Battle of Mechanisms / Properties
1/10
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
11 Terms
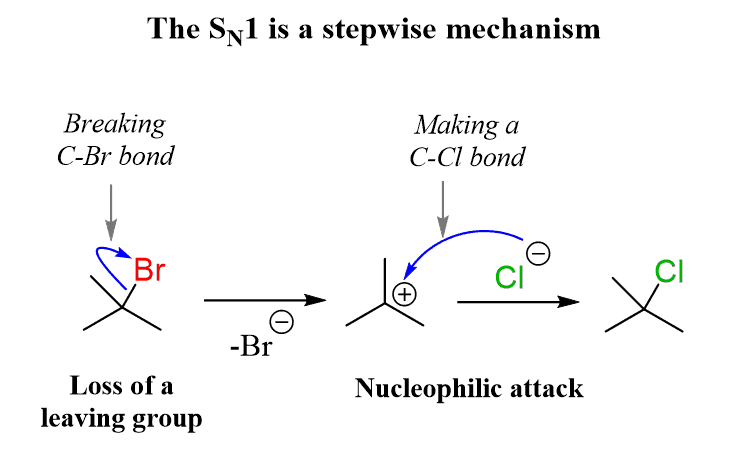
Sn1 Conditions / Mechanism
Mechanism: 2 Steps
Conditions:
Weak Nucleophile
Polar Protic Solvent (H bonds slow the nucleophile)
Electrophiles must be capable of stabilizing carbocation (good resonance/hyperconjugation)
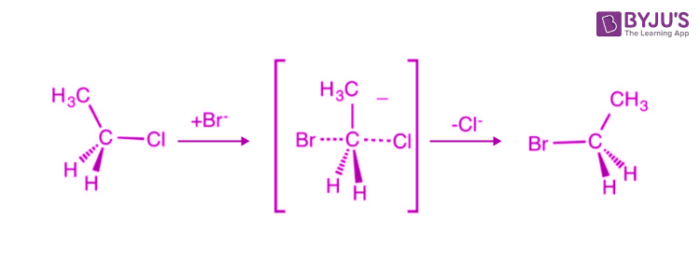
Sn2 Conditions / Mechanism
Mechanism: 1 Step
Conditions:
Strong Nucleophile
Polar Aprotic Solvent
Carbon electrophiles must have least steric hinderance and resonance withdrawing)
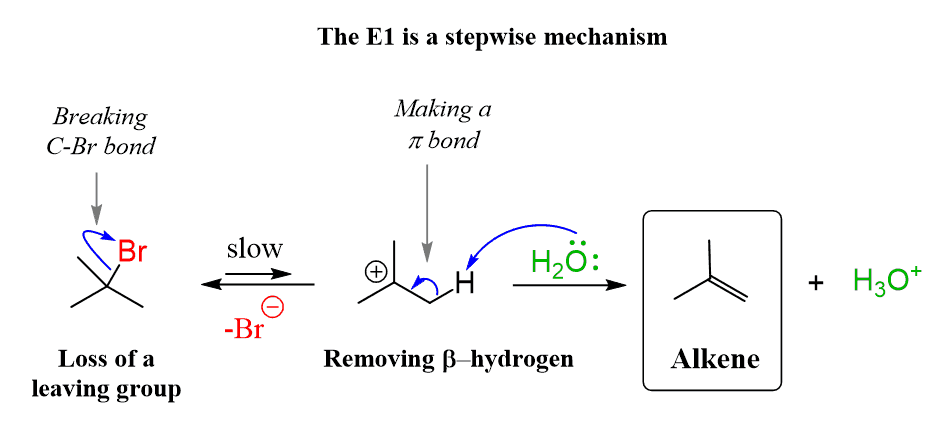
E1 Conditions / Mechanism
Mechanism: 2 Steps
Conditions:
Weak base
Heat
carbon electrophile needs electron donating (C-H) groups
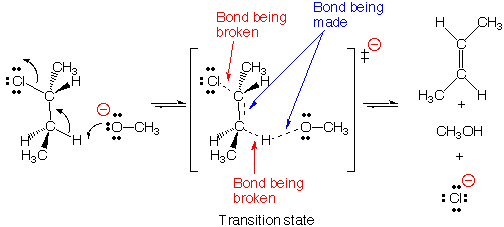
E2 Conditions / Mechanism
Mechanism: 1 Step
Conditions:
heat
carbon electrophile must have sigma beta C-H bond anti to sigma alpha C-LG bond)
Strong Base
Weak Base & Weak Nucleophile
Neutral, highly electronegative atoms
Weak Base & Strong Nucleophile
Large, Polarizable Atom
Resonance Stabilized negative charge on smallest atom
Strong Base & Strong Nucleophile
Negative charge on smallest atom without any resonance stabilization
Strong Base & Weak Nucleophile
Sterically hindered / BONUS if negative charge on smallest atom
pKa Factors
Electronegativity —> Higher the EN of atom directly bonded to H = Higher acidity
Hybridization —> Higher the S% character = Higher Acidity
Polarizability —> The larger/more distortable the electron cloud of atom directly bonded to H = Higher Acidity
Resonance —> if lone pair of conjugate base can be delocalized through resonance, more acidic
Induction —> the more Electronegative the atom near the H = higher acidity
Hyperconjugation —> more alkyl groups = lower acidity
Differences in Enantiomer Properties
How they interact with other chiral molecules
How they rotate plane polarized light in different directions
Types of Enantiomer Mixtures
Enantiomerically Pure —> Only one type of enantiomer = light rotated only to one direction
Enantiomer Excess —> Major Enantiomer % - Minor Enantiomer %
Racemic (50/50) —> No light rotation (cancels out)