General Chemistry 1 - Chapter 7: Periodic Properties of the Elements
1/15
Earn XP
Description and Tags
For exam study
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
Who developed the periodic table?
Dmitir Mendeleev and Lothar Meyer
What is periodicity?
The repetitive pattern of a property for elements based on atomic number.
What is effective nuclear charge?
The property of atoms that cause the outermost electrons, valence electrons, to be shielded by the other shells from the nucleus, causing the valence electrons to not feel the full force of the nucleus. This is a periodic property, meaning it increases across and period and slightly increases down a group.
What is the nonbonding atomic radius (aka van der Waals radius)?
Half the shortest distance seperating two nuclei duringa. collision of atoms.
What is the bonding atomic radius (aka covalent radius)?
Half the distance between nuclei in a bond.
What are the periodic trends of the bonding atomic radius?
It decreases from left to right across a period.
It increases from top to bottom of a group.
What properties does the size of an ion depend on?
Nuclear charge, number of electrons, and the orbitals in which the electrons reside.
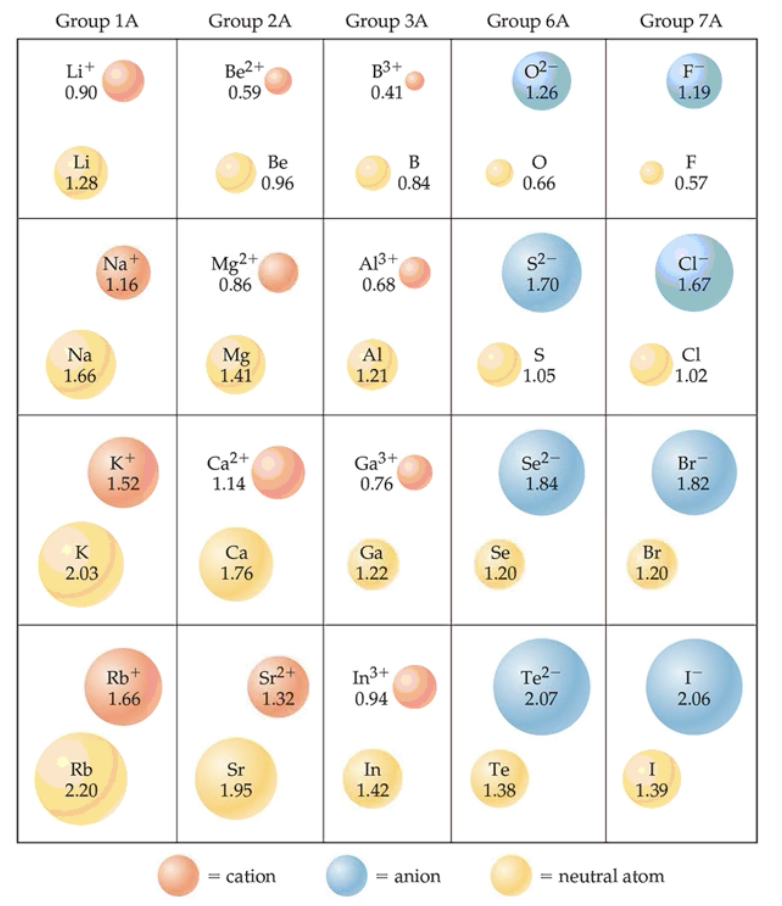
Cations are _______ than parent ions, and anions are ________ than parent ions.
Smaller, larger.
What is an isoelectronic series?
A series of atoms and ions that have exactly the same number of electrons.

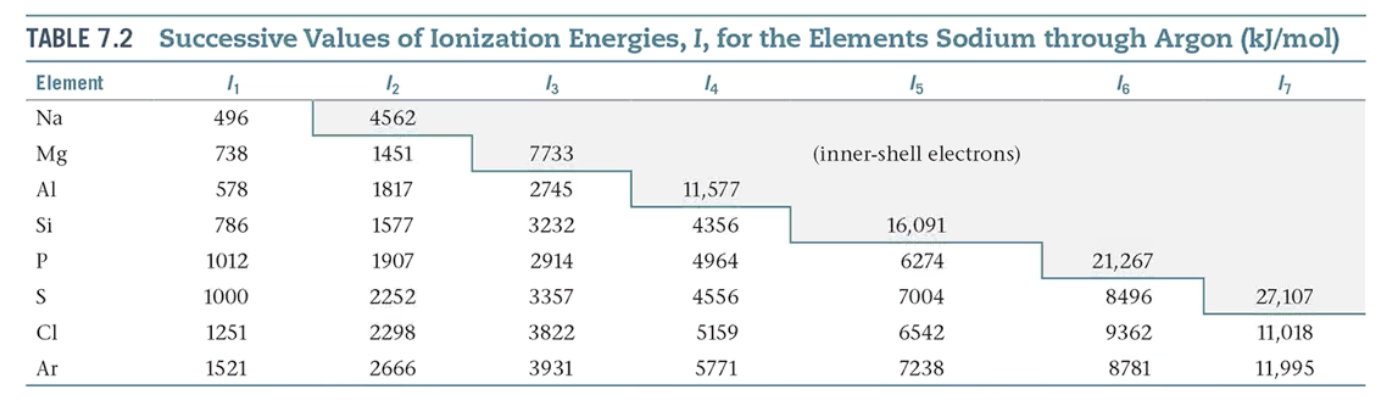
What is ionization energy? What is first and second ionization energy?
The minimum energy required to move an electron from the ground state of a gaseous atom or ion.
The first ionization energy is that energy required to remove the first electron.
The second ionization energy is that energy required to remove the second electron.
What is the standard formula for ionization energy of a standard element?
M(g) ——> M+(g) + 1e-
Where M = a standard element in gaseous form
What are the periodic trends in First Ionization Energy (I1)?
1.) Generally increases across a period.
2.) Generally decreases down a group.
3.) The s- and p- block elements show a larger range of values for I1.
4.) The d-block generally increases slowly across the period.
5.) The f-block generally show small variations.
What are some irregularities in the First Ionization Energy trend?
The trend is not followed when the added valence electron in the next element:
enters a new sublevel (higher energy sublevel)
is the first electron to pair in one orbital of the sublevel
What is electron affinity?
The energy change accompanying the addition of an electron to a gaseous atom (the opposite of Ionization energy).
What is the general equation for electron affinity?
X(g) + e- → X-(g)
What are the general periodic trends in electron affinity?
1.) Not much change in groups.
2.) Across a period, it generally increases with three notable exceptions:
Group 2A (s sublevel is full)
Group 5A (p sublevel is half-full)
Group 8A (p sublevel is full)