Chemistry 1B
0.0(0)
Studied by 5 peopleCard Sorting
1/42
Earn XP
Description and Tags
Last updated 2:44 AM on 9/29/22
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
43 Terms
1
New cards
Forward Reaction
Products only
2
New cards
Reverse Reaction
Reactants only
3
New cards
Dynamic Equilirbium
Forward reaction = Reverse reaction
4
New cards
equilibrium expression
K= ([product1]^(coefficient) X [product2]^(coefficient))/([reactant1]^(coefficient) X [reactant2]^(coefficient))
![K= ([product1]^(coefficient) X [product2]^(coefficient))/([reactant1]^(coefficient) X [reactant2]^(coefficient))](https://knowt-user-attachments.s3.amazonaws.com/e8f50c60e2fd436da2db356a2038472b.jpeg)
5
New cards
What states of matter should be included in equilibrium expression?
Aqeous, gas
6
New cards
What states of matter should NOT be included in the equilibrium expression?
Solids, liquids
7
New cards
If K = 1
Reaction doesn't favor either side
8
New cards
If K>1
Reaction is product favored
9
New cards
If K
Reaction is reactant favored
10
New cards
If K>10^10
Reaction is NOT reversible
11
New cards
If K
No reaction
12
New cards
You can use the equilibrium expression for what quantities?
Partial pressure and concentrations
13
New cards
Molarity equation
M= n/V
14
New cards
moles equation
n = PV/RT
15
New cards
Partial pressure and concentration equilibrium values
Kp = Kc (RT)^Δn
16
New cards
K relationships: Rxn reversed
Inverted K (1/K)
17
New cards
K relationships: rxn multiplied
Raise K by factor (K^x)
18
New cards
K relationships: Rxn divided
Root K by factor (x√K)
19
New cards
2+ Reactions added
Multiply K values (K1*K2)
20
New cards
Lechatliers Principle
If rxn is disturbed, it will shift to a new equilibrium

21
New cards
What is RDS
The slowest reaction is the Rate Determining Step in a series and determines rate law
22
New cards
Molecularity
number of molecules involved
23
New cards
Activation energy and RDS
RDS will have the highest activation energy
24
New cards
What does Rate law do?
Shows relationship between rate of rxn and concentration
25
New cards
How is Rate law determined
experimentally ONLY
26
New cards
Rate law equation
rate = K [A]^m[B]^n[C]^c (m,n,c determined experimentally)
![rate = K [A]^m[B]^n[C]^c (m,n,c determined experimentally)](https://knowt-user-attachments.s3.amazonaws.com/3034f07800404c39b7c65f74cec5e217.jpeg)
27
New cards
If:
A+B +C -> product
And you know:
rate =[A]^1*[B]^2*[C]^0
What happens if you double A?
A+B +C -> product
And you know:
rate =[A]^1*[B]^2*[C]^0
What happens if you double A?
Rate X 2
28
New cards
If:
A+B +C -> product
And you know:
rate =[A]^1*[B]^2*[C]^0
What happens if you double B?
A+B +C -> product
And you know:
rate =[A]^1*[B]^2*[C]^0
What happens if you double B?
Rate X 4
29
New cards
If:
A+B +C -> product
And you know:
rate =[A]^1*[B]^2*[C]^0
What happens if you double C?
A+B +C -> product
And you know:
rate =[A]^1*[B]^2*[C]^0
What happens if you double C?
no effect
30
New cards
If:
A+B +C -> product
And you know:
rate =[A]^1*[B]^2*[C]^0
What happens if you double A,B, and C?
A+B +C -> product
And you know:
rate =[A]^1*[B]^2*[C]^0
What happens if you double A,B, and C?
Rate X 8
31
New cards
What is the rate of chemical reactions?
How fast reactants make products
32
New cards
what happens to the concentration of a reactant over time in a reaction?
Concentration lowers over time
33
New cards
How to find instantaneous rate of change of a reactant? products??
Rate = -1/coefficient * (change in concentration)/(change in time)

34
New cards
What are the three techniques used to monitor reaction mixtures?
Polarimetry, spectrophotometry, pressure measurement
35
New cards
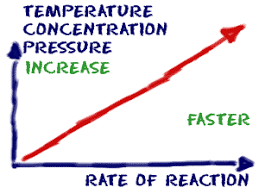
What raises rate of reaction?
↑ Temperature, ↑pressure, ↑Concentration, ↑ volume, ↑ surface area, + catalyst
36
New cards
integrated rate law
Equation showing relationship between concentration and time

37
New cards
First order integrated rate law
ln[A] = -K*t*ln[Ἁ]
![ln[A] = -K*t*ln[Ἁ]](https://knowt-user-attachments.s3.amazonaws.com/8ff07ebe272b43129d4b63782b5889c7.jpeg)
38
New cards
Second order integrated rate law
1/[A] = K*t + 1/Ἁ
![1/[A] = K*t + 1/Ἁ](https://knowt-user-attachments.s3.amazonaws.com/a6b395181c214fd2b38d31a590c6e8c8.jpeg)
39
New cards
Zero Order Integrated Rate Law
[A] = -K*t + Ἁ
![[A] = -K*t + Ἁ](https://knowt-user-attachments.s3.amazonaws.com/110b4e7304104b1a9e24b443d7a3668b.jpeg)
40
New cards
Half life of zero order
Ἁ/2K
41
New cards
Half life of first order
0.693/K
42
New cards
Half life of second order
1/KἉ
43
New cards
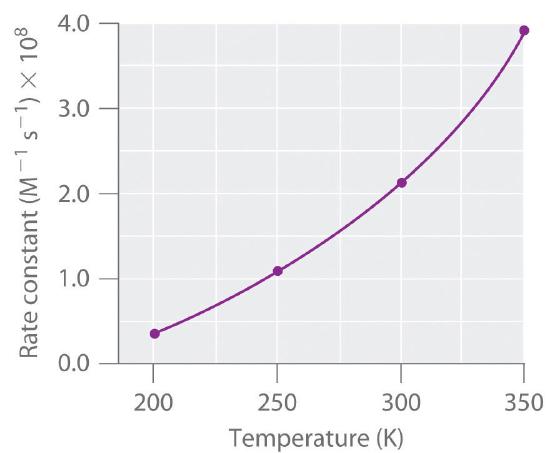
Temperature effect on Rate equation
ln(K) = -(Ea/R)*(1/T)+ln(A)
Ea= Activation energy
T = temperature
R = 8.314 (gas constant)
A = frequency factor
Ea= Activation energy
T = temperature
R = 8.314 (gas constant)
A = frequency factor