EXAM 3 Electrophilic Aromatic Substitution
1/58
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
59 Terms
Reagents for EAS
There are multiple different reagents these are the ones you would start off by putting on the Benzene ring.
Reagent for Chlorine
Cl2/FeCl3 = Cl+

Reagent for Bromine
Br2/FeBr3 = Br+

Reagent for Iodine
I2/H2O2 or Cu2+ = I+
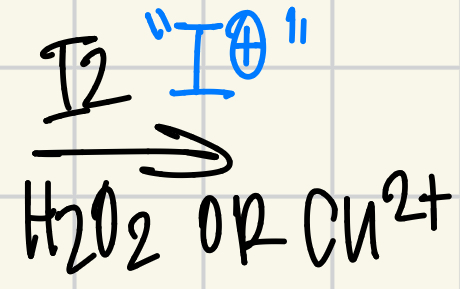
Reagent for Nitration
HNO3/H2SO4 = NO2+

Reagent for Sulfonation
SO3/H2SO4 = SO3H+

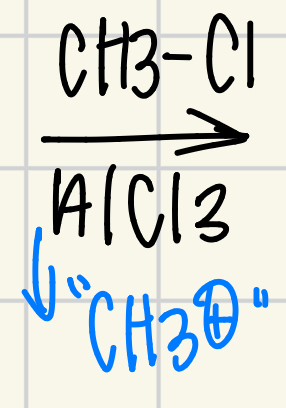
Friedel-crafts alkylation of benzene
CH3Cl, AlCl3 = CH3
Remember about major and minor products how can they be distinguished, Hint: Think zaitsev's rule
Products can be done with carbocation rearrangement
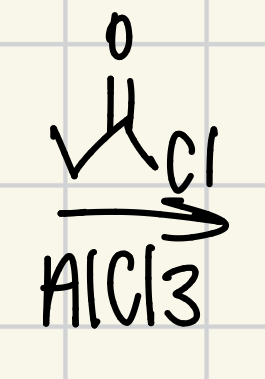
Friedel-Crafts Acylation
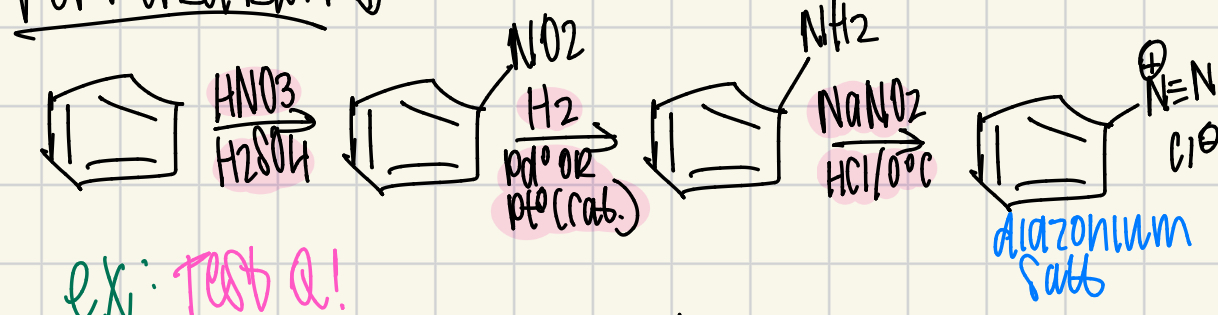
Reagent Pathway to make a Diazonium salt
Everything is on Benzene!!!
