Devlopmental Bio Exam 4
1/132
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
133 Terms
4 main regions of the neural plate
Cranial neural crest
Cardiac neural crest
Trunk neural crest
Vagal and sacral neural crest
Cranial neural crest
Neural crest cells in the future head region- differentiates into the cartilage, bone, cranial neurons, pigment cells, and connective tissues of teh face
Cariac neural crest
Subregion of the cranial neural crest- its cells develop into melanocytes, neurons, cartialge, and connective tissue
Trunk neural crest
Migrate ventrolaterally or dorsolaterally to become the dorsal root gangila containing the sensory neurons, sympathetic ganglia and adrenal medulla
Vagal
Generate the parasympathetic ganglia of the gut
Are neural crest cells multipotent or totipotent? How was this tested?
They are believed to be multipotent- Researchers used transgenic mice and traced 100 cell clones of migratory neural crest cells and showed that about 75% of them proliferated and their progeny shoed multiple tupes of lineages that differentiated into different cell types
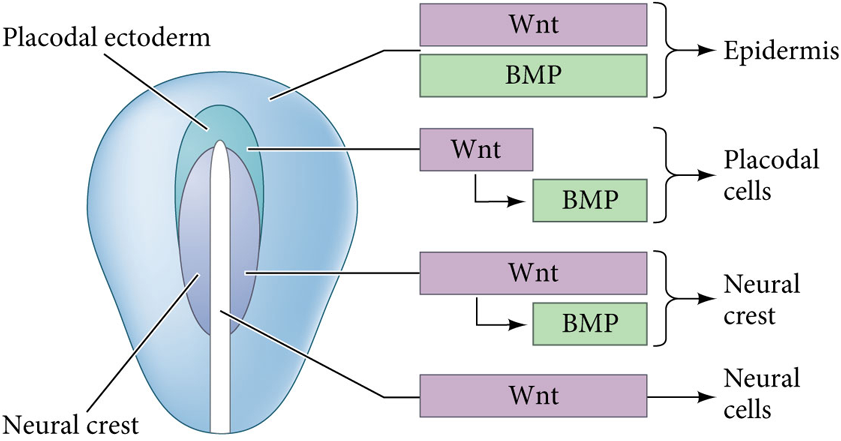
What happens if BMP and Wnt signaling are continuous in the the neural plate?
The fate of the ectoderm is epidermal
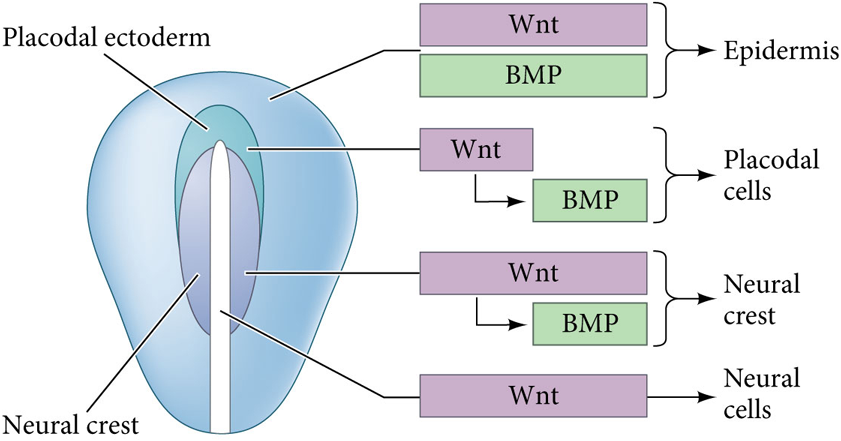
What happens if BMP signaling is blocked in the neural plate?
