Chapter 6i & 6ii - Organic chemistry (intro,alkanes,alkenes)
1/104
Earn XP
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
105 Terms
What is organic chemistry?
The chemistry of carbon compounds
How many bonds can carbon form?
Carbon can form 4 strong covalent bonds with itself
What is a hydrocarbon?
Molecules that contain only hydrogen and carbon
What is an unsaturated hydrocarbon?
A hydrocarbon that contains at least one double carbon-carbon bond
What is a saturated hydrocarbon?
A hydrocarbon that contains only single carbon-carbon bonds
What is a molecular formula?
Shows the actual number of atoms of each element in a molecule.
What is an empirical formula?
Shows the simplest whole number ratio of atoms in a compound.
What is a general formula?
Represents a homologous series using an algebraic expression.
What is a homologous series?
A homologous series is a group of organic compounds that have the same functional group, the same general formula and the same chemical properties
What is a structural formula?
Shows how atoms are arranged in a molecule without drawing bonds
What is a displayed formula?
Shows all bonds and atoms.
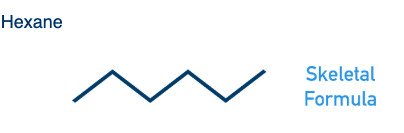
What is a skeletal formula?
Simplified representation where carbon atoms are at the ends and bends of lines, and hydrogens bonded to carbon atoms aren’t shown.
Write the molecular formula of hexane?
C6H14
What is the empirical formula of hexane?
C3H7
What is the general formula of hexane?
CnH2n+2
What is structural formula of hexane?
CH3CH2CH2CH2CH2CH3
What is the displayed formula of hexane?
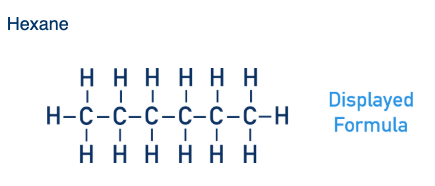
What is the skeletal formula of hexane?
What is a functional group?
A functional group is an atom or group of atoms within a molecule that gives the molecule its reactivity and can determine its physical and chemical properties. (e.g. the double C=C bond in alkanes)
What is the IUPAC naming system?
This is the International Union of Pure and Applied Chemistry system for naming organic compounds. It is an international language for chemistry that can name any organic compound.
What is nomenclature?
The naming of organic compounds
What are the prefixes for the first 10 organic compounds?
Number of C atoms | Molecular formula of straight-chain alkane | Name of alkane | Stem used in naming |
|---|---|---|---|
1 | CH4 | methane | meth- |
2 | C2H6 | ethane | eth- |
3 | C3H8 | propane | prop- |
4 | C4H10 | butane | but- |
5 | C5H12 | pentane | pent- |
6 | C6H14 | hexane | hex- |
7 | C7H16 | heptane | hept- |
8 | C8H18 | octane | oct- |
9 | C9H20 | nonane | non- |
10 | C10H22 | decane | dec- |
What are the steps in naming organic compounds?
Find the longest carbon chain (gives you the stem)
Identify the main functional group (determines the suffix or prefix).
Number the carbon chain to give the lowest possible numbers to functional groups. (if theres more than one longest chain, pick the one with the most side chains)
Any side-chains or less important functional groups are added as prefixes at the start of the name. Put them in alphabetical order, after the number of the carbon atom each is attached to.
If there's more than one identical side-chain or functional group, use di- (2), tri- (3) or tetra- (4) before that part of the name — but ignore this when working out the alphabetical order.
Use commas between numbers and hyphens between letters and numbers.
What are the prefixes and suffixes of the common functional groups?
Ester - alkyl- -oate
amine - alkyl- -amine
Nitrile - -nitrile
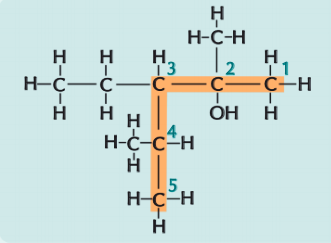
How would you name this?
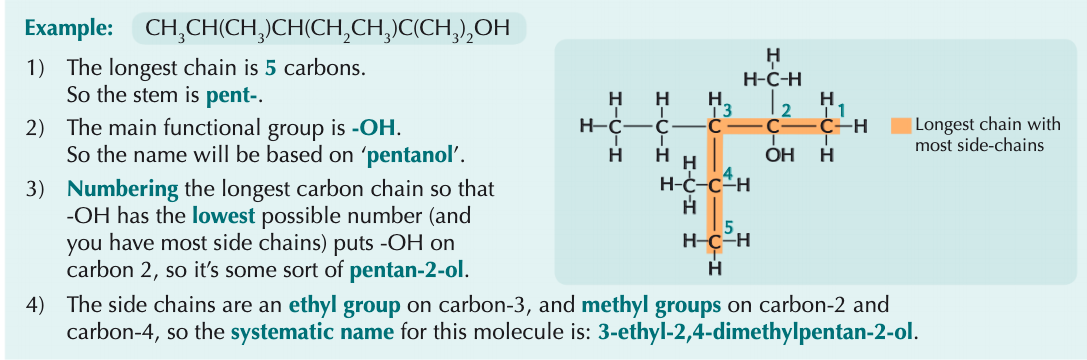
What is an addition reaction?
An addition reaction is an organic reaction in which two (or more) molecules combine to give a single product with no other products
What is a polymerisation reaction?
Where small monomers join to make a long-chain polymer
What is a substitution reaction?
A substitution reaction is a reaction that involves replacing an atom or group of atoms by another
What is an elimination reaction?
An elimination reaction is a reaction in which a small molecule (such as H2O or HCl) is removed from an organic molecule
What is a hydrolysis reaction?