1. HNO3/H2SO4 = NO2
2. H2/Pd or Pt = NH2
3. NaNO2/HCl/0ºC = N Triple Bond N or Diazonium salt
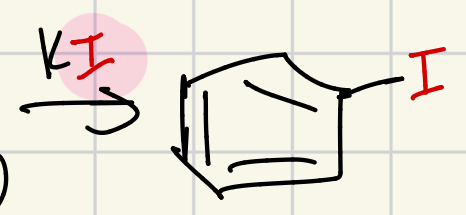
Iodine on the “wheel of doom”
KI = I
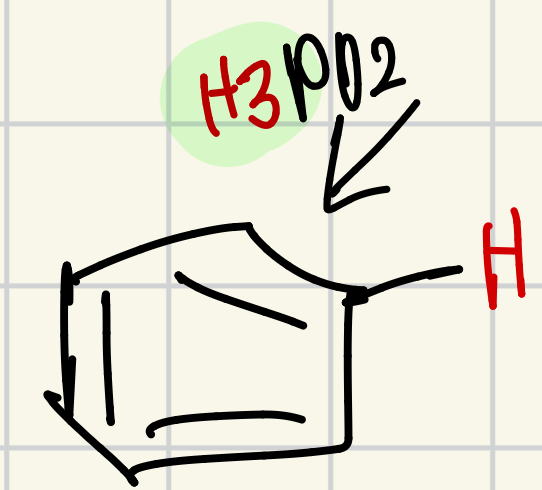
Hydrogen on the “wheel of doom”
H3PO2 = H
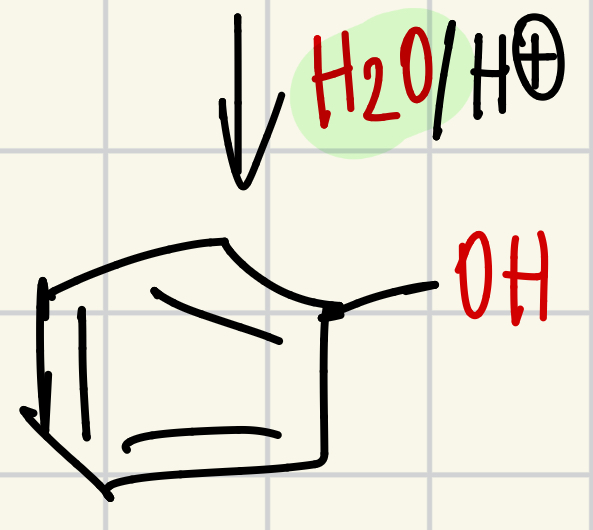
OH on the “wheel of doom”
H2O/H+ = OH
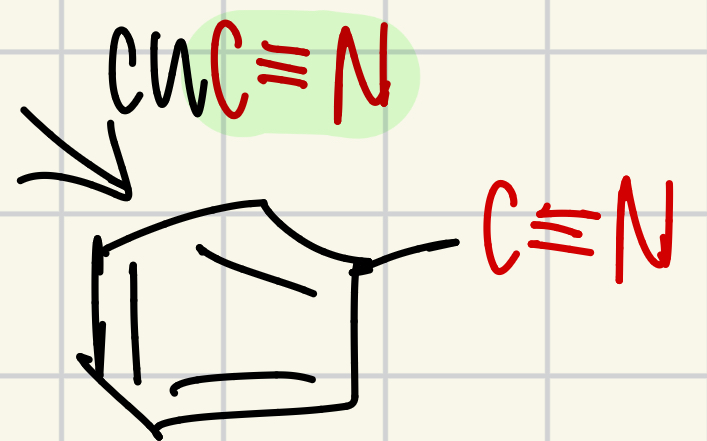
C Triple bond N on the “wheel of doom”
CuC Triple Bond Nitrogen
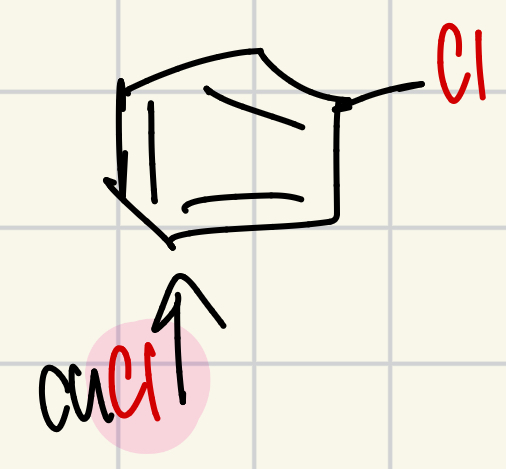
Chlorine on the “wheel of doom”
CuCl = Cl
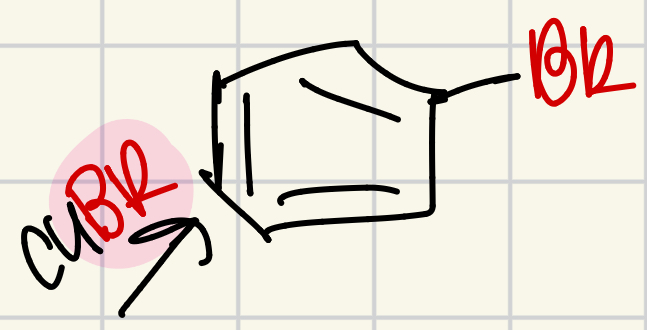
Bromine on the “wheel of doom”
CuBr = Br
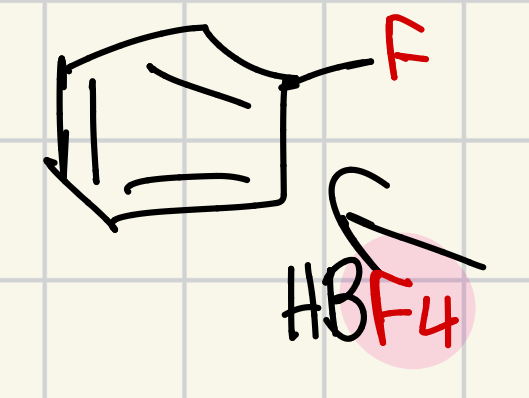
Fluorine on the “wheel of doom”
HBF4 = F
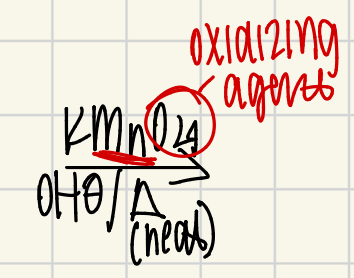
Side Chain Oxidation Reagent
K+MnO4-/OH-/Heat
only works in the presence of Hydrogens off of the alpha carbon!
