Mono and polysaccharides
1/96
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
97 Terms
How to find D and L stereochemistry of chiral compounds
R= D (put group on the right side of fischer projection)
S = L (put group on left side of fischer projection)
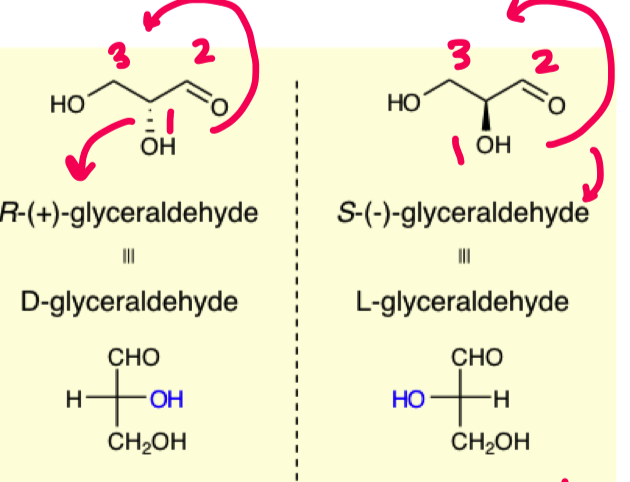
what stereochemistry do all naturally occuring sugars have?

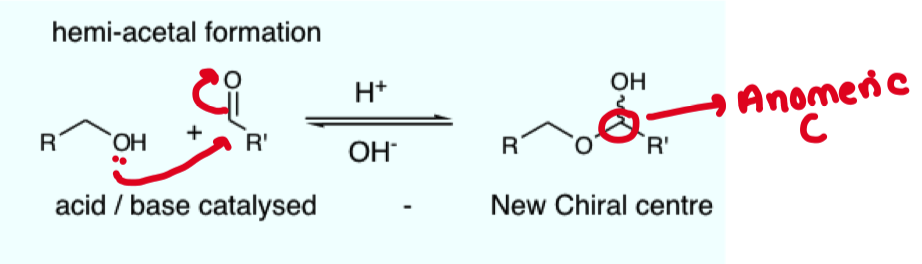
Give the general equation for hemiacetal formation (starting from alcohol and aldehyde)
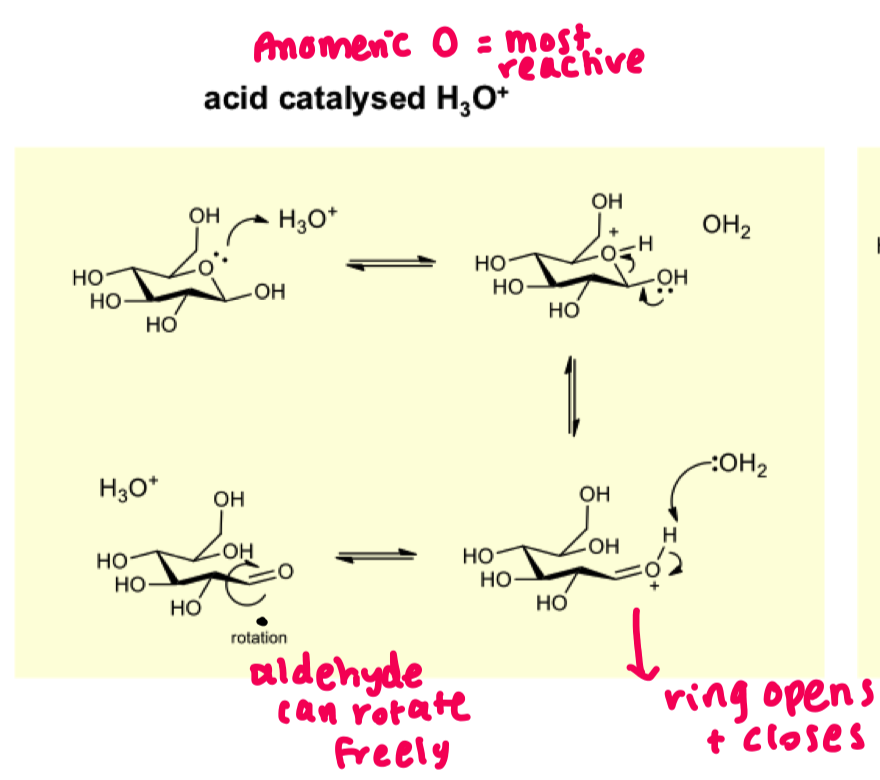
give the mechanism for acid catalysed hydrolysis of hemiacetal
give mechanism for the base catalysed hydrolysis of hemiacetal
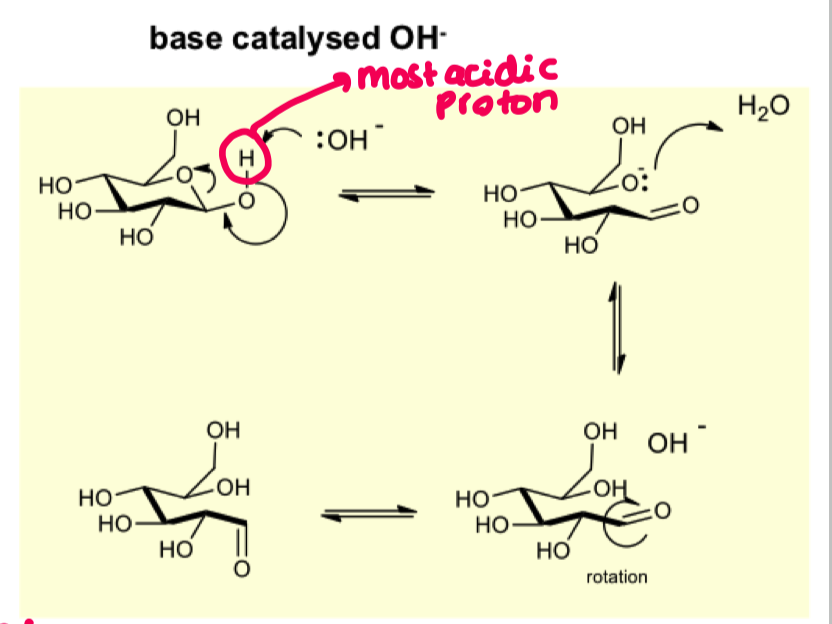
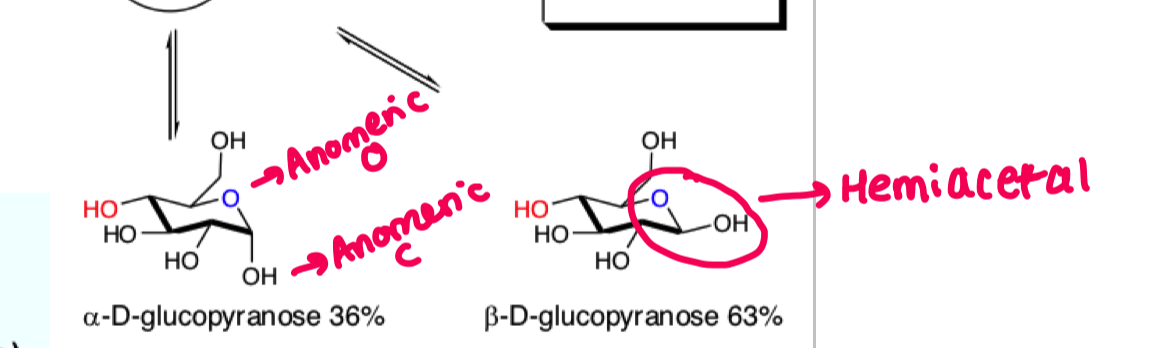
what is the consequence of the aldehyde being able to rotate freely?
you end up with alpha beta mixtures
these have different physical and chemical properties
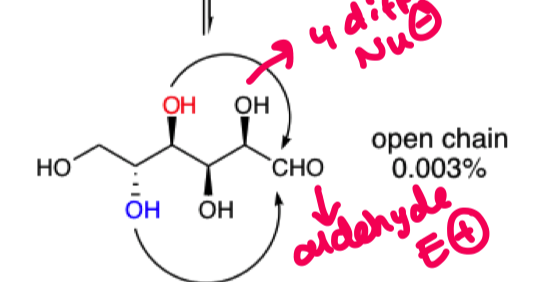
why do we only get attack from the red or blue OH on this chain?
as if the other OH were involved then the ring would be too small
thermodynamically favoured ring size is 5-6 membered.
what do we get if we attack from the red OH
form a 5 mem furanose ring
alpha and beta
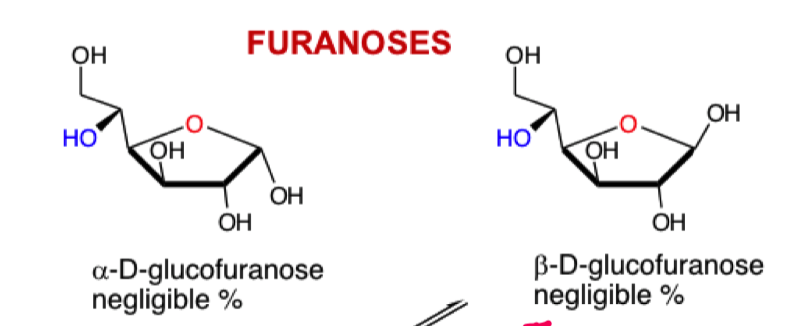
what do we get if we attack from the blue OH?
6 membered ring pyranose
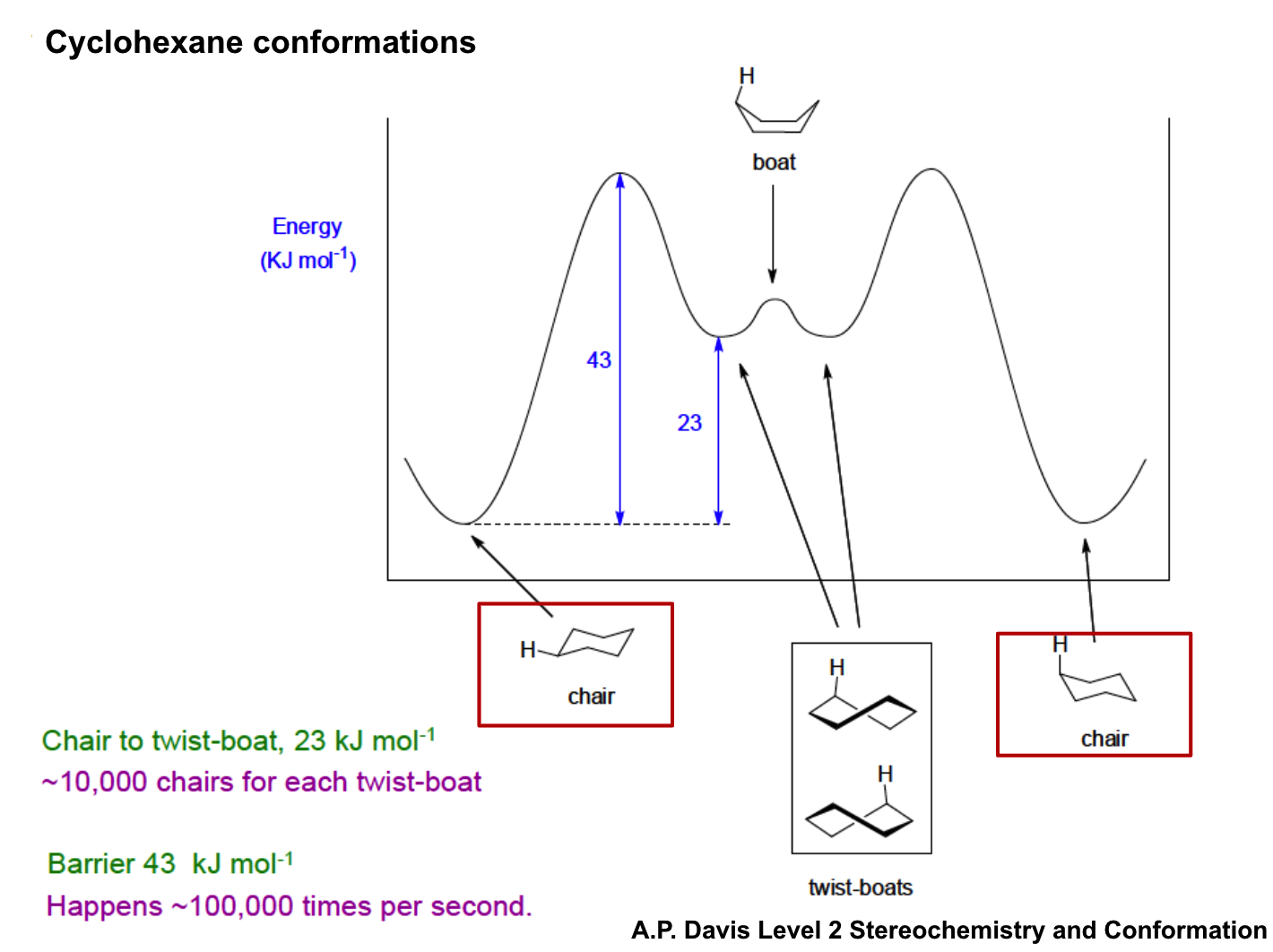
explain this graph
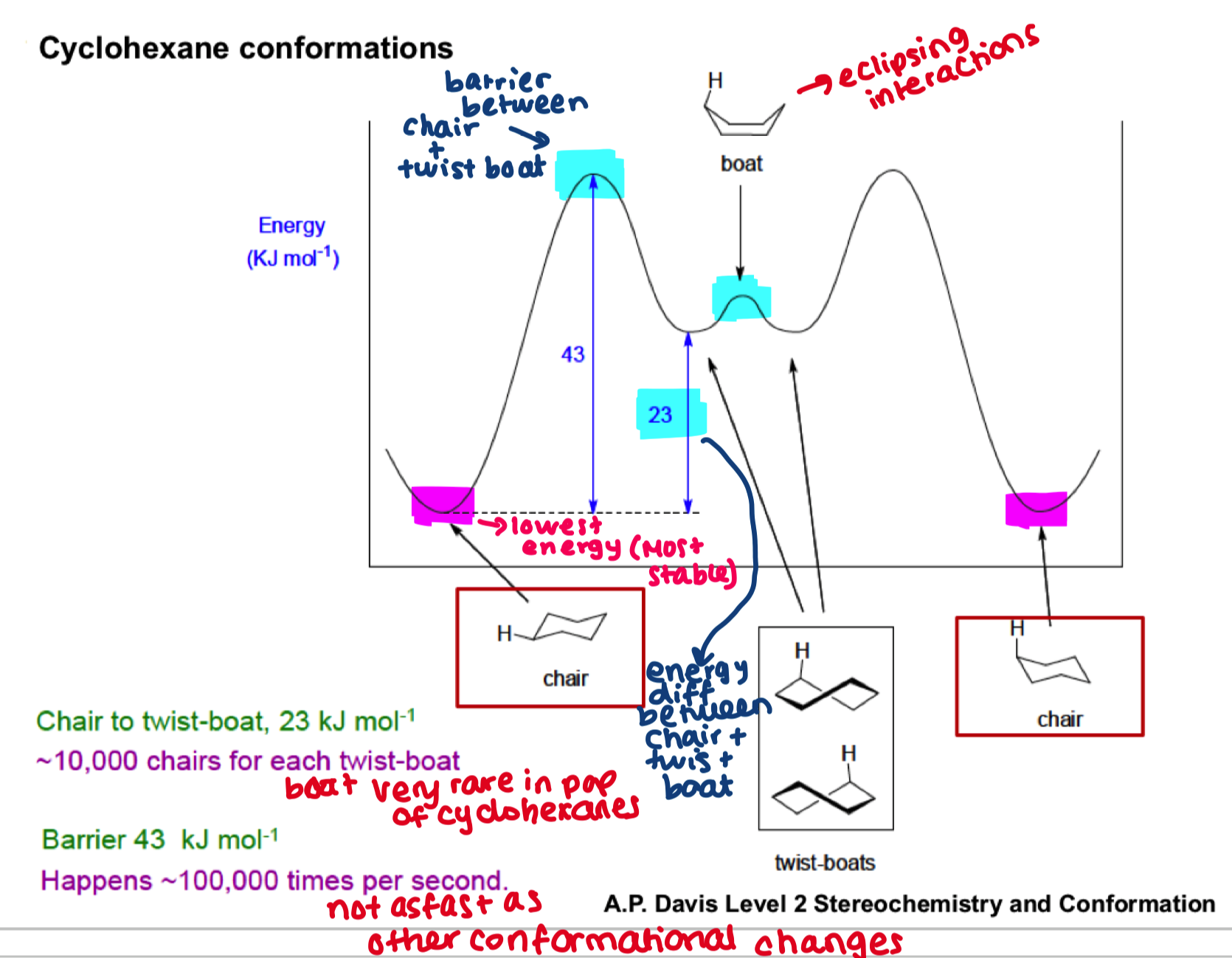
draw all axial and then all equatorial substituents around a cyclohexane ring
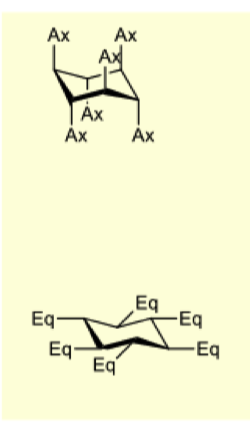
draw alpha D glucose and then beta D glucose
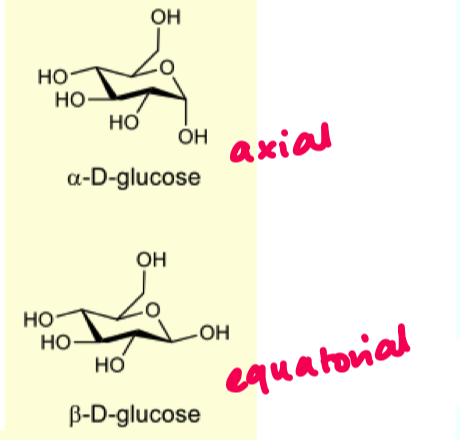
which are more preferred axial or equatorial?
