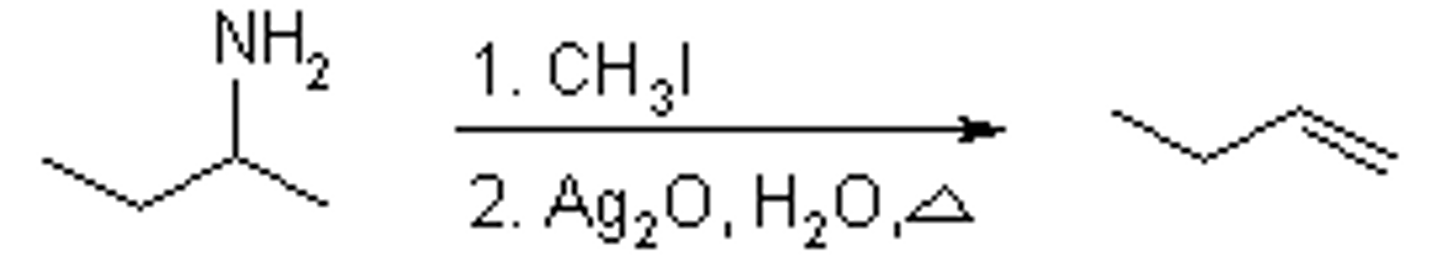reactions and mechanisms (amines) oat notes
1/14
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
15 Terms
