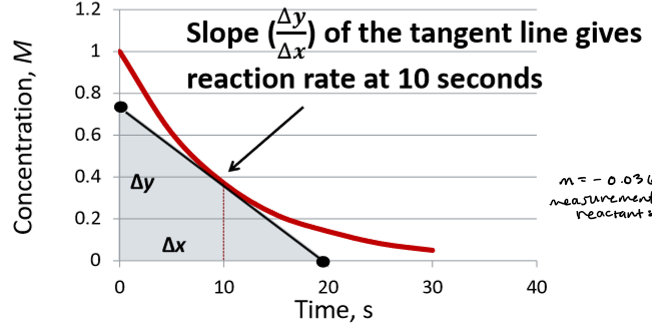
instantaneous reaction rate
equal to slope of line drawn tangent to curve at time t
reaction rates & stoichiometry
relative rates of disappearance of reactants or appearance of products depend on reaction stoichiometry
EX: 2HBr → H₂ +Br₂
2 moles HBr are consumed for every 1 mole H₂ and Br₂ that is formed
so, rate of range of [HBr] is double that of either [H₂] or [Br₂]
![<ul><li><p>relative rates of disappearance of reactants or appearance of products depend on reaction stoichiometry</p></li><li><p>EX: 2HBr → H₂ +Br₂</p><ul><li><p>2 moles HBr are consumed for every 1 mole H₂ and Br₂ that is formed </p></li><li><p>so, rate of range of [HBr] is double that of either [H₂] or [Br₂]</p></li></ul></li></ul><p></p>](https://assets.knowt.com/user-attachments/9f91a48b-ee9c-446f-94f5-0b8100165366.png)
what is a rate law
dependence of rate on concentration
ex. for reaction aA + bB → products, the rate law is given by (differential rate law):
rate = k * [A]^x * [B]^y
differential rate law
rate = k * [A]^x * [B]^y
x and y are orders of the reaction in [A] and [B] respectfully
x and y are usually small integers, but may be zero as well
k = specific rate constant
how to find over order of reaction using differential rate law?
overall order of the reaction is x + y
relationship btwn rate & concentration in differential rate law (mathematical)
-∆[R]/∆t = k[R]^x
integrated rate law
describes dependence of concentration on time
[R] = f(t)
what makes zero order rate law different from the others?
reaction rate is independent of the reactant concentration [R]
zero order integrated rate law
[R] = [R]₀ - kt
zero order differential rate law
k[R]⁰
first order differential rate law
rate = k[R]
first order integrated rate law
exponential: [R] = [R]₀e^(-kt)
linear: ln[R] = ln[R]₀ - kt
half-life
t½
time required for initial concentration to dec by 1/2
first order half-life
t = t½, then [R] = ½[R]₀
substitute and rearrange to solve for t½: t½ = .693/k
half-life of first order reaction is independent of concentration
what is the half-life of ¹⁴C?
t½ = 5730 years
second order differential rate law
rate = k[R]²
second order integrated rate law
1/[R] = 1/[R]₀ + kt
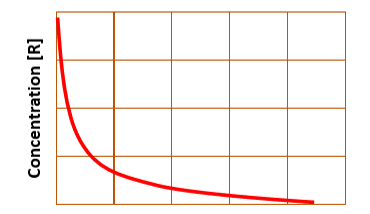
for integrated second order rate law, a plot of concentration vs. time yields…
a curve
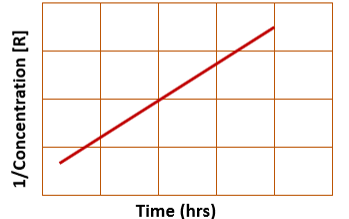
for integrated second order rate law, a plot of 1/concentration vs. time yields…
a straight line
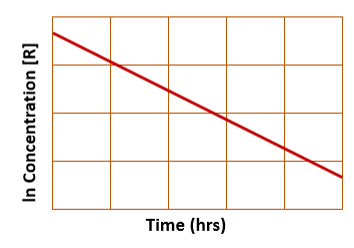
for integrated first order rate law, a plot of ln(concentration) vs. time yields…
a straight line
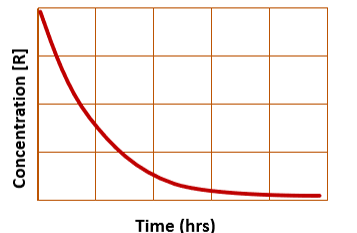
for integrated first order rate law, a plot of concentration vs. time yields…
a curve
how do the units of k change depending on the order?
