TM electrons
1/30
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
31 Terms
d-electron count definition
Number of electrons in the metal d orbitals after accounting for oxidation state
How to find d-electron count
Determine oxidation state, then subtract electrons from neutral metal configuration (remove 4s before 3d)
Example: Cr3+ d-electron count
Cr = [Ar] 3d5 4s1 → Cr3+ = d3
Does d-electron count equal number of unpaired electrons?
No, it only gives total d electrons, not their arrangement
What determines number of unpaired electrons?
d-electron count, geometry, and ligand field strength
What is ligand field strength?
Ability of ligands to cause electron pairing (strong-field vs weak-field)
High spin definition
Electrons occupy orbitals singly before pairing → more unpaired electrons
Low spin definition
Electrons pair in lower orbitals first → fewer unpaired electrons
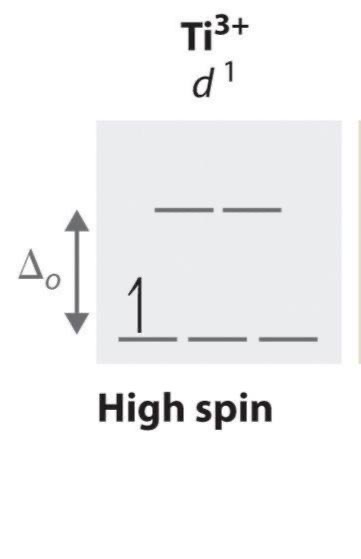
d1 configuration unpaired electrons
1 (always)
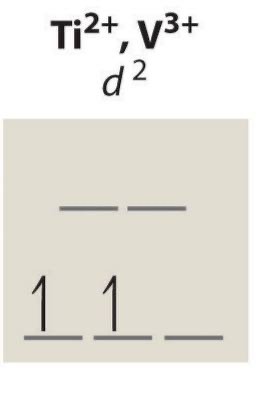
d2 configuration unpaired electrons
2 (always) HS
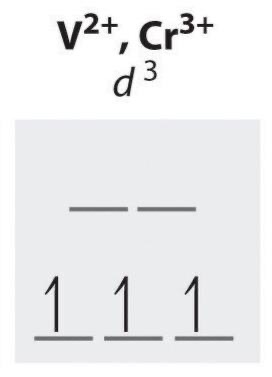
d3 configuration unpaired electrons
3 (always, octahedral) HS
d4 high spin unpaired electrons
4

d4 low spin unpaired electrons
2

d5 high spin unpaired electrons
5

d5 low spin unpaired electrons
1

d6 high spin unpaired electrons
4

d6 low spin unpaired electrons
0

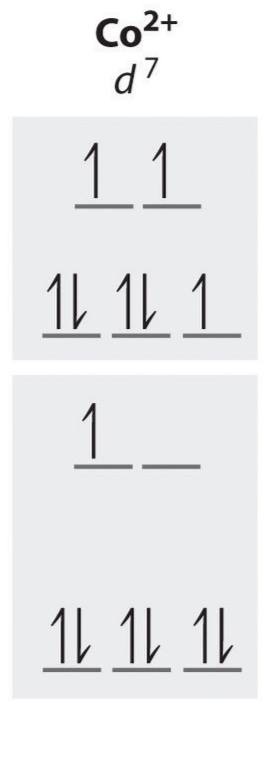
d7 high spin unpaired electrons
3
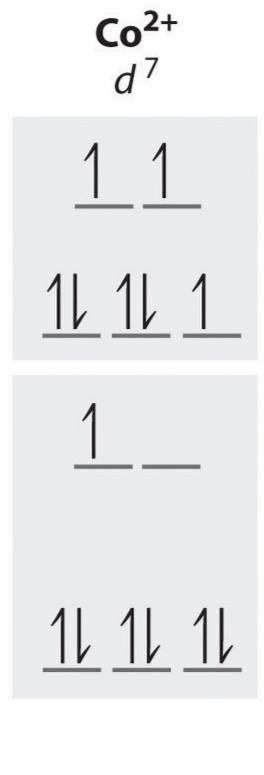
d7 low spin unpaired electrons
1
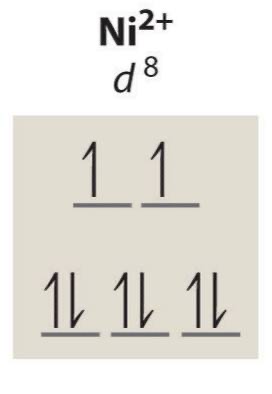
d8 typical unpaired electrons
Usually 0 (square planar) or 2 (octahedral)
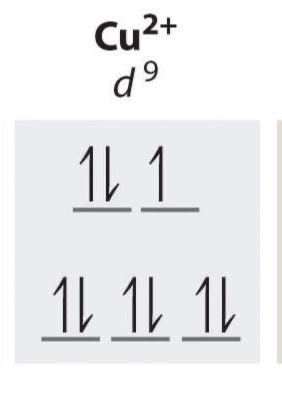
d9 unpaired electrons
1
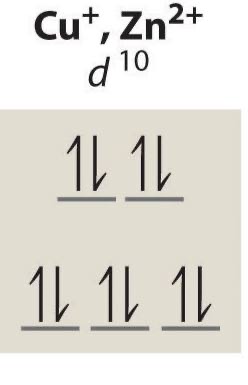
d10 unpaired electrons
0
Why d1–d3 are simple
No pairing needed → number of unpaired electrons equals d count
Why d4–d7 are complex
Electrons can pair or stay unpaired depending on ligand strength
Relationship between unpaired electrons and magnetism
Unpaired electrons → paramagnetic; none → diamagnetic
Spin-only magnetic moment formula
μ = √(n(n+2)) where n = number of unpaired electrons
Common mistake in d-electron counting
Forgetting to remove 4s electrons before 3d
Example: Fe2+ d-electron count
Fe = [Ar] 3d6 4s2 → Fe2+ = d6
Example: Co3+ d-electron count
Co = [Ar] 3d7 4s2 → Co3+ = d6
One-line rule for d-electron count
d count tells total electrons; unpaired depends on their arrangement