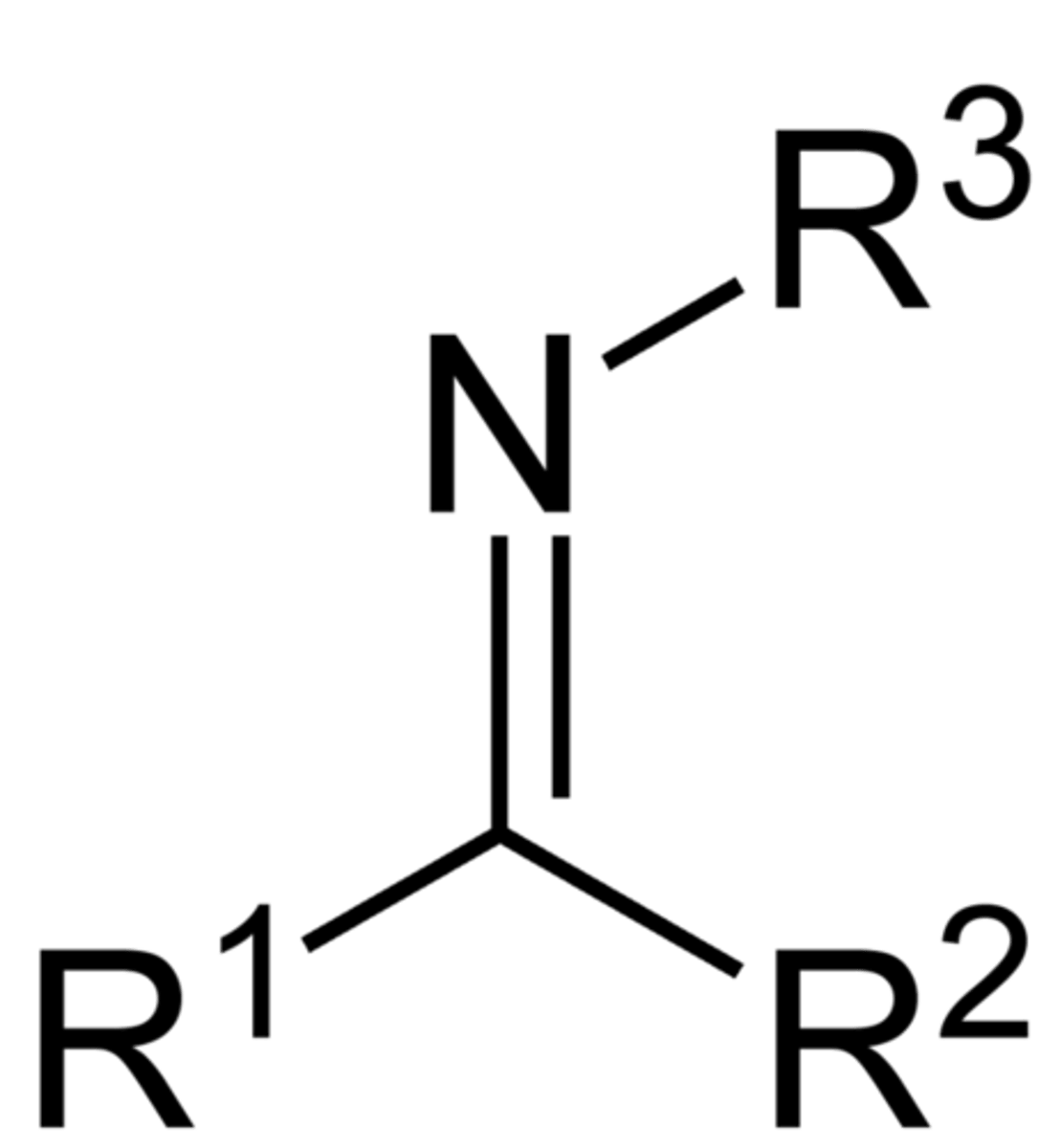
imines
nitriles
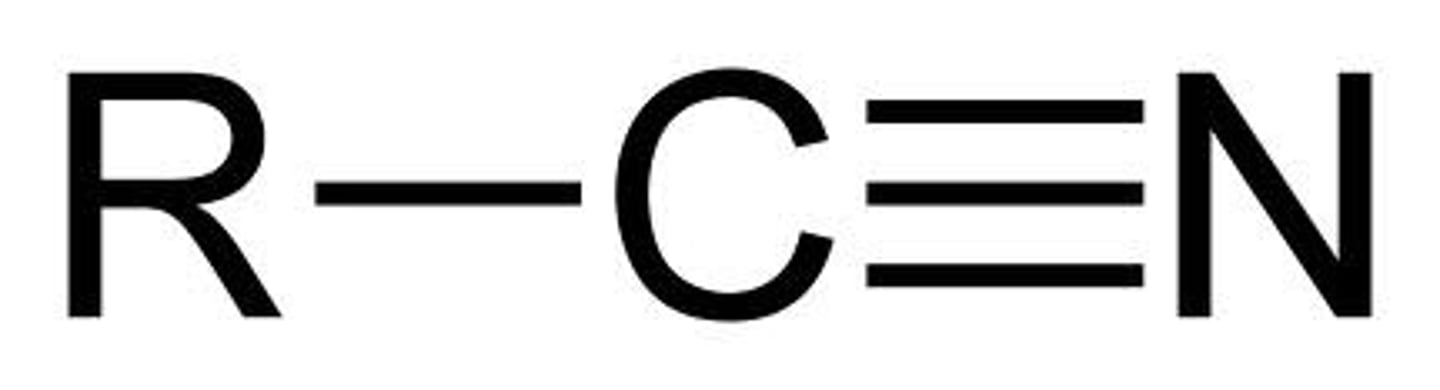
azide
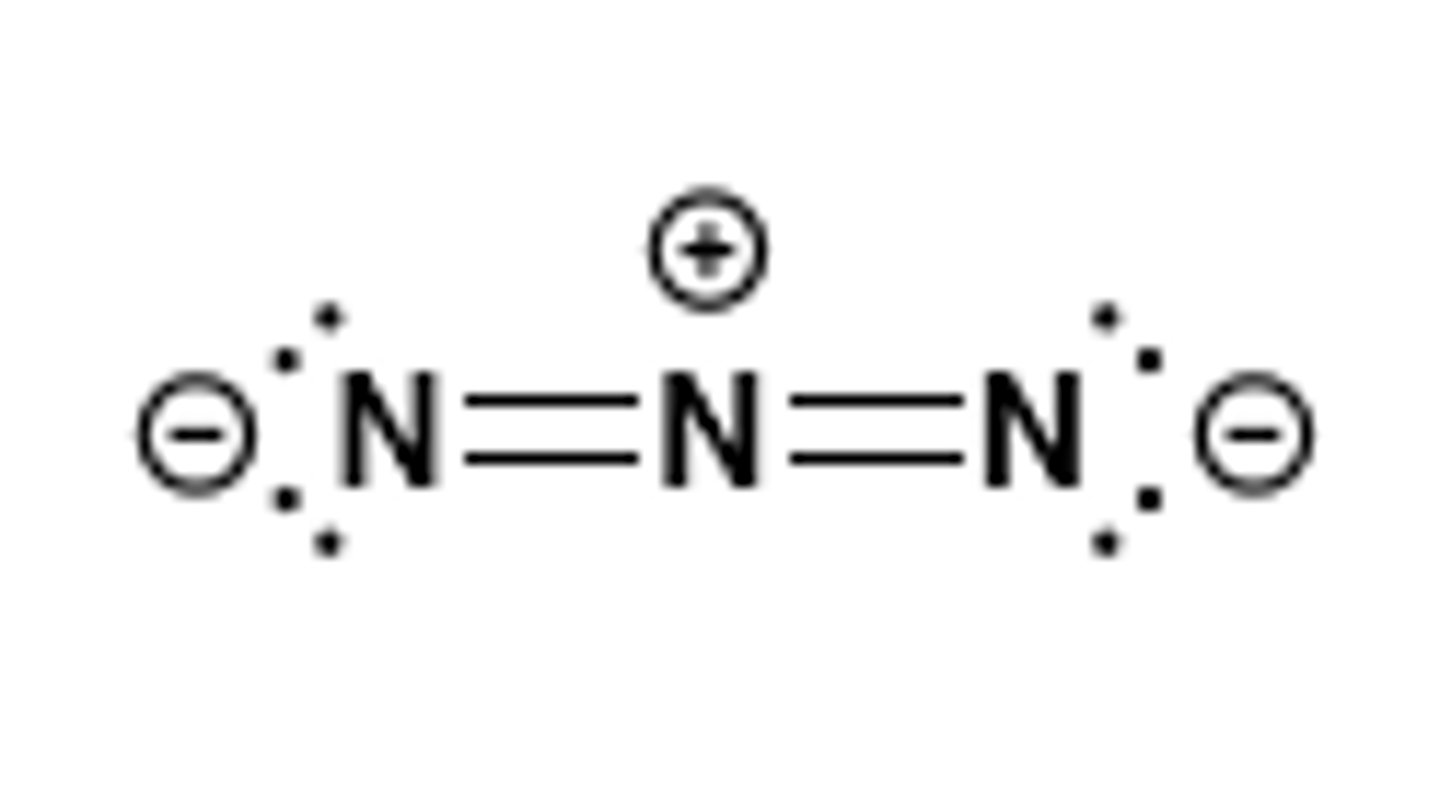
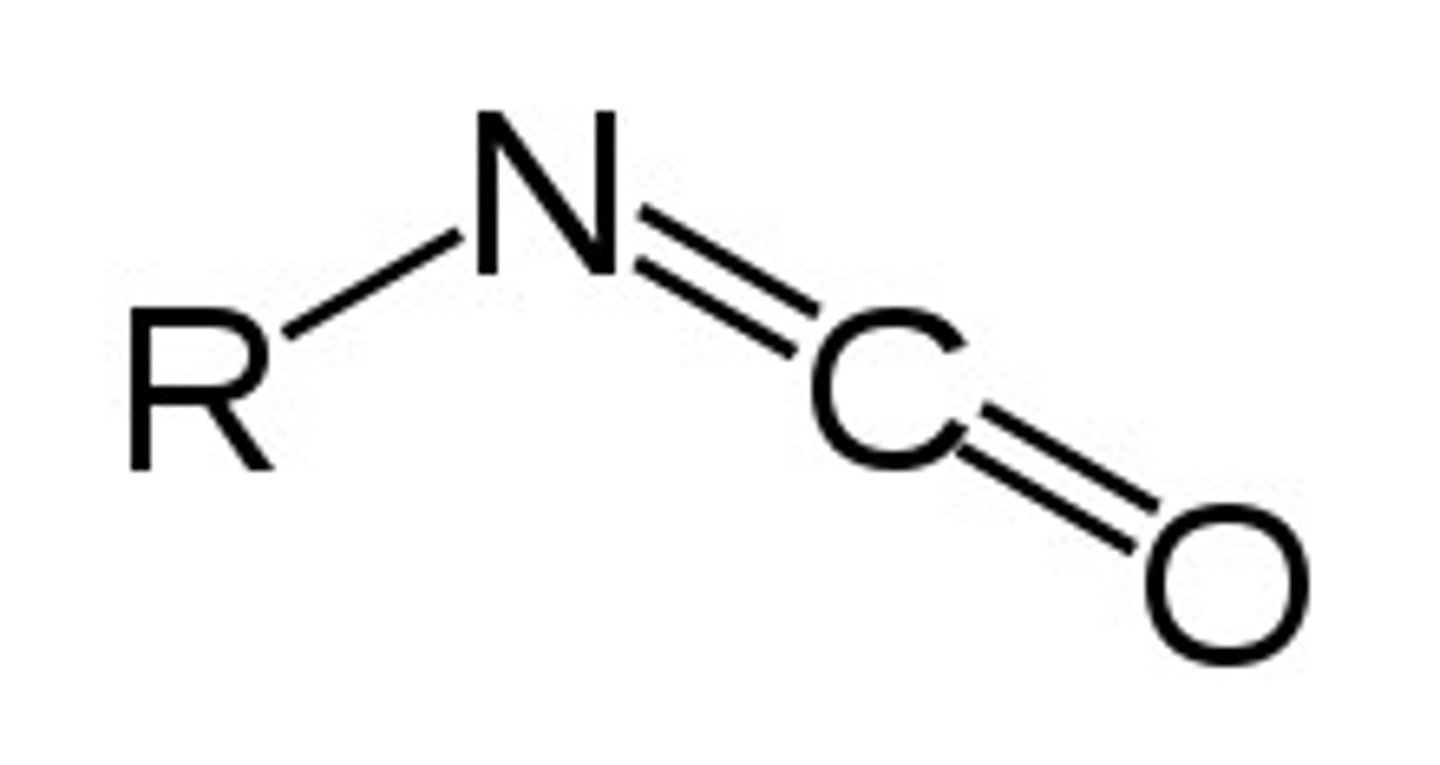
