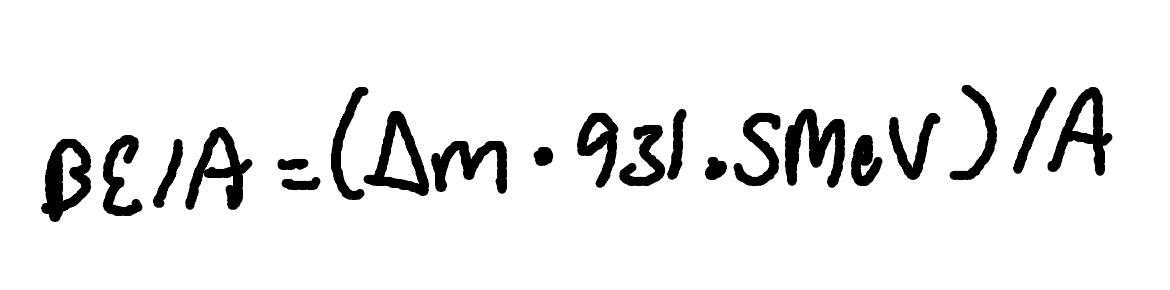PHY 132 T4 Formulas
1/33
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
Lorentz factor
where
γ = Lorentz factor
v = relative speed
c = speed of light (3×10^8m/s)
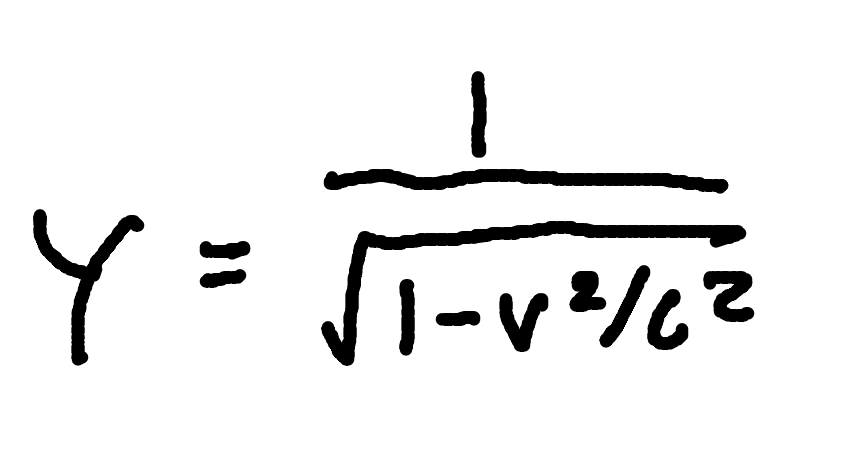
Time Dilation
where
Δt = dilated time
Δt0 = proper time.
γ = Lorentz factor
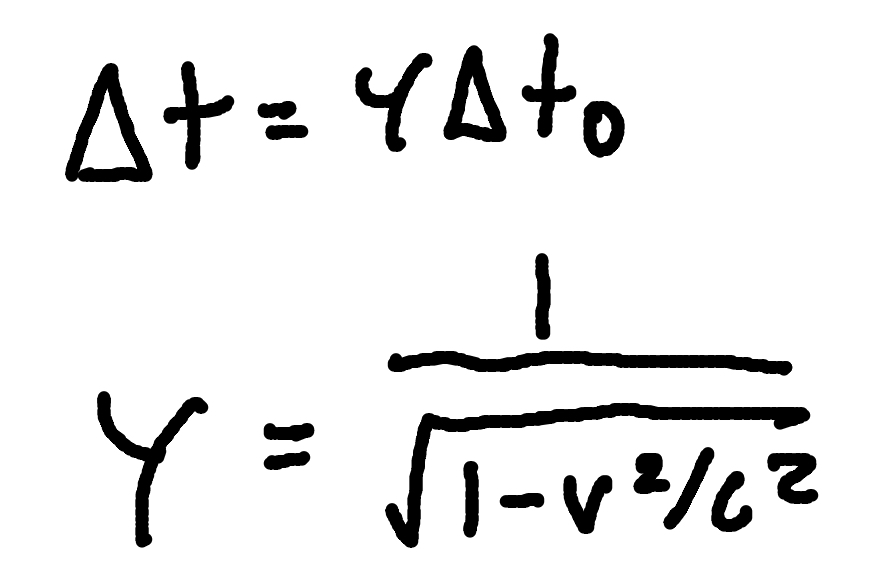
Length Contraction
where
γ = Lorentz factor
L0 = proper length
L = contracted Length
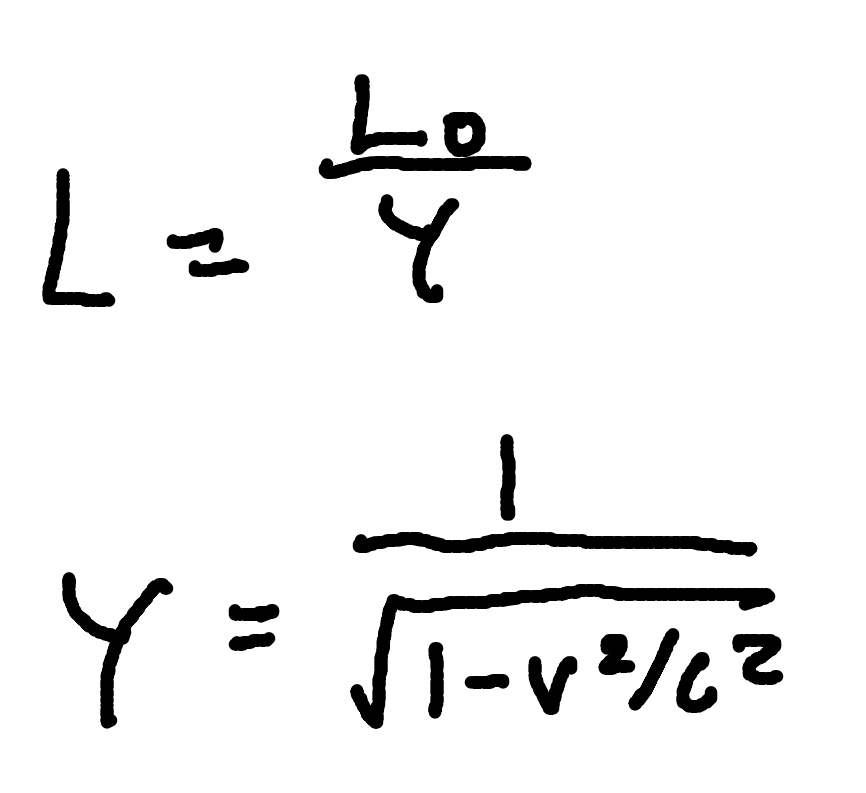
Lorentz Transformations
where
γ = Lorentz factor
x-hat = position in a moving frame
x = position in one frame
t= time
v = speed frame
t-hat = time in a moving frame