A hydrolysis reaction is a reaction in which a compound is broken down by water (it can also refer to the breakdown of a substance by dilute acids or alkali)
What is a condensation reaction?
A condensation reaction is a reaction in which two organic molecules join together and in the process eliminate small molecules (such as H2O or HCl)
What is an oxidation reaction?
The gain of oxygen or loss of hydrogen
What is a reduction reaction?
The gain of hydrogen or loss of oxygen
What are nucleophiles?
Electron pair donors.
They are often negatively charged or contain lone pairs. They are electron rich so are attracted to places that are electron poor. They are attracted to positive ions and delta positive charges
What are electrophiles?
Electron pair acceptors.
They are often positively charged. They are attracted to electron-rich areas such as a double bond, negative ions and atoms with lone pairs.
What are free radicals?
They have an unpaired electron and this makes them very reactive.
What are isomers?
They have the same molecular formula but a different arrangement of atoms. There are two types (structural isomers and stereoisomers)
What is a structural isomer
They are compounds that have the same molecular formula but different structural formulae. e.g. propene and cyclopropane
What are the three types of structural isomer?
Chain
Positional
Functional group
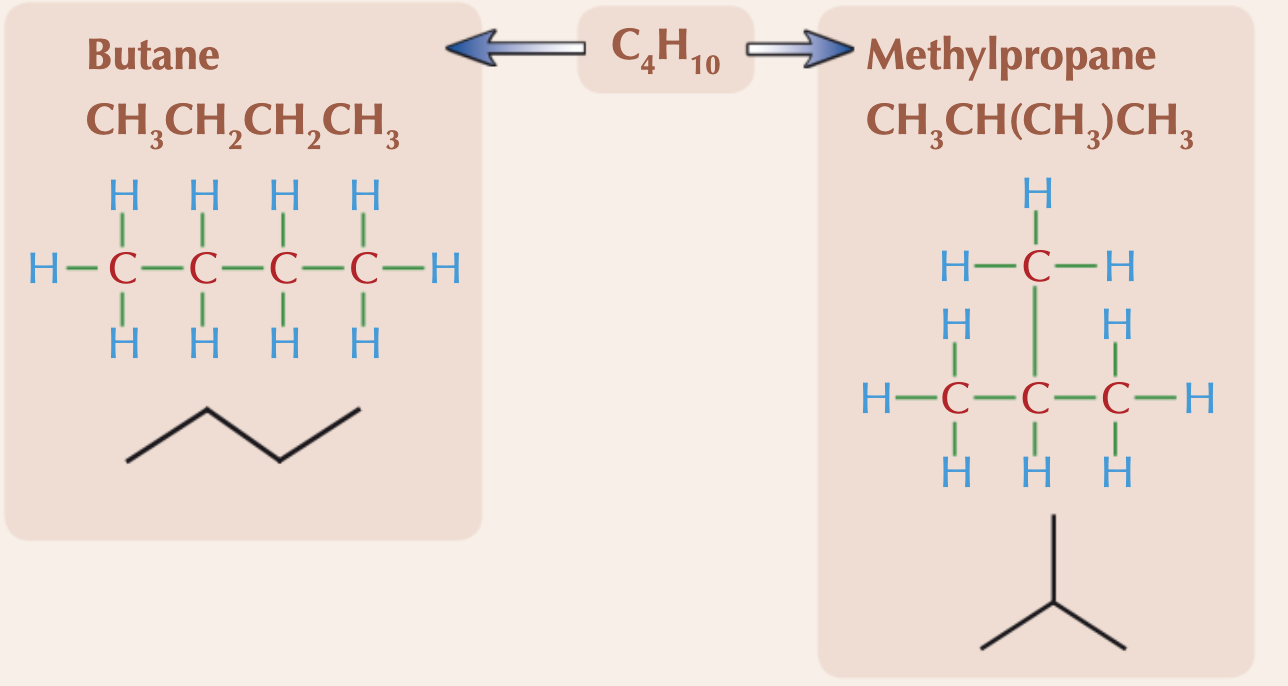
What is a chain isomer?
When compounds have the same molecular formula, but their longest hydrocarbon chain is not the same. This is due to branching. These will have similar chemical properties but their physical properties (e.g. melting points) will be different because of the different shapes.