isocyanate
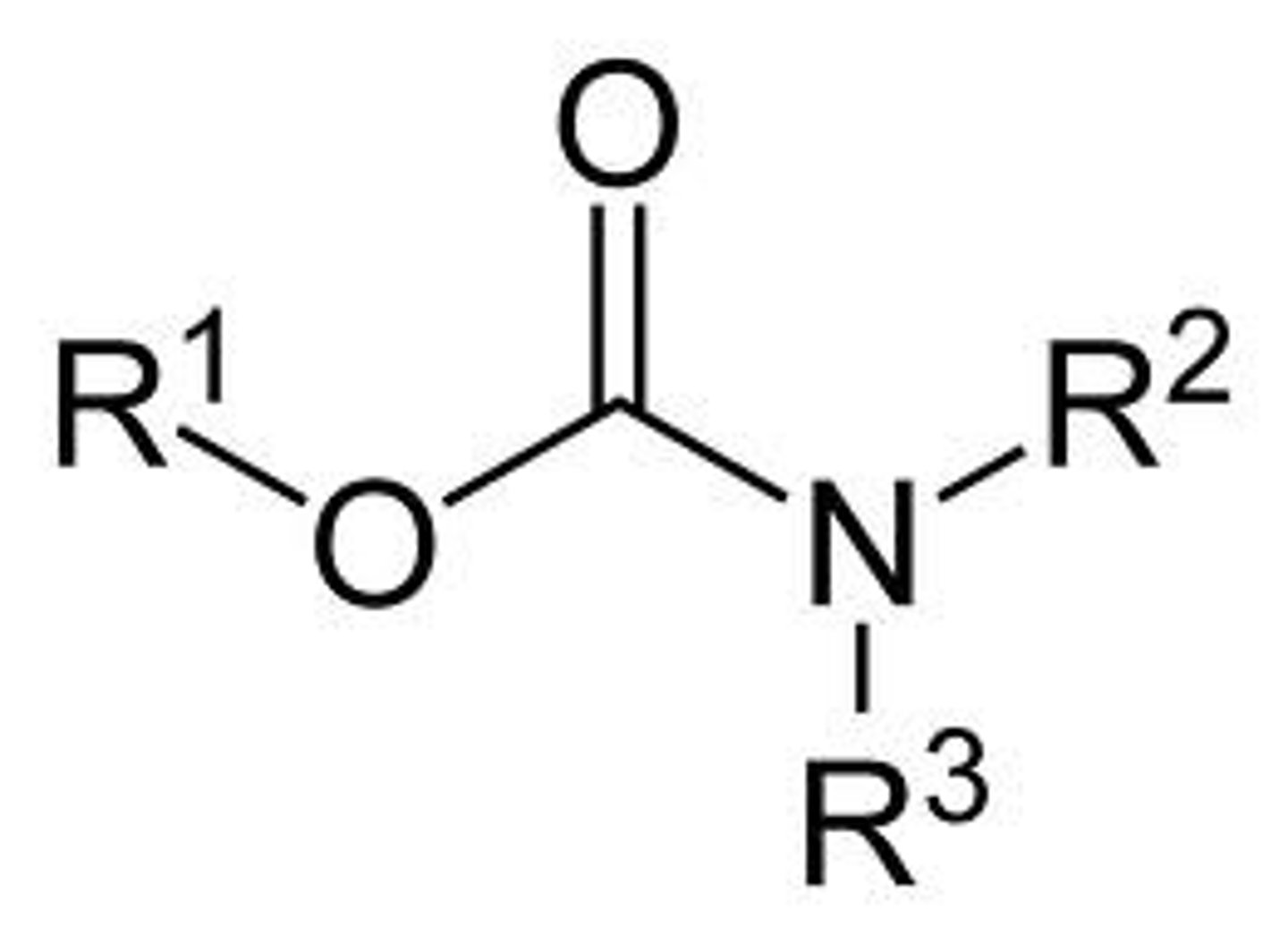
carbamates
nitrene
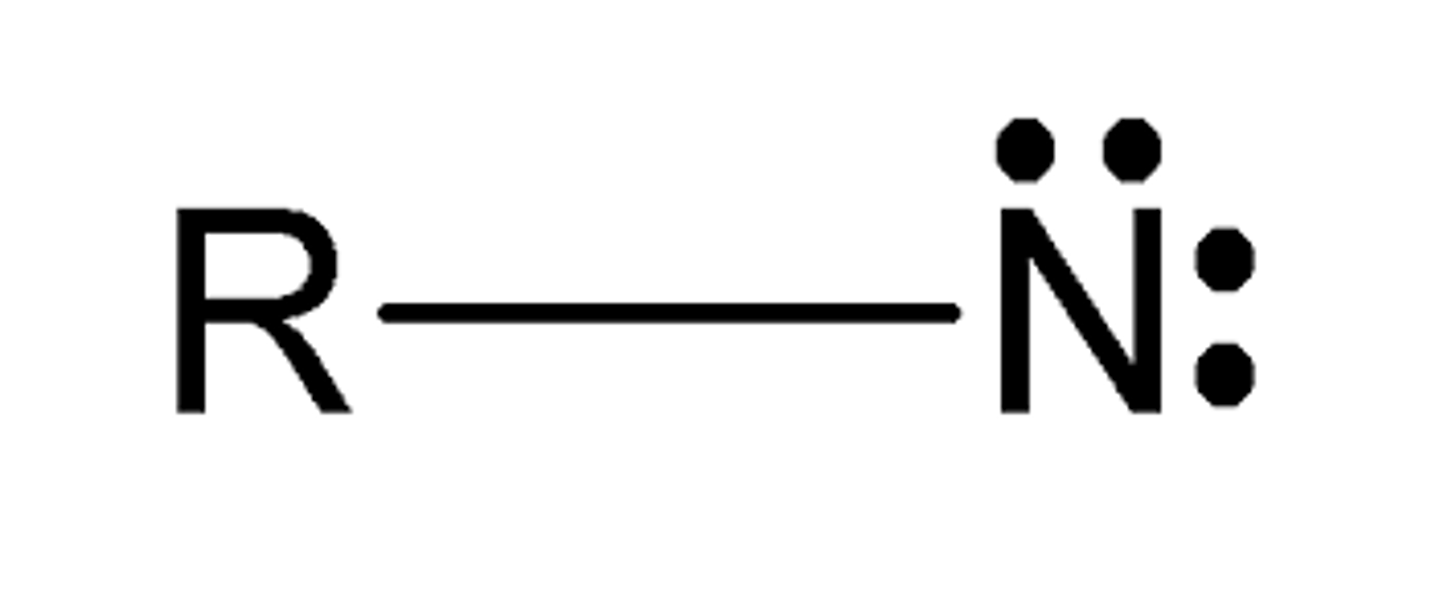
properties of amines
all but tertiary can form hydrogen bonds