QAC - Chapter 23 - What is chromatography
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
Chromatography
A versatile laboratory technique used to separate, identify, and quantify components in a mixture by passing it through a stationary phase using a mobile phase.
Elution
The process of passing liquid or gas through a chromatography column.
Chromatography
A versatile laboratory technique used to separate, identify, and quantify components in a mixture by passing it through a stationary phase using a mobile phase.
Mobile Phase
(A liquid or a gas flowing through the column)
The stationary phase (stays in place inside the column):
A viscous liquid chemically bonded to the inside of a silica capillary column
A viscous liquid coated onto the surface of solid particles packed in the column
The solid particles themselves
Elution
Packed Column
Filled with particles of the stationary phase.
Open Tubular Column
Narrow, hollow capillary with a stationary phase coated on the inside walls.
Absorption Chromatography
Solute is adsorbed on the solid particles.
Stationary Phase: Solid
Mobile Phase: Liquid or gas
The more strongly a solute is adsorbed, the slower it travels through the column (greater retention time, tR)
Partition Chromatography (Example: Gas Chromatography)
Stationary Phase: Liquid bonded to a solid support
Mobile phase: liquid or gas
Solute partitions between the stationary liquid and the mobile phase, which is a flowing gas in gas chromatography.
Ion-exchange chromatography
Stationary solid phase: : A resin containing covalently attached anions (-SO3-) or cations (–N(CH3)3+ ).
Mobile phase: Liquid
Ions of the opposite charge are attracted to the stationary phase by electrostatic force.
Molecular exclusion chromatography or gel filtration/permeation chromatography
Stationary phase: porous gel
Mobile phase: liquid or gas
This technique separates molecules by size, with larger solutes passing through most quickly.
Affinity Chromotography
Very selective/specific interactions
Partition Coefficient, KD
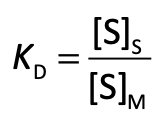
Chromatogram:
A graph of detector’s response as a function of elution time
Retention time, tR
For each component, it is the time between injection of the mixture onto the column and when that component reaches the detector.
Adjusted Retention time (t'R)
For a retained solute, it is the additional time required to travel the length of the column, beyond that required by the solvent
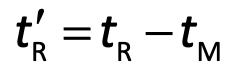
tm
The time that unretained mobile phase travels through the column (minimum possible time).
The greater α
The greater the separation between the two components.
α is independent of flow rate and can be used to help identify peaks when the flow rate changes.
Diffusion Coefficient, D
The rate at which a substance moves randomly from a region of high concentration to a region of lower concentration.

Plate height (H)
The constant of proportionality between the variance, σ2, of the band and the distance it has traveled, x.