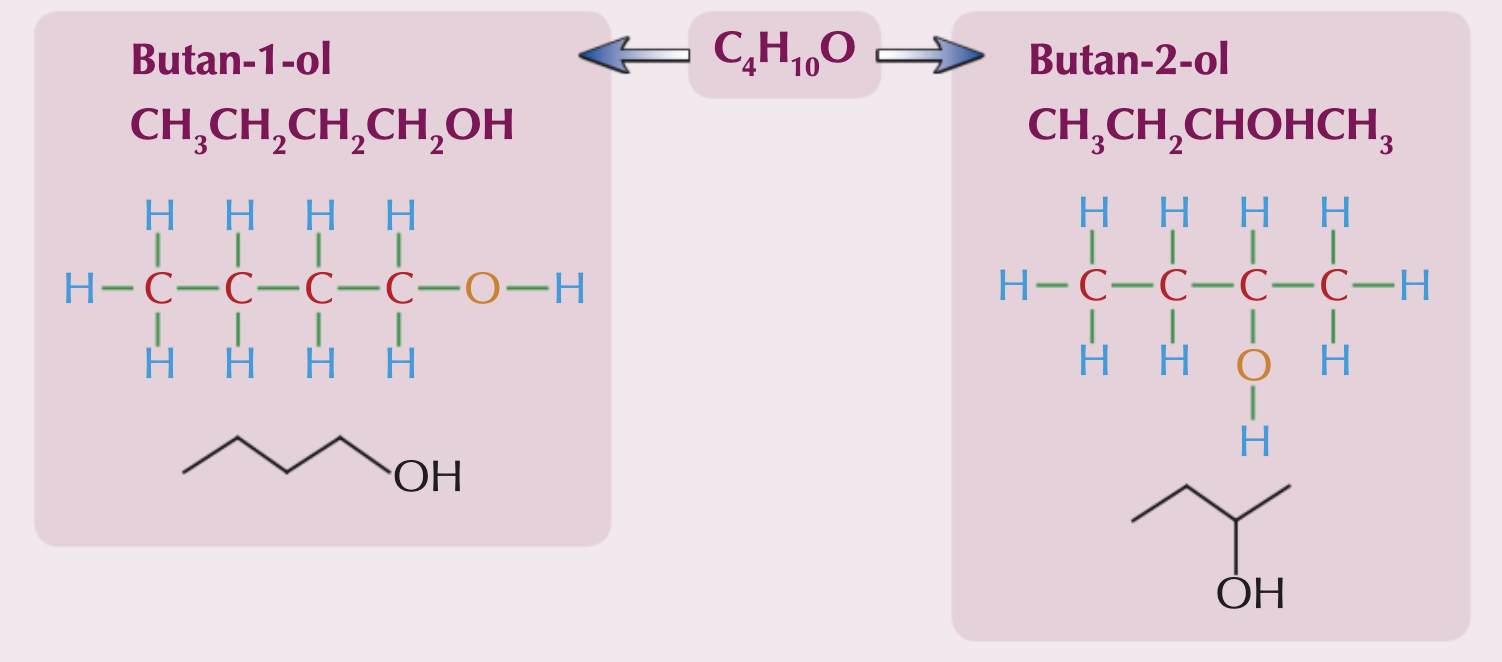
What is a positional isomer?
Positional isomers arise from differences in the position of a functional group in each isomer. The functional group can be located on different carbons. Their physical properties would be different and their chemical properties can be too.
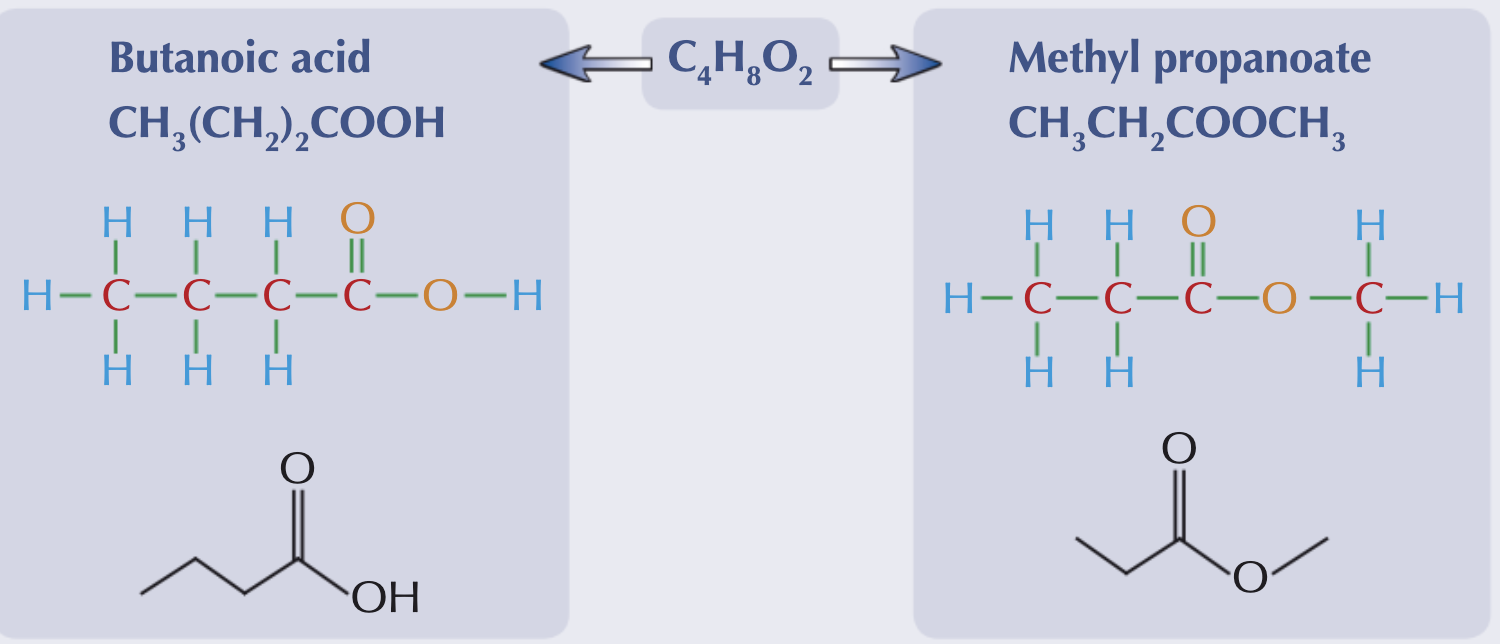
What is a functional group isomer?
Where the same atoms are arranged into different functional groups. These will have very different physical and chemical properties.
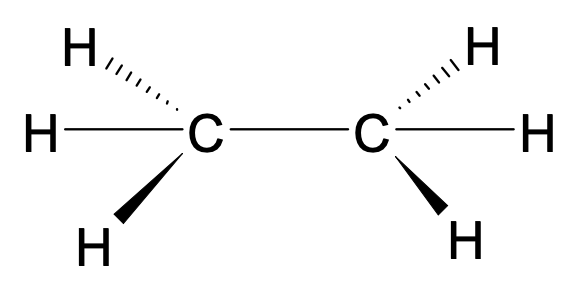
What is the bonding angle and shape for carbon atoms in a saturated hydrocarbon?