so higher bp than tertiary, sp3, needs 3 substituents, very weak acids
nitrogen inversion
an inversion of the sp3 orbital occupied by the lone pair
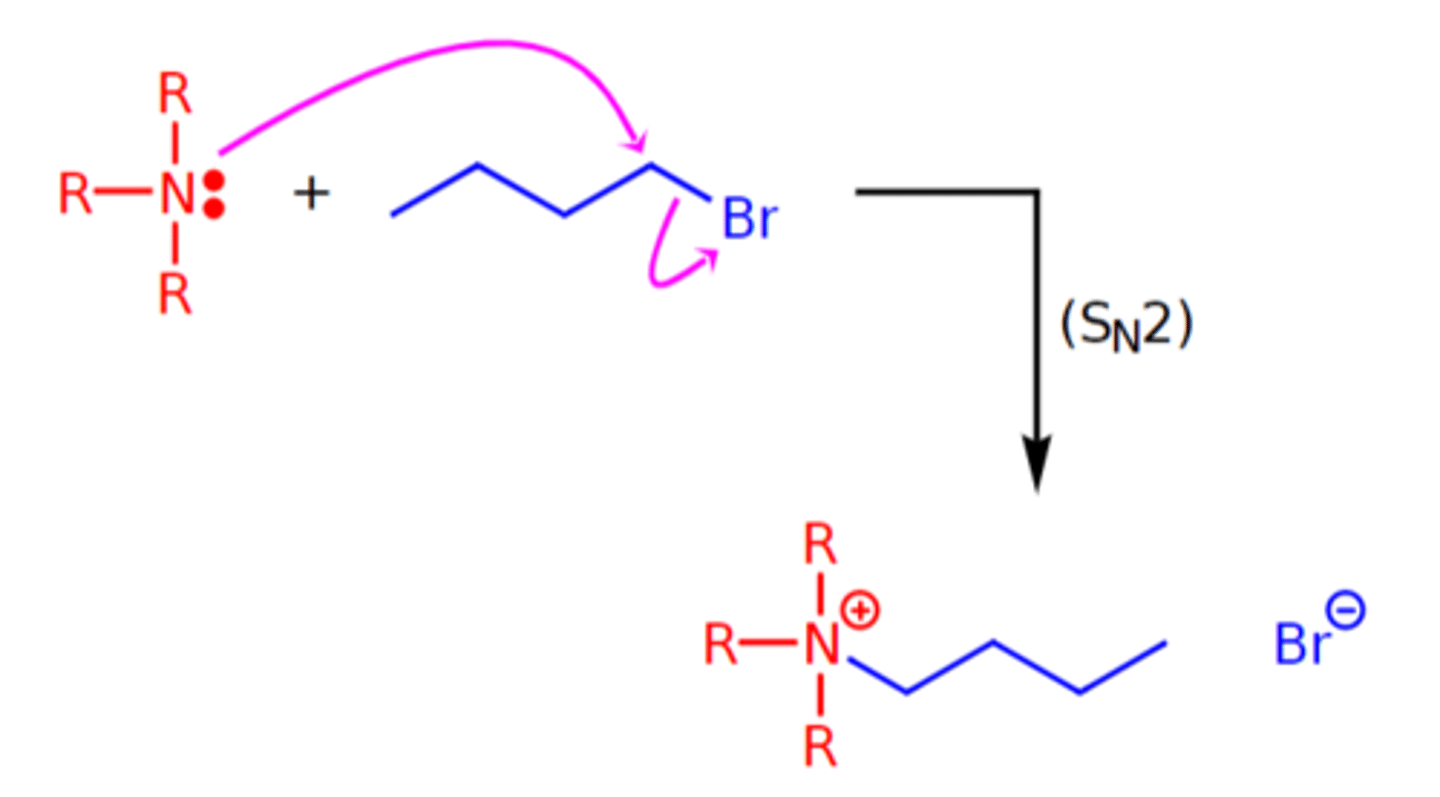