The ectoderm becomes neural
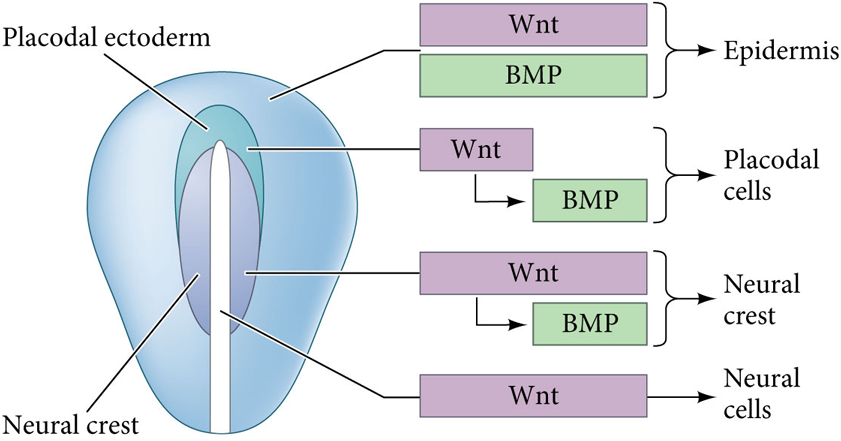
What happens if Wnts induce BMPs and Wnt signaling is turned off
The cells becomes committed to be anterior placodes
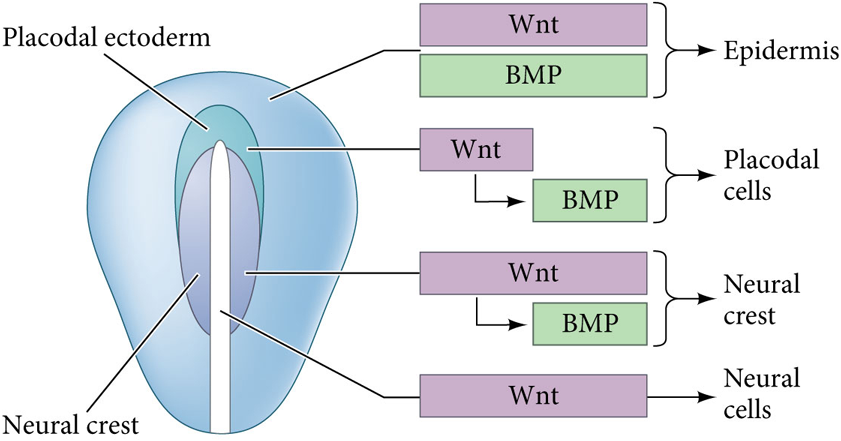
What happens if Wnt signaling induces GMPS but stays on?
The cells becomes capable of becoming neural crest
When is the first visible indication of neural crest cells?
After cell specification the first visible indication of neural crest cells is an epithelial-mesnchymal transition- preps for the neural tube
What triggers the epithelial-mesenchymal transition?
The activation of Wnt genes by BMP
How can different regions of the ectoderm area of the neural crest be identified?
By their expression of different cell-cell adhesion- Surface ectoderm express E-cadherin, premigratory neural crest express cadherin-6B, neural tube expresses N-cadherin
Sox2 function
Represses Snail2- expressed by cells of the neural ectoderm
Snail2 function
Represses N-cadherin
Sox2/Snail2 cross-transcriptional repression
In the nerual plate Sox2 repressess Snail2 to prevent the expression of neural crest specifiers (neural fate); In the dorsal-most region high BMP and Wnt induce the expression of Snail2 that overcomes Sox2 repression and represses it (Neural Crest fate)
What biomechanixal factoer might also help trigger epithelial to mesenchymal transition?
Mesodermal compaction by convergent extension provies a tissues stiffening force that is required for epithelial-mesenchymal transitions; this provides a greater force on the neural plate to promote EMT and a more stable substrate for a neural crest migration
Contact inhibition
Occurs when two migrating cells make contact which represses backward migration and results in forward migration of the leading edge of cells
Ventral pathway
Migration pathway of trunk neural crest cells that travel though the anterior of the sclerotome
Dorsolateral pathway
Pathway taken by trunk neural crest cells traveling dorsolaterally beneath the ecoderm to beome melanocytes
Semaphorin-3F
A protein that repels neural crest cells; blocks early migrating cells from traveling between the somites
Ephrins
A juxtacrine ligand that repells neural crest cell migration
Vagal and sacral neural crest function
These cells generate the parasymapthetic ganglia of the gut
What is the fate of late migrating neural crest cells?