creates carboxylic acids
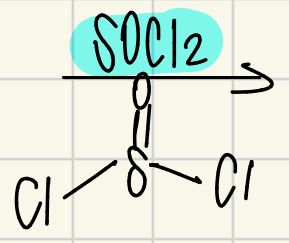
Reagent for converting a carboxylic acid to an acyl halide
SOCl2/ET3N (Tri-ethylamine)
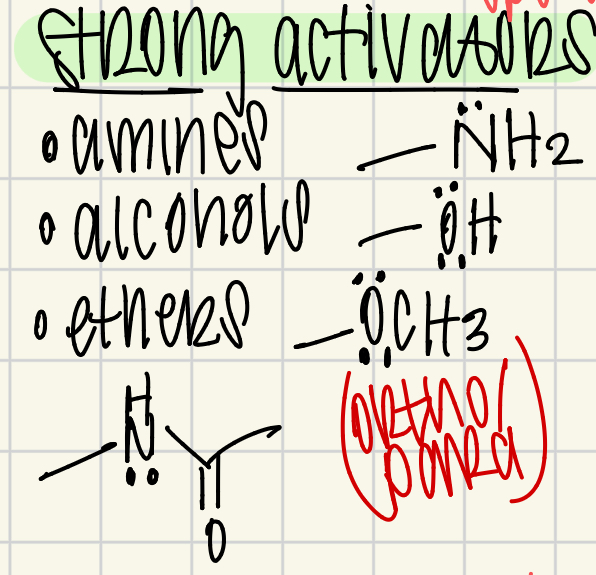
Strong Activators
NH2, OH, OCH3 (lone pair atoms)
ortho/para director
speed up EAS
highest
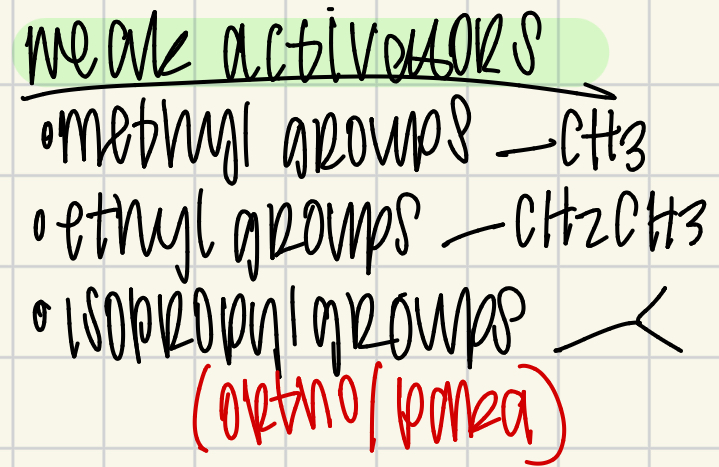
Weak activators
alkyl groups (ortho/para)
Weak deactivators
Halogens (F, Cl, Br, I)
ortho para directors

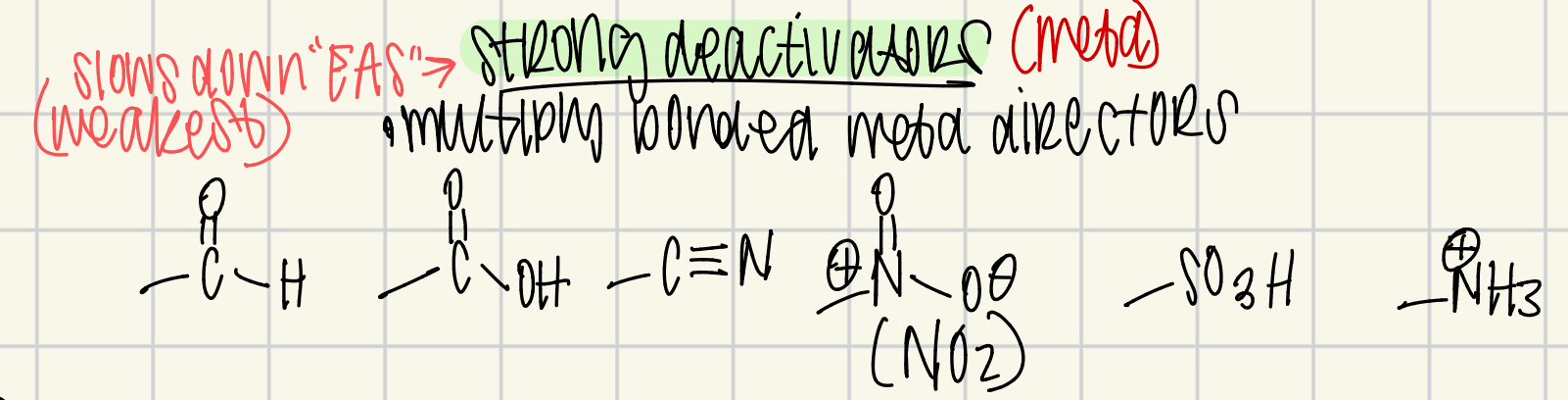
Strong deactivators
multiply bonded atoms
meta director
weakest
Nucleophilic Aromatic Substitution (NAS)
Replacement of a leaving group on an aromatic ring by a strong nucleophile. Usually takes place by an addition-elimination mechanism or by a benzyne mechanism.
