IB HL Physics (2023) - The particulate nature of matter
1/34
Earn XP
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
35 Terms
Density
Mass per unit of volume (Kgm-3)
Temperature ☆
A measure of the average random kinetic energy of particles in a substance (°C Or K)
Heat
The thermal energy transferred from a body of higher temperature to a body of lower temperature
Absolute Zero ☆
The lowest temperature theoretically possible at which particles have zero kinetic energy (0K or -273°C)
Thermal Equilibrium
When two bodies in thermal contact have the same temperature and no net flow of heat energy
Internal Energy
The sum of the kinetic and potential energies in ln the molecules of a body (J)
Specific Heat Capacity
The amount of thermal energy required to raise 1kg of a substance bu 1K (J Kg-1K-1)
Thermal Capacity
The amount of energy required to raise the temp of an object by 1K or 1°C (J K-1)
Specific Latent Heat of Fusion
The energy required to change 1kg of a solid into a liquid (JKg-1)
Specific Latent Heat Vaporisation
The energy required to change 1kg of a liquid into a gas (JKg-1)
Methods of Heat Transfer
Conduction - Through solids
Convection - Through fluids
Thermal Radiation - Transfer through the emission of photons
Black Bodies ☆
An object which can absorb and emit all wavelengths of EM radiation (reflects NO radiation)
Intensity
The amount of energy recieved per unit second, per cross sectional area (Wm-2)
Black bodies and Intensity
For black bodies (like stars) we can plot the intensity of each wavelength emitted on a graph and it always takes this shape
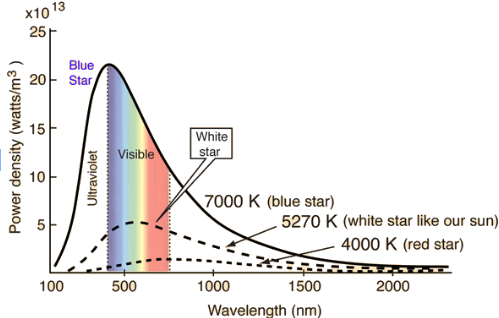
Wien’s Displacement Law ☆
The relationship between the peak wavelength and the absolute temperature of the surface of a black body
T at λmax = 2.898 x 10-3 mK (metres Kelvin NOT miliKelvin)
λmax is the wavelength at which maximum energy is radiated
Luminosity (L)
The Energy radiated by a star per second (W)
Stefan’s Law ☆
The total energy radiated per unit time by a black body is proportional to the fourth power of its absolute temperature and surface area
Apparent Brightness (b)
The amount of energy per second per unit area that arrives at a distance from the star (Wm-2)
Emissivity (e)
The ratio of power emitted from a body to the power emitted by a black body of the same size
e will always be between 0 and 1
a black body has an emissivity of 1
Albedo
The proportion of incident light that is reflected off a surface
Solar Constant ☆
The intensity of solar radiation across all wavelengths that is incident at the mean distance of earth from the sun
Greenhouse Gases
Gases which make up our atmosphere that trap heat and prevent it from escaping into space
Carbon Dioxide (CO2)
Methane (CH4)
Water Vapour (H2O)
Nitrous Oxide (N2O)
The Greenhouse Effect: Excitation ☆
Gas Molecules will absorb certain wavelengths of photons and temporarily gain more energy. This is known as excitation
Eventually they re-emit these photons in all directions
The Greenhouse Effect: Resonance ☆
The natural frequency of greenhouse gases is in the infrared region which means they are prone to absorbing the energy from infrared radiation
Then they later re emit the photons in all directions and sometimes back towards Earth
Energy Balance
the difference between the energy into the earth and the energy leaving the eart
Enhanced Greenhouse effect ☆
the additional radiative forcing resulting from increased concentrations of greenhouse gases induced by human activities
Pressure
The amount of force per unit area that acts perpendicular to the surface of an object (Pa)
Pressure in Solids and liquids
Solids - weight/ area of face
liquids density*gravity*height
State of a Gas
The specific physical condition of a gas sample at a given time
Ideal Gas Assumptions ☆
D - duration of collisions are negligible
R - random motion by particles
I - Intermolecular forces not present between particles
V - volume of particles are negligible
E - elastic collisions between particles
Real Gases and Ideal Gases
Real gases approach ideal gas behaviour at:
Low pressure and High Temperature
Ideal Gas Laws
Boyle’s Law - Pressure-Volume
Charles’s Law - Temperature-Volume
Gay-Lussac’s Law - Pressure-Temperature
Gay-Lussac’s Law ☆
For a fixed mass of gas with constant volume the pressure is directly proportional to the Absolute temperature of the gas
P/T = constant
higher temp=higher avg speed
particles collide with the walls more frequently
rate of change of momentum increases
greater average force
Boyle’s Law ☆
For a fixed mass of gas at a constant temperature the pressure is inversely proportional to the volume
PV = Constant
with a smaller volume the frequency of collisions increases leading to more force due to an increased rate of change of momentum
Charles’ Law ☆
For a fixed mass of gas kept at a constant pressure the volume is proportional to the temperature
V/T= Constant
When the temperature increases avg speed of molecules increase and so there are more frequent collisions with the side of the container, leading to the volume increasing