alkylation of amines
Lower amines can be alkylated by reacting them as nucleophiles with alkyl halides
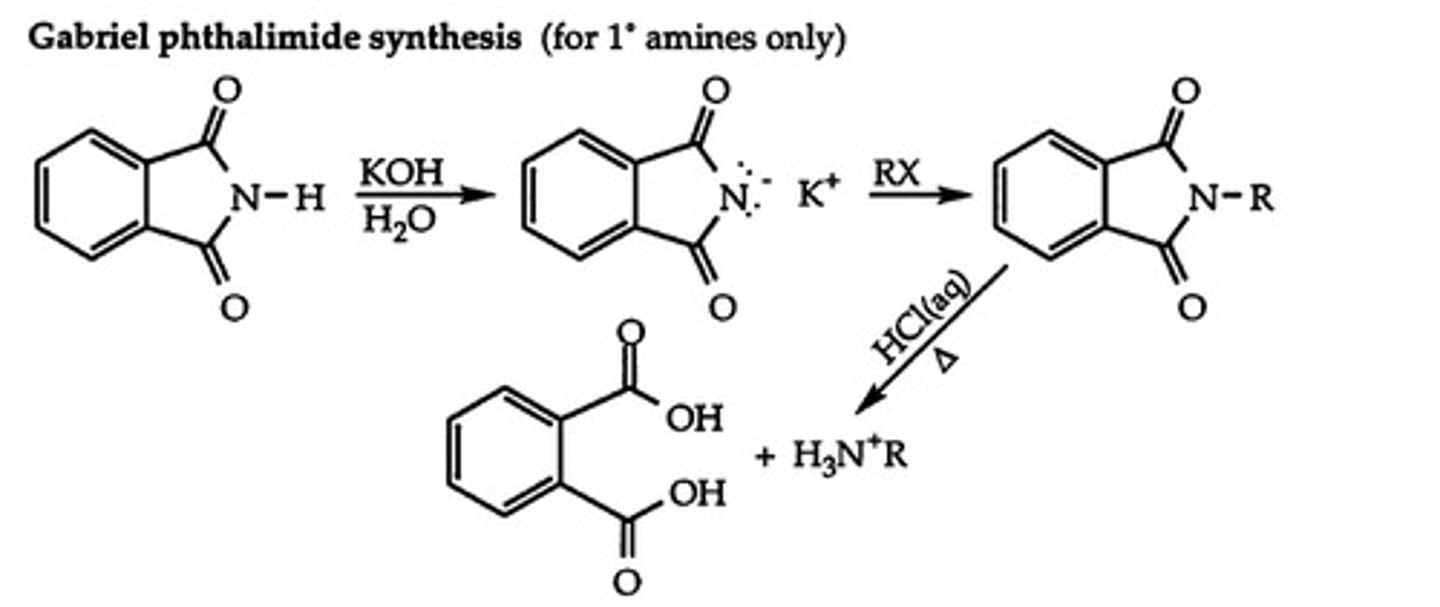
Gabriel synthesis
converts primary alkyl halide into primary amine using phthalimide intermediate
reduction of nitro compounds
Converts nitro compounds to amines using hydrogen.