c = speed of light (3×10^8m/s)
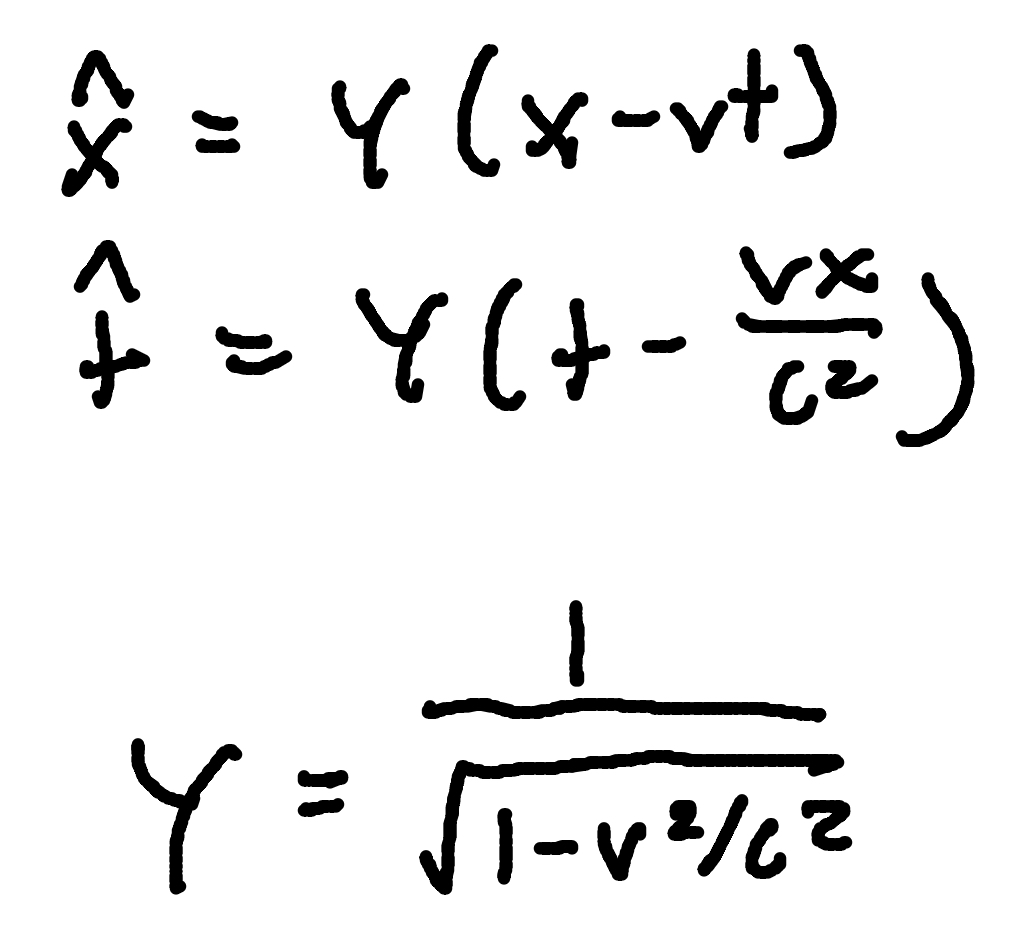
Relativistic velocity addition
where
u = velocity in one frame
u’ = velocity in moving frame
v= frame speed
c= speed of light (3 × 10^8m/s)
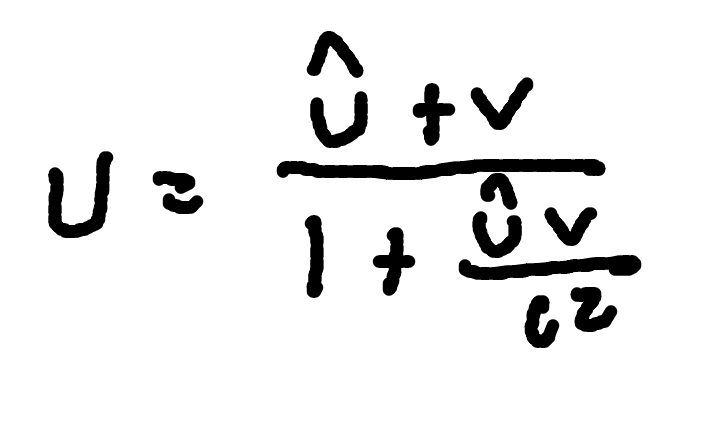
Relativistic Momentum
where
p = relativistic momentum
γ = Lorentz factor
m0 = rest mass
v = speed
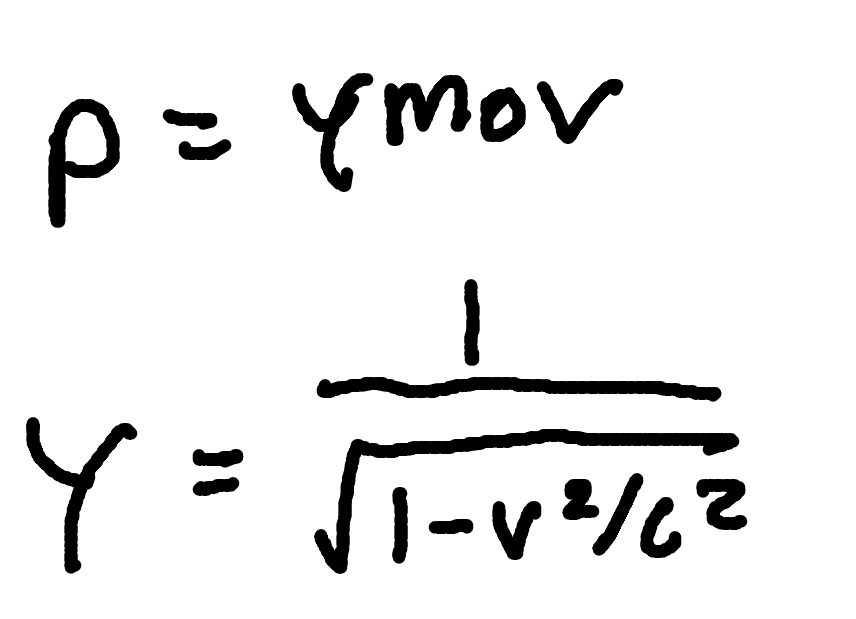
Relativistic Kinetic Energy
where
γ = Lorentz factor
K = Kinetic Energy
m0 = rest mass
c = speed of light (3×10^8m/s)
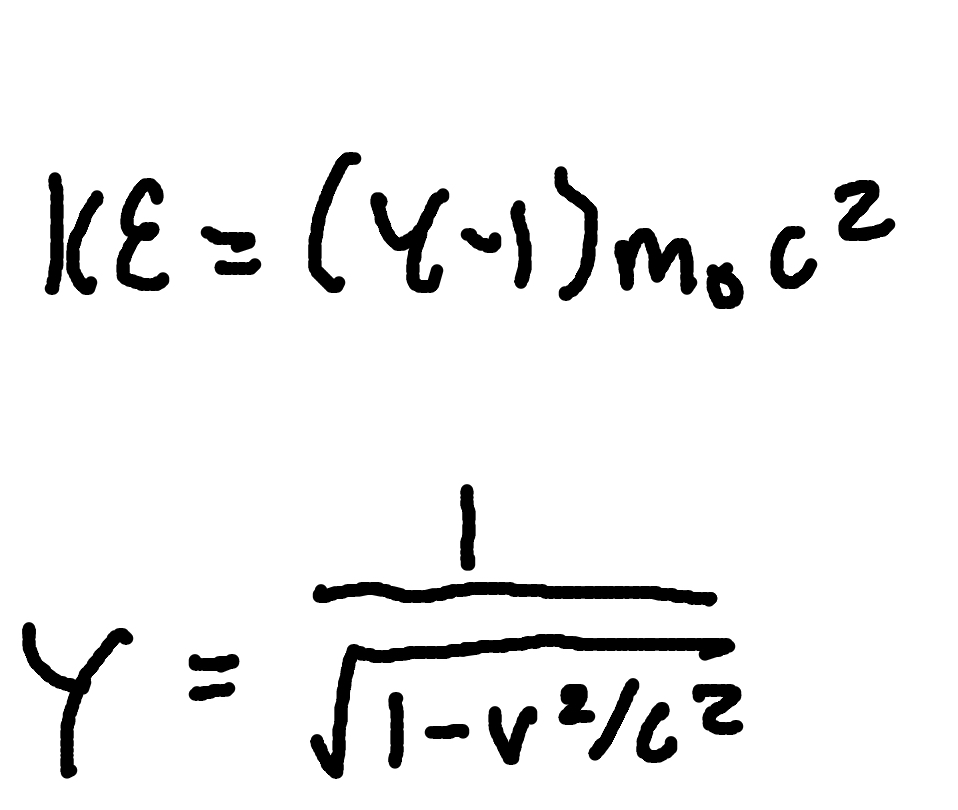
Total Energy
where
γ = Lorentz factor
E = total Energy
m0 = rest mass
c = speed of light (3 × 10^8m/s)
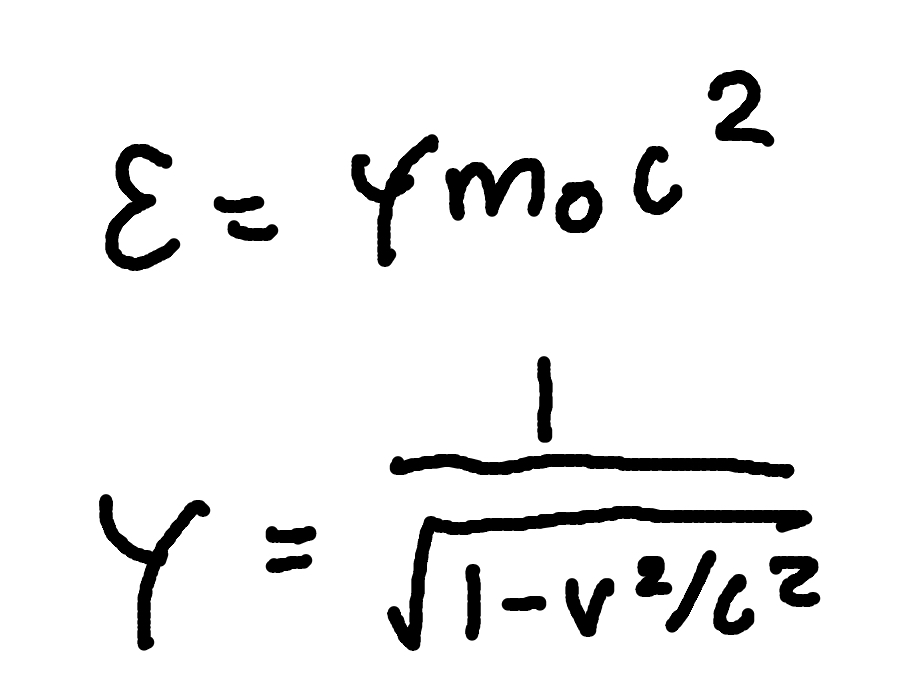