These cells enter the dorsolateral pathway and remain above the neural tube to become melanoblasts
What controls the swith between cells that follow the ventral pathway and dorsolateral pathway?
Foxd3 transcription factor: Represses the gene for MITF which is necessary for melanoblast specification
How are cells guided in the dorsolateral pathway?
The melanoblasts upregulate the receptor for ephrin; ephrin expressed along the dorsolateral migration pathway stimulates the migration of melanocyte precursors
Placode
Thickening of the surface ectoderm that give rise to a variety of tissue-specific epithelial and neuronal cell types; formed from non-neural ectoderm
Genes that we need to know for sure
tbx5, t genes, focus on big picture genes, Fgf (x?), hox genes,
will be question on
the eye
What parts of the gene regulatory network should we know
only things covered in class (Pax6, fox3, ftx- six genes in the section) MATHB
Lens placode (anterioro placode)
Invaginates to form the lens vesicle which later forms the lens of the eye- do not generate sensory neurons
Olfactory placode (anterior placode)
Gives rise to the sensory neurons involved in smell
Otic placode (posterior placode)
Gives rise to the sensory epithelium of the inner ear and to neurons that form the cochlear-vestibular ganglion
Lateral line placodes (posterior placode)
Generate unique sensory cells call neuromasts that contain mechanosensory hair cells
Are tissues determined at the neural plate stage?
No
What signaling factors influence the induction of the pre-placodal region?
Activation of Wnt and BMP and repression of Fgf
What does Fgf influence in the otic system?
Fgf induces otic-epibranchial precursors to form from the posterior pre-placodal region
How does Wnt signaling influence the otic system?
Wnt signaling from the neural plate promotes otic identity while repressing epibranchial development
How does BMP signaling influence the otic system?
BMP signaling supports the specification of epibranchial neurons
Steps of otic-epibranchail placode induction
Cells of the posterior pre-placodal region are induced to become otic-epibranchial progenitors by Fgf signaling
Additionaly Fgfs are supplied
Wnt signaling promotes otic but represses epibranchial fate
Continued Fgf signaling activates the epibranchial program and BMPs initiate epibranchial neurogenesis
Otic morphogenesis steps
Basal expansion
Apical constiction forms otic cup
Edges of the otic cup come together to form the otic vesicle
How does sonic hedgehog signaling (shh) contribute to axis determination of the otocyst?
Shh signaling within the inner ear develops the ventrally derived cochlear cell types
How does sonic hedgehog retinoic acid contribute to axis determination of the otocyst?
Posterior regions are developed in high concentration of retinoic acid
What three things cause invagination the develops the eye
The cells of the lens placode extend adhesive filopodia that contact the optic vesicle
The cells at the edge of the invaginiating layer undergo basal constriction
The cells at the center of the invaginating layer undergo apical constriction
Why is six3 so important to retinal development?
Six3 is a major coordinator or retinal patterning preceding differentiation of the retina over neural tissue
What’s the latest stage where lens ectoderm are only specified?
Stage 18: neural fold has just formed and the early retina is just approaching the surface ectoderm where the lens will form
What is the earliest stage where lens ectoderm are determined?
Stage 21: neural tube has just fused and the retina regions are first coming in contact to the lens area
What genes are required for lens ecoderm determination?
MafB, Sox2, Sox3
What is the importance of Pax6?
Pax6 integrates positional info (from Otx2) and the competence (from Sox2) to officially specify the lens
What is the importance of MafB?
Turn on the actual lens proteins and stop the cell cycle what allow cells to be determined