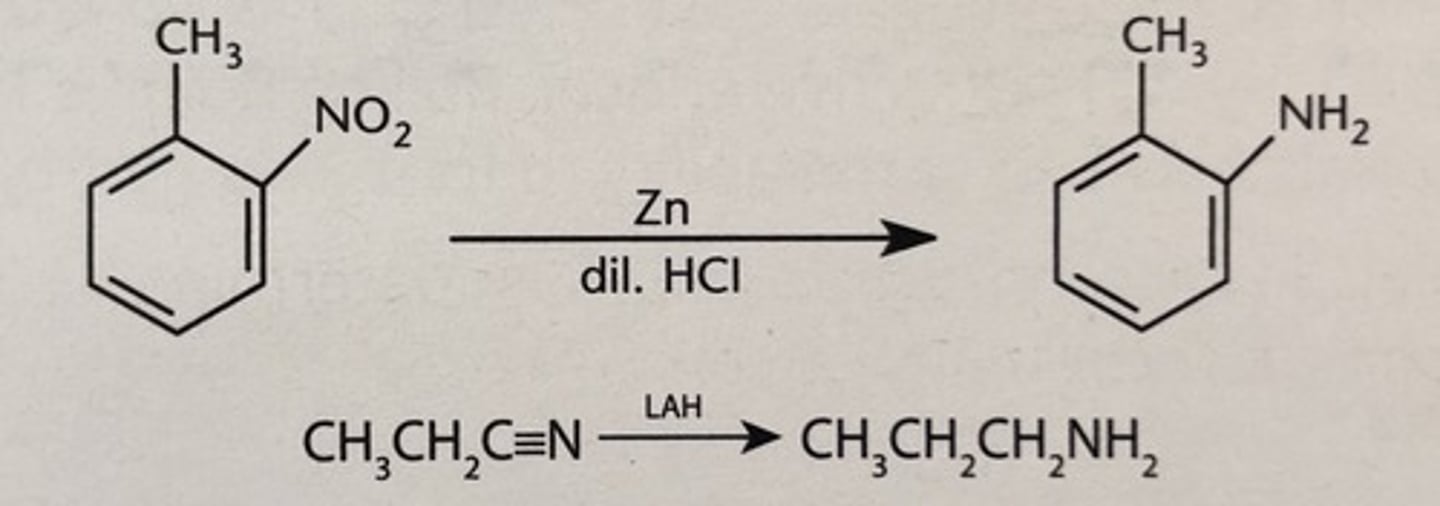
reduction of nitriles
Nitriles reduced to amines
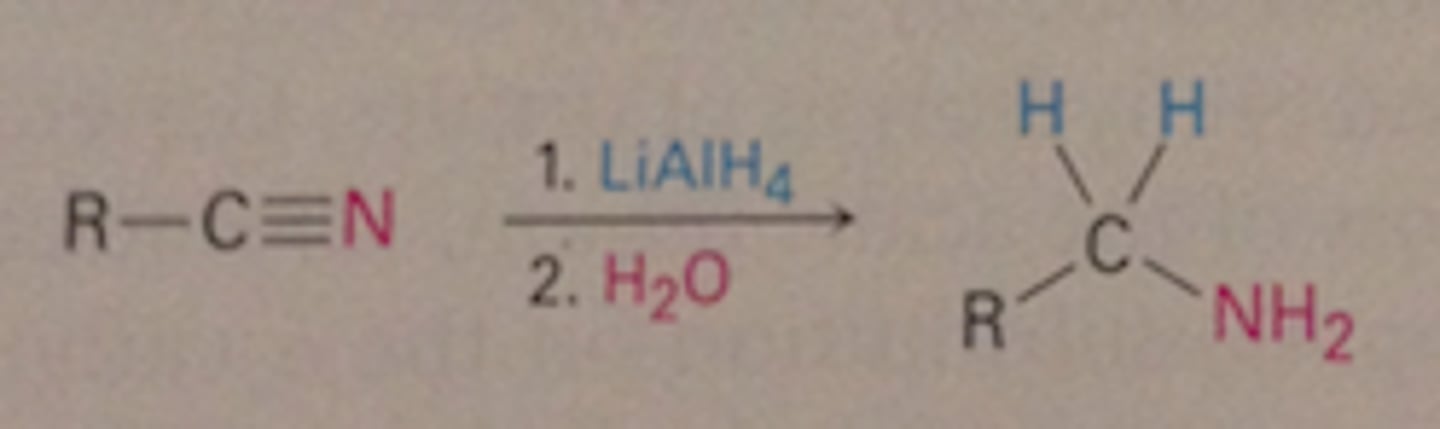
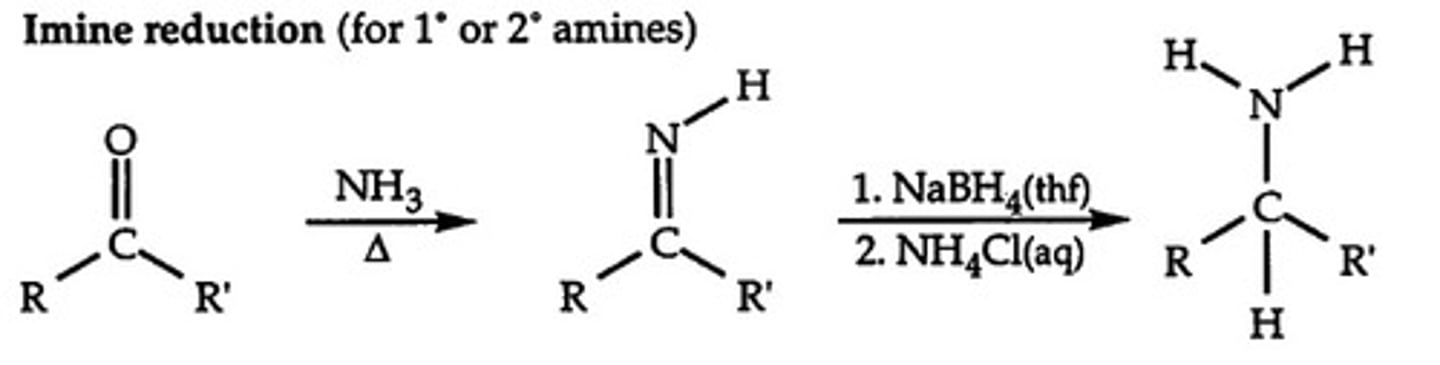
reduction of imines