Rest Energy
Where
E0 = Rest energy
γ = Lorentz factor
m0 = rest mass
c = speed of light (3 × 10^8m/s)
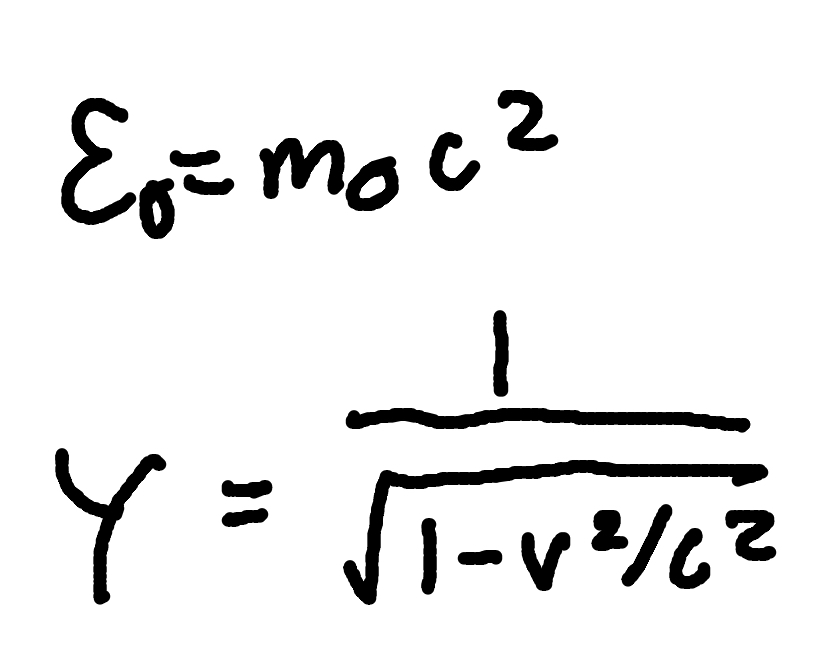
Planck and Photon Energy
where
E = Photon Energy
h = Planck’s Constant (6.626 × 10^-34 J x s)
f = frequency
c = speed of light (3 × 10^8 m/s)
lambda = wavelength
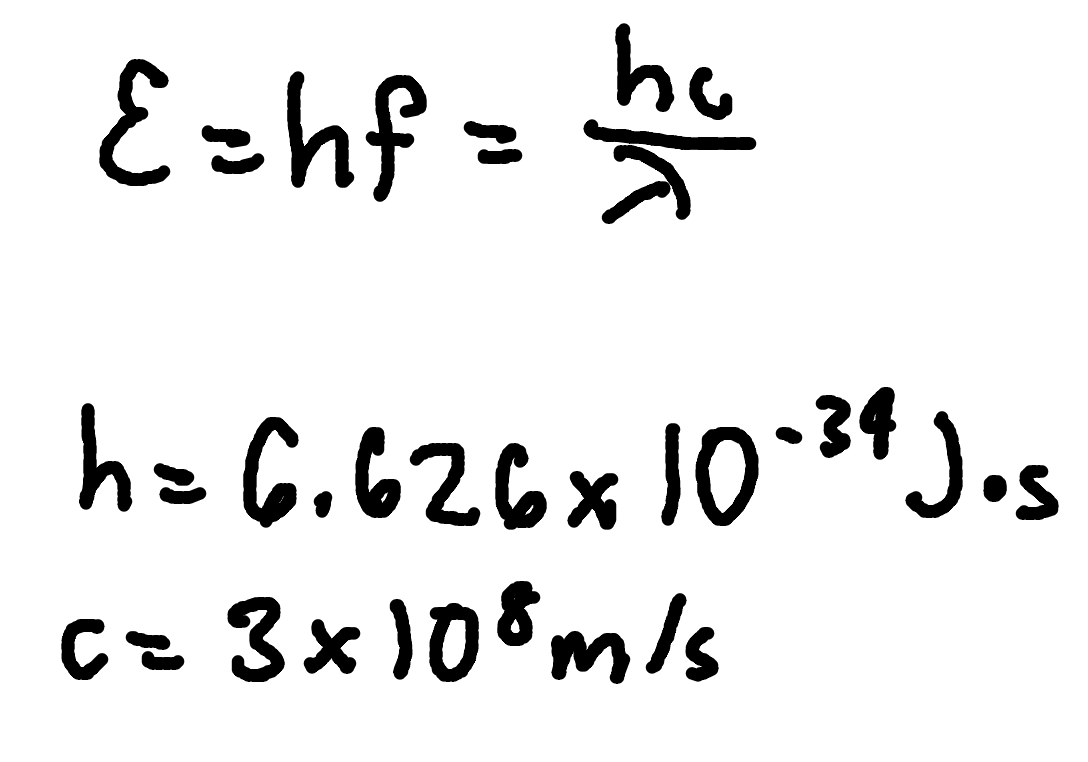
Photon Momentum
where
p = photon momentum
E = photon energy
c = speed of light (3 × 10^8m/s)
h = Planck’s Constant (6.626 × 10^-34 J x s)
f = frequency
lambda = wavelength
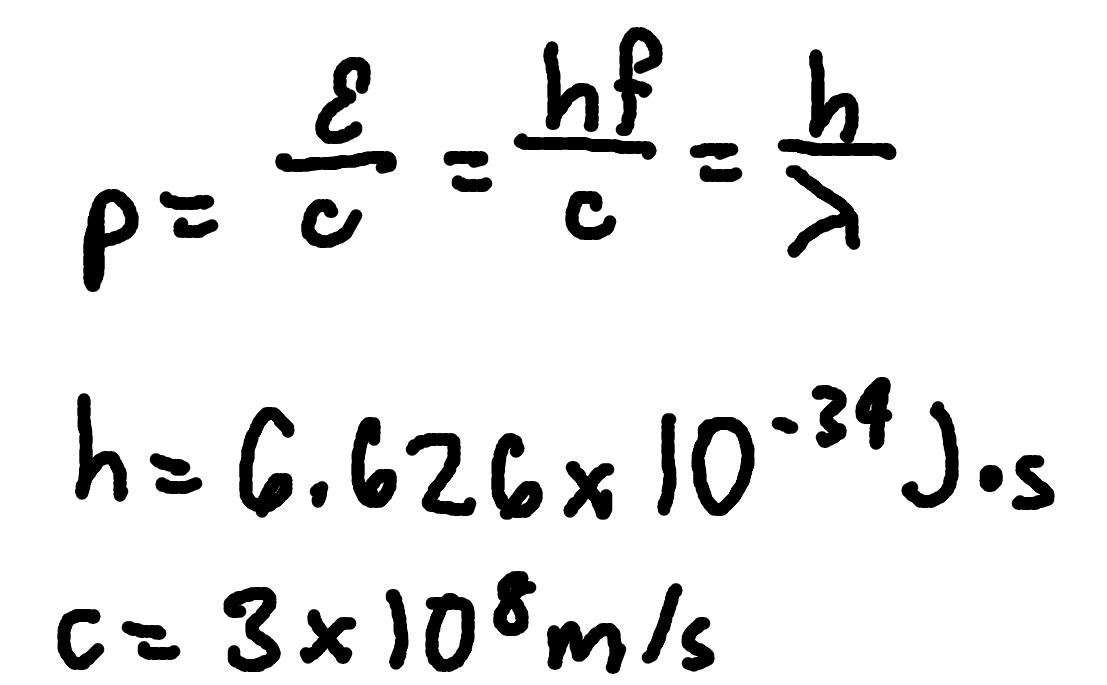
Photoelectric effect
where Kmax = max electron kinetic energy,
ϕ = work function,
f = frequency.