Tetrahedral 109.5o
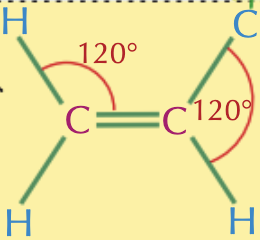
What is the bonding shape and angle around a carbon atom in an unsaturated hydrocarbon?
Trigonal planar 120o
Can the carbon atoms in a double bond rotate?
NO!
What are stereoisomers?
Compounds that have the same structural formula but a different arrangement in space. Alkenes have stereoisomers and this is due to the lack of rotation around the double bond.
What is the difference between E and Z isomers?
Z- isomers have the same/ the same priority groups on the same side of the double bond (top or bottom)
E-isomers have the same / the same priority groups on different sides of the double bond
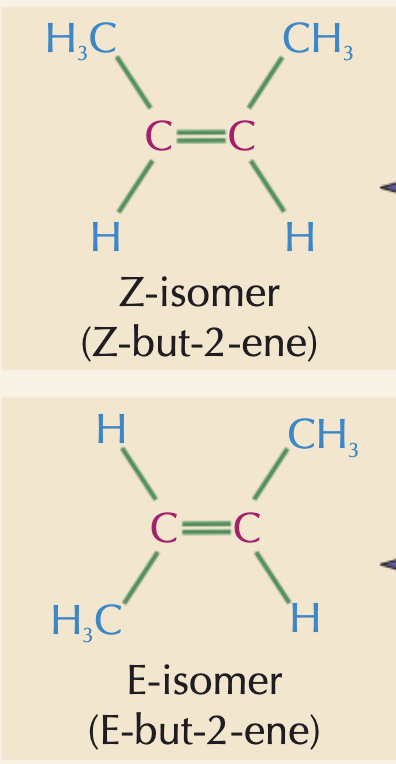
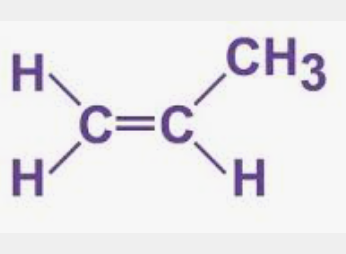
Can there be stereoisomers of propene?
No, as each carbon isn’t bonded to 2 different groups
When can you have E/Z isomerism?
There is restricted rotation around the C=C double bond.
There are two different groups/atoms attached both ends of the double bond
How can you find the type of E/Z isomer when the groups attached to the carbon are all different?
Step 1: Look at the atoms directly attached to the C=C bond.
Step 2: Assign higher priority to the atom with the higher atomic number. If the highest priority atoms bonded to each carbon are the same (for example C) you then assign the highest priority of the groups bonded to that atom (for example CH2CH3 group has higher priority than CH3 group).
Step 3:
If the higher priority groups are on the same side, it is Z.
If the higher priority groups are on opposite sides, it is E.
When can you use the cis/trans naming system as well as the E/Z system?
When there is at least one group in common with the carbons.

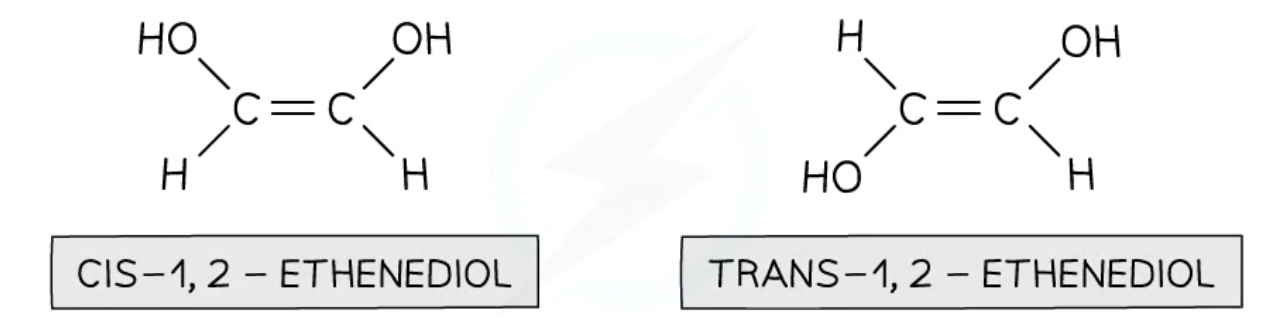
What is “cis” used to describe?
Where the same group is on the same side of the double bond.
What is ‘trans’ used to describe?
Where the same group is on different sides of the double bond.

Which of these is cis and which is trans?
What are alkanes?
Saturated hydrocarbons
What is the general formula of alkanes?
CnH2n+2
Are alkanes polar?
No. They are also insoluble in water.
What are cycloalkanes?
Cycloalkanes are also saturated hydrocarbons but have ring structures. Their general formula is CnH2n.
How do we obtain alkanes?
Alkanes are obtained from the fractional distillation and cracking of crude oil
What is crude oil?
Crude oil is a mixture of hydrocarbons containing alkanes, cycloalkanes and arenes (compounds with a benzene ring). It is extracted from the earth in a drilling process and transported to an oil refinery where it is separated into useful fuels by fractional distillation.