reduction of amides
1. LiAlH4
2. H2O
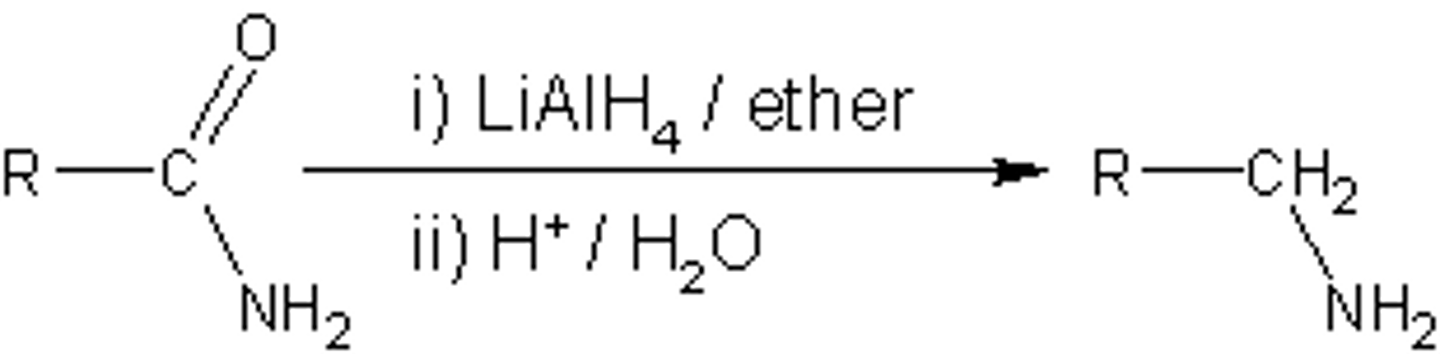
exhaustive methylation
The reaction of an amine with sufficient methyl iodide to convert the amine into a quaternary ammonium iodide