Glucose Homeostasis
1/37
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
38 Terms
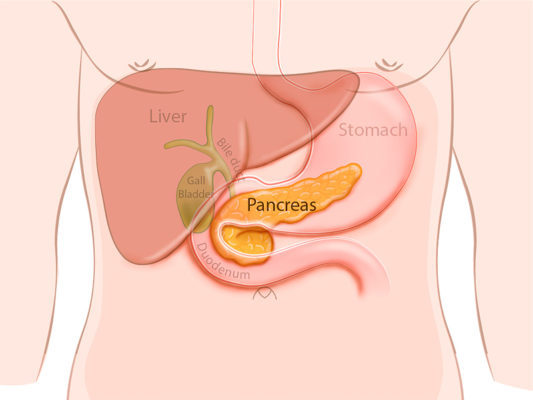
What is the exocrine and endocrine function of the pancreas?
Exocrine: Secreting digestive enzymes into ducts
Endocrine: Releasing hormones into the blood
Name the cells in the islets of Langerhans
α-cells
β-cells
δ-cells
What hormone does α-cells in islet of Langerhans secrete?
Glucagon
What hormone does β-cells in islet of Langerhans secrete?
Insulin
What hormone does δ-cells in islet of Langerhans secrete?
Somatostatin
What’s the difference between endocrine signalling and paracrine signalling?
Endocrine signalling acts over long distances, using the bloodstream to transport hormones to distant target cells.
Paracrine signalling acts locally (signaling molecules diffuse through the extracellular matrix to affect nearby neighboring cells)
What are the effects of insulin on plasma metabolite (small molecules circulating in the blood plasma, resulting from metabolic processes like breaking down food, drugs, etc)?
Insulin decreases plasma glucose, amino acids, and free fatty acids
What are the effects of insulin on metabolism?
Insulin promotes anabolic processes such as uptake and storage in liver, muscle, and adipose tissue
What are the effects of glucagon on plasma metabolites and metabolism?
Glucagon increases plasma glucose and ketones
Glucagon promotes catabolic processes such as the breakdown of energy stores (mainly in the liver)
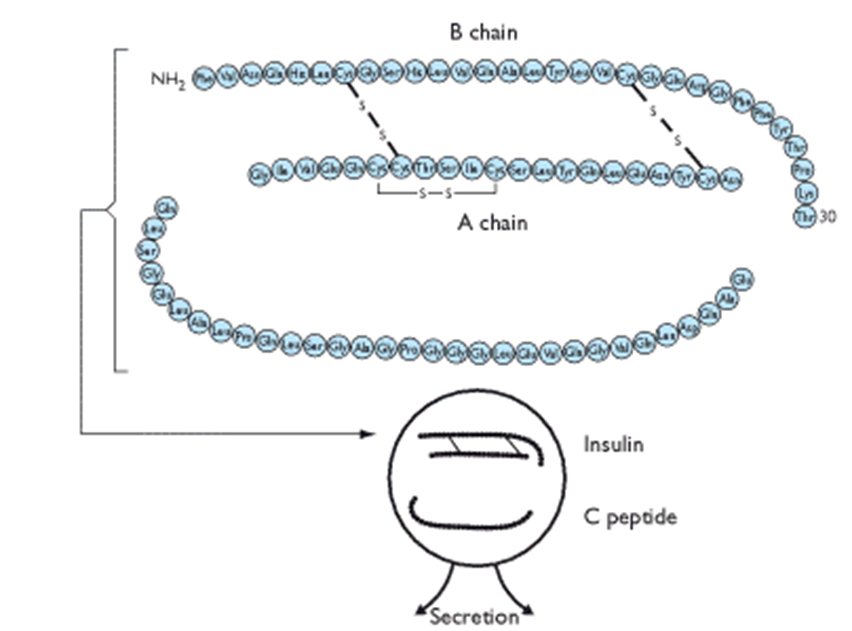
What is the structure of insulin?
The B and A chain are biologically active
The C-peptide is inactive
What degrades insulin?
Insulinase
Is insulin a highly conserved structure across many species or not?
It is highly conserved
Insulin is secreted in response to what?
Changes in glucose/ATP
How is insulin secreted from β-cells?
Via calcium-dependent exocytosis
Glucose enters β-cells
Glucose is metabolised to produce ATP
Increased ATP closes ATP sensitive K+ channels, causing depolarisation
This opens voltage-dependent Ca2+ channels, allowing Ca2+ influx, triggering insulin release by calcium dependent exocytosis
Sulfonylureas
A class of oral medications used to treat type 2 diabetes by stimulating the pancreas to release more insulin, lowering blood sugar levels
How does sulfonylureas stimulate insulin secretion?
Sulfonylureas closes ATP-sensitive K+ channels, leading to depolarisation. This opens voltage-dependent Ca2+ channels, allowing Ca2+ influx, triggering insulin release by calcium dependent exocytosis
When are we in absorptive state?