equatorial
because 1,3 diaxial interactions are disfavoured
also to avoid gauche butane interactions
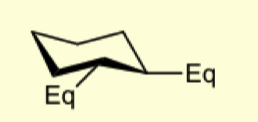
what are 1,2 eq substituents to eachother?
trans
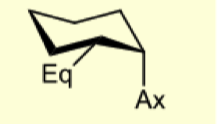
what are 1,2 eq/ax substituents to eachother?
cis
draw the alpha D glucose and flip it to show all substituents
flipped chair = L configuration
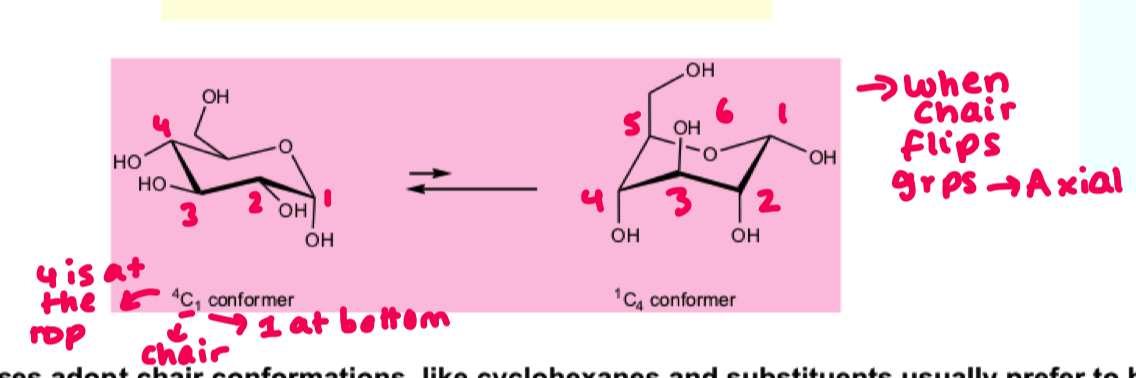
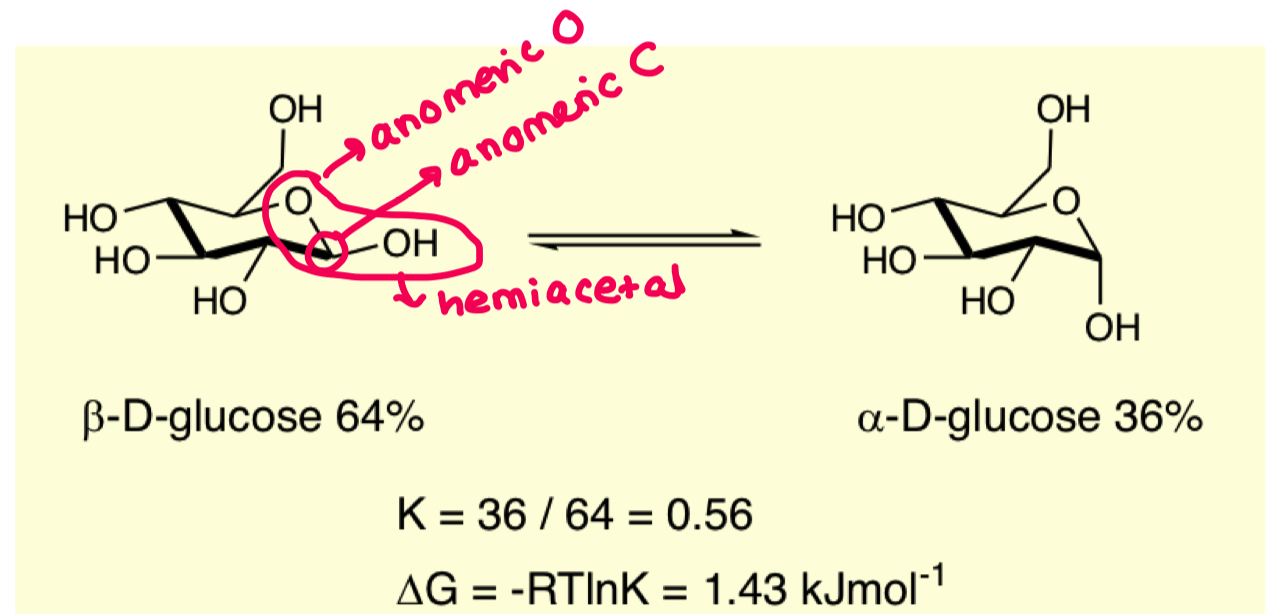
we see a preference for the beta equatorial position but only just. why?

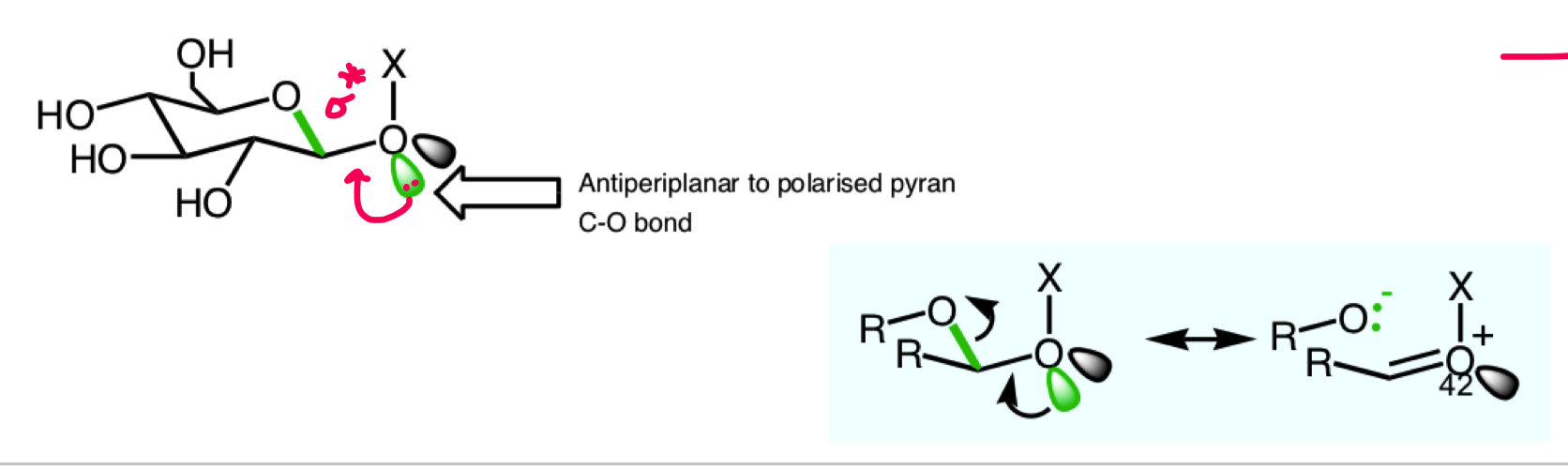
what is the anomeric effect?

why does the anomeric effect occur? draw the 2 stabilising interactions
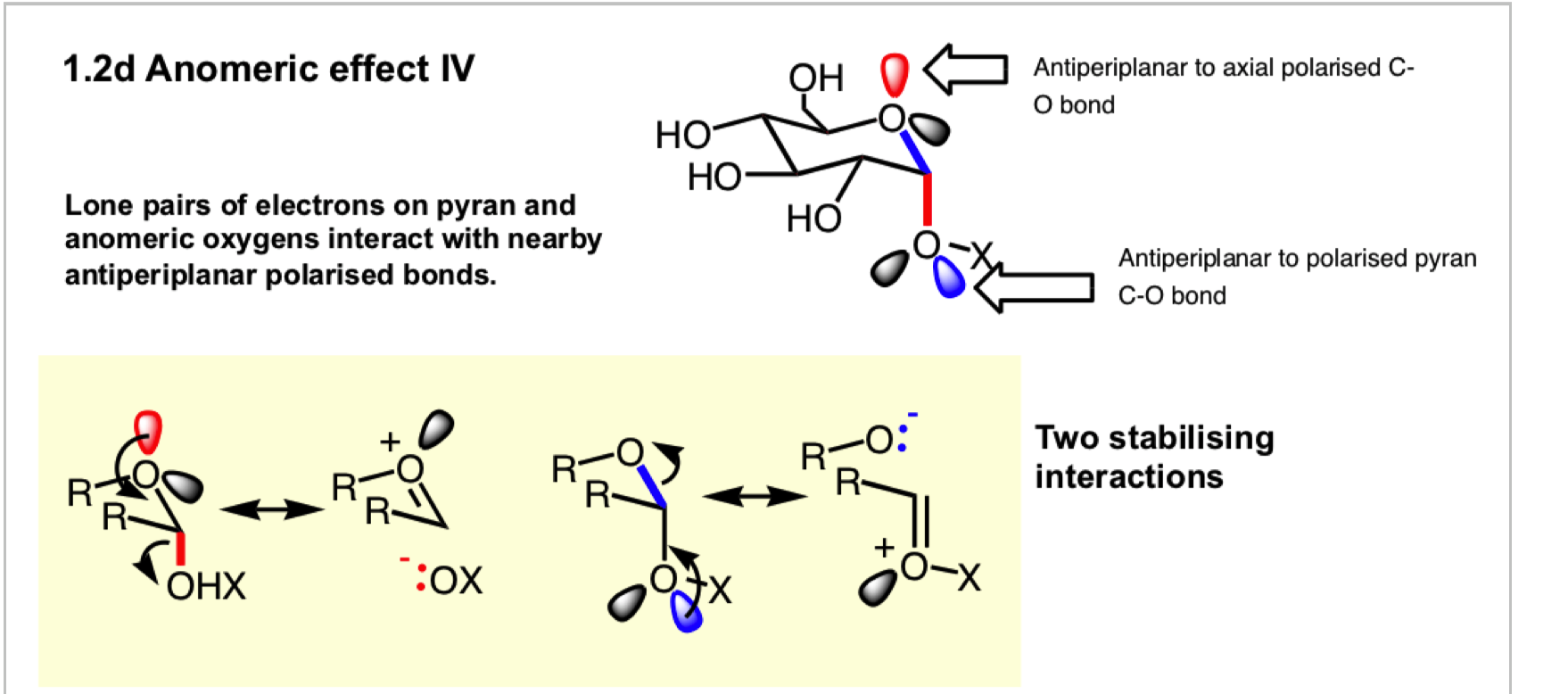
describe the small stabilisation that we see for an equatorial substituent on the ring
why are protecting groups useful?