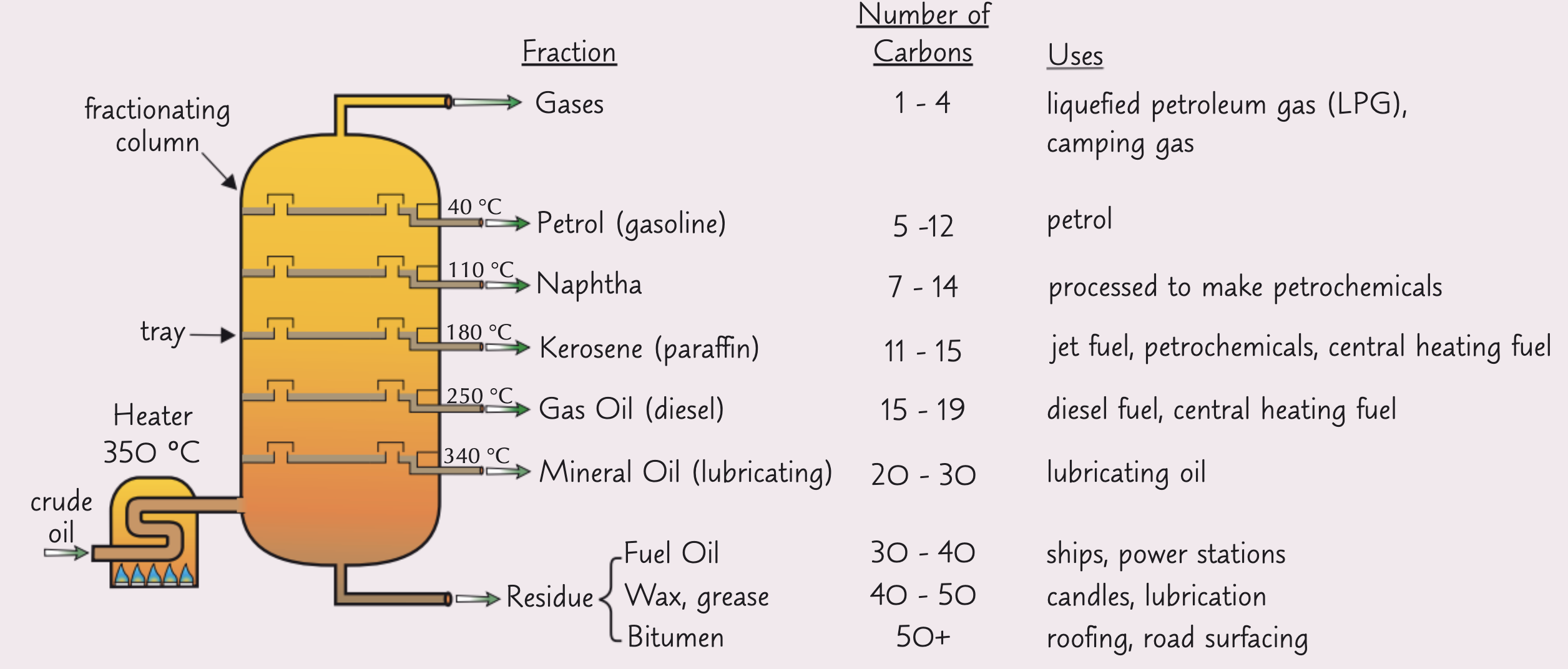
How does fractional distillation work?
First, the crude oil is vaporised at about 350 °C.
The vaporised crude oil goes into a fractionating column and rises up through the trays. The largest hydrocarbons don't vaporise at all, because their boiling points are too high - they just run to the bottom and form a gooey residue.
As the crude oil vapour goes up the fractionating column, it gets cooler. Because the alkane molecules have different chain lengths, they have different boiling points, so each fraction condenses at a different temperature. The fractions are drawn off at different levels in the column.
The hydrocarbons with the lowest boiling points don't condense. They're drawn off as gases at the top of the column.
Why is cracking useful?
The lighter fractions (short chain alkenes and alkanes) are more in demand as they make better fuels and can easily be used to form plastics
What is cracking?
Cracking us breaking long-chain alkanes into smaller hydrocarbons
What are the two types of cracking?
thermal
catalytic
How does thermal cracking work?
It requires high temperatures (up to 1000 oC) and high pressure (up to 70 atmospheres) and produces alkanes and a lot of alkenes. The alkenes are good as they can be used to form polymers
How does catalytic cracking work?
Catalytic cracking uses a zeolite catalyst (hydrated aluminosilicate) or aluminium oxide, at a slight pressure and high temperature (about 450 °C).
It mostly produces aromatic hydrocarbons and motor fuels.
Using a catalyst cuts costs, because the reaction can be done at a low pressure and a lower temperature. The catalyst also speeds up the reaction, saving time.
Aromatic compounds contain benzene rings. Benzene rings contain a ring of 6 carbon atoms with delocalised ring of electrons
What is reforming and why is it useful?
Most vehicles run on petrol which is contains a mixture of alkanes and other things. Some of these alkanes are straight chained and these are more likely to have issues with knocking (where alkanes explode rather than combust. Adding branched and cyclic hydrocarbons means knocking is less likely so makes the fuel more efficient. Reforming often uses a platinum catalyst.
Reforming is described as the processing of straight-chain hydrocarbons into branched-chain alkanes and cyclic hydrocarbons for efficient combustion.
Why are alkanes useful as fuels?
Alkanes make good fuels as they readily undergo combustion, releasing heat energy.
What is complete and incomplete combustion?
Complete combustion occurs when there is enough oxygen present and carbon can be fully oxidised, forming carbon dioxide as a product (and water)
e.g. methane —> CH4 + 2O2 → CO2 + 2H2O
Incomplete combustion occurs when there is limited oxygen present and carbon can’t be fully oxidised, meaning carbon monoxide (CO) or carbon (soot) gets formed as a product (and water).
e.g. CH4 + O2 → C + 2H2O OR CH4 + 1.5O2 → CO + 2H2O
Why is burning fossil fuels bad?