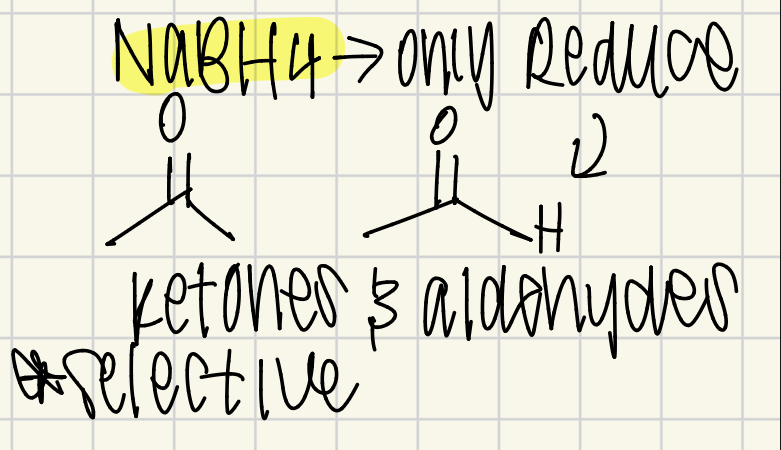
NABH4 H- reacts with?
Selective - Aldehydes and Ketones!!!!!!
LiAlH4 can react with?
Non-selective = Reduces everything!!!
What's the purpose of a Ketals/acetals
To protect Ketones and Aldehyde functional groups
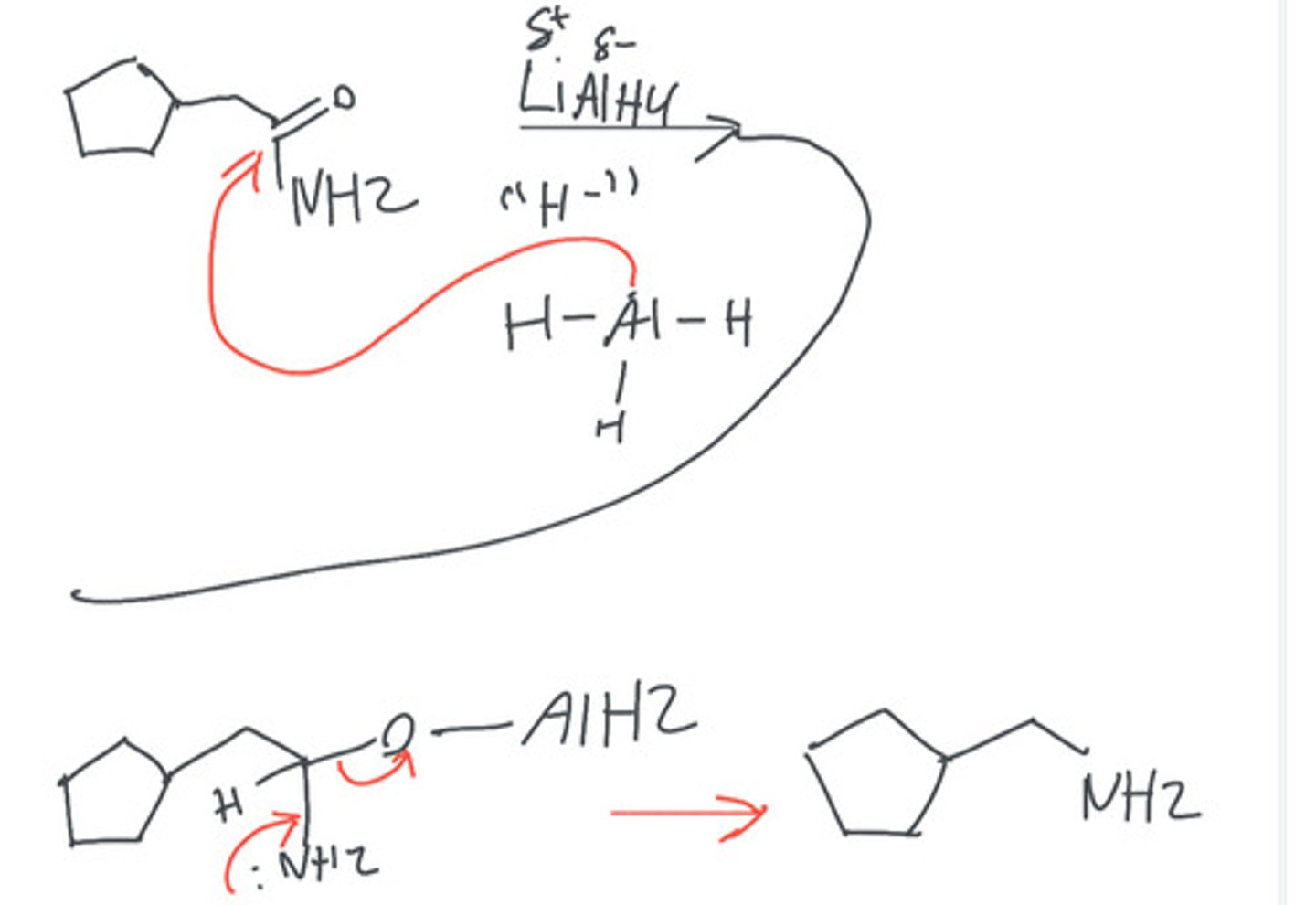
Product when LiAlH4 reacted with an Amide
1º Amine/It works by having the Oxygen bind with the Aluminum make it the leaving group
NaBH4 has to be done what again
Repeated if used on Aldehydes and Ketones
Benzyne Reagents