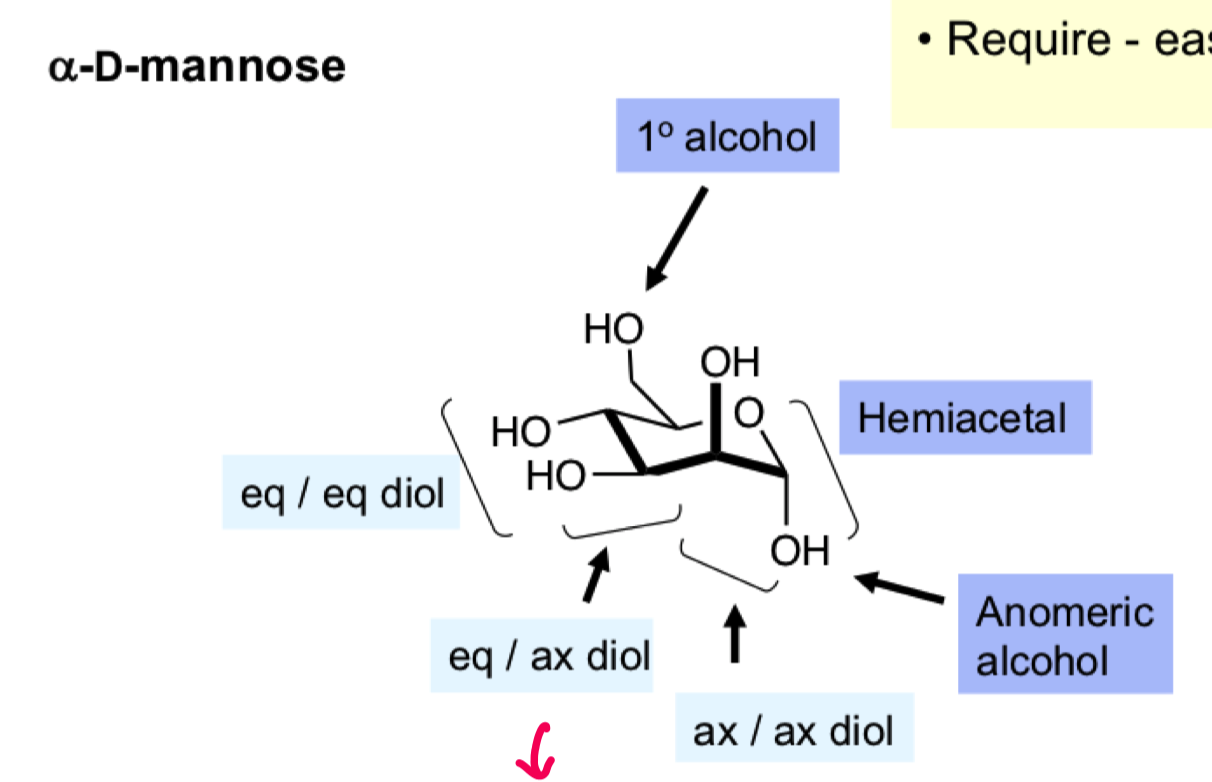
Annotate a- D glucose with the different functional groups
what is an orthogonal set?

describe reagents/reaction for the protection of alcohol with OAc?
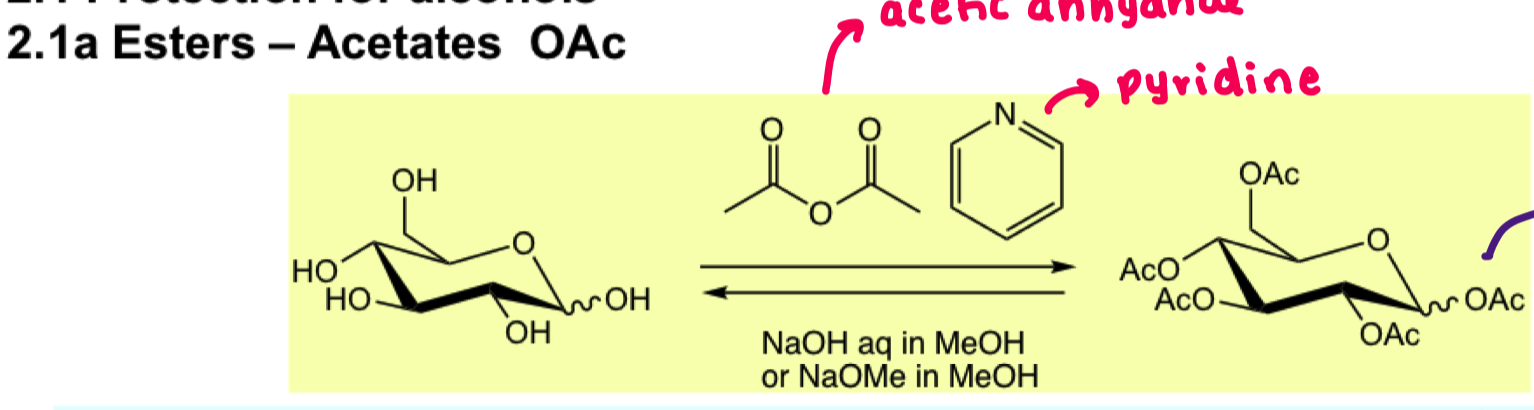
why do we NEED to use pyridine?
because OH nucleophile on its own is not nucleophilic enough
pyridine is used to generate a good electrophile
could also use ET3N
pyridine also works as a solvent
draw the mechanism for alcohol to OAc
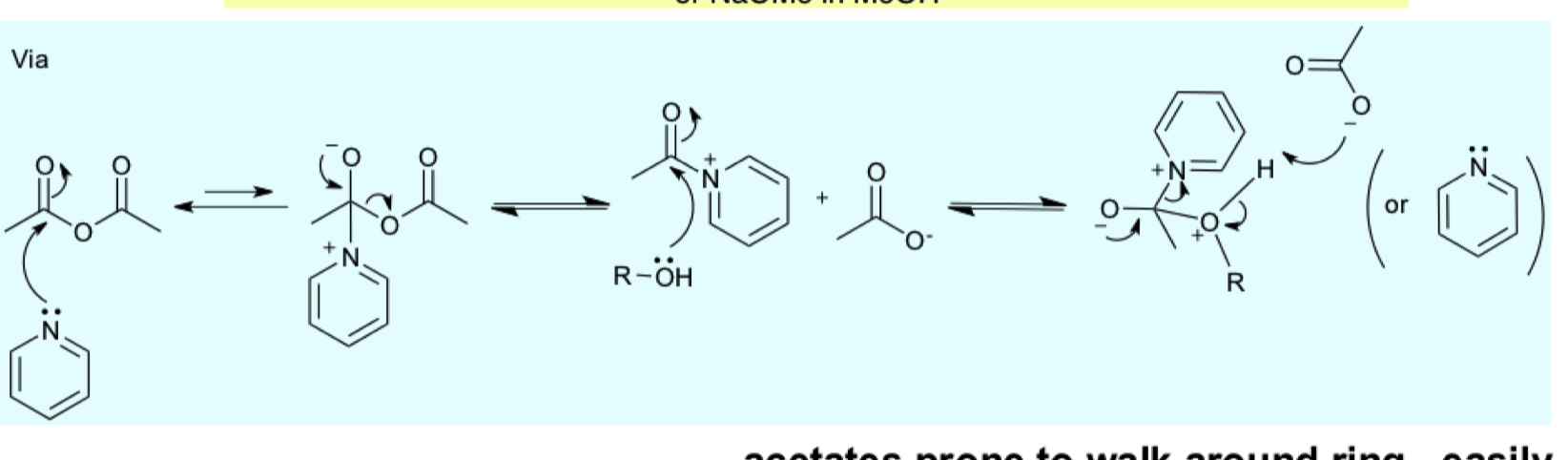
what is a con to acetate groups
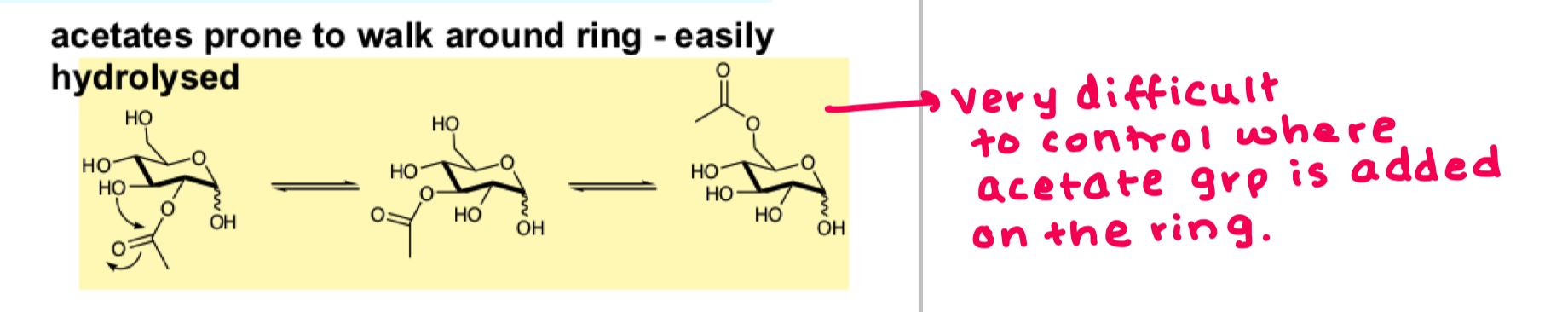
why can acetate not move to the 3 position?
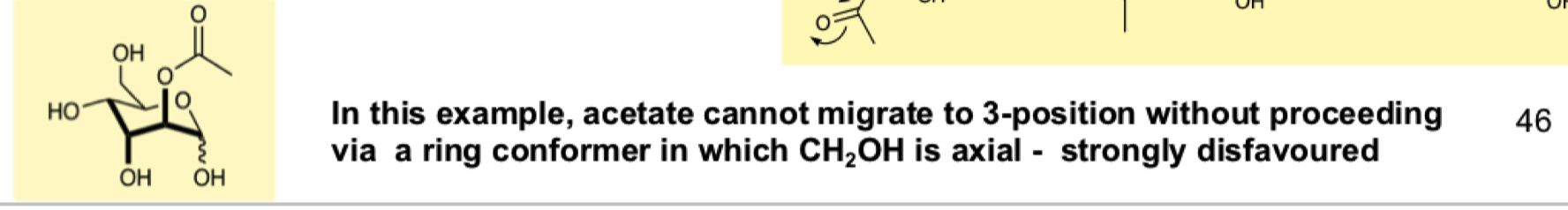
draw the mechanism for the reverse reaction of OAc to OH
Draw the mechanism for the addition of a benzoate group, what functional group does benzoate contain?
ester
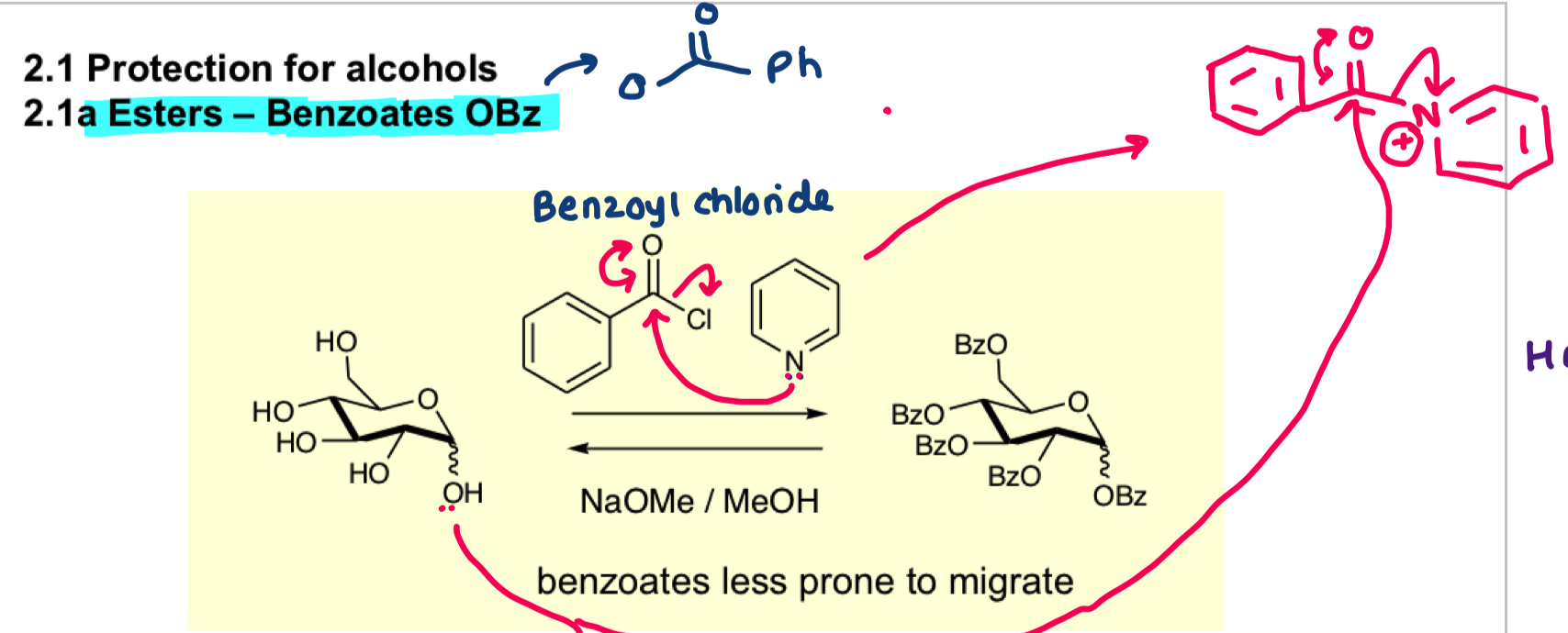
draw a benzyl group and what functional group does it contain?
-ether
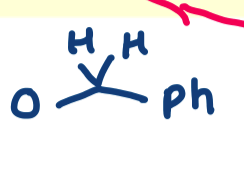
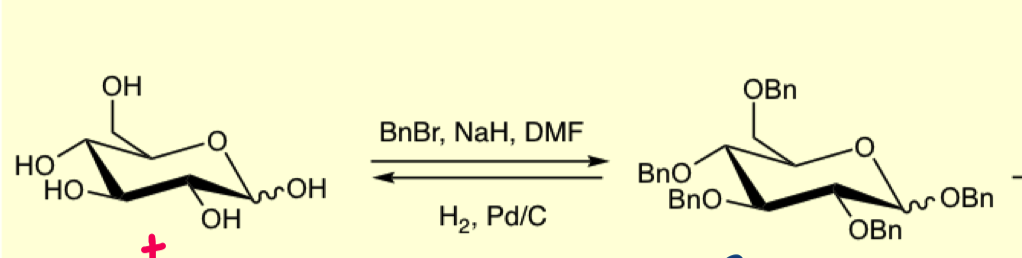
why can we not use BnBr alone? why is DMF used?
must use NaH as BnBr on it own is not nucleophilic enough
DMF is a polar solvent used to solubilise the substrate
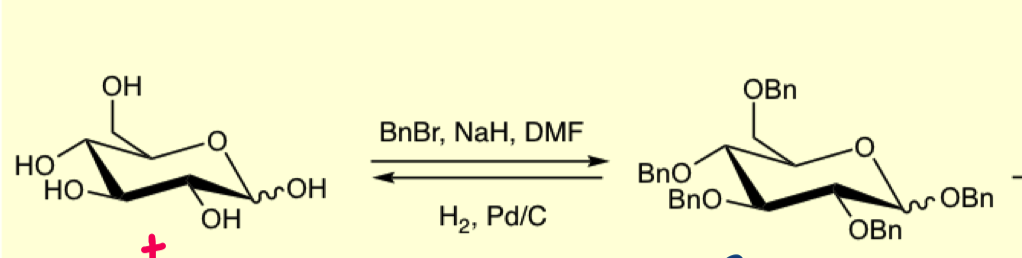
draw the mechanism for benzyl groups
forms an alkoxide which is now nucleophilic enough to attack BnBr
why do we need different protecting groups?
so that we can selectively deprotect one in the presence of the other
how can we deprotect only the anomeric carbon?
