(3A) Non-Linear Pharmacokinetics
1/30
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
31 Terms
What drugs have Nonlinear behavior
Phenytoin (Dilantin)
Alcohol
LINEAR PK
Processes =
constant
Means:
Same mechanism regardless of dose
LINEAR PK
KEY IDEA:
Dose ↑ → concentration ↑ proportionally
INTERMITTENT INFUSION SHORTCUT
If SAME:
Dosing interval
Infusion time
Concentration type
👉 Then:
Can use simple proportion
If NOT same:
Must do:
Step-by-step PK math
I THINK WE WILL HAVE QUESTION ON INTERMITTENT INFUSION
NONLINEAR PK DEFINITION
Processes:
NOT constant
Change with:
Dose
Concentration
NONLINEAR PK
MAIN CAUSE
Saturation of enzymes/proteins
OTHER CAUSES
Change in urine pH
Altered hepatic blood flow
Disease states
👉 Not always enzyme-based
HOW NONLINEAR HAPPENS
At low dose:
At high dose:
At low dose:
Plenty of enzyme
Normal metabolism
At high dose:
Enzyme gets saturated
Drug cannot be metabolized properly
👉 System changes
💡 CONCEPT: NOT ONLY METABOLISM
Can also affect:
Absorption
Distribution
Excretion
Linear behavior
Dose ↑ → concentration ↑ proportionally
Half-life constant
Clearance constant
Nonlinear behavior
No predictable relationship ❌
Can see:
Huge increase (↑↑ concentration)
Plateau (no increase)
have no dose-proportionally
involve one or more zero-order process
Drug-drug interactions ↑
Cannot use superposition ❌
No single population parameter
HALF-LIFE CHANGES
Can:
Increase (drug accumulates)
Decrease (less absorption or faster removal)
👉 Depends on cause
DOSE PROPORTIONALITY
Linear →
Nonlinear →
Linear → proportional
Nonlinear → lost proportionality ✅⭐
ZERO-ORDER PROCESS
Occurs when:
System is saturated
Constant rate (not dependent on concentration)
CONCEPT: POPULATION VALUES
Instead of one value:
You may see:
Ranges
Natural (saturable) drug metabolism - STARTS FROM HERE
Michaelis-Menten kinetics
Originally developed:
NOT for drugs
From test tube dye reactions
👉 Later applied to:
Drug metabolism
MICHAELIS-MENTEN KINETICS
We have:
E = enzyme
S = drug (substrate)
MICHAELIS-MENTEN KINETICS
PROCESS
Step 1:
Enzyme + Drug → bind
→ form:
👉 Enzyme-substrate complex (ES)
Step 2:
Binding is reversible
Can bind and unbind
👉 Represented by:
K₁ (forward)
K₋₁ (backward)
Step 3:
Complex → metabolized
→ produces:Metabolite (product)
Enzyme regenerated

MICHAELIS-MENTEN KINETICS
IMPORTANT
Once metabolite forms:
❌ Cannot go back to original drug
MICHAELIS-MENTEN KINETICS
RATE CONSTANTS
K₂ = rate of metabolism
Also represents:
👉 Maximum rate (Vmax concept later)
MICHAELIS-MENTEN KINETICS
Enzyme is:
Released after reaction
Can be reused again
WHERE MICHAELIS-MENTEN KINETICS CAME FROM?
Developed in:
Controlled lab environment
Conditions:
Fixed enzyme
Fixed drug
Clean system
WHEN MICHAELIS-MENTEN FAILS
1. Enzyme concentration changes
Reaction changes enzyme amount
2. Body changes enzyme production
Upregulation
Downregulation
👉 Very common in drugs
3. Drug can be regenerated Example: Phase II reactions
Drug + something (e.g., sugar)
WHEN MICHAELIS-MENTEN FAILS
example
Estrogen:
Gets conjugated (sugar added)
Gut bacteria remove sugar
Drug becomes active again
Gets reabsorbed
👉 This breaks model ❌
KEY PARAMETERS of Michaelis–Menten model - overview
Km = capacity
Vmax = speed
Vmax
Maximum rate of metabolism
Fastest body can metabolize drug
👉 Cannot go faster than this
Km
Concentration threshold
Point where system is:
Getting saturated
INTERPRETATION
If concentration > Km:
If concentration < Km:
If concentration > Km:
System ≈ saturated
Working near Vmax
zero-order (constant rate)
If concentration < Km:
System NOT saturated
Has extra capacity
pseudo-first-order
TWO SCENARIOS in Michaelis–Menten model?
CASE 1: UNSATURATED (LOW CONC)
CASE 2: SATURATED (HIGH CONC)
CASE 1: UNSATURATED (LOW CONC) - equation you use
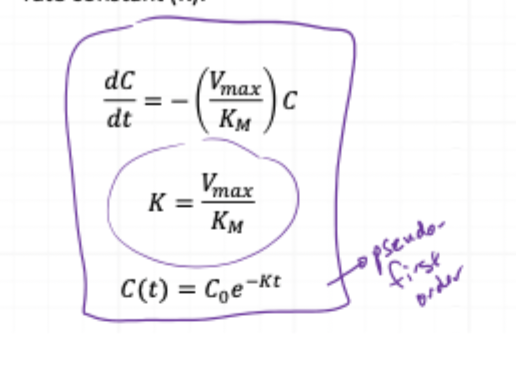
CASE 2: SATURATED (HIGH CONC) - equation you use
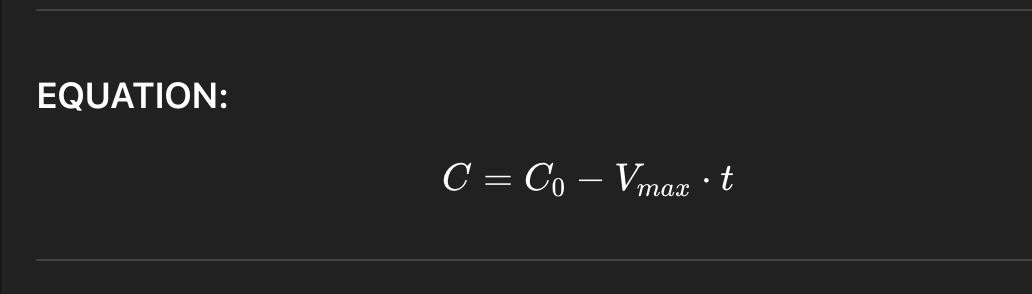
Michaelis–Menten model
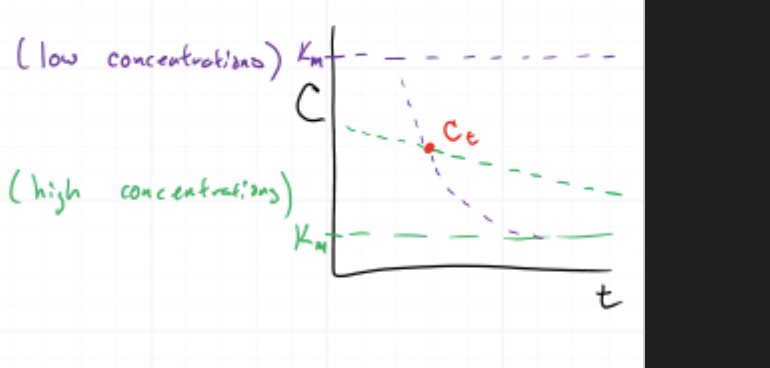
GRAPH THINKING
At any point:
Look at concentration (Ct)
👉 Ask:
Is it:
Above Km?
Below Km?