Although they do produce lots of energy they also produce very harmful waste products including toxic gases such as carbon monoxide (CO), oxides of nitrogen (NO/NO2) and volatile organic compounds (VOCs)
What problems can carbon monoxide cause?
CO is a toxic and odourless gas
It can bind better to haemoglobin than oxygen so oxygen cannot bind to it so less oxygen is transported around the body
can cause dizziness, loss of consciousness and eventually death
What problems can nitrogen oxides cause?
Oxides of nitrogen (NOx) are produced when the high pressure and temperature in a car engine cause the nitrogen and oxygen in the air to react together. When oxides of nitrogen (NO,) escape into the atmosphere, they dissolves in moisture and are converted into nitric acid, which can fall as acid rain.
What problems can sulfur dioxide cause?
Acid rain can be caused by burning fossil fuels that contain sulfur. The sulfur burns to produce sulfur dioxide gas which then enters the atmosphere, dissolves in the moisture, and is converted into sulfuric acid.
What problems can carbon particulates cause?
Solid carbon particles (or particulates) released from incomplete combustion clump together to form soot which gradually falls back to the ground
Particulates can cause several problems:
If they are inhaled they can damage the lungs and cause respiratory problems
They can cover buildings and statues, making them look unclean and accelerating corrosion
They can reflect sunlight back into space reducing the amount of light reaching the earth, this is called global dimming
What do catalytic converters do?
Catalytic converters sit quietly in a car exhaust and stop some pollutants from coming out.
Without catalytic converters, cars spew out lots of bad stuff, like carbon monoxide,
oxides of nitrogen and unburnt hydrocarbons.
Catalytic converters get rid of theses pollutants by using a platinum catalyst to change them to harmless gases, like water vapour and nitrogen, or to less harmful ones like carbon dioxide.
e.g. nitrogen monoxide and carbon monoxide an be converted to nitrogen and carbon dioxide 2NO(g)+2CO(g) —> N2 (g) + 2CO2 (g)
What are the renewable alternatives to fossil fuels?
Biofuels are fuels made from living matter over a short period of time:
bioethanol is ethanol (an alcohol) made by the fermentation of sugar from crops such as maize,
biodiesel is made by refining renewable fats and oils such as vegetable oil,
biogas is produced by the breakdown of organic waste matter.
These fuels do produce CO, when they're burnt, but it's CO, that the plants absorbed while growing, so biofuels are usually still classed as carbon neutral.
Biodiesel and biogas can also be made from waste that would otherwise go to landfill.
Biofuel production could provide money for less developed countries as they have the space to grow the crops required
What are some negatives of Biofuels?
CO, is still given out while refining and transporting the fuel, as well as making the fertilisers and powering agricultural machinery used to grow and harvest the crops.
One problem with switching from fossil fuels to biofuels in transport is that petrol car engines would have to be modified to use fuels with high ethanol concentrations.
Also, the land used to grow crops for fuel can't be used to grow food — this could be a serious problem
Developed countries (like the UK) will create a huge demand as they try to find fossil fuel alternatives.
Poorer developing countries (in South America, say) could use this as a way of earning money, and convert farming land to produce 'crops for fuels'. This may mean they won't grow enough food to eat.
What are the two ways a covalent bond can break?
Homolytic — bond breaks evenly so both atoms get one unpaired electron. This leads to the formation of free radicals
Hetrerolytic — bond breaks unevenly so one atom receives both electrons. This forms two ions
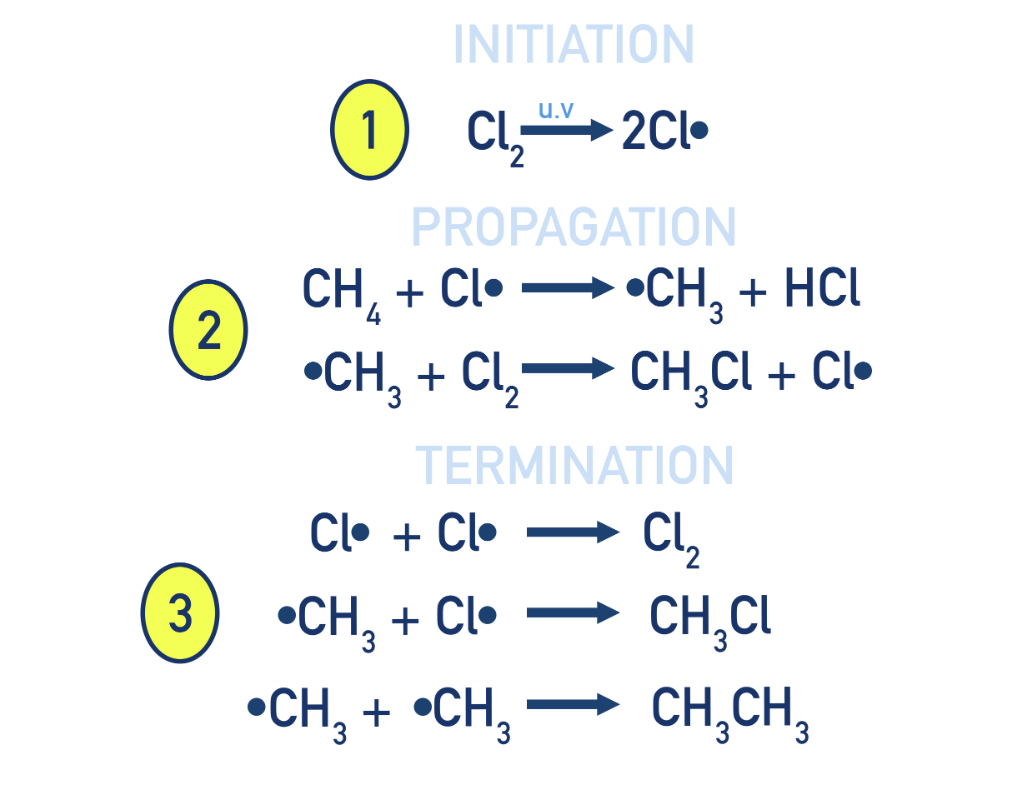
What are the stages of free radical substitution?
initiation: halogen bond homolyticly broken with UV light to form 2 free radicals
propagation: Radicals react to form new radicals in a chain reaction
termination: Radicals combine to form stable (non-radical) molecules, stopping the reaction.