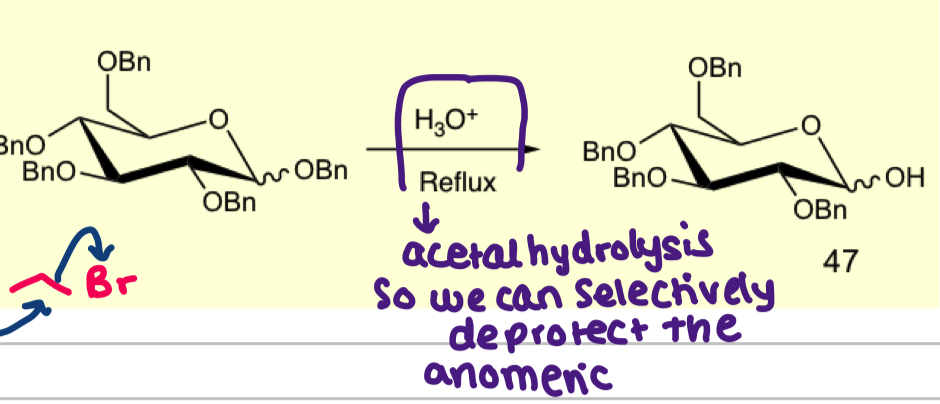
what is the purpose of using bulky ethers and give an example?
this is selective protection for primary alcohols ( very bulky and if u use 1 equiv then it will react with most reactive hydroxyl group)
example is OsiR3 or OTIPS
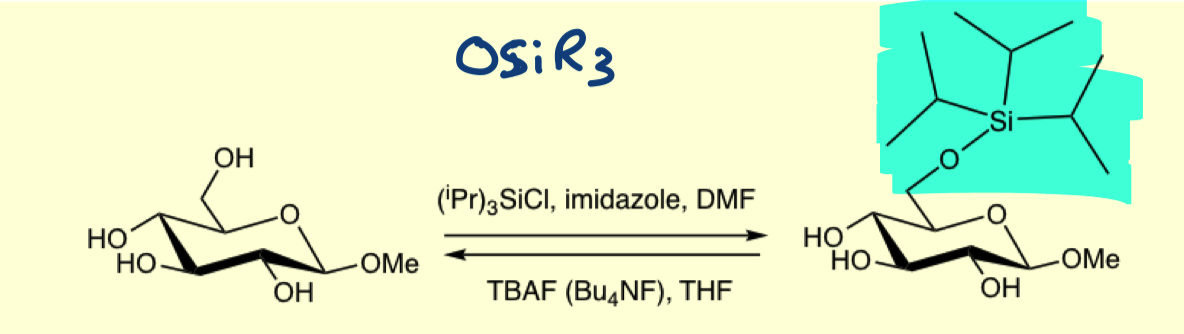
how can we protect all the groups with bulky ethers?
by using an excess of OSiR3
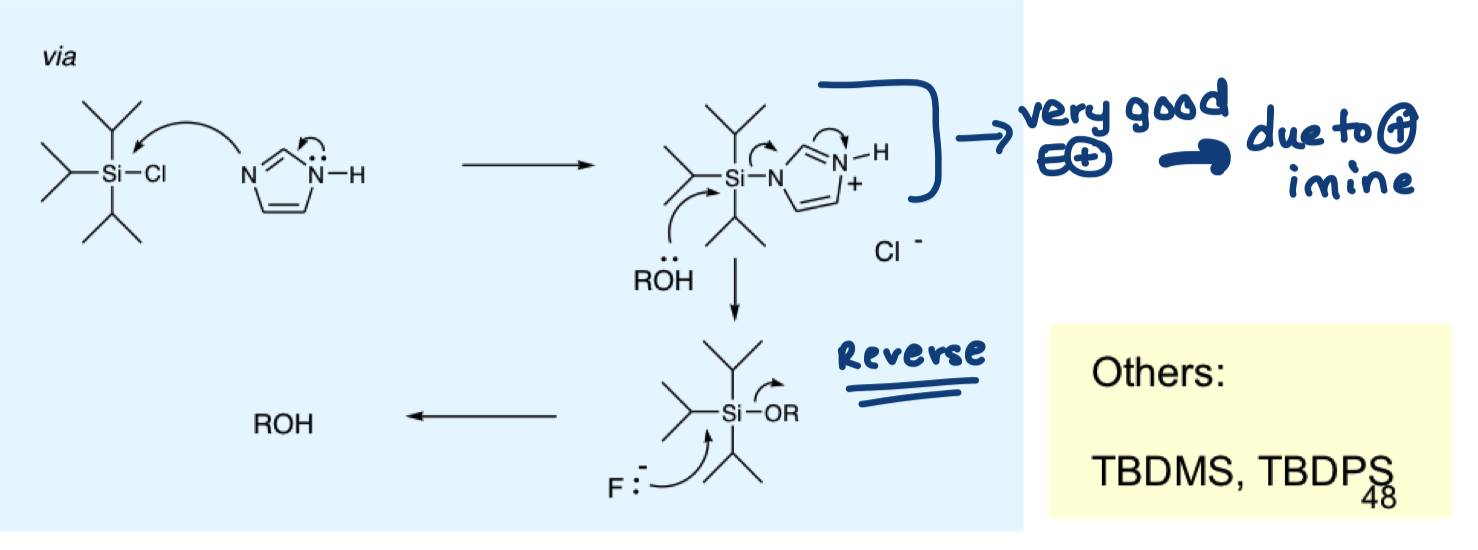
draw the mechanism of bulky ether
describe the purpose of using a TROC group?
— trichloroethylchloroformate is used to protect amines
which are more reactive: amines or hemiacetals?
amines are more nucleophilic
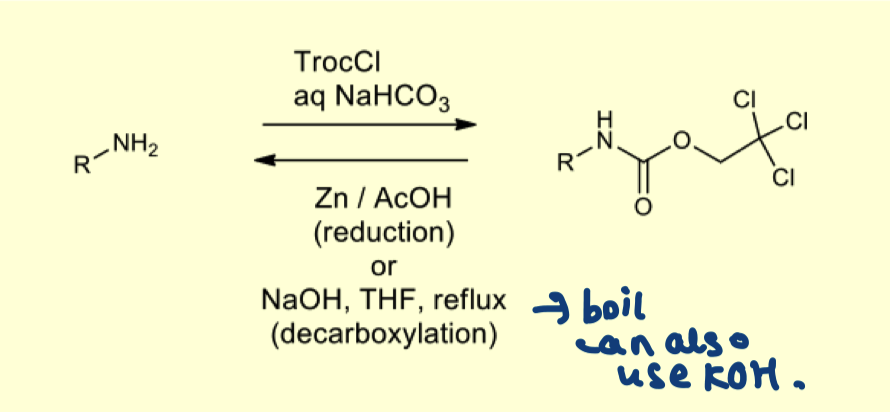
describe the mechanism of amine protection with TROC
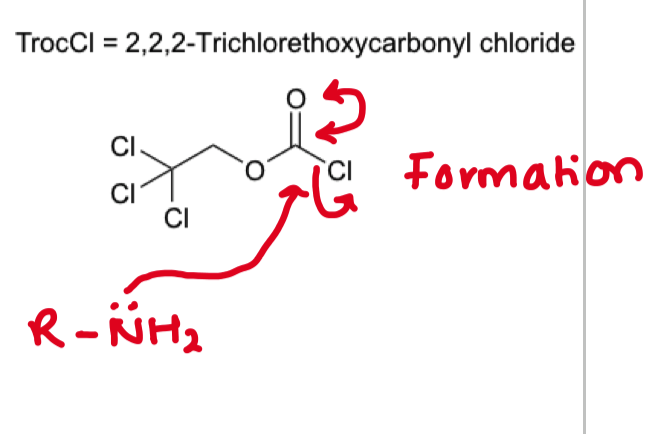
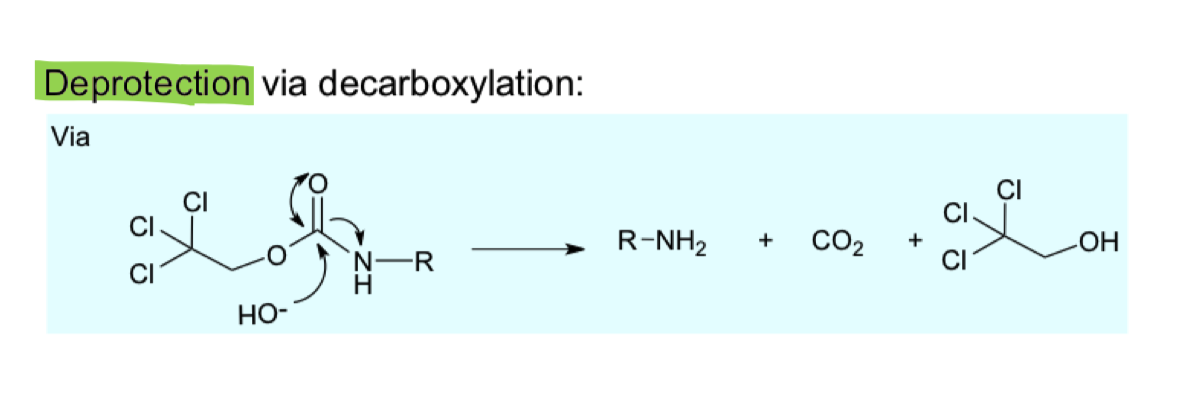
describe the mechanism of removing TROC from amine (reverse)
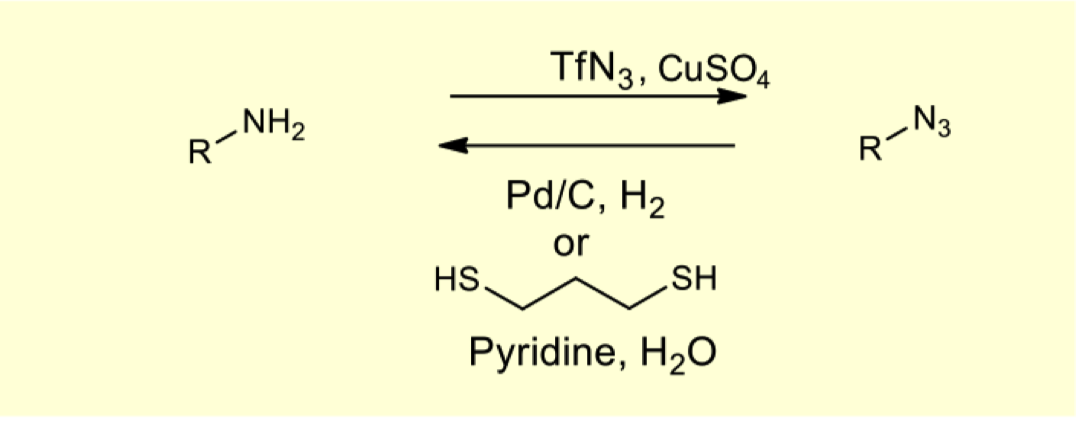
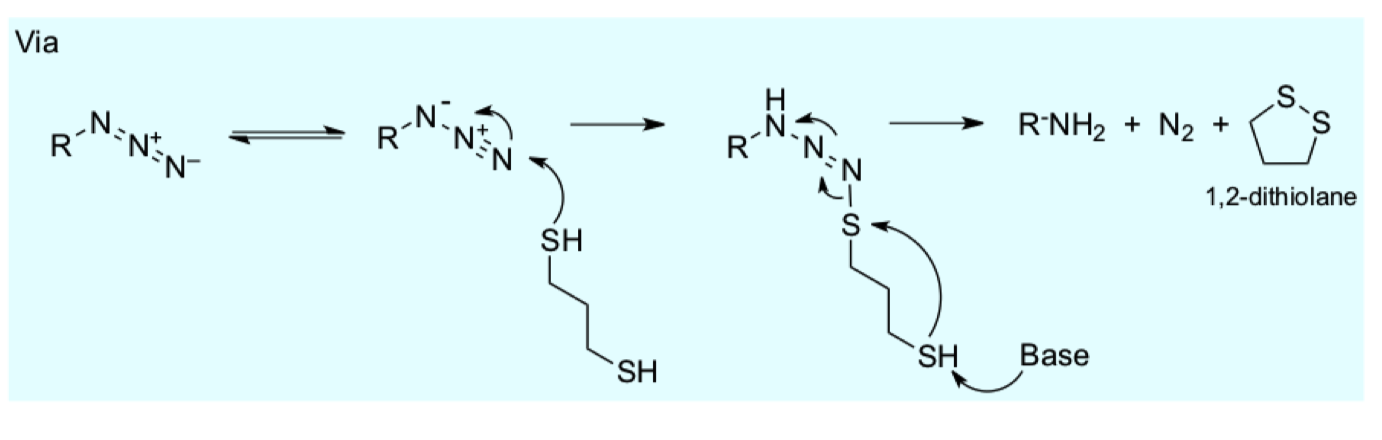
draw the mechanism for deprotection via azide reduction
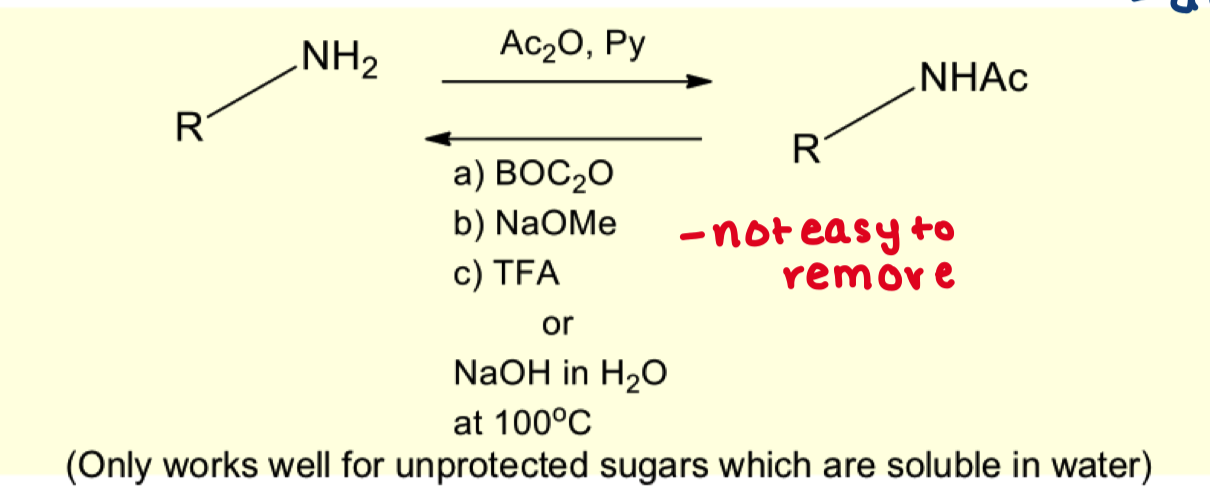
is it vital to use pyridine for this reaction? why is this reaction not commonly used?
no because the amine is nucleophilic enough to directly attack the Ac2O group.