h = Planck’s Constant (6.626 × 10^-34 J x s)
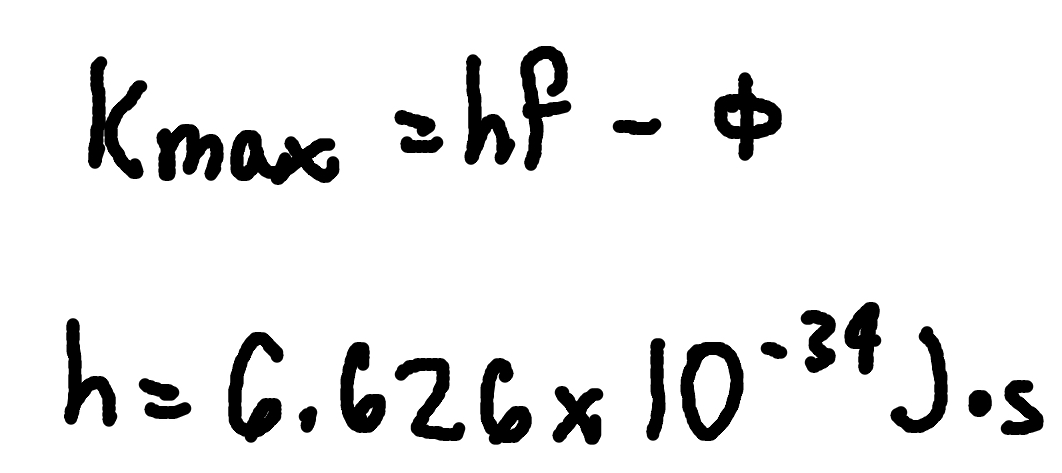
Threshold Frequency
where
f0 = threshold frequency
ϕ = work function
h = Planck’s Constant (6.626 × 10^-34 J x s)
Number of photons
Where
N = number of photons
Etotal = Total Energy
h = Planck’s Constant (6.626 × 10^-34 J x s)
f = frequency
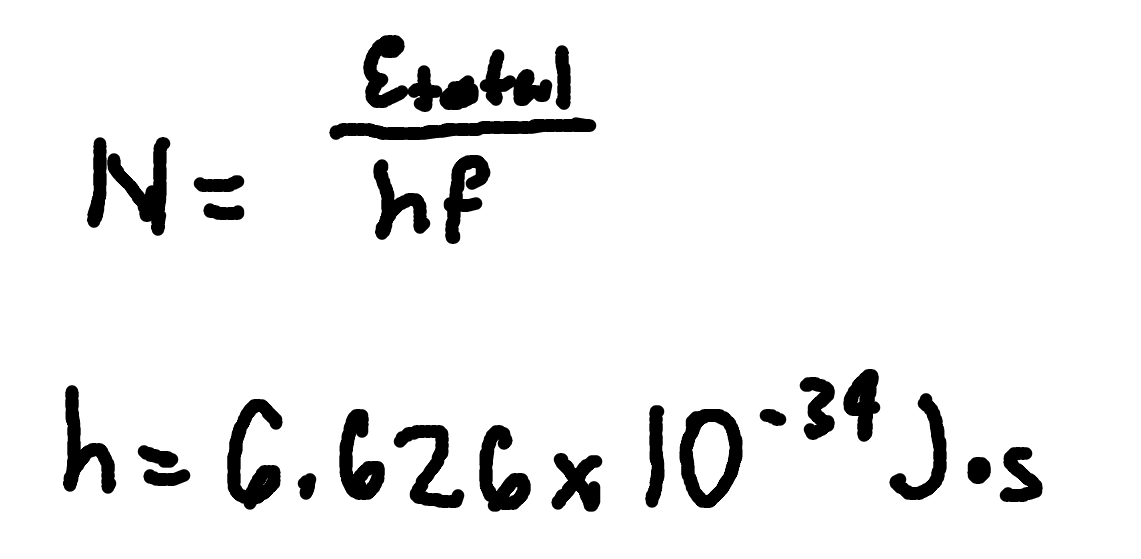
Photons per Second
where
N/t = photons per second
P = power
h = Planck’s Constant (6.626 × 10^-34 J x s)
f = frequency
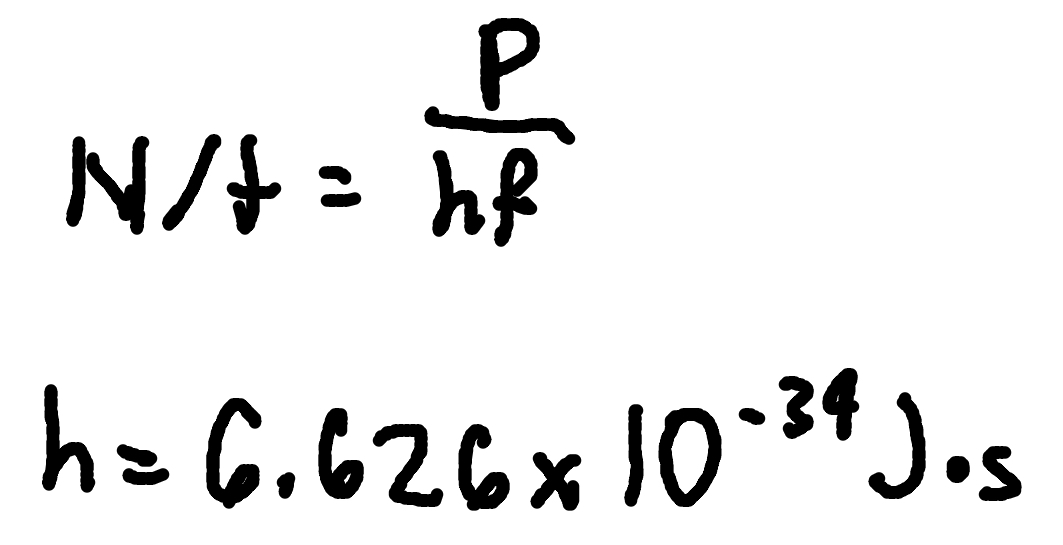
Compton Effect
where
λ′ = scattered wavelength,
λ = initial wavelength,
θ = scattering angle.
h = Planck’s Constant (6.626 × 10^-34 J x s)
me = electron mass (9.11 × 10^-31 kg)