After eating
Nutrients absorbed
Insulin level increases
When are we in post-absorptive state?
Fasting (between meals)
Insulin levels decrease
What happens to plasma insulin levels during the day?
Plasma insulin concentrations rise during the absorptive state and fall during the post-absorptive state
When and how is insulin secreted?
Insulin is secreted by β-islet cells via exocytosis in response to high glucose levels. Insulin acts by binding to specific receptors on the plasma membrane of target cells.
What does RTK stand for?
Receptor tyrosine kinase
What is RTK?
A type of cell surface receptor that has built in enzyme activity. It binds a signalling molecule (like insulin) outside the cell and then triggers changes inside the cell.
What’s the mechanism of action with a ligand?
When a ligand binds, the receptor forms a dimer and activates its tyrosine kinase activity. It then autophosphorylates (adds phosphate groups to its own tyrosine residues), which starts intracellular signalling pathways.
Why are RTKs important?
RTKs regulate key processes like cell growth, metabolism, and survival
The insulin receptor is an RTK and is essential for controlling blood glucose levels
What are the effects of insulin on carbohydrate metabolism?
Insulin facilitates glucose entry into muscle and adipose tissue via GLUT transporters
Insulin stimulates the liver to store glucose as glycogen
Insulin decreases the concentration of glucose in the blood
What are the effects of insulin on carbohydrate metabolism?
Insulin promotes synthesis of fatty acids in the liver when glycogen stores are saturated, increasing lipoproteins in circulation
Insulin inhibits breakdown of fat in adipose tissue
Insulin promotes glycerol synthesis from glucose and increases triglyceride synthesis
How does insulin stimulate glucose uptake in cells?
When blood glucose levels rise, insulin is released into the bloodstream
Insulin binds to insulin receptor (RTKs) on the cell membrane
This activates intracellular signalling pathways.
Glucose transporter-containing vesicles move to and fuse with the cell membrane
This increases the number of glucose transporters on the cell surface
As a result, more glucose enters the cell from the bloodstream
Insulin-sensitising drugs enhance this process and increase glucose uptake
What are the effects of insulin on skeletal muscle?
Insulin stimulates glucose uptake into muscle cells via GLUT4 transporters
Inside the cell, glucose is converted to glucose-6-phosphate
It can also be metabolised to produce energy or converted to lactic acid
Insulin promotes amino acid uptake into muscle cells
This supports protein synthesis and formation of structural proteins
Overall, insulin increases glucose utilisation and storage in skeletal muscle
What are the effects of insulin on the liver?
Insulin increases uptake and phosphorylation in liver cells (via glucokinase), forming glucose-6-phosphate
It stimulates glycogen synthesis from glucose-6-phosphate (via glycogen synthase)
It promotes glycolysis, converting glucose-6-phopshate to pyruvate
Pyruvate is further converted to acetyl-CoA, which is used for fatty acid synthesis
Fatty acids are packaged into lipoproteins and exported to adipose tissue
Insulin promotes uptake and use of substrates such as lactic acid and amino acids
It inhibits gluconeogenesis
Overall, insulin promotes storage of glucose and reduces glucose production by the liver
How does insulin promote triglyceride storage in adipose tissue?
Insulin increases glucose uptake, which is converted to glucose-6-phosphate, then pyruvate, then acetyl CoA, then fatty acids
Insulin increases production of glycerol-3-phosphate from glucose, providing the backbone for triglyceride formation
Insulin stimulates lipoprotein lipase, releasing fatty acids from circulating lipoproteins for uptake
Insulin increases amino acid uptake to support metabolic activity
Overall, insulin increases triglyceride synthesis and fat storage
What is glucagon?
Glucagon is a peptide hormone made of 29 amino acids.
It is synthesised and released by α-cells of the pancreatic islets.
When is glucagon released?
When blood glucose levels fall. It acts as a counter-regulatory hormone to insulin.
What are the main actions of glucagon?
Glucagon stimulates glycogenolysis in the liver, increasing glucose release
It promotes gluconeogenesis, producing new glucose
It increases the breakdown of fats (lipolysis)
Overall, it raises blood sugar
How would you describe insulin and glucagon?
Counter-regulatory hormones
What is hypoglycaemia?
Hypoglycaemia is defined as blood glucose less than 3 mmol/L (normal is 4-6 mmol/L)
Glucose supply becomes insufficient to maintain normal tissue function
What is sensitive to low glucose levels in hypoglycaemia?
The CNS is particularly sensitive to low glucose levels
Neuroglycopenic symptoms include:
Impaired vision
Slurred speech
Staggered walk
Mood change
Confusion
Coma
Death
ANS symptoms include:
Palpitations
Sweating
Shakiness
Hunger
What is diabetes mellitus characterised by?
Characterised by hyperglycaemia (blood glucose over 7mmol/L)
What is a major risk factor for developing type 2 diabetes?
Obesity