it deactivates the molecule
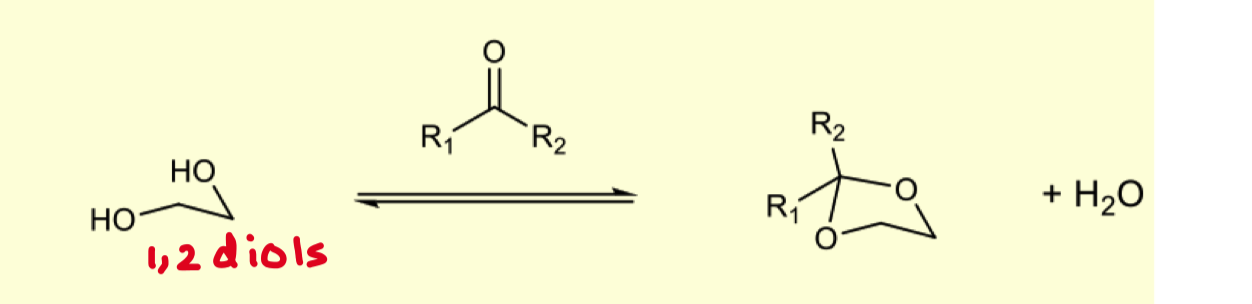
How can we protect 1,2 diols
solvent = excess of carbonyl
acid catalysis = H+/ZnCl2 or TsOH
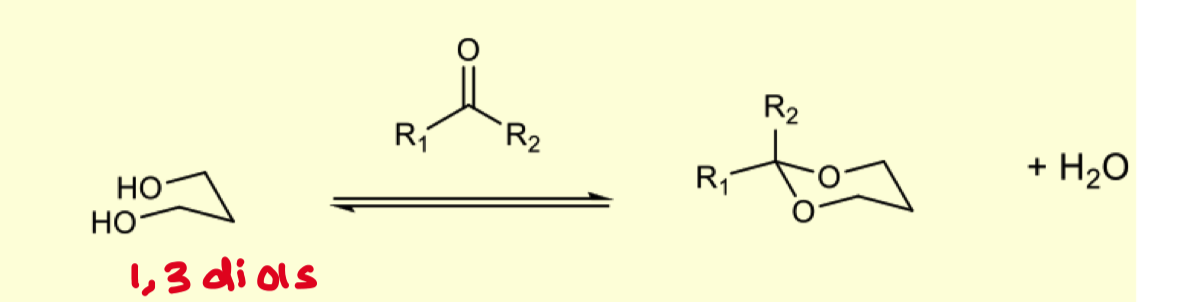
how can we protect 1,3 diols?
same reactants as 1,2
what do we use for the reverse reaction for diol deprotection
aqueous dilute acid
how to control whether we reacts a 1,2 diol vs a 1,3 diol?

how do cis diols and trans diols affect selectivity for diol protection?

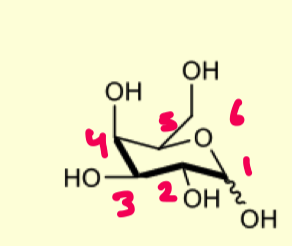
describe the protection of D-galactopyranose with acetonides
prefers 1,2 diol
prefers eq/ax relationship
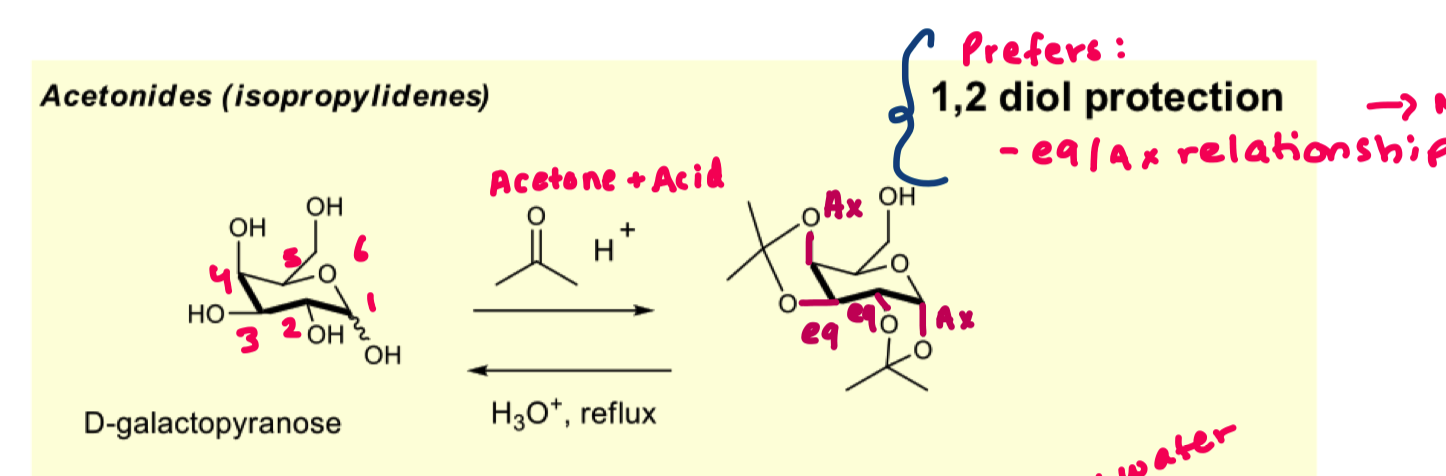
why does the OH at the anomeric prefer the axial position over the equatorial?
because we want to form the most thermodynamically stable product
we are using a ketone so it prefers ax/eq relationship
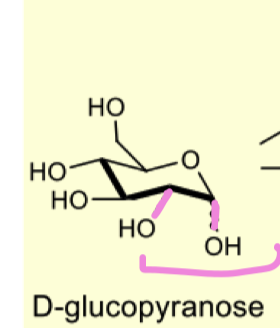
describe the protection of D-glucopyranose with acetonides and explain why it formed the 5 mem ring?
forms the 5 membered furanose ring
because there arent many ax/eq relationship therefore to make more we can treat c4 OH and C5 OH as a 1,3 diol
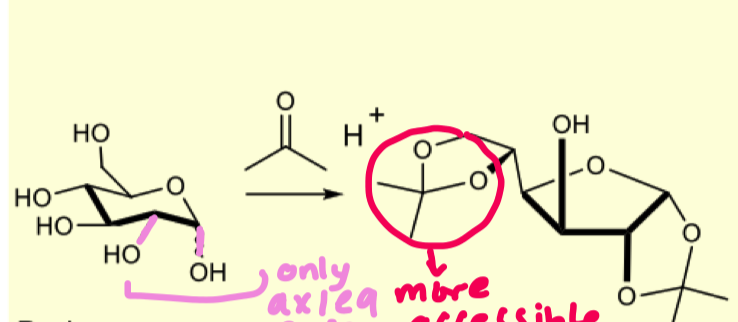
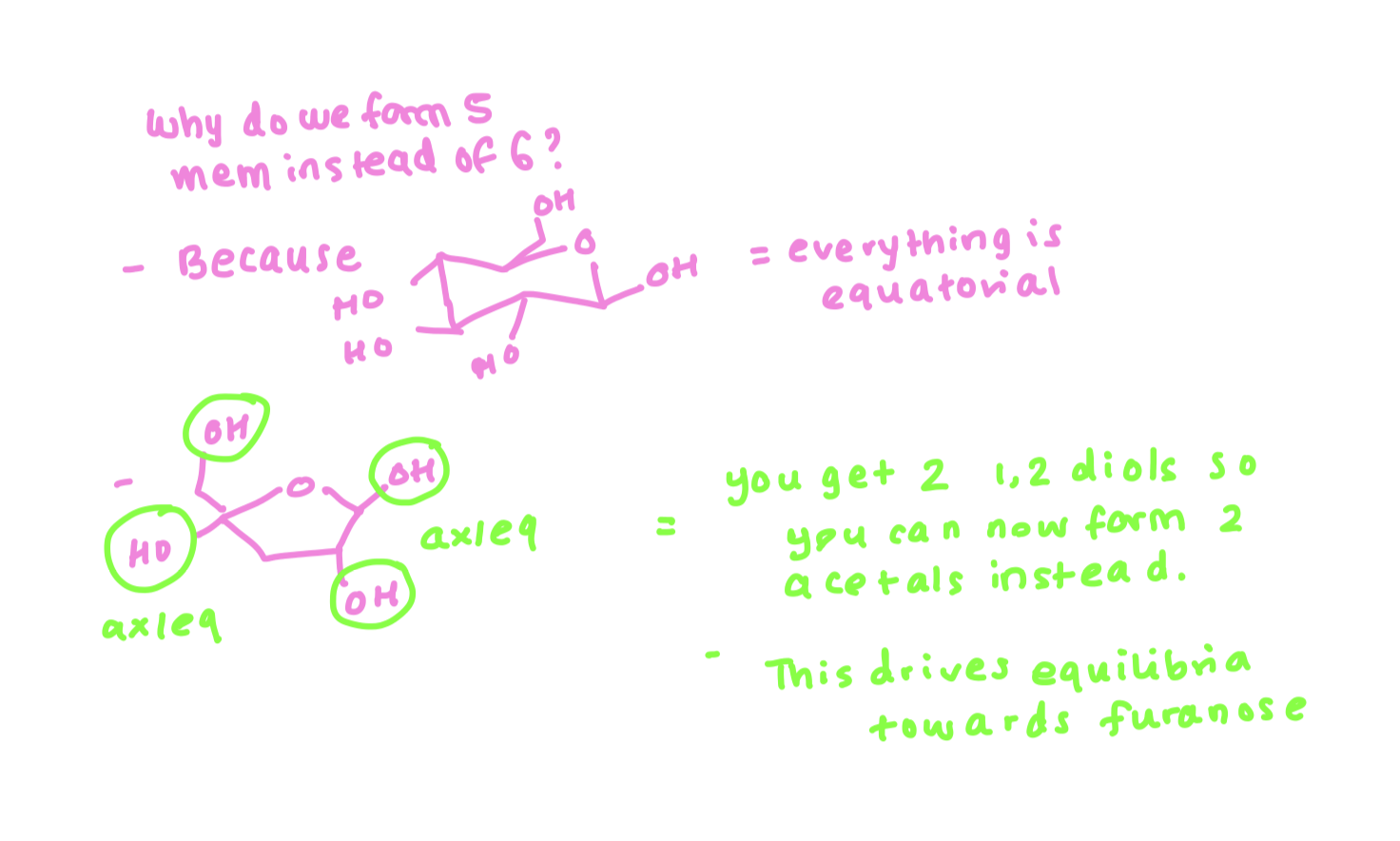
why does it form the 5 membered ring and not the 6 membered ring ?
hemiacetals are always in equilibrium with other species so finding axial/equatorial relationships pushes equilibrium towards forming furanose over pyranose
what are the reagents for the reverse reaction (deprotection of acetonides)?
H3O+
how can we selectively deprotect certain acetals?
you always hydrolyse the most accessible acetyl
the one on the anomeric is less accessible compared to others at the primary position
so for this use H3O+ (acetic acid and water at RT)
this is mild acid hydrolysis
how to deprotect all acetals?
you can use acetic acid and water but must leave for reflux and long time
you can use H2SO4 and HCL (much stronger acids)
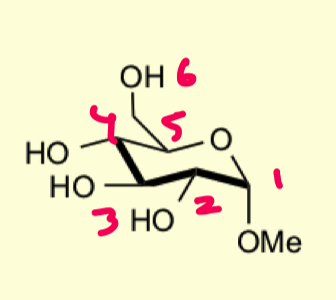
how to protect 1,3 diols to form benzylidene acetals?