NH2-/NH3 (Liquid death) The ONLY REAGENT FOR BENZYNE
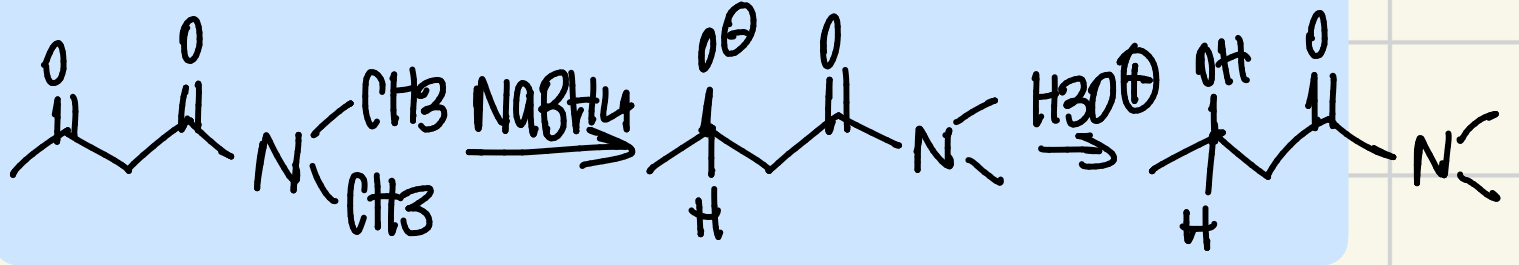
NaBH4 is NOT powerful enough to reduce carboxylic acids, esters, or amides
So you can selectively reduce a ketone or aldehyde in the presence of one of these groups.
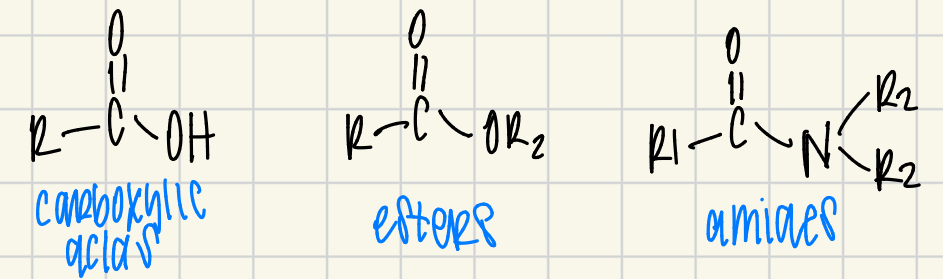
carboxylic acid
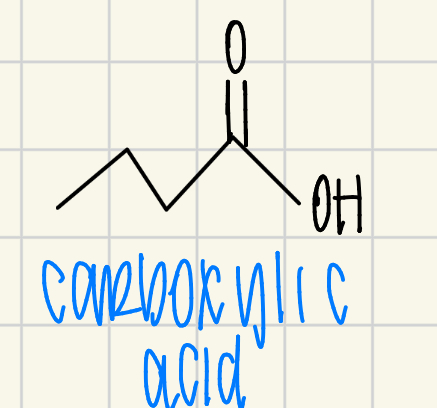
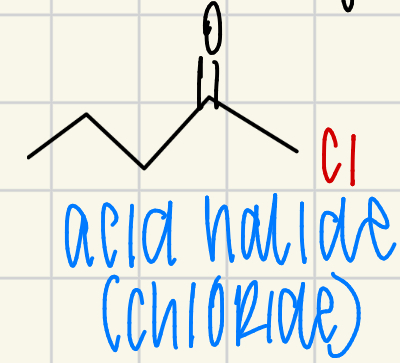
acid halide (chloride)
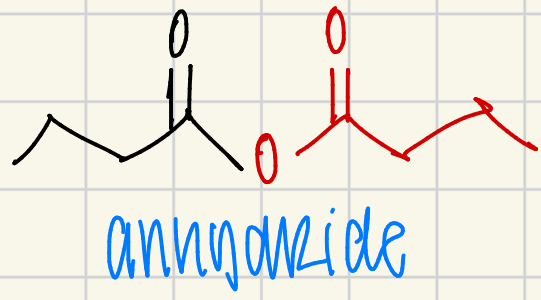
anhydride
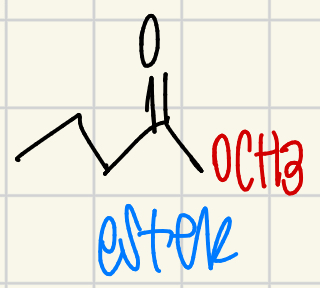