There are several possible termination reactions
What are the limitations of free radical substitutions?
impurities
further substitution
Substitution of different carbon atoms
What are alkenes?
Hydrocarbons containing at least one C=C double bond. (unsaturated like cycloalkanes)
What is the general formula of alkenes?
CnH2n
What bonds make up a C=C double bond?
The C=C double bond is made up of a strong sigma (σ) bond and a weaker pi (π) bond.
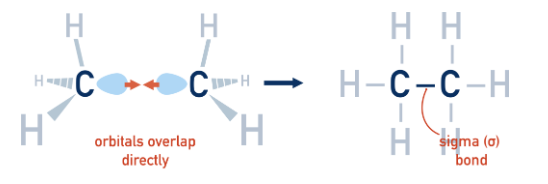
What is a sigma bond?
It forms a single bond. Sigma bonds are formed from the end to end overlap of atomic orbitals (s orbitals overlap this way as well as p orbitals). They allow free rotation around single C–C bonds (not across C=C).
What is pi bond?
Pi (π) bonds are formed from the sideways overlap of adjacent p orbitals
The two lobes that make up the π bond lie above and below the plane of the σ bond
This maximises overlap of the p orbitals
A single π bond is drawn as two electron clouds, one arising from each lobe of the p orbitals
The two clouds of electrons in a π bond represent one bond containing two electrons
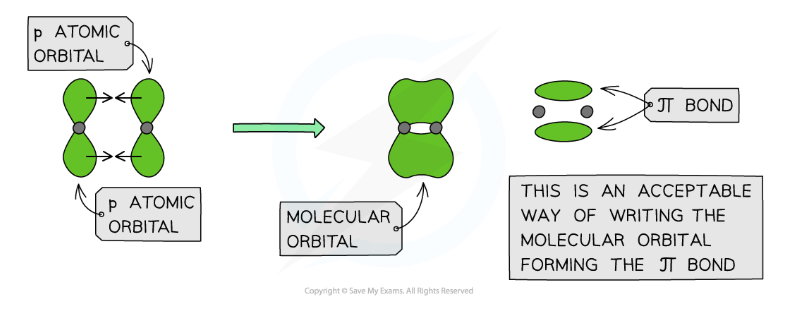
Is a pi bond weaker or stronger than a sigma bond?
Pi bonds are weaker than sigma bonds as they are exposed above and below the plane of the sigma bond and this means that the electrostatic attraction between the nuclei and the shared pair of electrons. They are therefore vulnerable to attack by electrophiles.
What is electrophilic addition?
An electrophilic addition is the addition of an electrophile to a double bond
What is hydrogenation?
Reaction with hydrogen (H2)
Alkene + H2 → alkane
Ni catalyst, ~150 °C.
Industrially used to hydrogenate vegetable oils and make margarine.
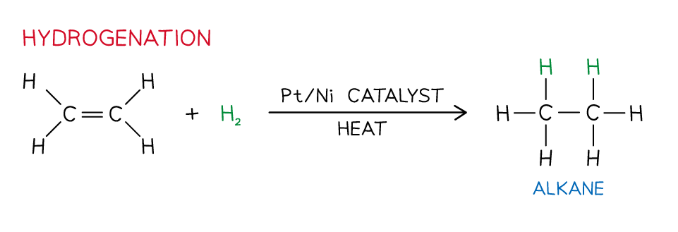
How is margarine made?
Hydrogenation is extensively used in industry to manufacture margarine
Naturally occurring vegetable oils are unsaturated and contain C=C double bonds
When these bonds react with hydrogen, some of the C=C double bonds become C-C single bonds
This process changes the properties of vegetable oil and converts it into a solid: margarine
How do alkenes react with halogens?
Halogens will react with alkenes to form dihalogenoalkanes — the halogens add across the double bond, and each of the carbon atoms ends up bonded to one halogen atom. It's an electrophilic addition reaction.
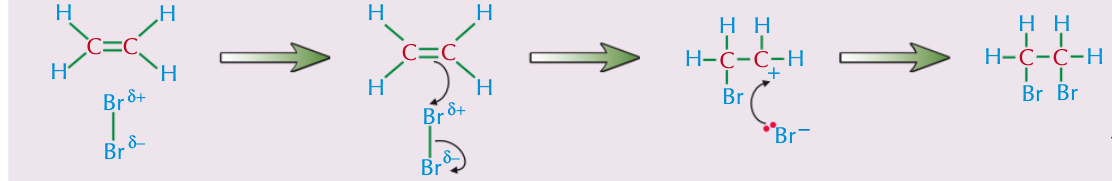
How can you test for alkenes and how does this work?
When you shake an alkanes with brown bromine water the solution will decolourise. This is because bromine is added across the double bond to form a colourless dibromoalkane.
How do alkenes react with steam?
Under the influence of a phosphoric(V) acid catalyst, alkenes react with steam at 300°C and 60-70 atm pressure, undergoing hydration to form alcohols.