zero = M * s⁻¹
first = s⁻¹
second = M⁻¹ * s⁻¹
etc…
at higher temperatures, reactions proceed at a _____ (faster/slower) rate
faster
rate constant inc exponentially as temp inc
collision frequency
Z = collision frequency, asks # of molecular collisions per second
reaction rate is proportional to collision frequency, Z
for reaction A + B → Products…
Z = Z₀[colliding species]
collision theory
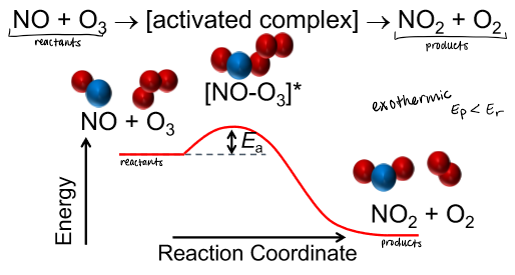
not all collisions result in formation of product; this is dependent on activation energy
activation energy
Ea
minimum collision energy required for reaction to occur
represents barrier to reaction proceeding
activated complex
least-stable (highest energy) arrangement of atoms that occurs in reaction
influence of temperature on kinetic energy
fraction of collisions w energy in excess of Ea equals e^(-Ea/RT)
rate of reaction is proportional to collision frequency times fraction of collisions w energy in excess of Ea
steric factor
steric factor, p, accounts for orientation of reactants
rate = p * Z₀[colliding species] * e^(-Ea/RT)
Arrhenius Equation
exponential form: k = A * e^(-Ea/RT)
linear form: lnk - lnA - Ea/R * 1/T
point-slope form: ln(k₁/k₂) = -Ea/R(1/T₁ - 1/T₂)
catalyst
substance that inc rate of reaction but is not consumed in reaction
provides alternate reaction path w/ lower Ea
first appears on reactant side
homogenous catalyst
catalyst that’s present in same phase as reactant
heterogeneous catalyst
catalyst that’s present in diff phase from reactants
enzyme catlysis
enzymes are large molecules are catalyze reactions they are made specifically for
enzymes are active under mild reaction conditions
elementary step
chem equation that describes actual molecular-level collision
mechanism
sequence of elementary steps that leads from reactants to products
intermediate
substance produced in an early step and consumed in a later step
first appears on product side
molecularity
# of reactant species involved in certain elementary step
most steps are either unimolecular (involves one molecule) or bimolecular (involves two molecules)
rate laws for elementary steps
rate = k[reactant species]^(coeff of reactant in eq)
rate-limiting step
aka slow step
determines overall rate of multi-rate reaction
fast steps before slow step usually affect concentrations of reactant species in rate-determining step (but the fast steps after have no effect)
typically, a fast step reaches ____. since rates at _____ are equal, then…
equilibrium (for both blanks)
rate₁ = rate-₁
Michaelis-Menten mechanism (enzyme catalysis)
step 1: enzyme binds to substrate in rapid, reversible reaction to form complex; this step reaches equilibrium
E + S ⇌ ES
step 2: product forms from complex, and enzyme is released; this is the rate limiting step
ES → E + P
the reaction rate is ___ order in substrate, S (and explain why)
zero
under normal conditions, [S] is much greater than [E]₀
nearly all enzymes are bound to substrate, so [ES] ~ [E]₀, and rate = k₂[E]₀ = constant
change in enthalpy
aka ∆H
represents heat absorbed or released by system at constant temp & pressure
work
application of force thru a distance (force * distance)
when system does work, like lifting a weight against gravity, the sign for work is negative
work done by gases
pressure difference causing volume change is also work
w = -P∆V
w can be expressed in L * atm or J
1 L * atm = 101.3 J
w is ____ (neg/pos) when system does work on its surroundings
negative