c = speed of light ( 3 × 10^8 m/s)
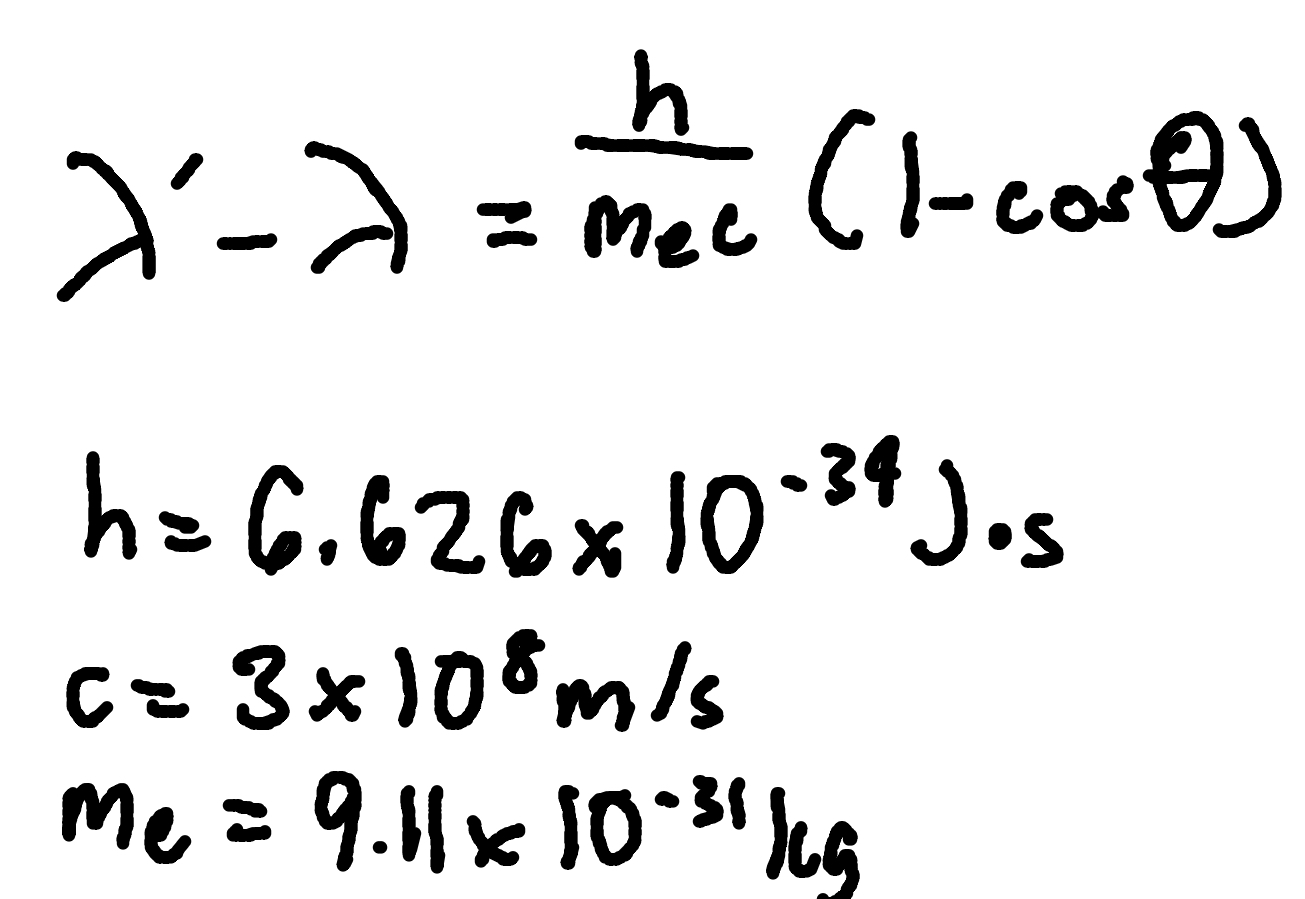
de Brogile Wavelength (Relativistic)
where
lambda = de Broglie wavelength
h = Planck’s Constant (6.626 × 10^-34 J x s)
p = photon momentum
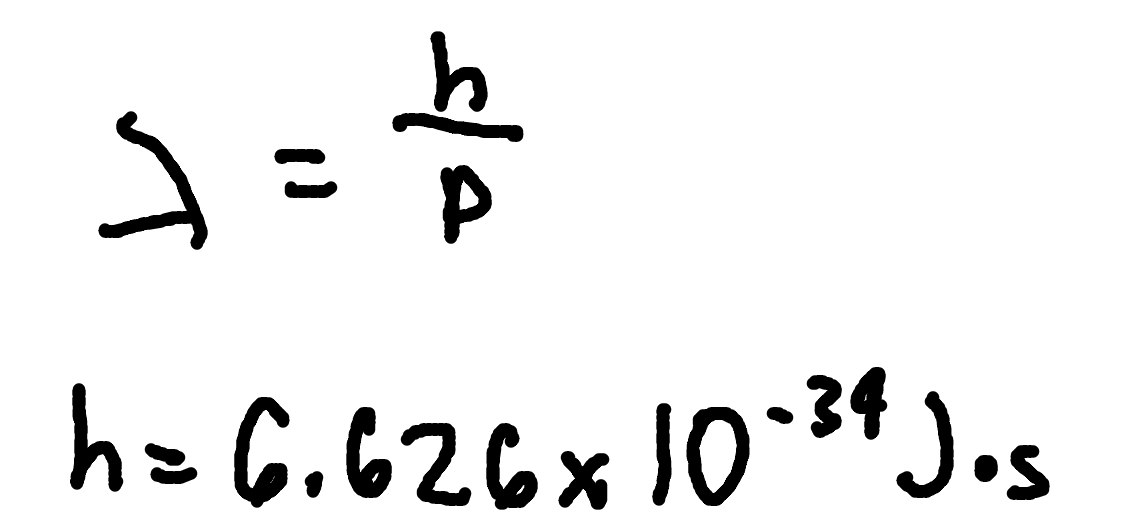
de Brogile Wavelength (non-relativistic)
where
lambda = de Brogile wavelength
m = mass
v = speed
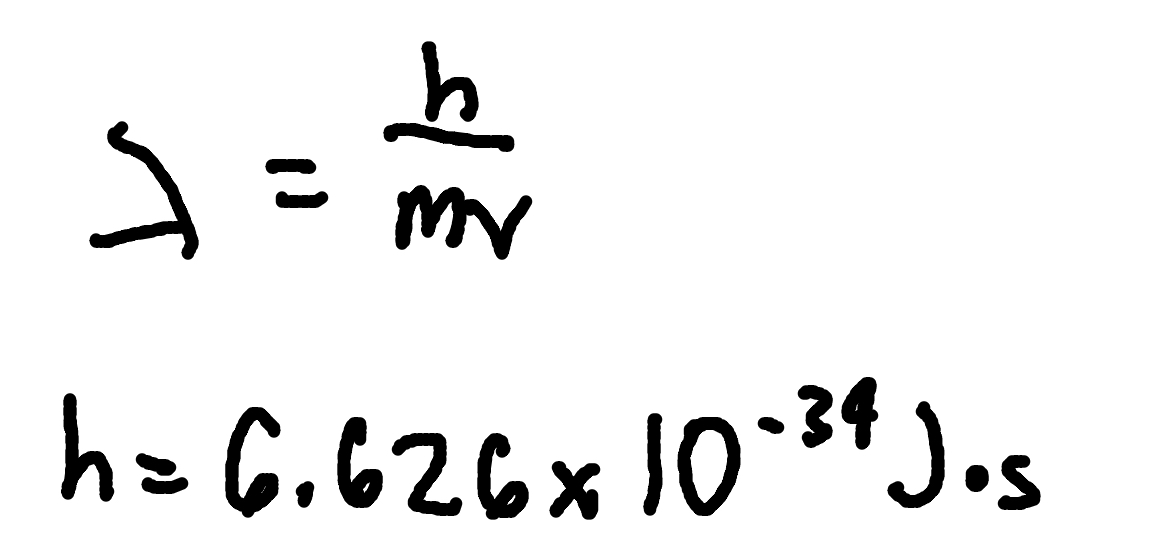
Hydrogen Spectral Lines
where
λ = emitted wavelength,
R = Rydberg constant(1.097 × 107 m−1)
nf = final level,
ni = initial level.
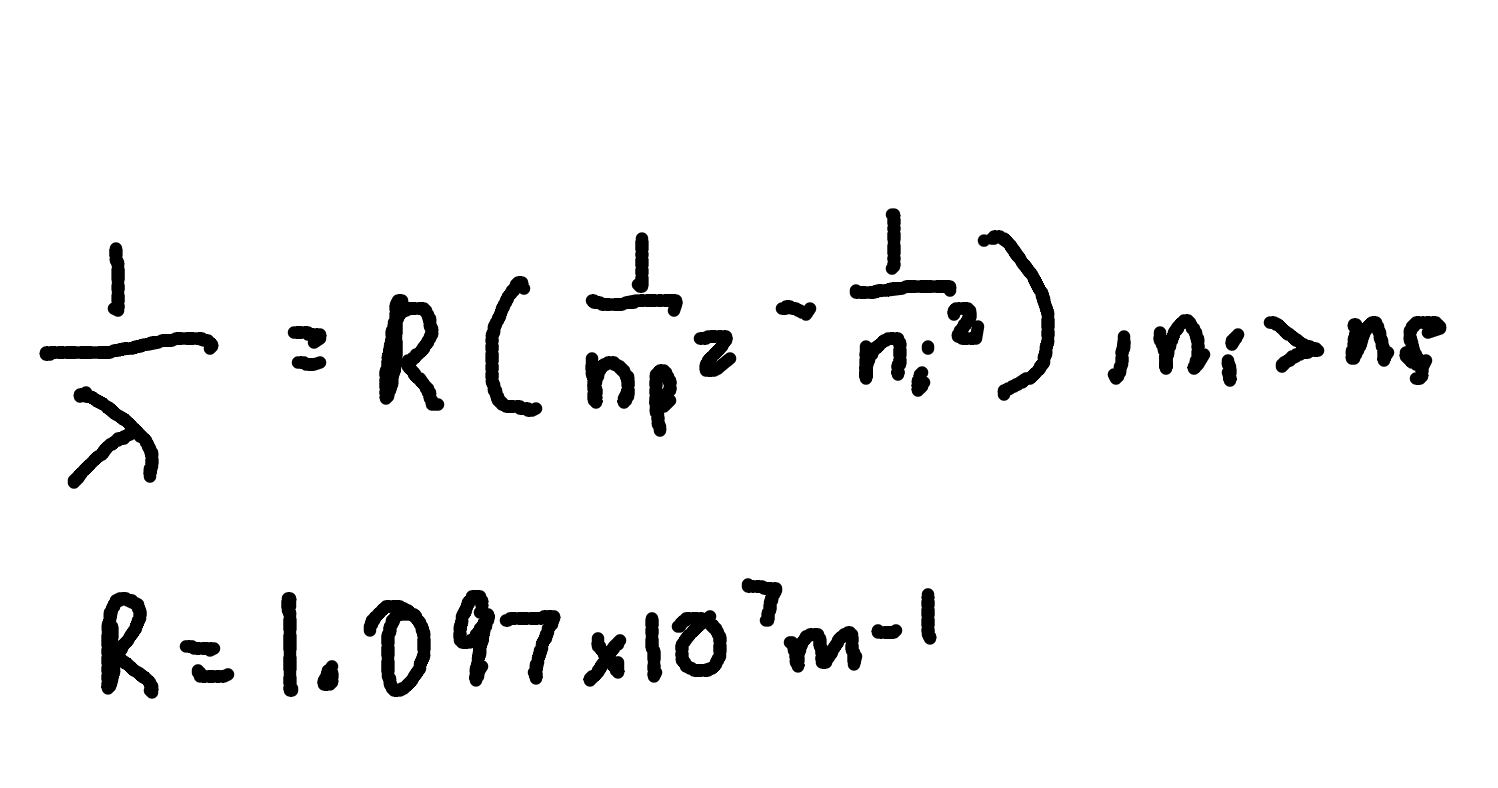
Angular Momentum Quantization
where
me = electron mass (9.11 × 10^-31 kg)
v = orbital speed,
rn = orbit radius,
n = principal quantum number.