This reaction is significant in the industrial production of ethanol from ethene.
A limitation is the reaction's reversible nature, with only about 5% yield. However, recycling unreacted ethene can increase overall yields to over 95%.
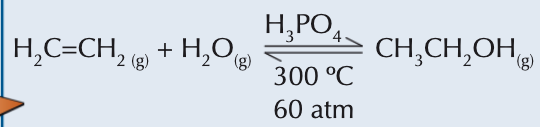
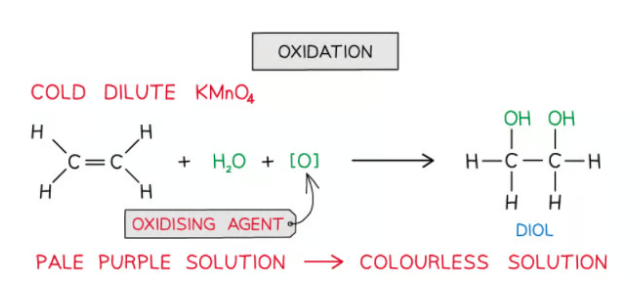
How are alkenes oxidised?
Alkenes can also be oxidised by acidified potassium manganate(VII) (KMnO4) which is a very powerful oxidising agent
When shaken with cold dilute KMnO4 the pale purple solution turns colourless and the product is a diol (an alcohol with two -OH groups)
This colour change means this reaction can be used, like bromine, to distinguish alkanes from alkenes ( alkanes do not have double bonds and so are not oxidised in this way)
How do alkenes react with hydrogen halides?
They react to form a halogenoalkane.
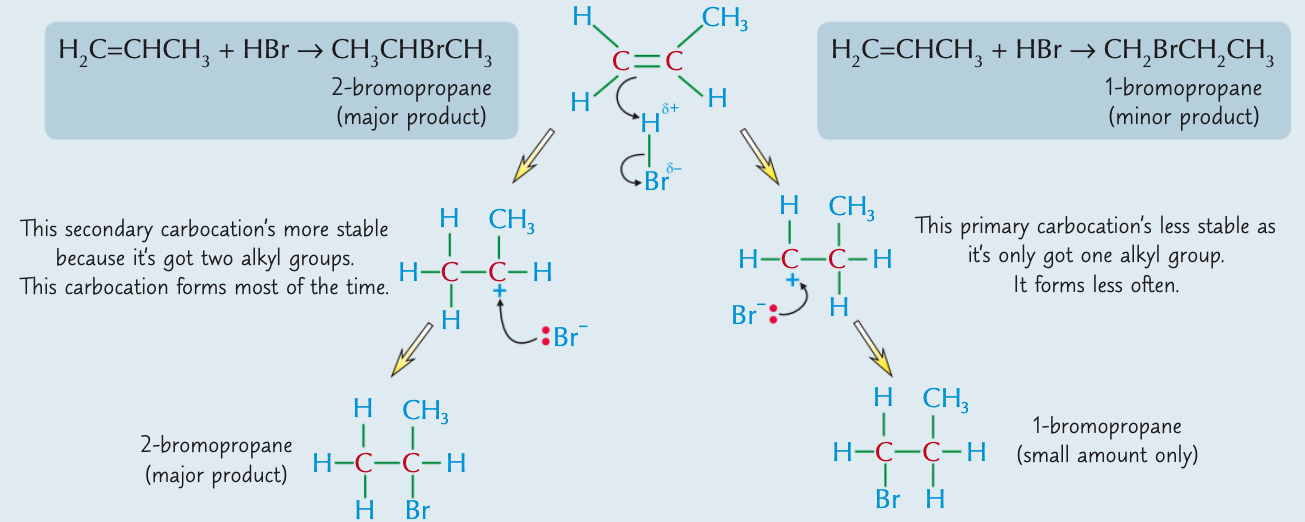
What will form if a hydrogen halide reacts with an unsymmetrical alkene?
It will form two products - the amount of each product depends on how stable the carbocation formed in the middle of the reaction is. The product formed the most is called the major product and the one formed the least is the minor product. The major product forms from the most stable carbocation.
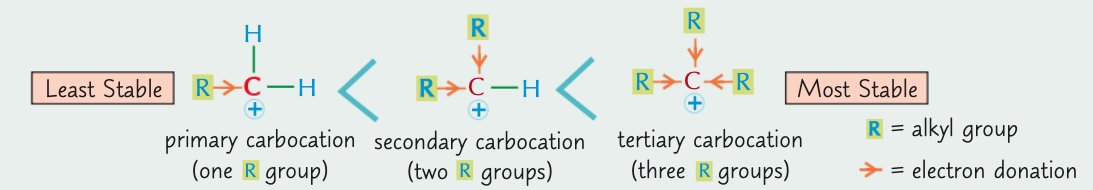
What is the most stable carbocation?
tertiary > secondary > primary. (tertiary is the most stable because the electron donating alkyl groups stab)
What is Markovnikov’s rule?
The major product from addition of a hydrogen halide (HX) to an unsymmetrical alkene is the one where hydrogen adds to the carbon with the most hydrogens already attached.
How would hydrogen bromide react with propene?
What is addition polymerisation?
Addition polymerisation is the reaction in which many monomers containing at least one C=C double bond form long chains of polymers as the only product
Just like in other addition reactions of alkenes, the π-bond in each C-C bond breaks and then the monomers link together to form new C-C single bonds
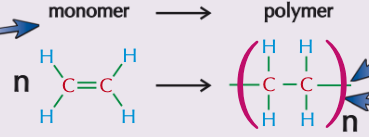
What are polymers?
A polymer is a long-chain molecule that is made up of many repeating units (monomers)