Clock Rate Reaction
1/40
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
41 Terms
Purpose
To determine the reaction rate law and effect of concentration for the redix reaction between peroxydisulfate and iodide ions
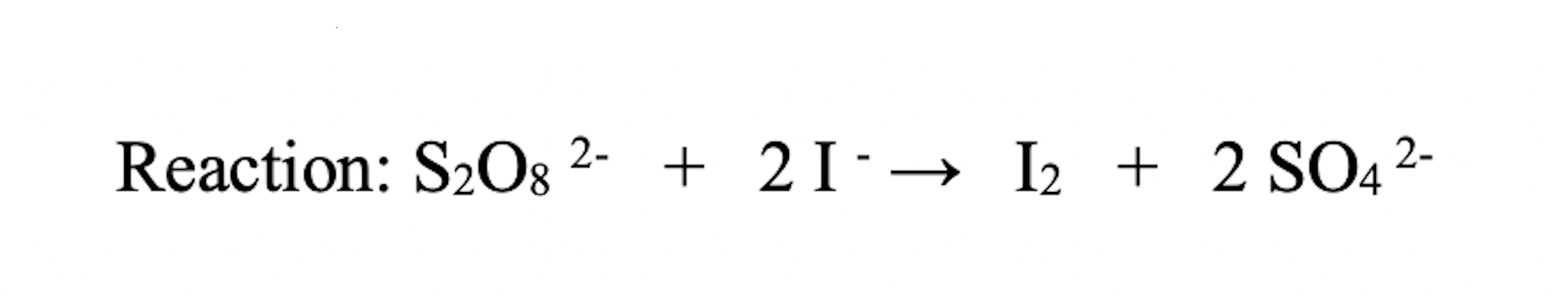
Procedure
Prepared 4 solutions as reactant 1 in a 250 mL erlenmeyer flask
In a separate beaker, prepared 4 solutions as reactant 2
Prepared 6 test tubes with 1.00 ml of thiosulfate
Placed a magnetic stir bar at the center in the reaction beaker
Poured reactant 2 into reactant 1 (100mL total) in reaction beaker then recorded time
Once blue-black color appeared, poured 1 test tube of thiosulfate and recorded time color change occured
Repeat until all 6 test tubes are used up
Repeat for next 3 solutions with 6 new test tubes of thiosulfate each
What were the 5 components in reactant 1 solution?
0.2M KNO3, 0.2M KI, 0.4M S2O32- , 1% starch, 0.1M EDTA
What was the role of Potassium Nitrate? (KNO3)
To have a constant number of ions
What is the role of Potassium Iodide? (KI)
KI contains I- which is reactant 1
What was the role of EDTA?
To remove metal impurities

What is the role of thiosulfate?
To reverse the reaction by destroying iodide
What was the limiting reagent?
Thiosulfate
What was the one component reactant 2?
Peroxydisulfate
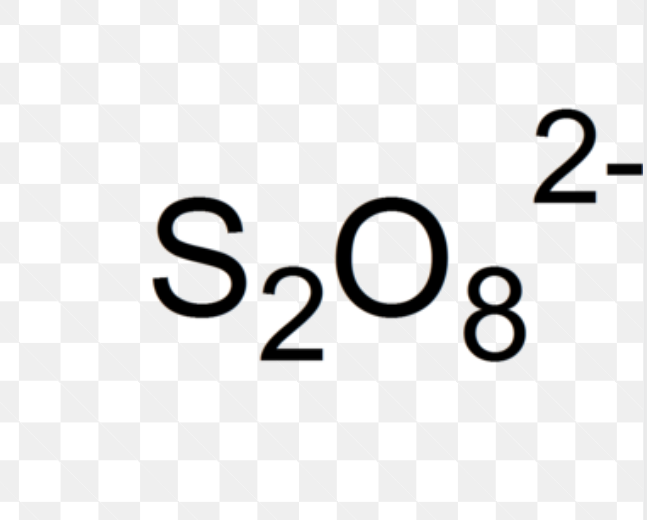
How many batches of thiosulfate for each experiment?
6
What was the color of the solutions containing K+ ions?
Colorless
What was the color of the solutions containing I- ions?
Brown
What was the color of the Iodide-starch complex?
Blue-Black
Reaction Rate
Change of concentration over time - negative for reactants, positive for products

What do rates of reactions depend on?
reactants, concentration of reactants, temperature, and presence of catalysts
Rate Law
The relationship of the rate of reaction K to the concentrations of the reactants raised to some powers

Are the order of reactants the same as the stochioemtry ratios?
NO
Total reaction order
sum of the exponents for the reactants
Reaction Order 1 example
Rate = [A][B]
If [A] is doubled in Rate = [A][B], how is the rate changed?
The rate is doubled
If Rate = [A][B]2 what is the overall reaction order?
A= 1
B = 2
1+2 = 3
If [B] is doubled in Rate = [A][B]2 how does the rate change?
The rate is quadrupled
What is the rate for this experiment?
Rate = K [I-][S2O8]
What is the overall reaction order for this experiment?
Second
What is the reaction order for [I-] in this experiment?
First
What is the reaction order for peroxidisulfate in this experiment?
First
What is the clock reaction?
First thiosulfate is added and solution is clear, then more Iodide forms and the solution is blue black, and finally more thiosulfate is added and solution is clear
Describe solution 1 and 2 in the experiment
Solutions 1 and 2 have the same amount of KI
Describe solution 3 in the experiment
Solution 3 had twice as much KI as 1 and 2
Describe solution 4 in the experiment
Solution 4 had 1/2 as much KI as 1 and 2
What was the x and y axis of the reaction rates graph?
X = Time in seconds, Y = moles of peroxydisulfate reacted
How to calculate rate of reactions for solutions 1-4
Slope/0.100L = Rate (mol x L-1 x s-1 )

How was rate measured in the experiment?
Measured in the intervals between the color changes
Which solutions measure the effect of [I-]?
Solutions 1 and 3 measured the effect of doubling [I-]
Which solutions measured the effect of [S₂O₈²⁻]?
Solutions 1 and 2 measured the effect of doubling [S₂O₈²⁻]
what does the blue-black color represent?
The completion of the clock reaction
Which chemical species is responsible for the blue black color change?
I2
What’s the reaction we’re studying?
S2O8 + 2I- —> I2 + 2SO4
peroxydisulfate + 2Iodide —> Pure Iodide + 2Sulfate
S2O8 role
Oxidize Iodide from I- to I2
Chemical Formula of Peroxidisulfate
S2O82-
Chemical Formula of Thiosulfate
S2O3 2-