use bulky aldehyde and lewis acid
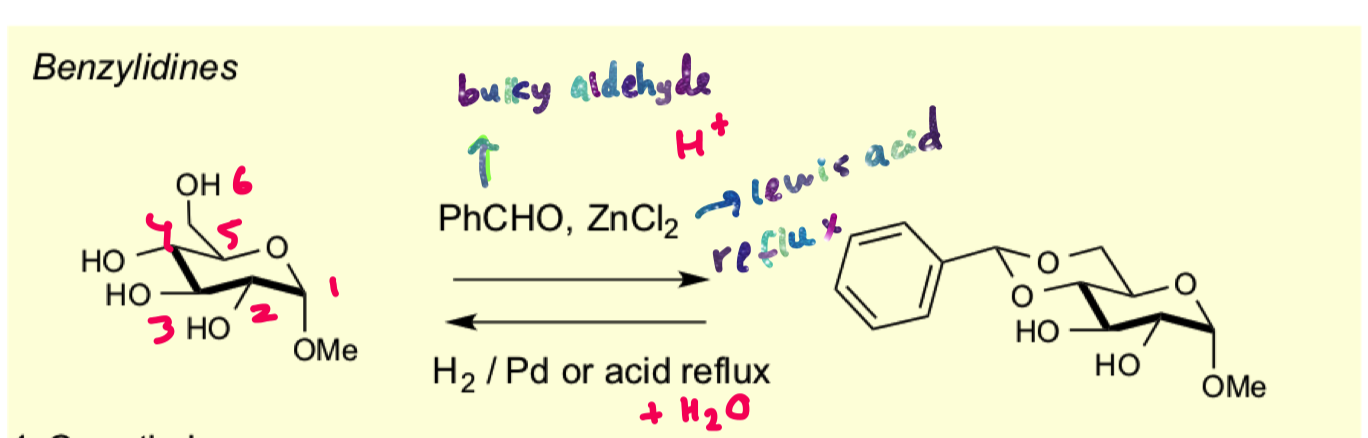
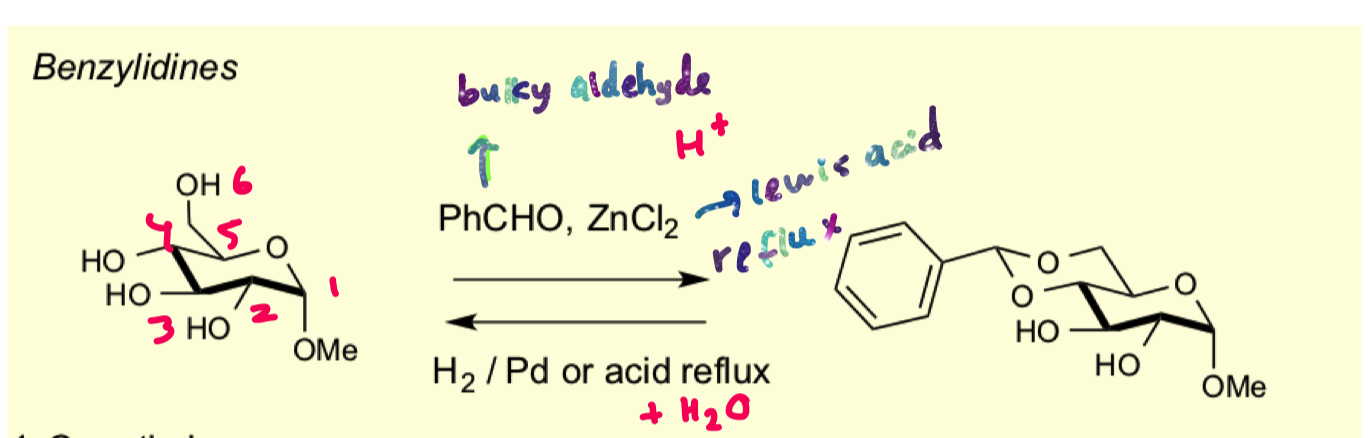
why does this reaction selectively protect 4,6
aldehydes prefer to form the transdecalin ring so that the phenyl group can be in the equatorial position

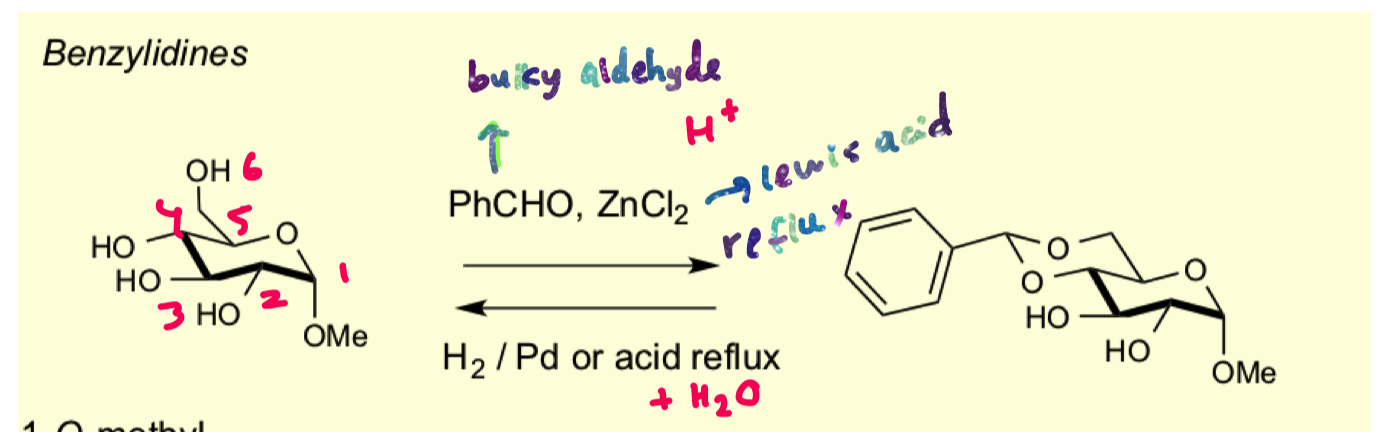
why do form the 6 membered ring and not the 5 membered ring for this reaction
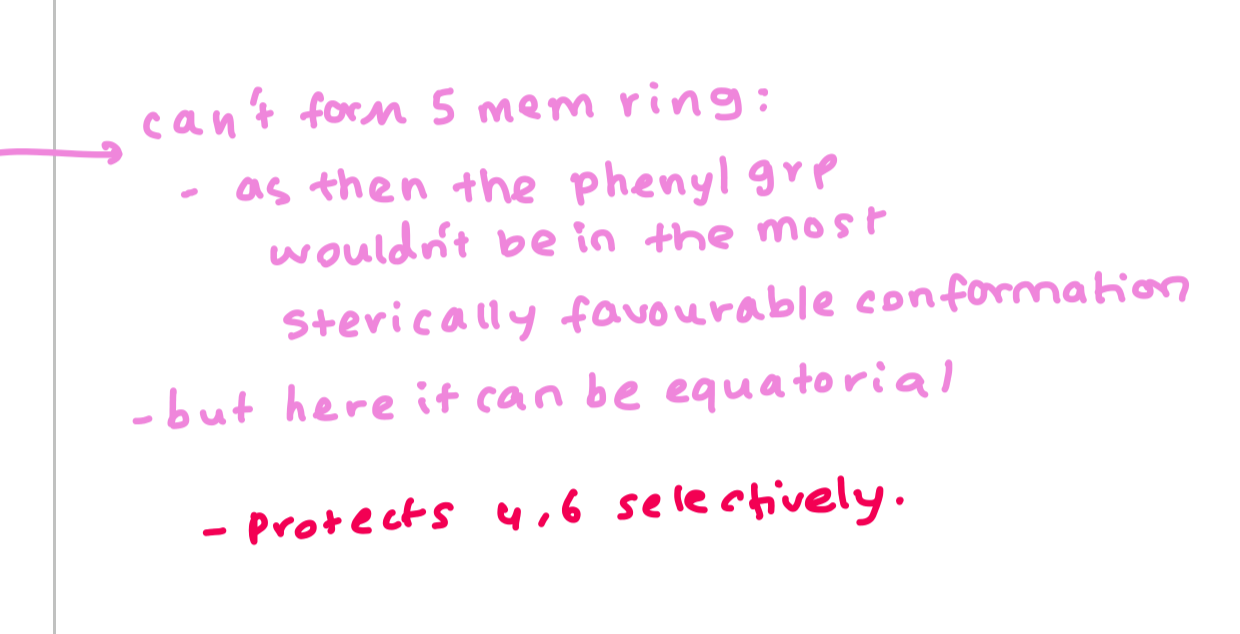
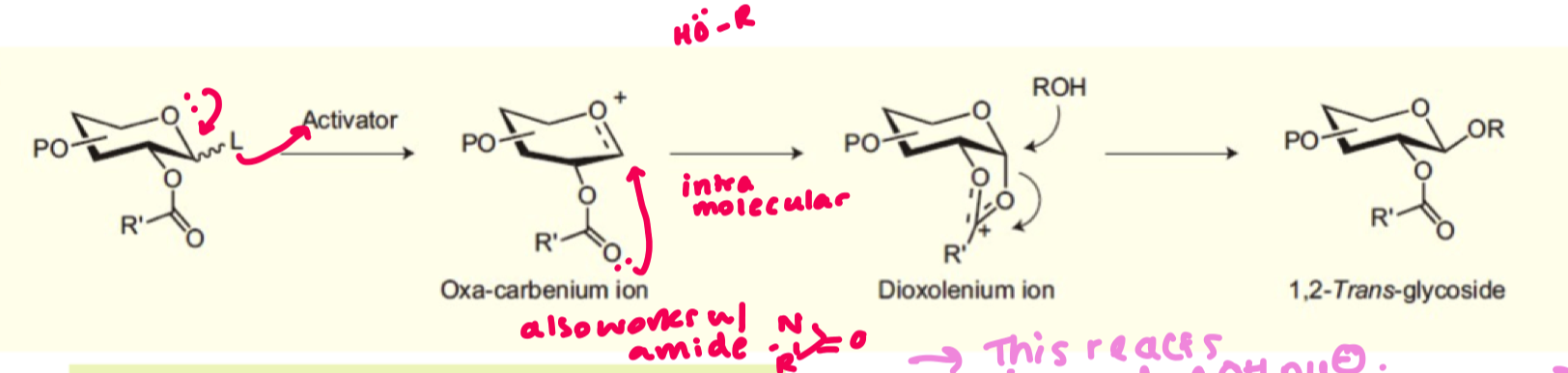
what is the general scheme for glycosylation reaction?
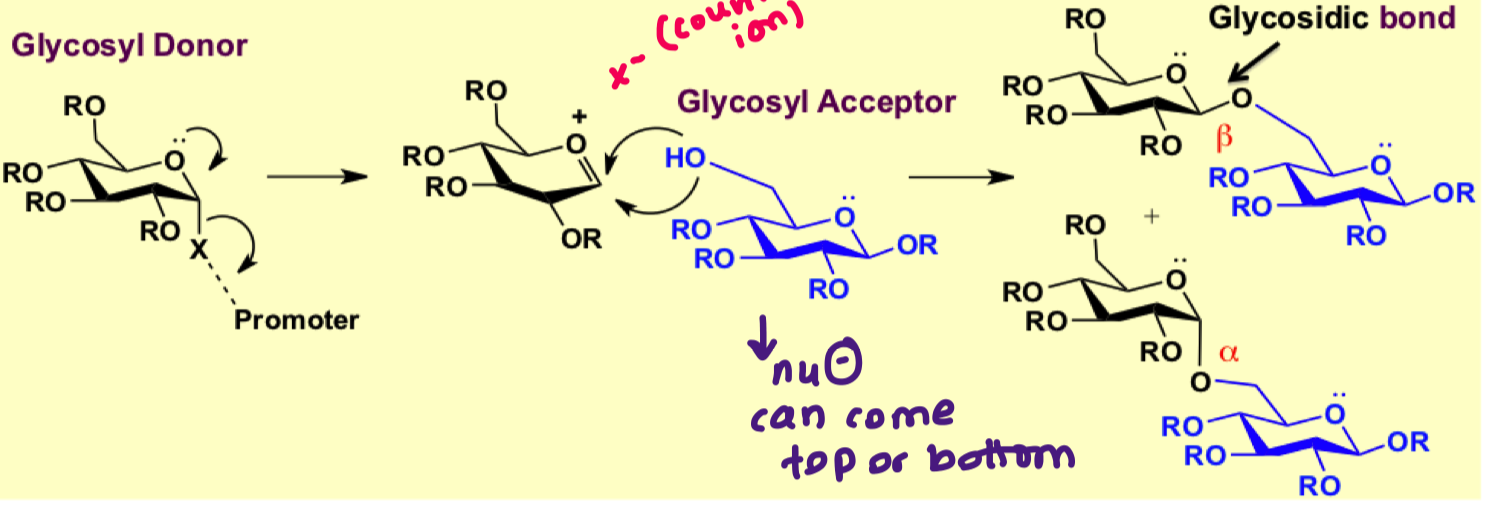
what is the role of a counter ion?
it helps stabilise the oxocarbenium ion
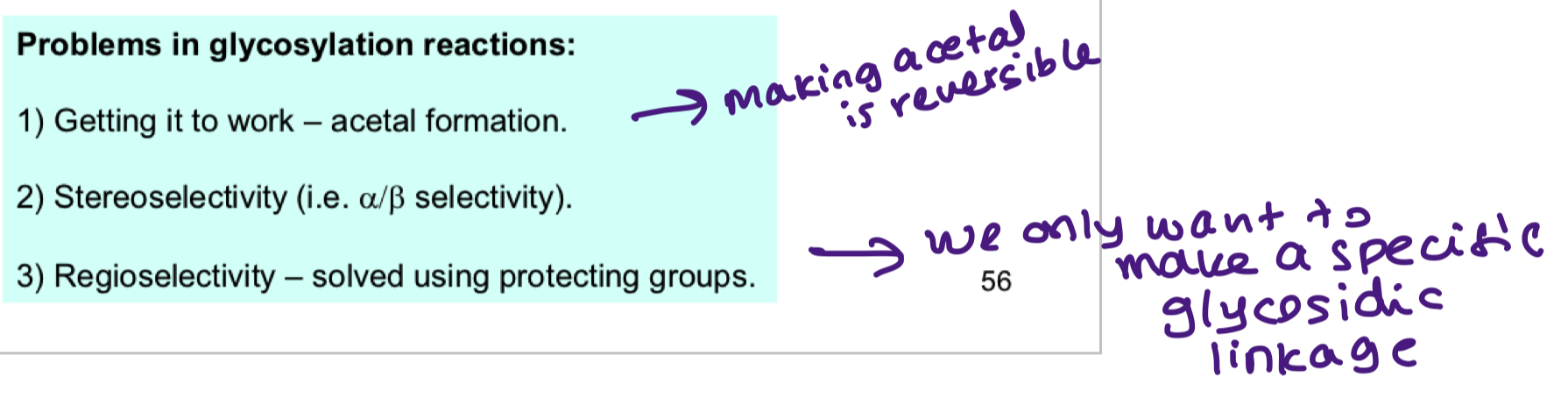
what are the problems associated with glycosylation reactions?
what properties should a good leaving group have?
easily introduced
stable when required
selectively activated
give examples of different leaving groups for glycoslyation

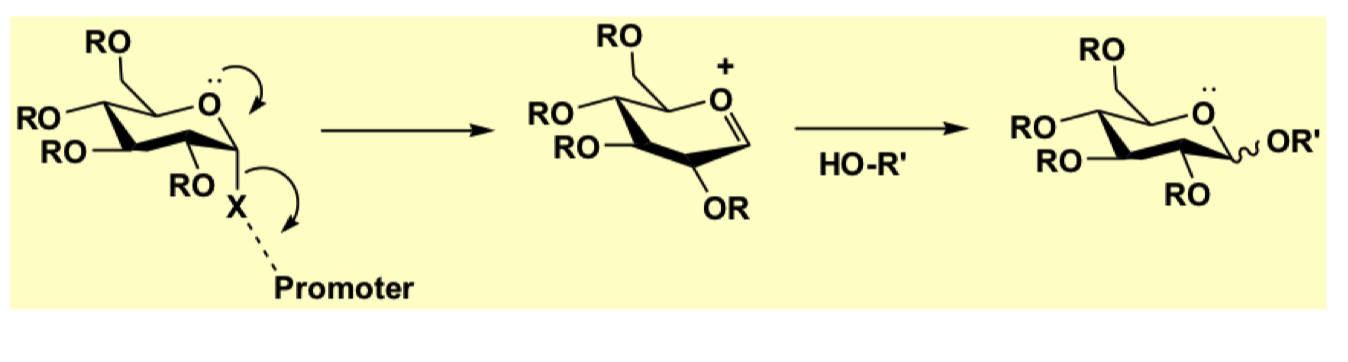
give the mechanism for glycosylation
why is it easier to make 1,2 trans glycosides compared to 1,2 cis ?
1,2 trans has groups further apart so easier for the nucleophile to come in and no sterics in the way
what is neighbouring group participation ?
groups which help to activate the leaving group
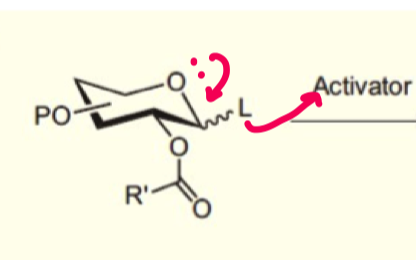
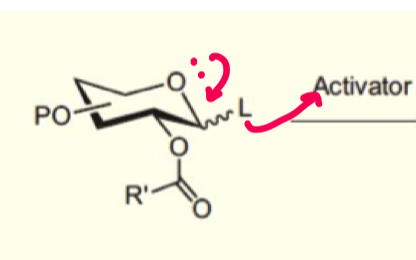
explain how we can form beta-glycosides using ester neighbouring group participation?
alcohol nucleophile is poor so ester can attack intramolecularly instead at the anomeric carbon.
this form the 1,2 trans ring as it is favoured (eq/ax) - stabilised carbocation
this allows the nucleophile to attack and as axial position is occupied, equatorial formation is very favoured
the only exception is if the ester group is axial first
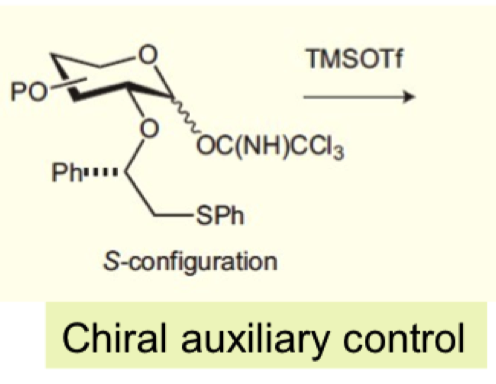
explain how we can form alpha glycosides using chiral auxillary control?