ester
amide
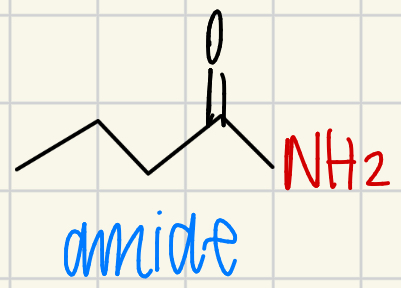
General mechanism of carboxylic acid derivatives with strong nucleophiles
Negative attacks C=O carbon
Texas carbon
O- swings down and kicks off the leaving group
Reform C=O bond
Replace the leaving group with the reagent
Leaving group floats in parentheses
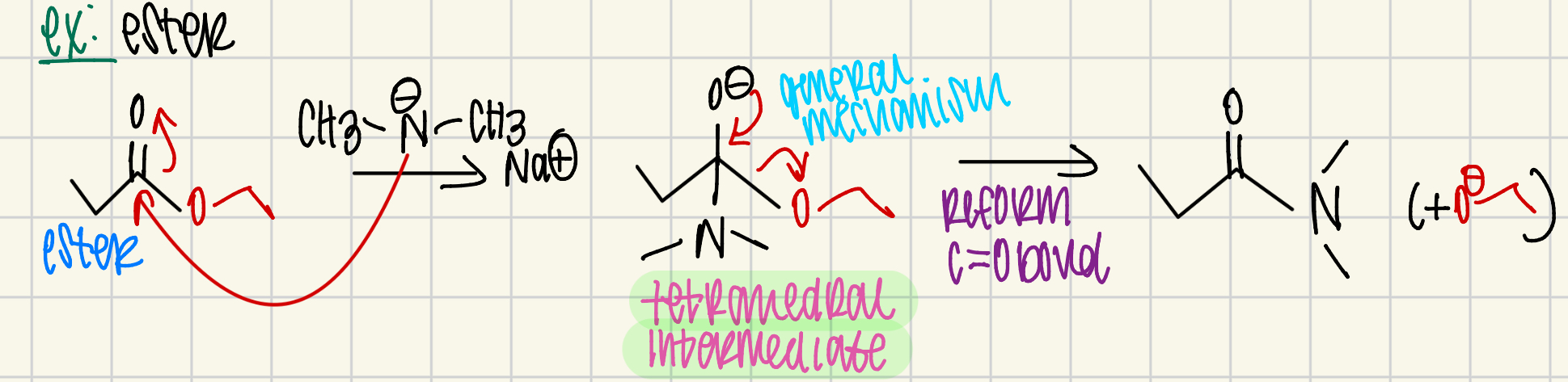
most reactive to least reactive…
acid halide > anhydride > ester > amide
T or F: reactions that turn more reactive derivatives into less reactive ones are ok!