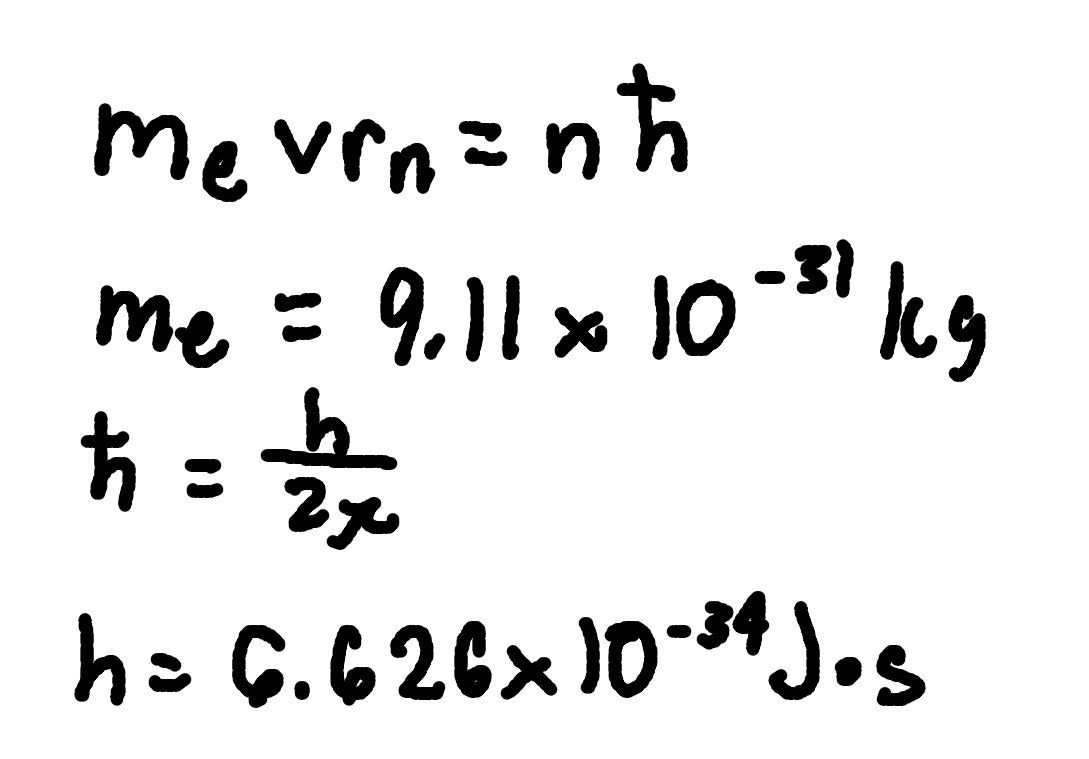
Allowed orbit radii for Hydrogen
where
rn = orbit radius,
n = quantum number,
aB = Bohr radius (.529 × 10^-10m)
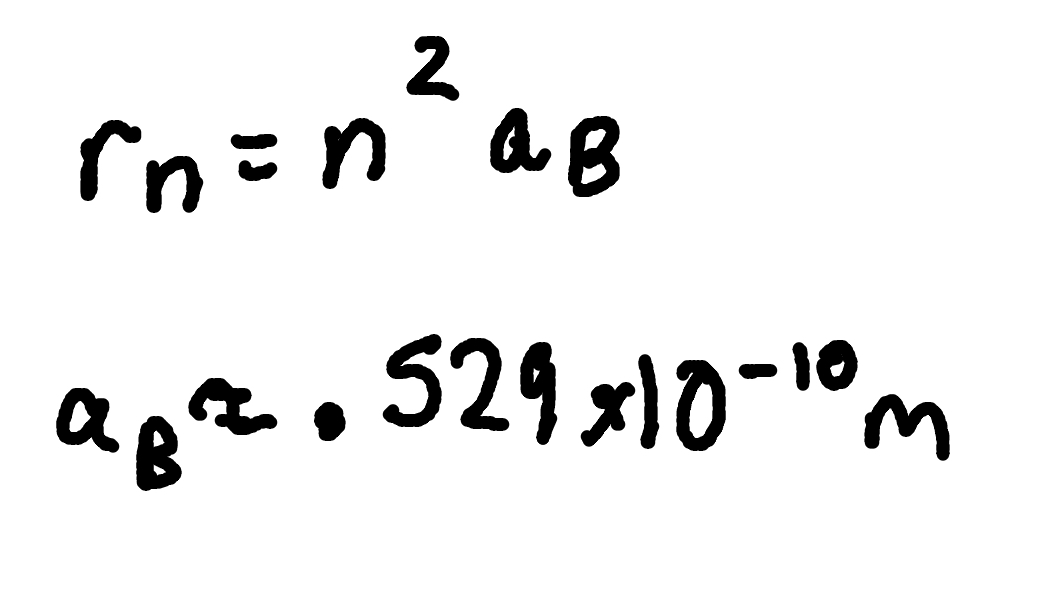
Hydrogen Energy Levels
where
En = energy of level n,
n = principal quantum number.
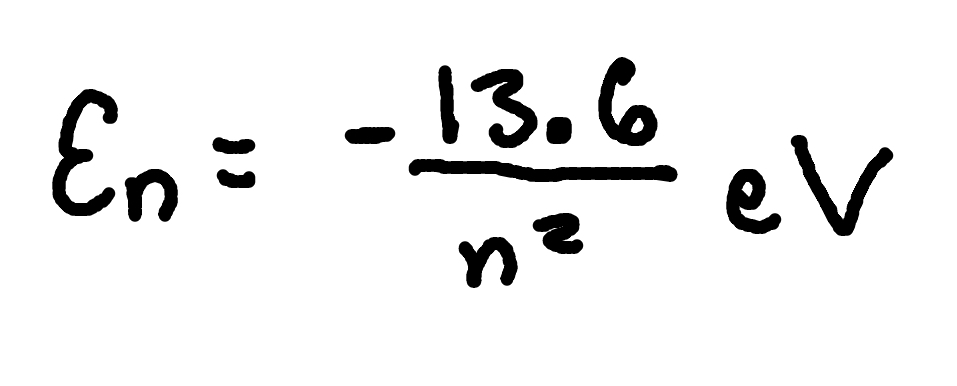
Ionization energy
For hydrogen from ground state: 13.6 eV.
Where (from level n):
E ionize from n = Ionization energy for n state
En = Energy level n
n = principal quantum number
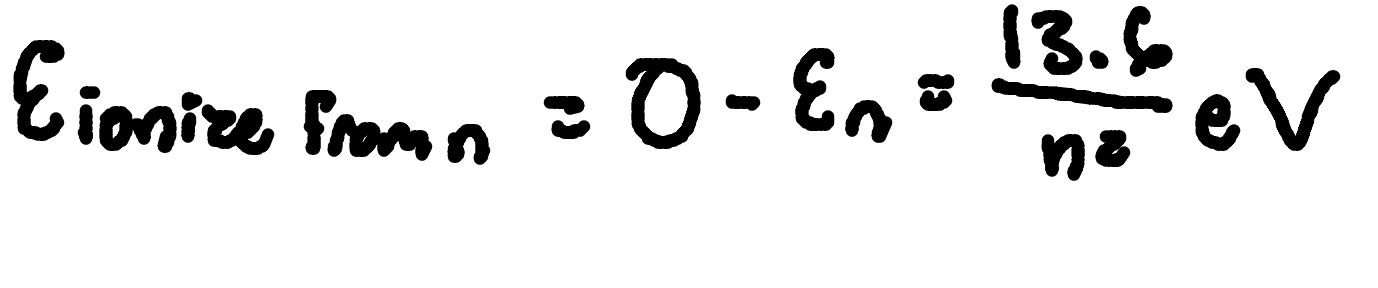
Transition Energy and Emitted Photon
where
ΔE = transition energy,
Ei = initial energy,
Ef = final energy,
f = frequency,
λ = wavelength.
h = Planck’s Constant (6.626 × 10^-34 J x s)
c = speed of light (3 × 10^8m/s)
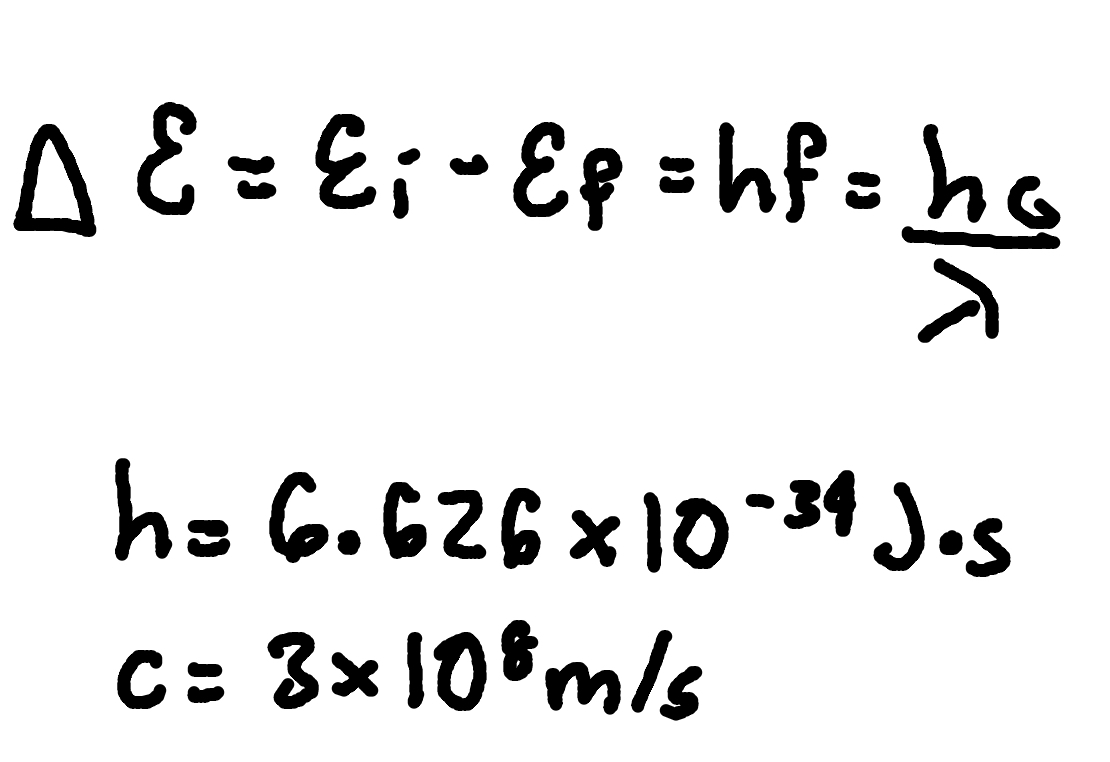
Orbit Radius (Hydrogen-like ion)
where
rn,Z = orbit radius for hydrogen-like ion,
Z = nuclear charge number.