SPh is very nucleophilic so is able to attack the anomeric carbon and forms the transdecalin chair
this blocks the equatorial face with a bulky group allowing for alpha product
Ph must be starting from S configuration
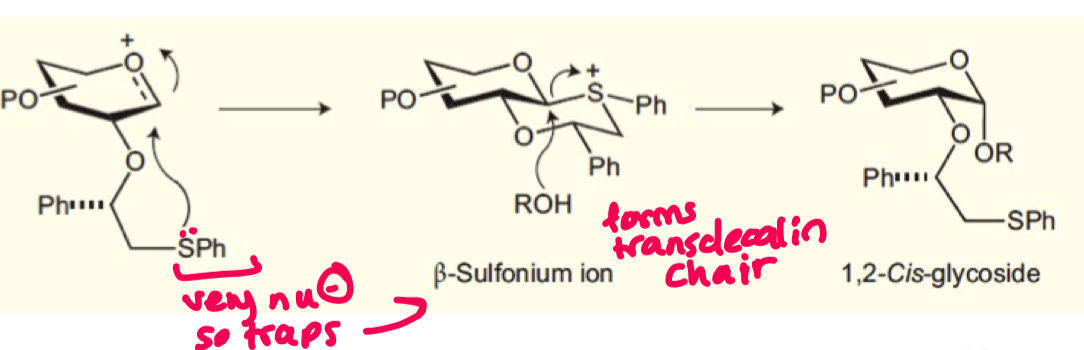
how does this favour beta glycosides?
if Ph group is (R) then cisdecalin would form
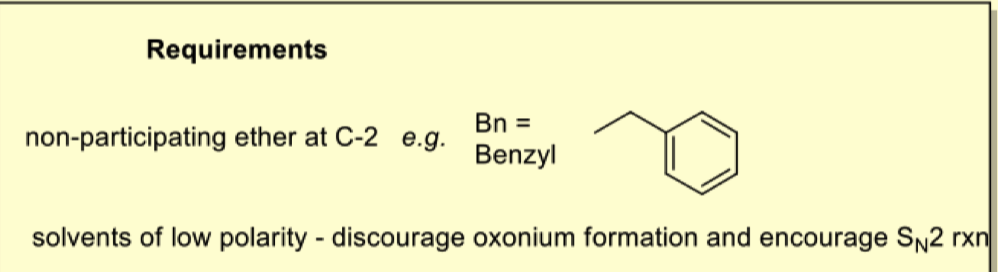
what is in-situ anomerisation?
another way of making alpha glycosides (1,2 cis)
what are the requirements for in-situ anomerisation
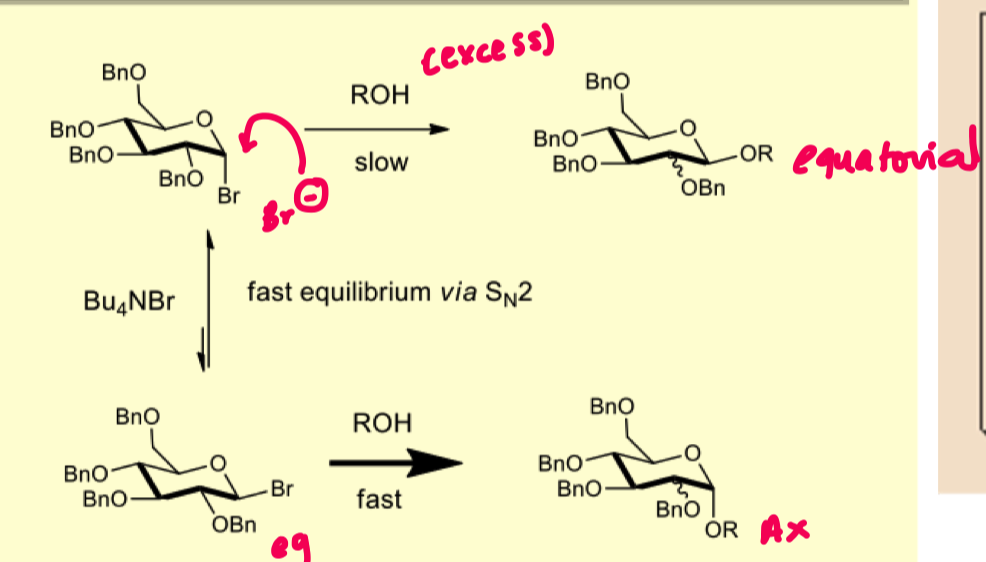
describe how in-situ anomerisation works
allows for the formation of equilibria between alpha and beta monomers
beta anomer is more reactive as due to the anomeric effect the equatorial bromide must flip to form the boat conformation for stability.
the boat conformation isnt stable compared to the chair which allows it to react faster with the nucleophile (R-OH) forming more of the alpha glycoside
when can we use solvent effects for alpha and beta selectivity?
when we are limited by the number of protecting groups we can use
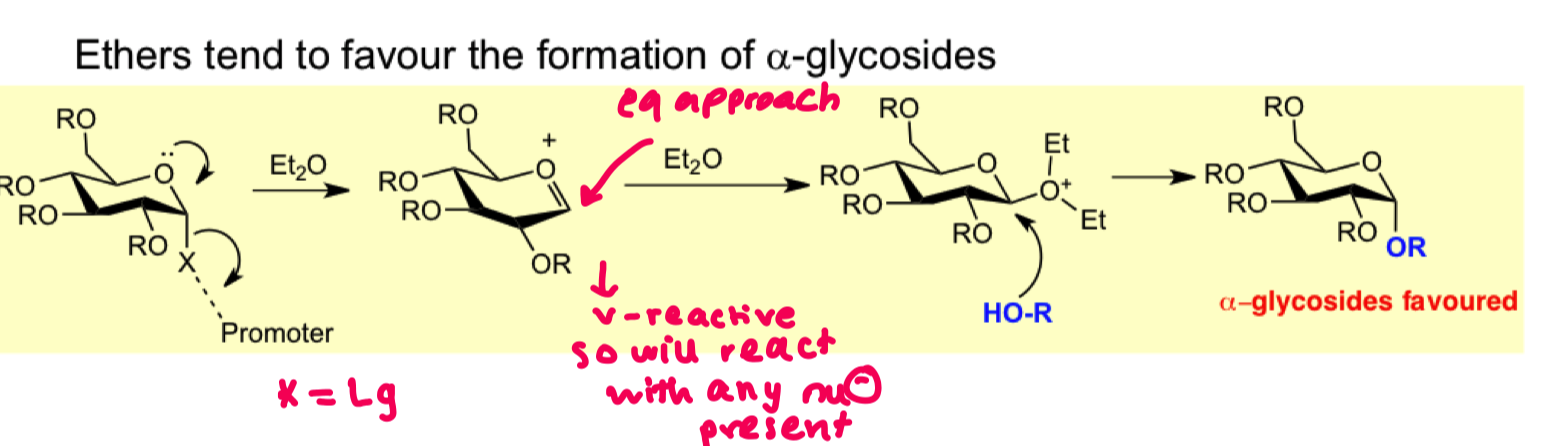
How can we use solvent effects to favour formation of alpha glycosides? draw the mechanism
-ethers
but we get a mixture not pure selectivity
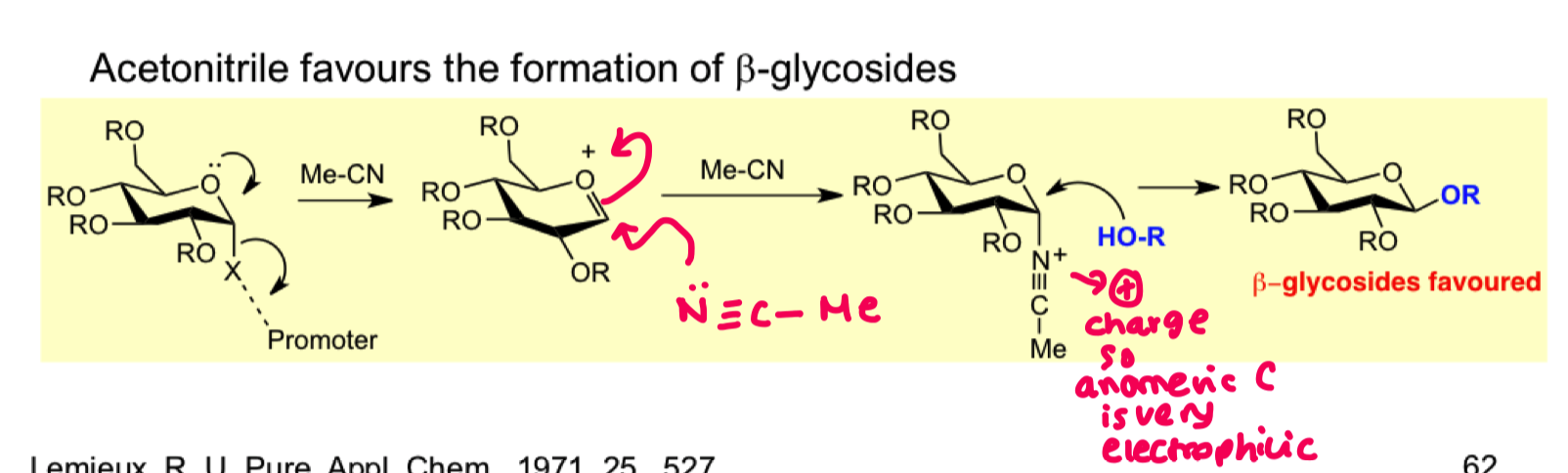
How can we use solvent effects to favour beta formation? draw the mechanism
acetonitrile
which leaving group is better and why? OAc or Br?
Br as it is more electrowithdrawing
how to form an activated glycoside using glycosyl halides on OAc? draw the mechanism
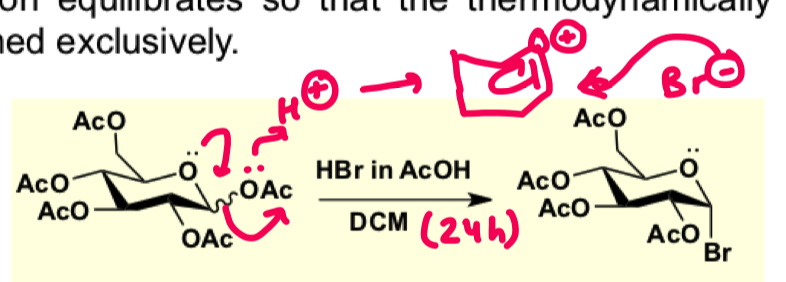
why does glycosyl halide form the axial glycoside?
In the presence of alot of acid you get the thermodynamic product due to the anomeric effect
if u quench the reaction after 5 minutes then you get a mixture of alpha and beta due to neighbouring group participation
how to form an activated glycoside using glycosyl halides on OBn? draw the mechanism
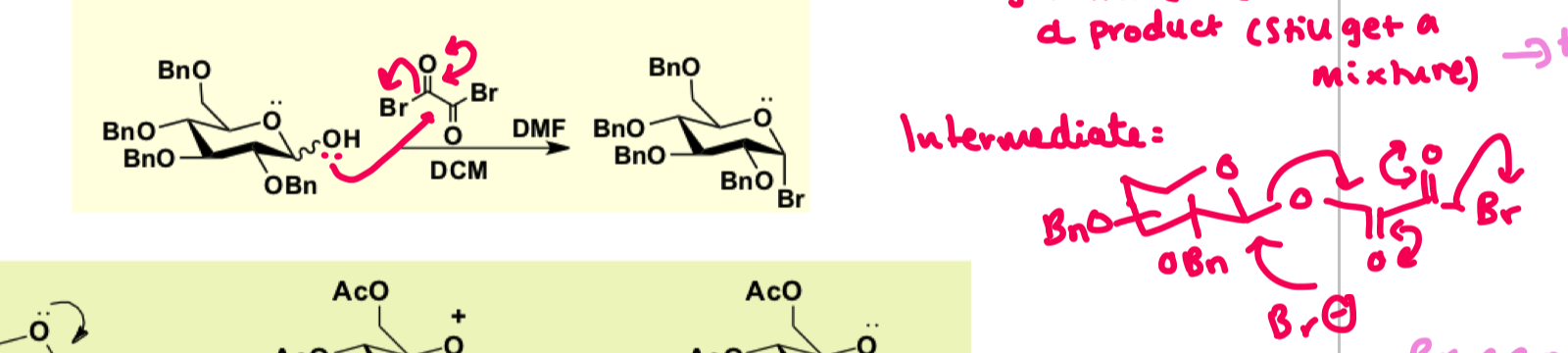
why do we have to put the Br group on last?
once Br group is attached than you cant modify the rest of the OAc groups as Br is too reactive of a leaving group therefore we must put on other protecting groups first.