TRUE

what does hydrolysis mean or saponification?
water breaking
ester hydrolysis
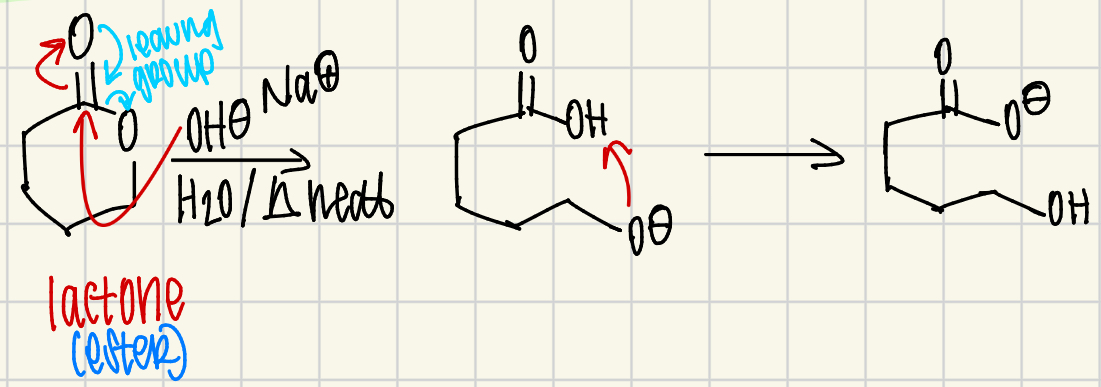
What is the Gabriel Synthesis?
Used to make primary (1°) amines
ortho/para directors
-NH2, -OH, -OCH3, -F, -Cl, -Br, -I
Groups with lone pair atoms directly attached to benzene cause EAS to occur
meta directors
-C=O, -SO3H, -C triple bond N, -NO2:
Groups with a multiply-bonded atom directly attached to benzene cause EAS to occur
what happens when groups have about (~) equal activating power?
let steric hinderance be your guide
what happens when you have conflict between 2 groups
determine which is stronger (SWWS)
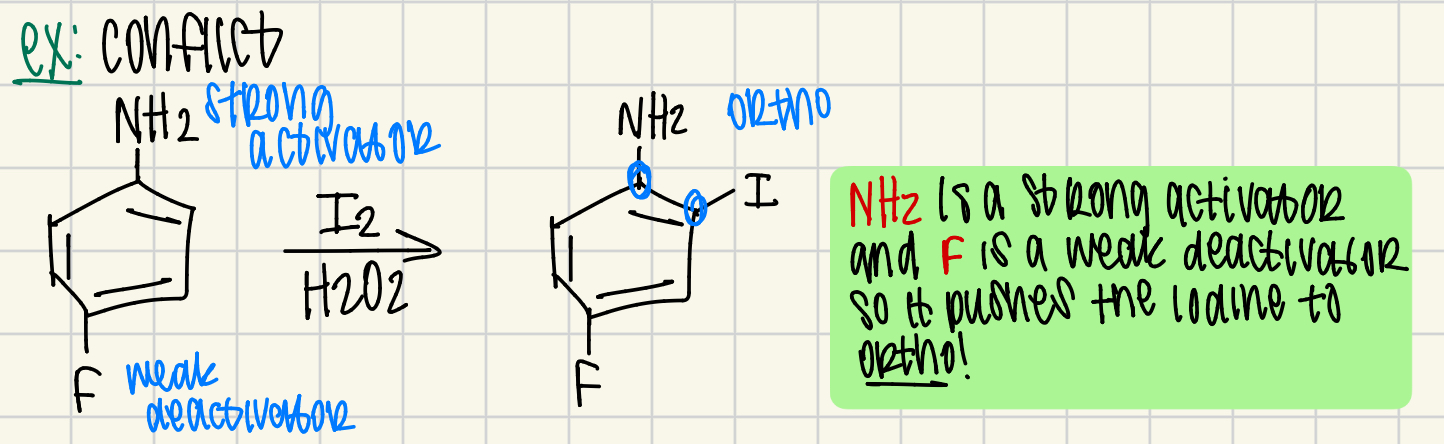
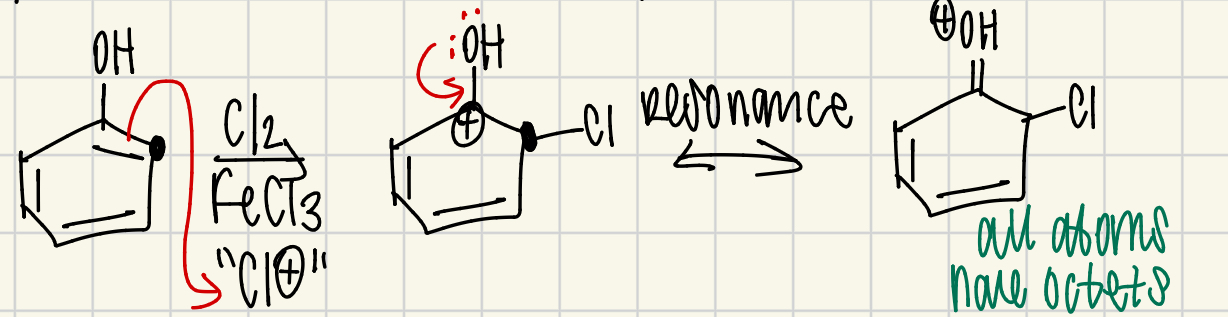
why do ortho/para directors work?
resonance
T or F: start an attacker on a negative thing and end on a positive
TRUE
what happens when you have 2 or more strong deactivators in an EAS rxn?