n = principal quantum number
aB = Bohr radius (.529 × 10^-10m)
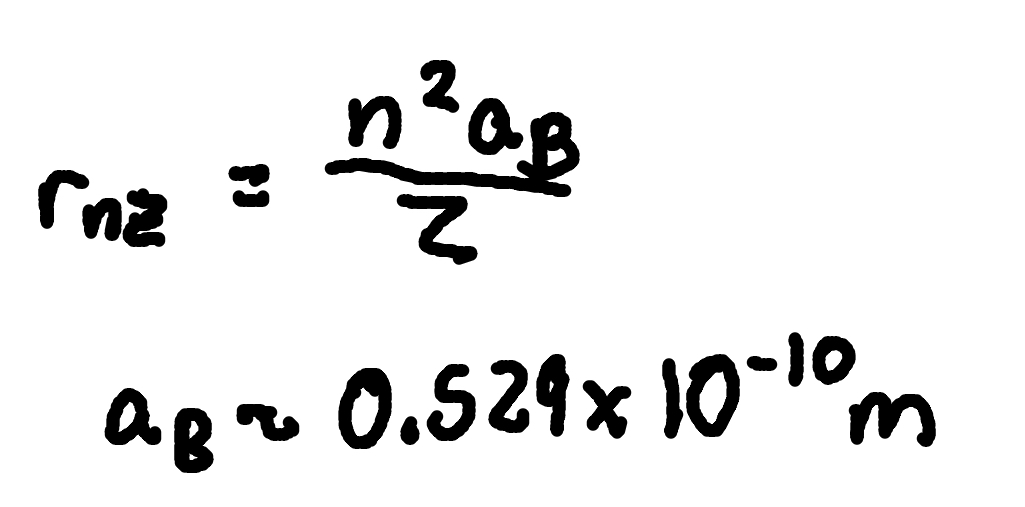
Energy Levels (Hydrogen-Like ions)
where
En,Z = energy level for hydrogen-like ion,
Z = nuclear charge number.
n = principal quantum number
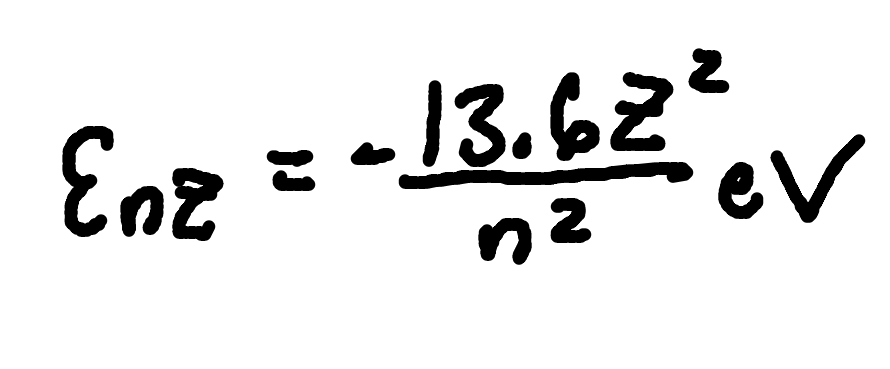
Total Energy (relativistic momentum)
Where
E = total energy
p = relativistic momentum
c = speed of light (3 × 10^8 m/s)
m0 = rest mass
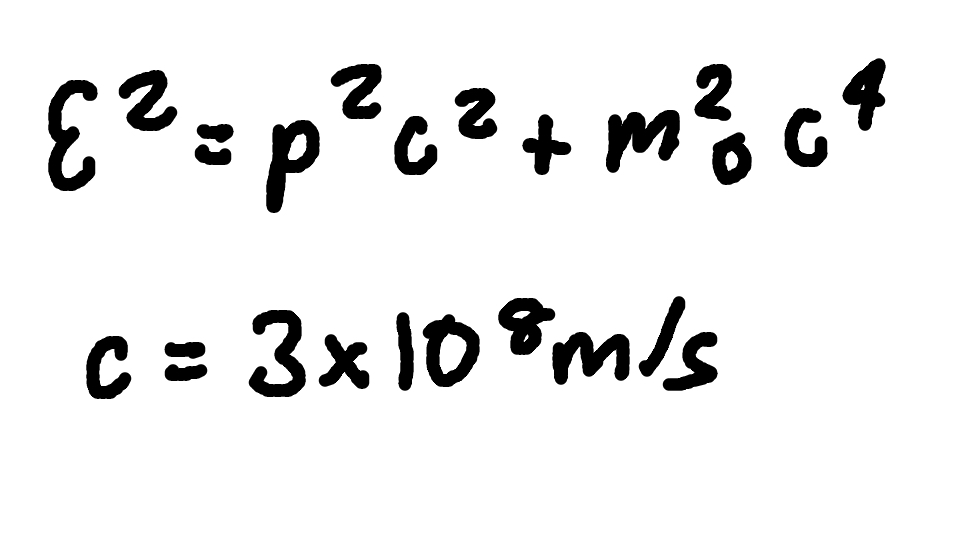
Bohr’s radius
where
aB = bohr’s radius
me = electron mass (9.11 × 10^-31 kg)
k = Coulomb constant (9 × 10^9 Nm²/C²)
e = charge magnitude (1.6 × 10^-19 C)
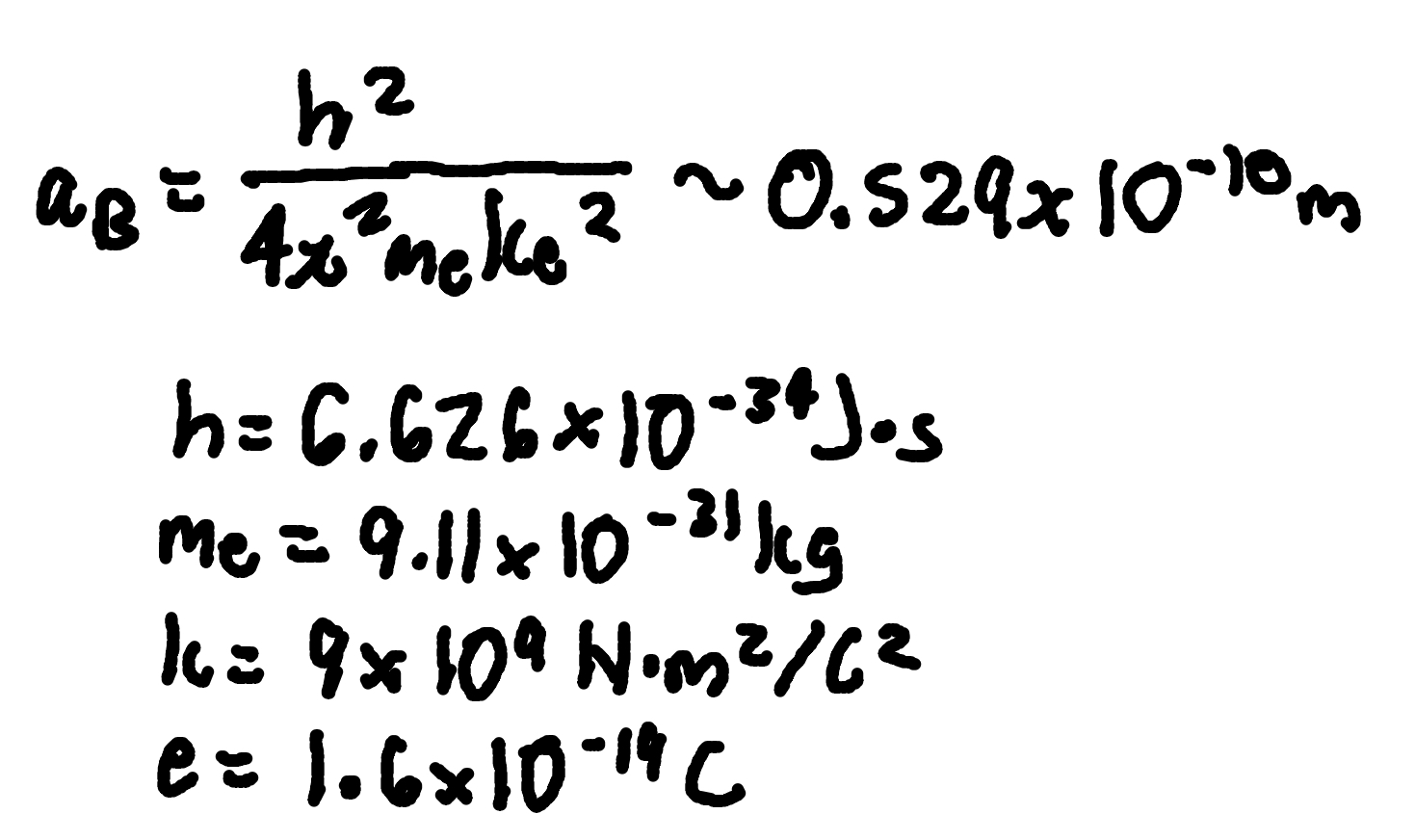
de Brogile frequency
where
f = frequency
h = Planck’s constant (6.626 × 10^-34 J x s)
E = total energy
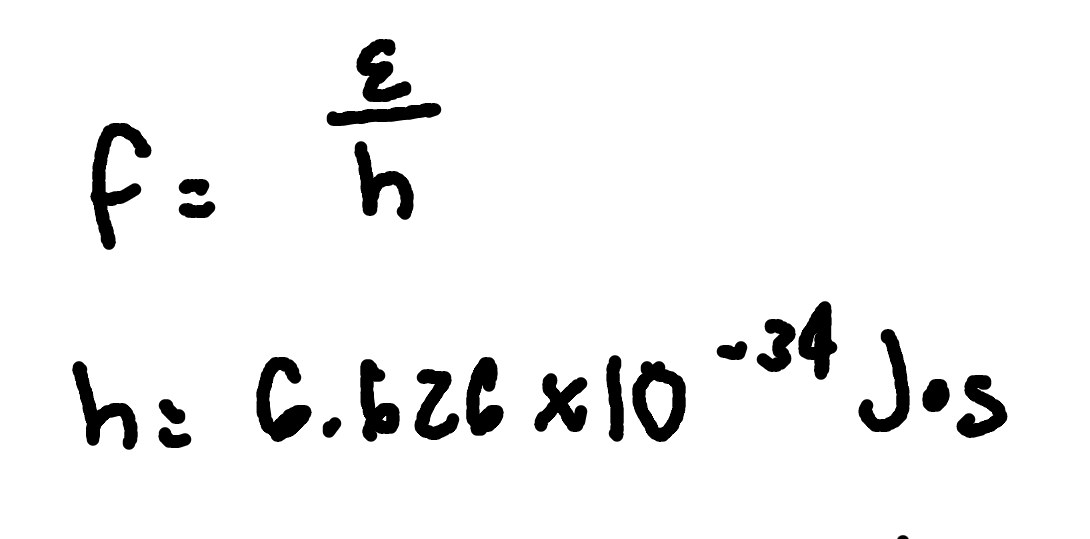
Element Notation