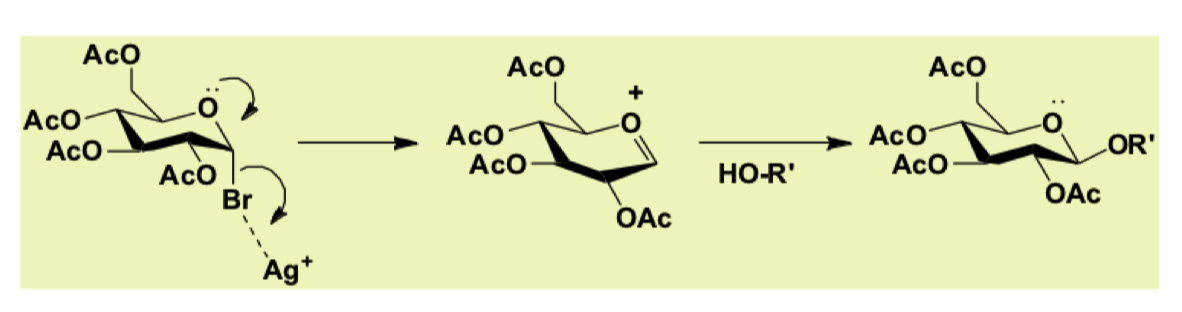
How can we activate the Br leaving group?
using silver salts

draw mechanism for activating Br
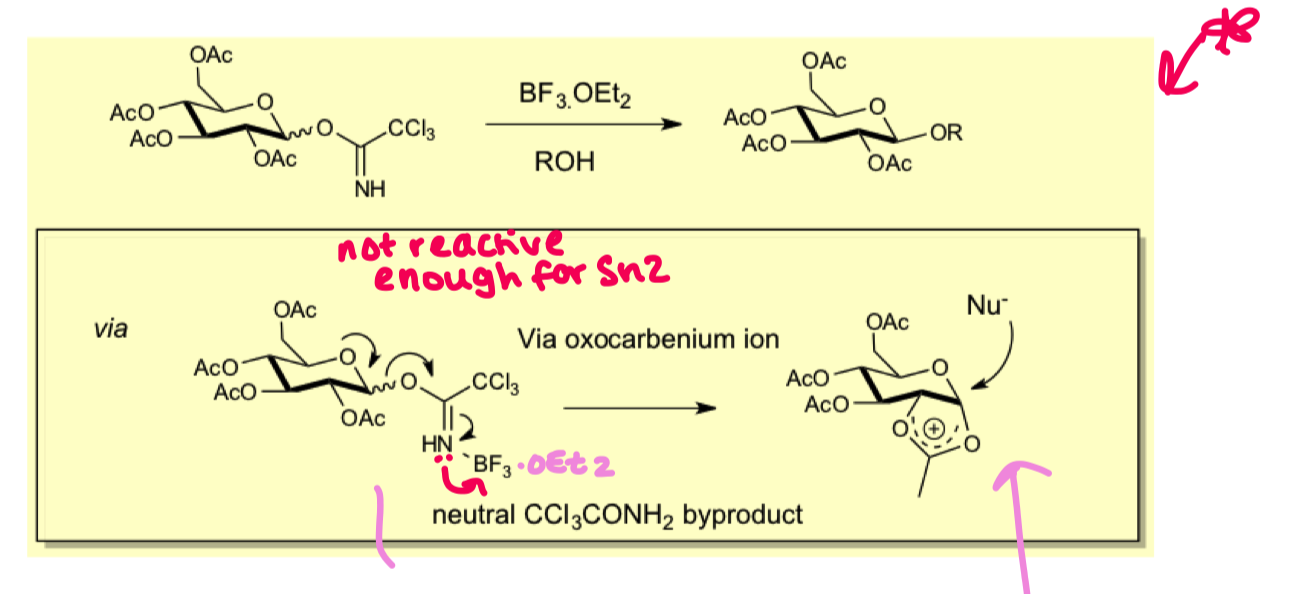
How to activate glycosides with trichloroacetimidates?
uses base (depending on this we can form alpha or beta)
then uses lewis acid

draw the mechanism of lewis acid BF3.OET2 to form the beta glycoside
forms via sn1 reaction, although some strong nucleophiles can react via sn2 reaction
forms via oxocarbenium ion (on the slide)
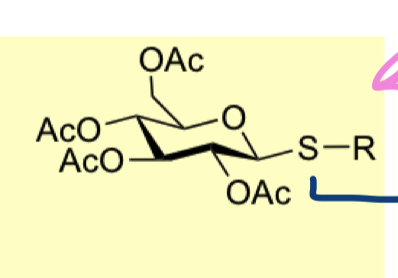
what are pros of using thioglycosides
able to generate mixed acetals which are stable to acid and base so we can do protecting group manipulations
draw the activation mechanism for thioglycosides
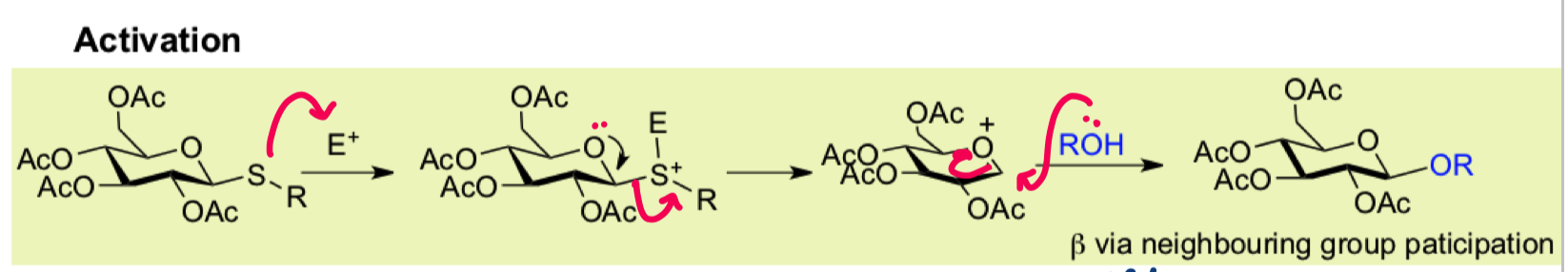
draw the promoter source I+ from NIS/TfOH
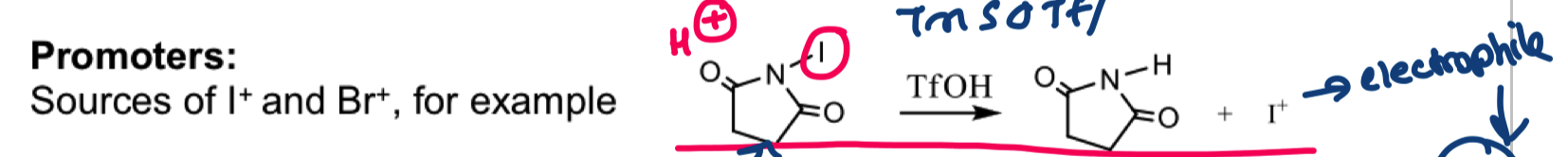
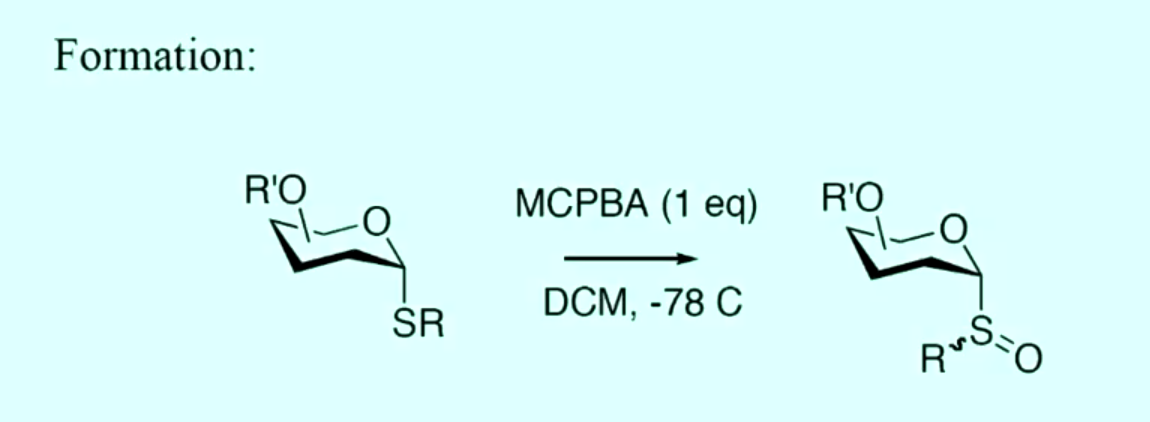
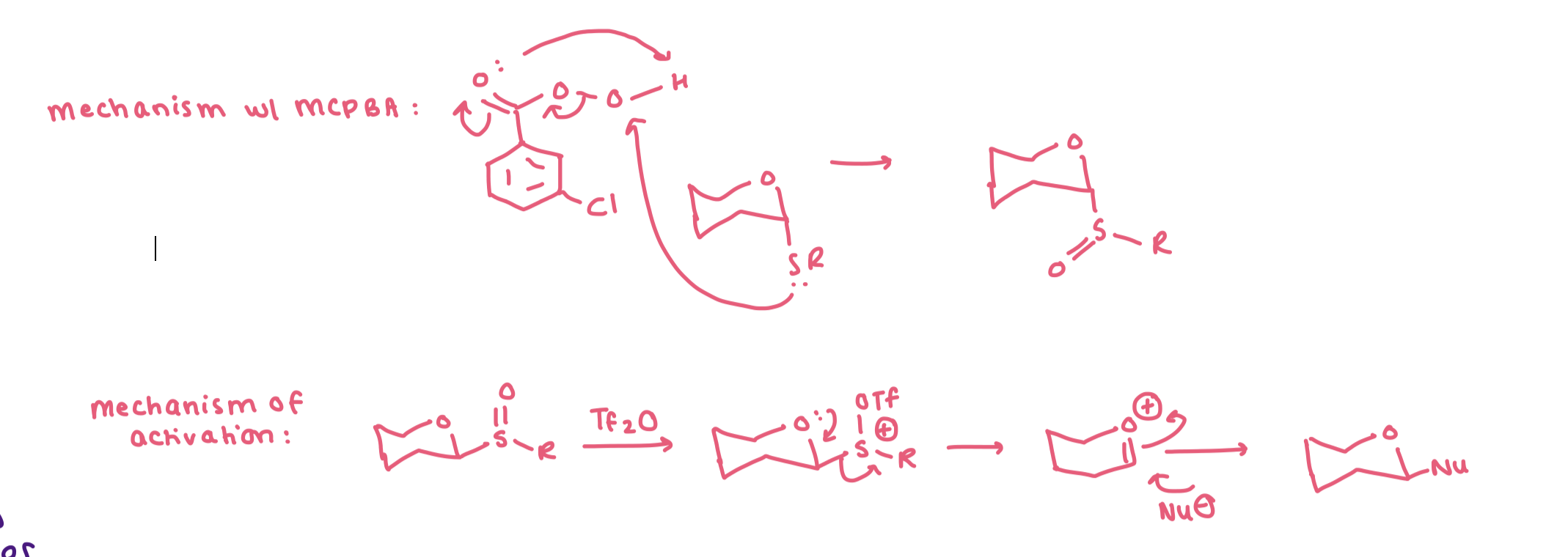
describe the mechanism for the formation of glycosyl sulfoxides with mcpba and the mechanism of activation with Tf2O
why is formation of glycosyl sulfoxides formed at -78 degrees?
requires low temps as sulfoxides are very reactive
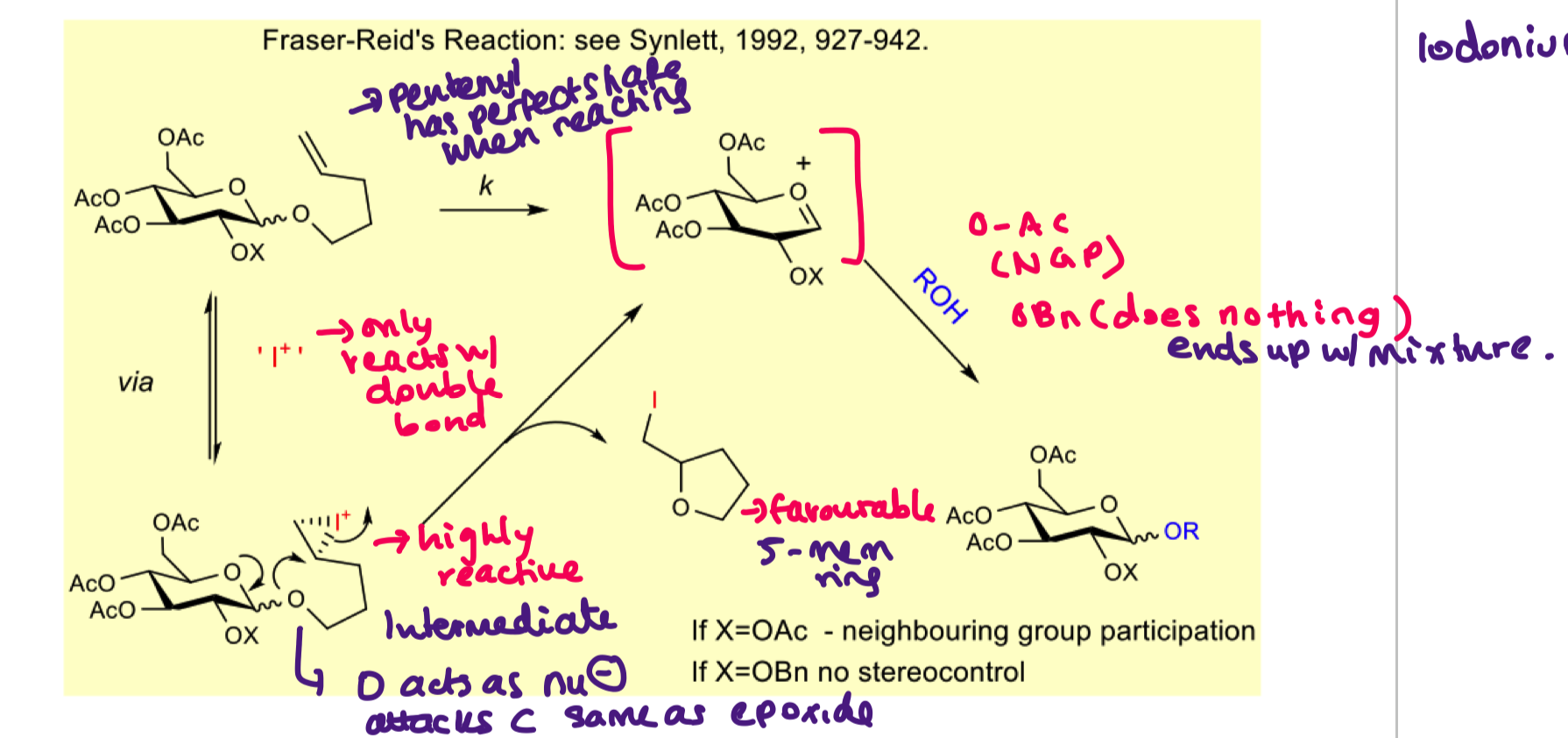
how can we use pentenyl glycosides for activated glycoside
use activation to form I+
I+ reacts with double bonds only (nothing else in the molecule will react)
draw the mechanism of pentenyl glycosides
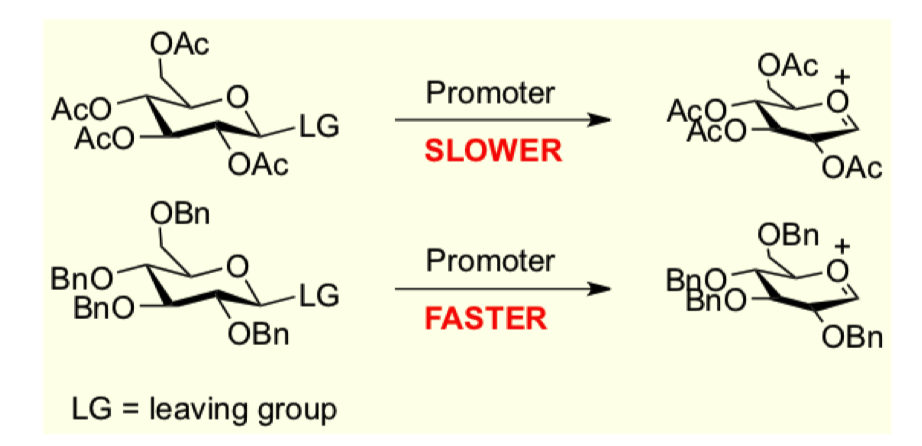
what does armed and disarmed glycosides describe?

what are disarmed glycosides?

what are armed glycosides?

why is the lower reactions faster?
has EDG
EWG pulls electron density away so slower
between BnO and BzO which forms faster reaction?
BnO (benzyl ether) as its EDG