NO REACTION!
T or F: activators make the attacker more negative
TRUE
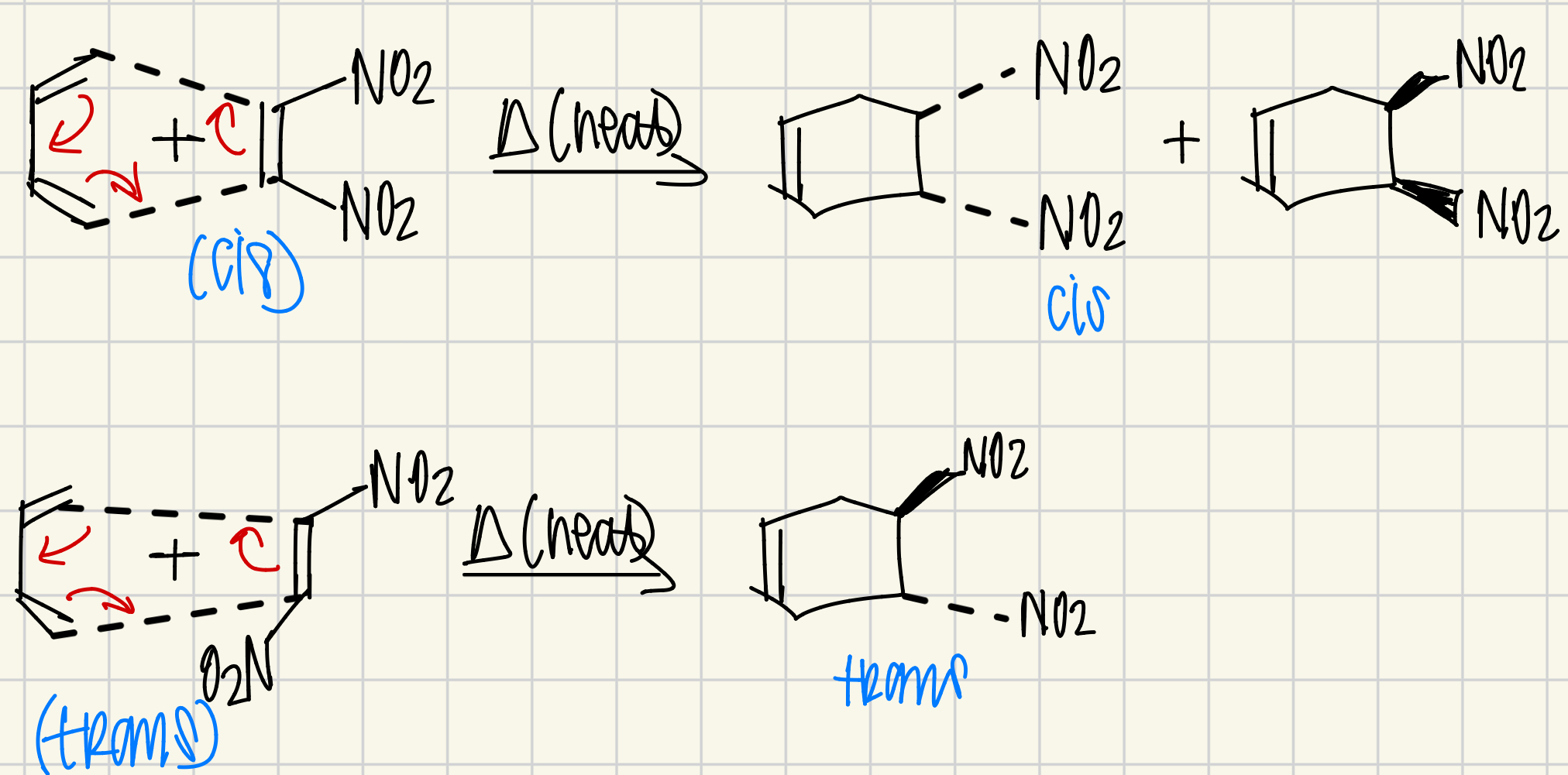
what mechanism looks like a carousel?
Diels-Alder Reaction
T or F: esters are more reactive than amides
TRUE
T or F: a successful haloform reaction produces a purple precipitate
FALSE
T or F: the 1st step of electrophilic aromatic substitution (“door opening”) is faster than the 2nd step
FALSE
what does “Orange Mice Sleep Gently After Parties” mean?
oxalic, malonic, succinic, glutaric, adipic, and pimelic
T or F: The product of a malonic ester synthesis is… malonic ester
FALSE
T or F: NaBH4 will reduce carboxylic acids but not ketones
FALSE
T or F: The side chain of ter-butylbenzene is easily oxidized by KMnO4/OH-
FALSE
T or F: The amino acids in proteins are connected via amide bonds
TRUE