where
Z = atomic number
A = number of nucleons
X = chemical symbol for element
N = number of neutrons
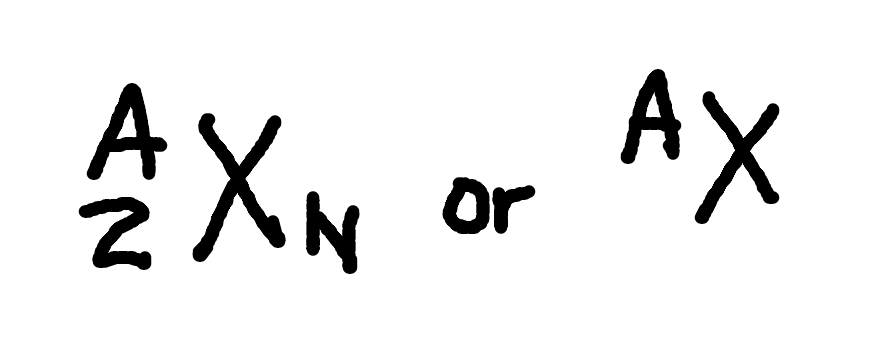
Radioactive Decay
where
N(t) = number of nuclei at time t
N0 = initial number of nuclei (i.e. at t = 0)
lambda = “decay constant” (related to t1/2 by lambda = ln2/t1/2)
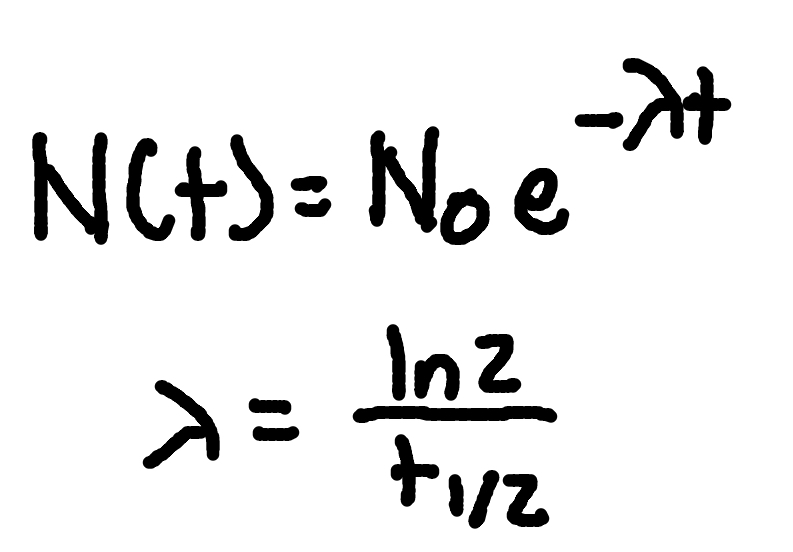
Relativistic mass
where
m(v) = relativistic mass
γ = Lorentz factor
v = velocity
c = speed of light (3×10^8m/s)
Mass defect
where
delta m = mass defect
Z = number of protons
mp = proton mass (1.00728amu)
N = number of neutrons
mn = neutron mass (1.00867 amu)
1 amu = 1.66054 × 10^-27kg “=“ 931.5MeV
Mnucleus = nucleus mass
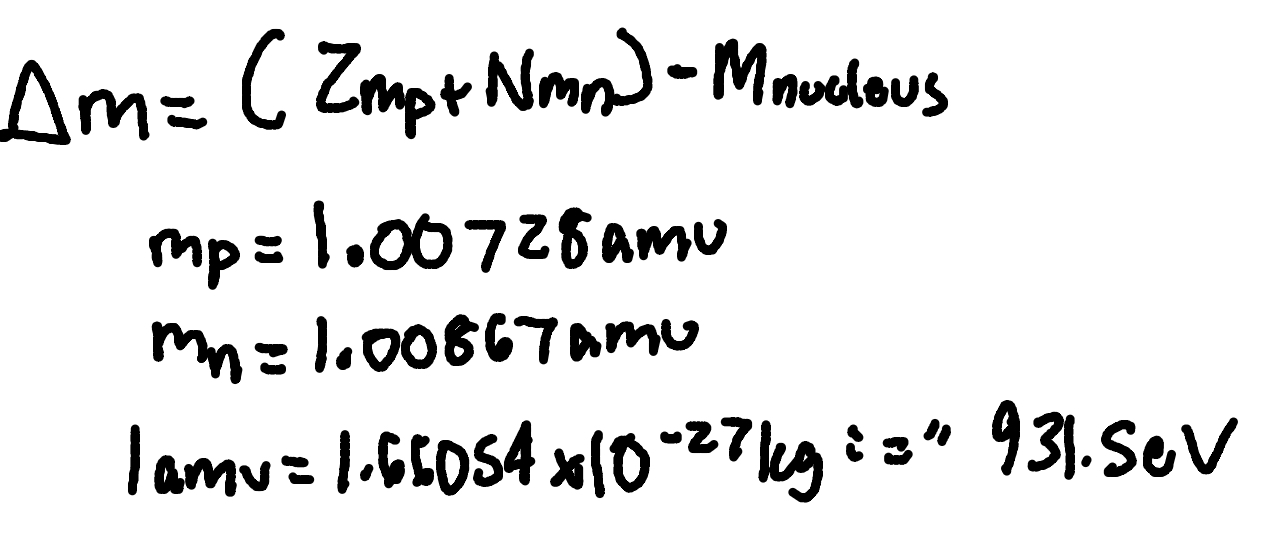
Binding energy per Nucleon
Where
BE/A = Binding energy per nucleon
A = Number of nucleons (Mass number)